Abstract
Mesenchymal stem cells have therapeutic properties that are related to their potentials for trans-differentiation, immunomodulation, anti-inflammatory, inhibitory effect on tumor proliferation, and induction of apoptosis. This study was performed to analyze the role of mesenchymal stem cells as an alternative for cellular signaling growth factors involved in the pathogenesis of leukemogenesis in rats. Treatment of rats with 7,12-dimethyl benz [a] anthracene induced leukemogenesis appeared as a significant decrease in hematological parameters with concomitant significant increase in bone marrow oxidative and inflammatory indices (transforming growth factor beta and interleukin-6) in comparison with normal groups. On the contrary, Western immunoblotting showed a significant increase in the signaling growth factors: PI3K, AKT, mTOR proteins and a significant decrease in PTEN in 7,12-dimethyl benz [a] anthracene–treated group. In addition, a significant increase in the transcript levels of B cell lymphoma-2 protein gene in the 7,12-dimethyl benz [a] anthracene group, while that of C-X-C motif chemokine receptor-4 and B cell lymphoma-2 protein associated x-protein were significantly downregulated compared to controls. Meanwhile, therapeutic mesenchymal stem cells treatment predict a significant improvement versus 7,12-dimethyl benz [a] anthracene group through the modulation of growth factors that confront bone marrow dysplasia. In the same direction treatment of 7,12-dimethyl benz [a] anthracene group with mesenchymal stem cells, it induced apoptosis and increased the homing efficacy to bone marrow. In conclusion, mesenchymal stem cells improve hematopoiesis and alleviate inflammation, and modulated PI3K/AKT signaling pathway contributed to experimental leukemogenesis.
Keywords
Introduction
Mesenchymal stem cells (MSCs) can restore and repair injured tissues. Its use in preclinical and clinical evidence showed the therapeutic benefit in various medical conditions. Currently, MSCs are the most commonly used cell-based therapy in clinical trials because of their reconditioned effects. 1
Leukemia is a malignant disease listed in the blood-forming organs through the production of increased numbers of immature or abnormal leukocytes, leading to anemia and other symptoms. Acute myeloid leukemia (AML) characterized by the proliferation of immature transformed myeloid blasts in the bone marrow (BM) moves to the blood stream and other parts of the body.2,3
Leukemias are hematopoietic processes initiated by rare leukemia stem cells engender from the transmutation of hematopoietic stem cells (HSCs) or compelled ancestor cells. 4 During malignant conversion, leukemia stem cells have the ability of self-renewal, proliferation, and differentiation through continuous genetic and epigenetic permutation and clonal variegation. 5 Acute amyloid leukemia involving different signaling pathways are expected to be involved in the clinical treatment of acute amyloid leukemia. 6
The PI3K/AKT/mTOR signaling pathways are detectable and activated in most cases of AML. Inhibition of PI3K/AKT or mTORC1 signaling pathways suppresses the proliferation in vitro. 7 In this study, we describe a novel mechanism by which MSCs can inhibit PI3K/AKT pathway that is up regulated in leukemia to provide guidance to develop an efficient and safe targeted therapy using MSCs.
Materials and Methods
Isolation of MSCs
According to Alhadlaq and Mao, 8 MSCs were isolated as follows:
BM was harvested by flushing the tibiae and femurs of 6-week-old male Wistar rats with Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum.
Nucleated cells were isolated with a density gradient (Ficoll/Paque (Amersham Pharmacia, Uppsala, Sweden)) and re-suspended in complete culture medium supplemented with 1% penicillin streptomycin.
Cells were incubated at 37°C in 5% humidified CO2 for 12–14days as primary culture or after the formation of large colonies.
When large colonies have developed (80%–90% confluence), cultures were washed twice with phosphate buffered saline, and the cells were trypsinized with 0.25% trypsin in 1 mM ethylenediaminetetraacetic acid (EDTA) for 5 min at 37°C.
