Abstract
Searching for new sources of safe nutraceuticals antitumor drugs is an important issue. Consequentially, this study designed to assess the antitumor activity of Pulicaria undulata extract in vitro in the treatment of hepatocellular carcinoma HepG2 cell line. Aerial parts of P. undulata plants were collected, used for phytochemical analysis, and assessed for anticancer activity. The antitumor activity was evaluated through studying the cell viability and apoptotic pathway. The gas chromatography–mass spectrometry phytochemical analysis revealed that P. undulata is a promising new source of several known antioxidant and antitumor compounds which could participate in drug development and exploration of alternative strategies to the harmful synthetic antitumor drugs. P. undulata stifled HepG2 cell viability in a concentration-dependent manner. Meanwhile, P. undulata tempted substantial apoptosis in HepG2 cells and enhanced the expression of miR-34a. However, the mRNA expression level of antiapoptotic B-cell lymphoma-2 was markedly decreased by P. undulata treatment. Moreover, P. undulata increased the protein expression of proapoptotic p53 and caspase 3/9 with reducing B-cell lymphoma-2 protein expression level. Thus, P. undulata induced apoptosis in the HepG2 cells by overexpression of miR-34a which regulates p53/B-cell lymphoma-2/caspases signaling pathway. These findings were well appreciated with morphological studies of cells treated with P. undulata. In conclusion, P. undulata could be a probable candidate agent for the initiation of cell apoptosis in HepG2 and thereby can serve as promising therapeutic agent for treatment of hepatocellular carcinoma which should attract further studies.
Keywords
Introduction
Hepatocellular carcinoma (HCC) represents the sixth common malignancy in the world and the second major sponsor to cancer death. 1 HCC denotes nearly 11.75% of all the gastric cancers, and around 1.68% of the whole malignancies in Egypt. 2 Failure of the apoptosis is witnessed in several humanoid tumors, which may lead to conversion of a normal cell to a tumor cell. 3
There are several management possibilities for HCC as curative resection, radiofrequency ablation, liver transplantation, trans arterial chemoembolization, radio embolization, and systemic targeted agents such as sorafenib. 4 Though the short-term survival of patients with HCC has been enhanced, these treatments have many side-effects, which are lethal and hurtful for patients, such as emotional distress, fatigue, pain and anemia, beside their high cost. 5 Programmed cell death (apoptosis) is a target for some conventional anti-cancer medications. Drugs can induce fatal intracellular damage, which often activates a down-stream cascade of molecular events. 6
MicroRNAs (miRNAs) have been shown to regulate the expression of numerous genes, including tumor suppressor genes and oncogenes such as p53 and Bcl-2. 7 However, despite extensive research, the functional role of miRNAs is not completely understood. Many miRNAs play a role in regulating apoptosis. Studies have demonstrated relationships between changes in apoptosis-related gene expression and miRNA expression during cancer therapy. 8 Further, following expression of miR-34a/p53, caspase 9 was activated, after which apoptosis occurred via the mitochondrial pathway. 9
There is now a tendency to the use of complementary remedies for treating and decreasing cancer symptoms and pain. 10 Since the earliest eras, natural products had been consumed as conventional remedies in various parts of the world including Egypt. 11 These plant products had been used for the treatment of numerous diseases counting cancer, as they have anticancer effect against diverse kinds of cancer. 12 These natural products have diverse mechanisms of action including the inhibition of cell development, variation in cell differentiation and initiation of apoptosis. 13
Pharmaceutical plants are rich source of secondary metabolites with outstanding biological activities. Henceforth, these secondary metabolites have a vital source with a variation of structural properties and arrangements.14–16 The genus Pulicaria, belonging to the Asteraceae family is utilized in traditional medication as antispasmodic, tonic, and antihyperglycemic drugs. 17 Also, they have antimicrobial, antioxidant, 18 and anticancer 19 properties.