After centrifugation, cells were re-suspended with serum-supplemented medium and were incubated in 50 cm2 Falcon culture flasks.
The resulting cultures were referred to as first passage cultures. On day 14, the adherent colonies of cells were trypsinized and counted.
Identification of MSCs Cells was by their morphology, adherence using flow cytometry.
Animals
Male Wistar rats at 27 days of age and weighing 25–45 g were used in this study. The animals were housed in plastic cages, under standard conditions of 12 h light/12 h dark cycle, normal temperature, good ventilation, and humidity range. The animals were provided water and food ad libitum throughout the study.
All the experimental procedures were carried out according to the principles and guidelines of the Ethics Committee of the National Research Center conformed to “Guide for the care and use of Laboratory Animals” for the use and welfare of experimental animals, published by the US National Institutes of Health (NIH publication no. 85–23, 1996).
The animals were randomly divided into three groups, each consisting 10 animals as follows:
Control group: 10 rats in this group were kept as control.
DMBA group: 10 rats were given three intravenous 35 mg/kg body weight 7,12-dimethyl benz [a] anthracene (DMBA) in dimethyl sulfoxide (DMSO) at biweekly interval beginning at the 27th day after birth. 9
DMBA + MSCs group: 10 rats were given three intravenous 35 mg/kg body weight 7, 12 Dimethyl Benz [a] anthracene in DMSO at biweekly interval beginning at the 27th day after birth and were treated once with MSCs (3.5 × 106 MSCs) till the end of the experiment.
At the end of the experimental period, all animals were sacrificed using diethyl ether, and the blood was collected via heart puncture. Part of the whole blood was collected in EDTA tubes for hematological study used for complete blood count (CBC). The other part was collected as serum for the biochemical parameters estimations. Both femurs were removed flushing of BM for the biochemical estimations. Also part of femur was rapidly excised for histopathological studies.
Levels of interleukin-6 (IL-6) and transforming growth factor (TGF)-β1 were determined using the corresponding enzyme-linked immunosorbent assay (ELISA) kit following protocols provided by the manufacturers (My Biosource Inc, San Diego, CA, USA).
Western immunoblotting
Western immunoblotting of BM tissue proteins quantified as previously described. 10
In brief, 20 µg of protein per lane were separated with 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membranes. The membranes were incubated for 2 h at room temperature with blocking solution then incubated overnight at 4°C with the primary antibodies toward PI3K, AKT, mTOR, and PTEN with β-actin as loading control. After washing three times with the blocking solution, the membranes were incubated with the secondary monoclonal antibodies conjugated to horseradish peroxidase at room temperature for 2 h and then membranes were washed four times with the same washing buffer. Membranes were developed and visualized by hemi luminescence using Amersham detection kit according to the manufacturer’s protocols, then exposed to X-ray film. Quantification of PI3K, AKT, mTOR, and PTEN proteins were performed by densitometric analysis of the autoradiograms using a scanning laser densitometer (Biomed Instrument Inc, Clinton Charter Township, Michigan, USA). Results were expressed as arbitrary units after normalization for β-actin.
Molecular investigation
mRNA gene expression of C-X-C motif chemokine receptor-4 (CXCR-4), B cell lymphoma-2 protein (Bcl-2), and B cell lymphoma-2 protein–associated x-protein (Bax) were determined by Quantitative Real Time polymerase chain reaction (qRT-PCR) in BM tissue. The sequence of the primers used for RT-PCR was observed in Table 1. Relative expression of target mRNA was calculated using the comparative Ct method using the Pfaffl 11 method.
Sequence of the primers used for real-time PCR.

Bcl-2: B cell lymphoma-2; Bax: B cell lymphoma-2 protein–associated x-protein; CXCR-4: C-X-C motif chemokine receptor-4.
Histopathological assessment
Femur (BM samples) were taken from rats in different groups and fixed in 10% formal saline for 24 h. Decalcification procedure was completed, and the tissues were washed in distilled water and then in serial dilutions of alcohol and were dehydrated. Specimens were cleared in xylene and embedded in paraffin. Paraffin blocks were sectioning at 4 μm thickness by sledge microtome and collected on glass slides deparaffinized and stained by hematoxylin and eosin (H&E) stain for examination through the light electric microscope.
Statistical analyses
All data were expressed as the mean ± standard error (SE). One-way analysis of variance (ANOVA) with least significant difference (LSD) was used to test for differences in means of variables between groups. A probability of
Results
Immunophenotyping of MSCs
The expression of cell surface markers of MSCs were evaluated by flow cytometry. As shown in Figure 1, the BM-MSCs weakly expressed CD34 (≤4%), (a specific cell surface markers of HSCs). However, CD29, CD105, and CD90 (≥90 %), which are specific cell surface markers of MSCs, were strongly expressed.
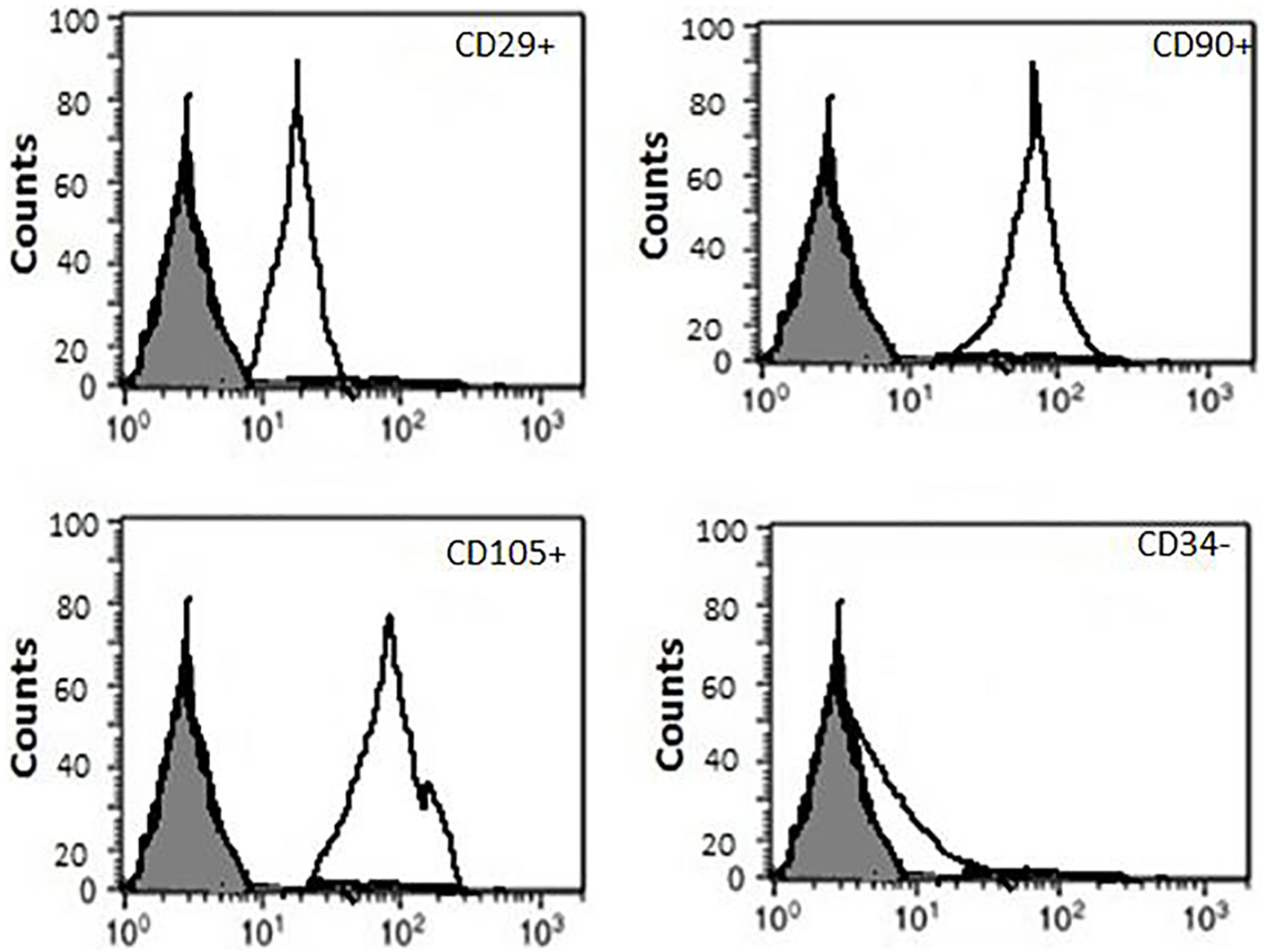
Flow cytometric characterization of the expression of surface antigens.