Consequently, the aim of this study was to assess the anticancer effect of Pulicaria undulata on HepG2 cells, and to characterize the fundamental mechanism of P. undulata-induced apoptosis in HepG2 cells.
Materials and methods
Plant material
Collection and identification of plant materials
P. undulata aerial parts were collected from Wadi Hagol arid habitat, Egypt, during the flowering period (April). The plant specimen was then recognized by Professor Dr Hemmat Khattab and documented in Botany Department, Faculty of Science, Ain Shams University. Plant name has been checked with http://www.theplantlist.org. A voucher specimen was acquiesced at the herbarium of Botany department.
Preparation of plant extracts
The aerial parts leaves/stem, and flower heads were cleaned, dehydrated in the dark and then crushed to ash by mechanical grinders. The powdered plant material was soaked in different polar solvents including distilled water or ethanol, methanol, petroleum ether, ethyl acetate, or hexane at 4°C. After 72 h, the extracts were sieved, and then filtrates were concentrated on rotating evaporator under reduced pressure at 30°C. The crude concentrated extracts for every solvent were completed to ending volume with distilled water and then exposed to phytochemical examination after discharging their colors using active charcoal. The qualitative phytochemical study was carried out to detect the most appropriate solvent which was capable to harvest greater amounts of phytochemicals.
Qualitative phytochemical tests
Phytochemical screening was conducted to detect tannins, flavonoids, terpenoids, saponins, alkaloids, quinones, glycosides, glycosinolates, and anthroquinones in all plant extracts following standard protocol described by Makkar et al., 20 Harborne, 21 Francis et al., 22 and El-Olemy et al. 23
The tannins and total phenolics were measured using Folin–Ciocalteu method; the flavonoid contents by aluminum chloride colorimetric method, total saponins by vanillin reagent, and glycoside by Buljet’s reagent. The total antioxidant capacity was measured by the phosphomolybdenum method according to Prieto et al. 24
1,1-Diphenyl-2-picrylhydrazyl radical scavenging activity
The antioxidant activity of the extracts was predicted using 1,1-diphenyl-2-picrylhydrazyl (DPPH) method. 25 IC50 was measured from % inhibition versus different concentrations of standard ascorbic acid or a plant extract concentrations (1–500 μg/mL) and calculated according to the following equation
% of radicle scavenging = (absorbance of blank − absorbance of (antioxidants scavenging activity of sample)/(absorbance of blank) × 100
Antioxidant reducing power capacity
The reducing power of the extracts was determined by ferric reducing/antioxidant power (FRAP) according to the method of Benzie and Strain. 26 Results were expressed as nmol Fe2+/g dry weight.
Gas chromatography–mass spectrometry analysis
The gas chromatography–mass spectrometry (GC–MS) investigation was conducted by using Agilent 7890B GC system coupled to an Agilent 5977A MSD with a capillary column (0.6 m × 100 μm × 0 μm) (Agilent Technologies, Santa Clara, CA, USA). Helium gas was used as carrier gas with constant flow rate of 1.5 mL/min. The injector temperature and ion source temperature were established at 250°C and 230°C, respectively. The oven temperature was established at 40°C for 2 min, then 10°C/min to 180°C for 5 min followed by 10°C/min to 250°C for 10 min. The total GC process time was 38 min. The GC/MS was run in Scan/SIM mode and identification of sample’s constituents were achieved by Agilent Mass Hunter software (NIST14.L).