Hematological parameters
It has been established that DMBA can induce leukemia and myelosuppression, which is characterized by cytopenia associated with anemia and bleeding. Table 2 shows the effect of DMBA and MSCs on hematological parameters. DMBA produces a significant decrease (
Effect of MSCs on WBCs, platelets counts, RBCs, MCV, and Hb content in DMBA-treated rat.
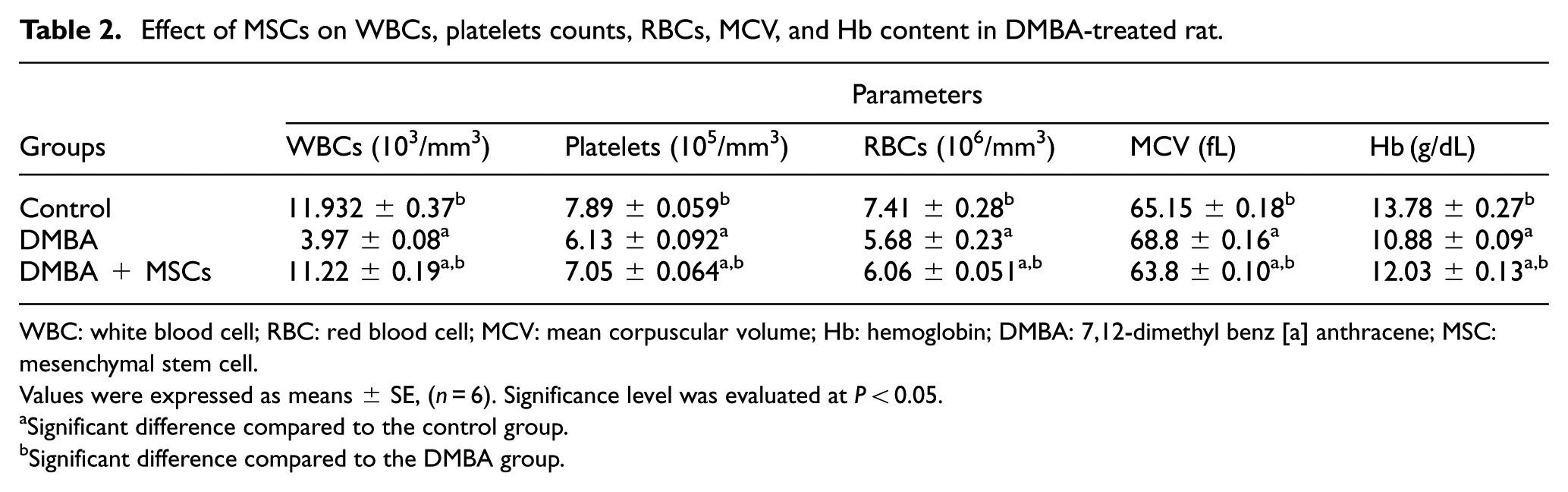
WBC: white blood cell; RBC: red blood cell; MCV: mean corpuscular volume; Hb: hemoglobin; DMBA: 7,12-dimethyl benz [a] anthracene; MSC: mesenchymal stem cell.