Cell culture
Human hepatocellular carcinoma cell lines (HepG2) were acquired from VACSERA Tissue Culture Unit. HepG2 cells were proliferated in Dulbecco’s Modified Eagle’s medium (DMEM) accompanied with 10% heat-inactivated fetal bovine serum and 1%
Cytotoxicity assay
Growth inhibitory effect of P. undulata was assessed using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Briefly, cells were seeded at a density of 1 × 104 cells/well in 96-well plates, afterward overnight incubation. The next day, the media were detached, and the cells were treated with either vehicle as a negative control or several concentrations of P. undulata (0–250 mg/mL) and incubated for 24 h. After incubation, 10% of MTT solution (2 mg/mL, Sigma, cat. no. M2128) was added per well, and the cells were incubated for another 4 h at 37°C in dark. The formazan crystals dissolved in dimethyl sulfoxide (DMSO) (100 μL/well, Sigma, cat. no. D2160) added to each well with continuous shaking for 5 min. Plate absorbance was read with a microplate reader (SunRise; TECAN Inc., San Jose, CA, USA) at 540 nm. The proportion of viability was calculated from equation [1 − (ODt/ODc) × 100%], where ODt is the optical density of wells treated with the investigated sample and ODc is the mean optical density of untreated cells. Triplicate wells were evaluated for each investigation. The concentration essential to cause fatal effects in 50% of intact cells (IC50) was assessed from graph plots of the dose response curve for each concentration using Graph Pad Prism software (San Diego, CA, USA).
Morphology study
After treatment of HepG2 cells with various concentrations of P. undulata, the HepG2 cells were stained with crystal violet stain. The treated cells were observed, compared to untreated cells, and photographed by inverted light microscopy.
Quantitative reverse transcription polymerase chain reaction
Total RNA, including miRNAs, was isolated from HepG2 cells using Trizol reagent (Invitrogen; Thermo Fisher Scientific, Waltham, MA, USA) according to the instructions of the manufacturer. Expression of miR-34a was analyzed with the mirVana™ PARIS™ Kit (Qiagen, Germantown, MD, USA), according to the manufacturer’s protocol; RNA was reverse transcribed into cDNA using the TaqMan® MicroRNA reverse transcription kit. Mature miR-34a expression was quantified using SYBR green master mix and small nuclear RNA U6 was used for normalization. For the analysis of B-cell lymphoma-2 (Bcl-2) and caspase 9 expression, cDNA was synthesized using the Thermo Scientific RevertAid™ First Strand cDNA Synthesis Kit (no. K1621) as described by the manufacturer. The housekeeping gene glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used for normalization.
The primers used were as follows: Bcl-2: 5′-ATG TGT GTG GAG AGC GTC AA-3′ and 5′-ACA GTT CCA CAA AGG CAT CC-3′; Casp 3: 5′-TGT TTG TGT GCT TCT GAG CC-3′ and 5′-CAC GCC ATG TCA TCA TCA AC-3′; Casp 9: 5′-AGC CAG ATG CTG TCC CAT AC-3′ and 5′-CAG GAG ACA AAA CCT GGG AA-3′; p53: 5′-TGT GGA GTA TTT GGA TGA CA-3′ and 5′-GAA CAT GAG TTT TTT ATG GC-3′; GAPDH: 5′-CCA GGG CTG CTT TTA ACT C-3′ and 5′-GCT CCC CCC TGC AAA TGA-3′; mature miR-34a: 5′-TGG CAG TGT CTT AGC TGG TTG T-3′; U6 snRNA: 5′-CGC TTC GGC AGC ACA TAT ACT AA-3′ and 5′-TAT GGA ACG CTT CAC GAA TTT GC-3′.
All the reactions were run in triplicate. Fold changes in gene expression were calculated using 2−ΔΔCt method quantification of miR-34a, Bcl-2, P53, Casp 3 and Casp 9 mRNA expression levels.