Values were expressed as means ± SE, (
Significant difference compared to the control group.
Significant difference compared to the DMBA group.
Oxidative stress markers
DMBA induced leukemia and BM injury throughout the significant increase in bone marrow lipid peroxidation (MDA) level and significant decrease in glutathione (GSH) activity in experimental group compared with control group. Meanwhile, MSCs treatment showed statistically significant improvement versus the DMBA group. A significant modulation in studied parameters that record the leukemia and BM toxicity (Table 3).
Effect of MSCs on bone marrow lipid peroxidation (MDA) and glutathione (GSH) in DMBA-treated rats.

DMBA: 7,12-dimethyl benz [a] anthracene; MSC: mesenchymal stem cell; MDA: marrow lipid peroxidation; GSH: glutathione.
Values were expressed as means ± SE, (
Significant difference compared to the control group.
Significant difference compared to the DMBA group.
Inflammatory markers
Figure 2 showed that DMBA injection produces a significant increase in the pro-inflammatory cytokines TGF-β and IL-6 levels as compared with the control group. Meanwhile, treatment of DMBA group with MSCs produces a significant modulation in both cytokines (TGF-β and IL-6) levels as compared with the control and DMBA groups was predicted.
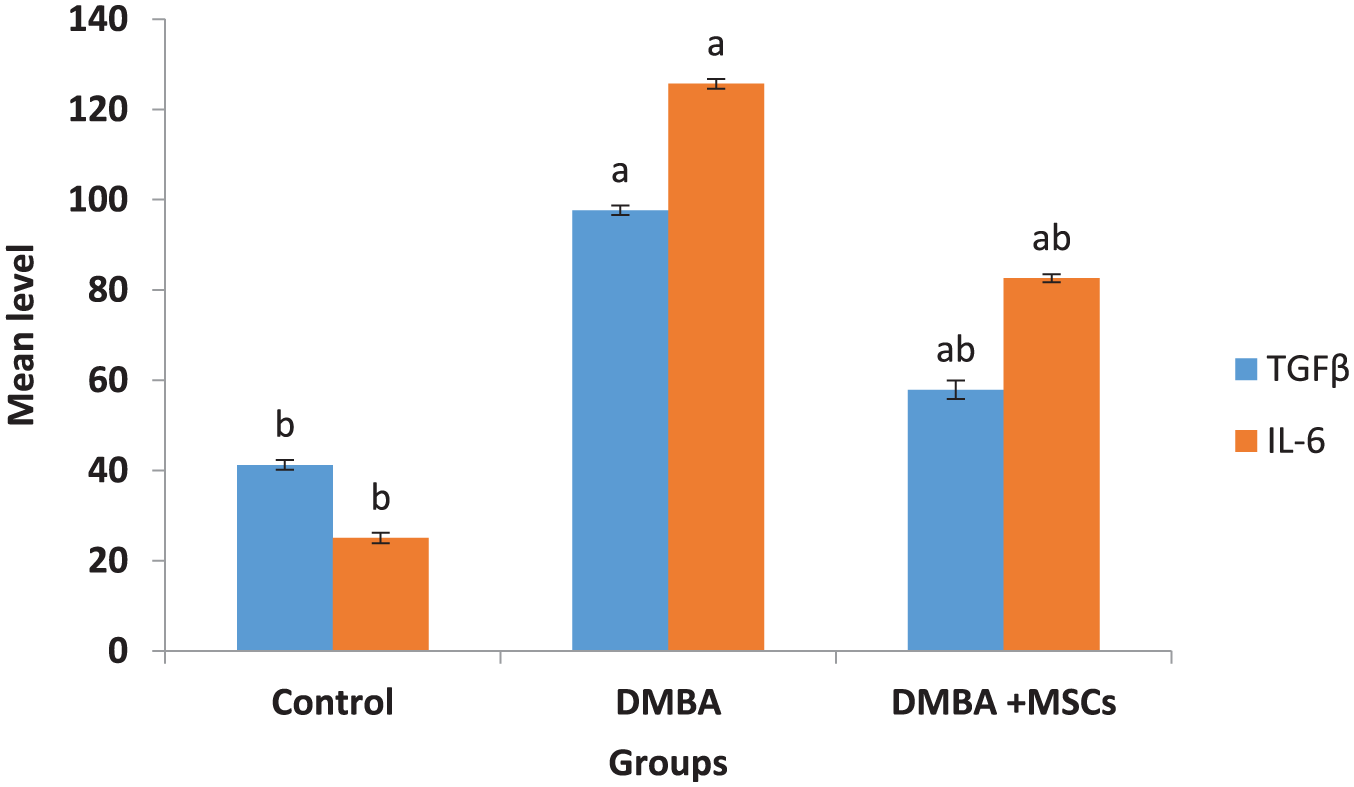
Effect of MSCs on inflammatory cytokines.
Western immunoblotting
Figure 3 showed that DMBA treatment induced a higher significant level of the signaling: PI3K, AKT, mTOR, and lower level of PTEN proteins. Meanwhile, therapeutic infusion of MSCs showed a significant improvement versus the DMBA group. These data suggest that PI3K/AKT/mTOR/PTEN signaling pathways are essential for inducing AML.