Western blot to detect the expression of p53 and apoptosis-related proteins (Bcl-2, caspase 3, and caspase 9)
Western blotting technique was used to estimate protein expression levels of p53, Bcl-2, and caspase 3/9. Western blot analysis was performed using standard techniques as described previously. 27 After treatment of HepG2 cells (1 × 104) with 0, 7.8, 31, 62.5, and 250 mg/mL of P. undulata for 24 h, cell lysates were prepared. The cells were harvested, washed with phosphate buffer saline (PBS), and lysed in protein lysis buffer containing protease inhibitors (10 μg/mL leupeptin, 2 mM phenylmethylsulfonylfluoride (PMSF), and 2 mM Na3VO4). Cell lysates were centrifuged at 13,000g at 4°C for 10 min to extract proteins. The protein concentration was determined by Bradford 28 assay. Subsequently, proteins (20 µg/lane) were fractionated by 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to a polyvinylidene difluoride (PVDF) membrane. Later, membranes were blocked with 5% skimmed milk in Tris-buffered saline Tween-20 (0.1% TBST) for 1 h at room temperature and incubated with monoclonal primary antibodies for p53, Bcl-2, caspase 3, caspase 9, and β-actin as loading control at 4°C overnight then incubated with appropriate horseradish peroxidase (HRP)-conjugated secondary antibodies at room temperature for 2 h on a rotary shaker. Finally, membranes were washed in 10 mM Tris–Cl, pH 7.5, 100 mM NaCl, and 0.1% Tween 20. At last to visualize the blots, chemiluminescence detection was performed with the Amersham detection kit according to the constructer’s protocols and exposed to X-ray film. Results of protein expression levels were normalized to β-actin and the bands were scanned and quantified by densitometric analysis.
Statistical analysis
All statistical investigations were carried out using SPSS 18.0 software. Student’s t-test or one-way analysis of variance (ANOVA) were used to compare alterations between groups. p value <0.05 is considered statistically significant.
Results
Phytochemical screening of P. undulata
The qualitative phytochemical screening of crude extracts of P. undulata aerial parts was performed to detect the presence of nutraceuticals bioactive metabolites and the most suitable solvent. The extracts of P. undulata aerial portions revealed the presence of flavonoids, phenols, tannins, coumarins, triterpenoids, alkaloids, steroids, glucosinolates, and glycosides. The best appropriate results are assayed in the alcoholic extracts particularly methanol (Supplementary Table 1). Consequently, the secondary metabolites quantities were assayed in the aerial portions of P. undulata methanol extract.
The quantitative study of the total phytoconstituents of P. undulata revealed the presence of total phenols 4.130 µg/g dry matter, 1.61 µg of flavonoids/g dry matter and 1.2 µg of total tannins/g dry matter. Moreover, 261.0 µg of total saponins, 14.72 µM of glycosides, 85.1 µmol glucosinolates, and 4.3 mg total alkaloids were determined in 1 g of dry matter (Table 1).
Quantitative phytochemical analysis of P. undulata methanolic extract.

DW: dry weight; DPPH: 1,1-diphenyl-2-picrylhydrazyl; FRAP: ferric reducing/antioxidant power.
Each value is a mean of three different replicates + SE.
In addition, the antioxidant activity of the test extract was assayed by DPPH radical-scavenging and total antioxidant capacity. Methanol extract showed potential free-radical activity (DPPH) of 28.1% and total antioxidant capacity (FRAP) of 119.1 nmol Fe2+/g dry weight, respectively (Table 1).
In the meantime, the GC-MS analysis revealed that, 47 major different phyto-components were recognized in the methanolic extract of P. undulata aerial portions along with their retention time (RT), molecular formula, and peak area (Figure 1 and Supplementary Table 2). The major identified bioactive constituents (Supplementary Table 2) are characterized by the antimicrobial, antiulcerogenic, antifungal, anticancer, antioxidant, and anti-inflammatory properties.

GC-MS spectrum of phytochemical constituents of P. undulata methanolic extract.
Cytotoxic effect of P. undulata in HCC cell line
We examined the cytotoxic effect of P. undulata in HCC cell line (HepG2). Exposure of the tumor cells for 24 h to several concentrations of P. undulata caused a concentration-dependent reduction in cell viability (Figure 2). The mean IC50 value of the HepG2 cell line was found to be 27.7 mg/mL.

Cytotoxic effect of P. undulata on human hepatocellular carcinoma cell line.
Morphology study
In order to explore the effect of P. undulata on tumor cell growth, the morphological variations of malignant cells were observed with the inverted light microscope after staining with the crystal violet stain. As shown in Figure 3, after treatment with different concentrations of P. undulata for 24 h, the density of cells diminished and morphology altered. For instance, fragmentation, cell shrinkage, and rounding can be observed. The treated HepG2 cells showed a decrease in the cell counts with increasing concentration of P. undulata. The results showed that P. undulata can obstruct the proliferation and growth of cancer cells.

Effect of P. undulata on the HepG2 cell morphology.
Effect of P. undulata on miRNA-34a expression level in HepG2 cells
The effect of P. undulata on miRNA-34a expression was determined using quantitative reverse transcription polymerase chain reaction. Our results showed that P. undulata could expressively increase miRNA-34a expression level in HepG2 cells as compared to untreated cells (Figure 4).

Effects of P. undulata on the expression level of miRNA-34a in HepG2 cells detected by RT-qPCR.
Effect of P. undulata on apoptosis-related gene expression levels in HepG2 cells
P. undulata treated HepG2 cells showed up-regulation of pro-apoptotic gene p53 and significant decrease of anti-apoptotic Bcl-2 (Figures 5 and 6, respectively). Also, it resulted in up-regulation of caspases 3 and 9 mRNA expression levels compared to untreated HepG2 (Figure 7). These results indicated that P. undulata can inhibit HepG2 cell proliferation and activate apoptosis pathway.
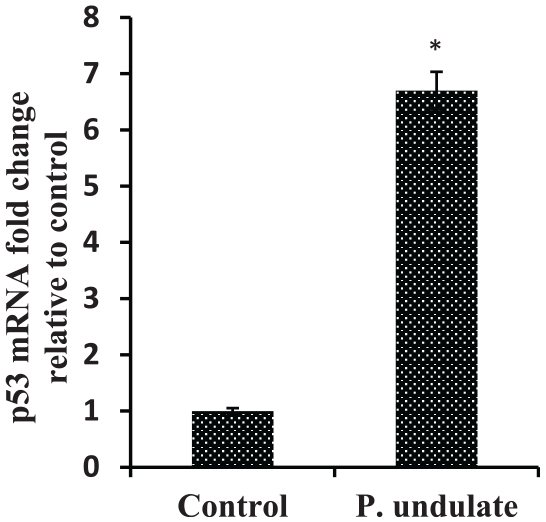
Effects of P. undulata on the expression level of p53 in HepG2 cells detected by RT-qPCR.

Effects of P. undulata on the expression level of Bcl-2 in HepG2 cells detected by RT-qPCR.
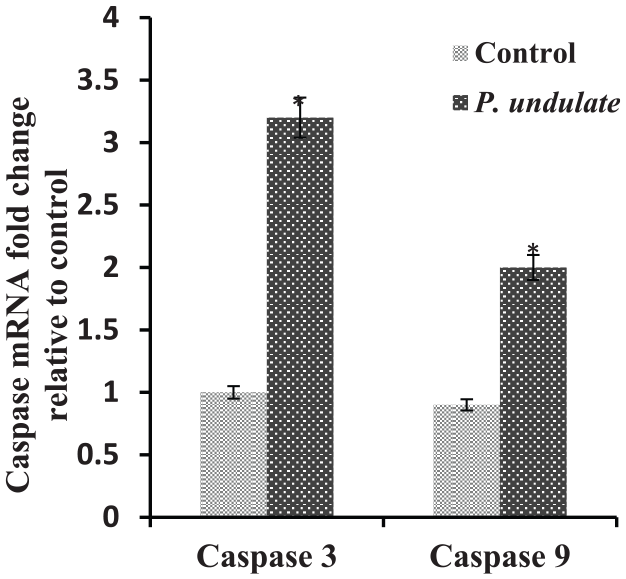
Effects of P. undulata on the expression level of caspases 3 and 9 in HepG2 cells detected by RT-qPCR.