Influence of MSCs administration on phosphorylation of PI3K, AKT, mTOR and PTEN: (a) Western blot analysis and immuno-blots for each protein from one representative experiment and (b) the accumulated, normalized data from five independent experiments are shown for PI3K, AKT, mTOR, and PTEN phosphorylation.
Levels of CXCR-4, Bcl-2, and Bax
By RT-PCR expression of CXCR-4, Bcl-2 and Bax genes were analyzed. The transcript levels of Bcl-2 were up regulated; however, the expression of both CXCR-4 and Bax were downregulated in DMBA-treated group in comparison with control. In the treatments using MSCs, the Bcl-2 gene expression was significantly downregulated and that of CXCR-4 and Bax were up regulated in the levels from control (Figures 4 and 5).

Relative gene expression of CXCR-4 in bone marrow of rats in different groups.

Effect of MSCs on apoptosis.
Histopathology
Histological appearance of control BM displays the normal BM structure and its various component including adipose tissue (AT), blood sinusoid, megakaryocyte, megakaryoblast, and normoblast (Figure 6(a) and (b)). When experimental rats were injected with DMBA, the BM biopsy showed markedly hypo-cellularity along with increased immature granulocytic precursors, megakaryocytes, and lower erythroid precursors (Figure 6(c) and (d)). Conversely, the BM biopsy of rats injected with DMBA and treated with MSCs showed normal appearance of marrow cells (Figure 6(e)).