Effect of P. undulata on protein expression of p53 and apoptotic markers
Treatment with different concentrations of P. undulata extract increased the expression of p53 and caspase 3/9 proteins while decreased the protein expression of antiapoptotic Bcl-2 protein in a dose-dependent manner (Figure 8) compared to control group. Thus, Western blotting revealed that P. undulata ameliorated tumor suppressor and apoptosis proteins.

Western blot analysis of proteins p53, Bcl-2, caspase 9, and caspase 3 relative to β-actin in HepG2 cells (control) and HepG2 cells treated with P. undulata (7.8, 31, 62.5, and 250 μg/mL) for 24 h. The bottom panels represented quantification of the immunoblot by densitometry.
Discussion
Currently, there is an increase call of safe nutraceutical drugs. The traditional pharmaceutical plants are important economic source of raw materials for the drug industry. Meanwhile, number of wild plants revealed antioxidants, antimicrobial, anti-inflammatory, and antitumor properties.29–31 Medicinal plants research in the family of Asteraceae has revealed anti-cancer efficacy in pre-clinical models.32,33 The genus Pulicaria, belonging to the tribe Inuleae of the Asteraceae family, consists of 100 species with a distribution from Europe to North Africa and Asia, particularly around the Mediterranean. 34
P. undulata has been used formerly as a conventional treatment and it contains varies classes of compounds as flavonoids, phenols, glucosinolates, cardiac glycosides, alkaloids, steroids, terpenes, and coumarins (Table 1), which are consequently, among the most potential mediators that could be used to treat chronic diseases. The GC–MS analysis (Supplementary Table 2) has shown the presence of 47 different phytochemical compounds in P. undulata methanolic extract. These phytochemicals are characterized by free radical scavenging skills; therefore, they have been recommended as anti-inflammatory, 35 antibacterial, 36 and antitumor activities. 31
In this study, the methanolic P. undulata extract exhibited DPPH radical scavenging potential and total antioxidant capacity. Such results pointed out that the methyl alcohol extracts of P. undulata, might be used as a powerful source of natural antioxidants. The positive associations between phenolic compounds and antioxidant activities (DPPH and FRAP) of plant extract 37 suggests the possible uses of P. undulata, in the amendment and treatment of some human diseases. 38 The existence of other compounds such as saponins, cardiac glycosides, and alkaloids in the methanol extract of P. undulata could be also involved in the antioxidant biological activity and thereby, medicinal uses. 39
Natural products are great source of new therapeutic agents which have been broadly explored as anti-cancer drugs from plants, marine organisms, and microorganisms. 40 Numerous researches have shown that natural components are suitable to stop and heal cancer by pointing to its vital hallmarks. 41 Some natural herbal drugs are also established to treat HCC. 42 In the existing study, MTT assay showed that P. undulata significantly reduced the cell viability and proliferation of HCC cell line in a concentration-dependent manner, suggesting that P. undulata could represents a novel candidate agent for curing HCC.
Also, it was reported formerly that the aqueous methanol extract of P. undulata contains kaempferol, kaempferol 3O-b-glucoside, quercetin, quercetin 3,7-di OCH, quercetin 3-O-b-galactoside, quercetin 3-O-b-glucoside, and caffeic acid. 43 Polyphenolic compounds, like quercetin, have been shown in experimental studies to amend the expression of numerous cancer-related miRNAs, including the cancer-associated let-7 family. 44 Previous studies have shown that a treatment with quercetin prompted several cellular actions such as cell cycle arrest, p53 activation, and induction of caspase-mediated apoptosis in HCC cells.45,46
miRNAs are being considered as striking goals for cancer prevention and treatment due to their tumor suppressor or oncogenic activities. 47 Many studies have proposed that the modulation of miRNAs serves as one of the vital machineries in the anticancer activities of a variety of natural agents. Moreover, mechanistic studies have revealed that they exert their anti-proliferative and/or pro-apoptotic effects to prevent the existence and/or spread of many malignancies by targeting several key elements in intracellular signaling network involved in carcinogenesis. 46
miRNA-34a (miR-34a) is a tumor suppressor that has attracted considerable attention in recent years. It modulates cancer cell invasion, metastasis, and drug resistance, and has also been evaluated as a diagnostic and/or prognostic biomarker. 48 miRNA-34a shows divergent expression in numerous cancer types like colon cancer, 49 prostate, 50 and pancreatic cancer. 51 Moreover, deregulation of miR-34a affects many biological processes like apoptosis, senescence, cell cycle, differentiation and growth.52,53
The strategy of miR-34a replacement has been investigated in clinical trials as the first attempt of miRNA application in cancer treatment. However, emerging outcomes promote the re-evaluation of existing knowledge and urge the need for better understanding the complex biological role of miR-34a. The targets of miR-34a encompass numerous regulators of cancer cell proliferation, survival, and resistance to therapy. miR-34a expression is transcriptionally controlled by p53, a crucial tumor suppressor pathway, often disrupted in cancer. Moreover, miR-34a abundance is fine-tuned by context-dependent feedback loops. The function and effects of exogenously delivered or re-expressed miR-34a on the background of defective p53 therefore remain prominent issues in miR-34a-based therapy. 54
Genetic mutations of p53 gene are the most common alteration detected in HCC. 55 Significantly, p53 is a vital target for miR-34a and it is shown that expression of miR-34a in mutant p53 is lower as compared to those cell lines bearing wild type p53. It is recommended that role of miR-34a in tumor development may be closely linked with p53 mutation and is inversely connected to Bcl-2 expression. 34
Bcl-2 is an anti-apoptotic protein playing a key role in the apoptotic signaling pathway. Bcl-2 can hinder cell apoptosis and recover cell proliferation. 56 In the apoptosis pathway, miR-34a was supposed to transduce the apoptosis signal from the p53 to other apoptosis-related proteins, such as cell survival-related gene Bcl-2. 57 The cysteine proteases activation, particularly caspases, is a crucial intracellular regulator of cell apoptosis. 58 Caspase 3 is a significant mediator of apoptosis 59 that is triggered by a diversity of activators including caspase 9. 58
Targeting Bcl-2 family proteins and tumor suppressor proteins is crucial in molecular target that ultimately leads to anti-cancer drug discovery. To analyze the mechanism underlying the P. undulata-induced apoptosis, we tested the expression of p53, Bcl-2, and caspase 3/9.
This data showed that up-regulation of miRNA-34a and its target gene p53 while, down-regulation of Bcl-2 after P. undulata treatment of HepG2 induces mitochondrial dysfunction by discharging cytochrome c and inducing the apoptosis of cancer cells through caspases 3 and 9 activation. The expression levels of caspase 9 and caspase 3 were significantly upregulated in the HepG2 cells succeeding P. undulata treatment.
To the best of our knowledge, this is the first study illustrating the anti-apoptotic effect of P. undulata on HepG2 cell line, which also highlights the possible mechanism underlying the cytotoxic effect of P. undulata. In conclusion, based on the above-mentioned results, this study suggests that P. undulata may be a candidate agent for the treatment of human HCC.
Supplemental Material
supp_table_1_and_2_pulicaria – Supplemental material for Assessment of anticancer activity of Pulicaria undulata on hepatocellular carcinoma HepG2 cell line
Supplemental material, supp_table_1_and_2_pulicaria for Assessment of anticancer activity of Pulicaria undulata on hepatocellular carcinoma HepG2 cell line by Manal A Emam, Hemmat I Khattab and Marwa GA Hegazy in Tumor Biology
Footnotes
Author contributions
All authors contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study does not involve any primary biological material from humans or animals.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