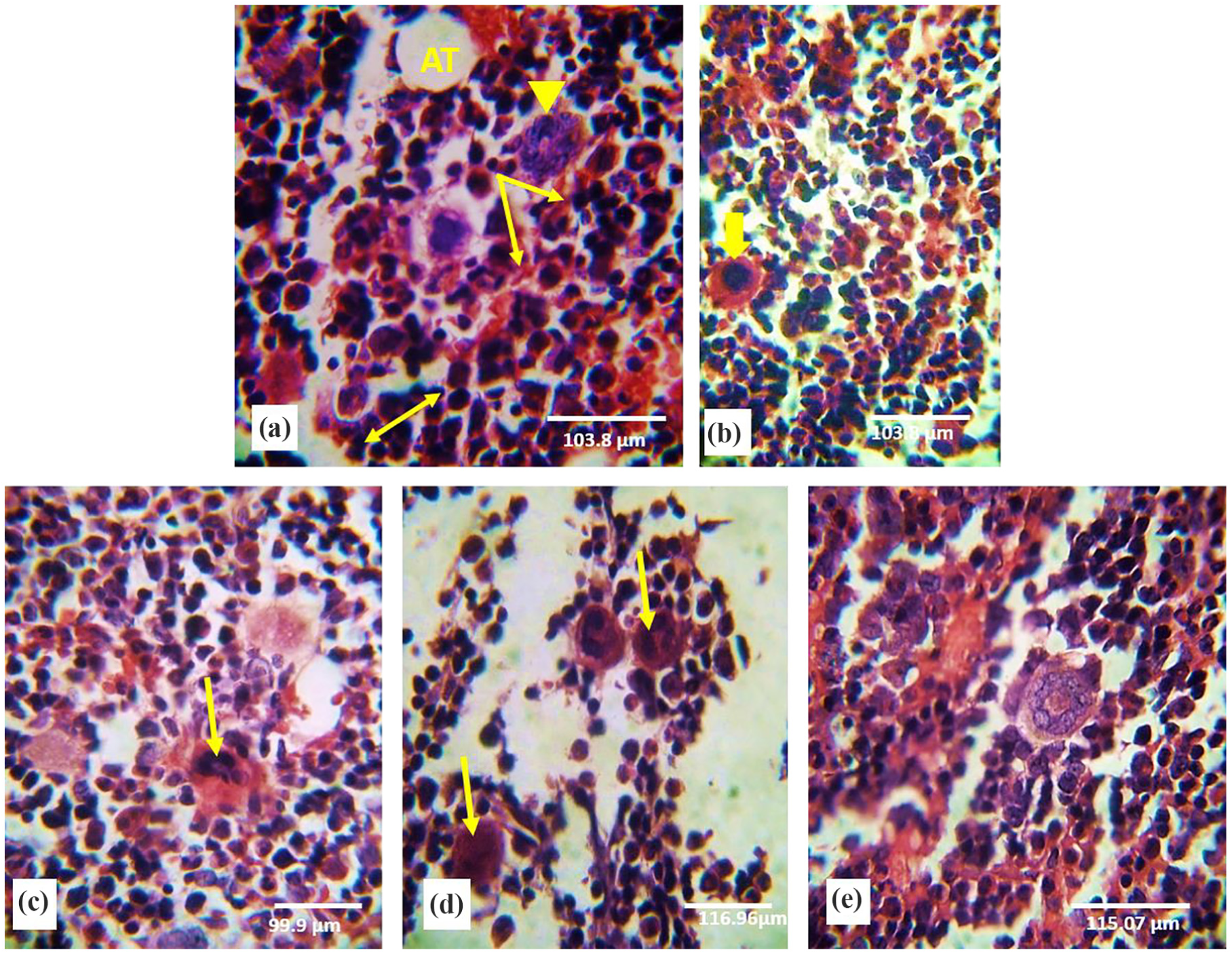
Photomicrograph of bone marrow biopsy stained with H&E: ( a) and (b) control bone marrow tissue sections represent adipose tissue (AT), blood sinusoid (↓), megakaryocyte (▾), megakaryoplast ( yellow blocked arrow) and normoplast (↔); (c) and (d) bone marrow tissue sections of rats treated with DMBA showing markedly increased immature granulocytic precursors and megakaryocytes (↓) but less erythroid precursors; and (e) bone marrow tissue sections of rats treated with DMBA and MSCs showed showing the normal appearance of marrow cells.
Discussion
In the present investigation, data showed that several pulse doses of intravenous DMBA injection exhibited signs of BM myelosuppression associated with changes and signs of earliest stage of leukemogenesis. Pulse DMBA injections damaged BM stem cells by reducing peripheral leukocyte count and produced leukemia in experimental rats.12,13
In this study, histopathological observations of BM biopsy showed markedly hypo-cellularity along with increased immature granulocytic precursors, megakaryocytes, and lower erythroid precursors when experimental rats injected with DMBA. On the other hand, DMBA damage was established by a decline in the peripheral blood cells and BM cells. Repeated biweekly dosing of rats with DMBA (potent carcinogens) induced cytotoxic genotoxic damage, carcinogenic and immune-toxic effects 14 in various species, tissues, and cell types; 15 especially in the BM through covalent binding of DMBA dihydrodiol epoxides to DNA. DMBA dihydrodiol epoxide DNA adducts in BM-cause mutations and depressions of DNA synthesis in hemopoietic cells, initiating leukemias and lymphomas. 16
Once leukemia was initiated, there was a gradual onset of BM depletion with concomitant depression in circulating blood cells (RBCs, WBCs, and platelets) 12 after DMBA injection. After single and multiple DMBA injections, preleukemic cells were observed in BM and peripheral blood. Failure in recovery from the intrinsic “physiological anemia” following DMBA injections seemed to be the important cause of leukemia.
Leukemia resulted in hematopoiesis impairment, which may be due to block of HSCs differentiation and decreased production rates of stem and progenitor cell. This was followed by severe peripheral blood pancytopenia, which is the reduction in the number of RBCs and WBCs, as well as platelets.17,18 However, treatment with MSCs exhibited rapid recovery in the level of WBCs, RBCs, platelets, and Hb in peripheral blood due to marked BM recovery, its increased cellularity in vivo, and improved hematopoiesis. 19 Moreover, MSCs alleviate the progenitor’s injuries by ameliorating the microenvironment and secreted cytokines; improve proliferation and differentiation of HSCs. 20
Even though MSC affect PI3K/PTEN/AKT pathway based on the preliminary WB data, it is unjustified to conclude that this pathway is essential in inducing AML.
It is known that PI3K/AKT/mTOR pathway plays a crucial role in many biological processes such as cell survival and proliferation, migration, cell cycle progression, and apoptosis. Furthermore, it is involved in many human cancers. 21 PI3K/AKT/mTOR has a central role in the pathogenesis of AML. This pathway is frequently activated specially in the immature leukemic population, and activation is reported in 50%–80% of AML cases. The degeneration and aberrant activation in the PI3K/AKT/mTOR pathway enhances survival, proliferation of the abnormal hematopietic progenitors (blast cells) and finally resulted in leukemia. 7
MSCs markedly enhance the recovery of hematopoiesis, alleviate the progenitor’s injuries by ameliorating the microenvironment, secreted cytokines and may also improve proliferation. 22 MSCs play a role in homing of HSCs in BM, support the long term of hematopoiesis and maintaining HSCs in an undifferentiated state. 20 Closing the contact between MSCs and HSCs props the differentiation of HSCs toward mature red cells. 20
MSCs provide soluble factors, contact-dependent interactions, and physical support to control HSCs self-renewal, quiescence, and differentiation 23 via direct cell contact and via the production of various cytokines, growth factors, and extracellular matrix (ECM) molecules. 24 MSCs are the precursor cells of mesenchymal cell types (adipocytes, osteoblasts, and fibroblasts). MSCs repaired the injuries of the hematopoietic niche caused by irradiation. 25
Toxicity of DMBA is associated with its oxidative metabolism leading to the formation of free radicals, covalently bind to nucleophillic sites on cellular macromolecules eliciting cancerous responses. Free radicals and their biochemical reactions are involved in cancer development estimating the risk of oxidative damage induced during carcinogenesis. 26
In this study, the carcinogenicity of DMBA in male rats was accompanied by significant deleterious changes in antioxidant status, as a response to the induced oxidative stress and production of reactive oxygen species (ROS). Furthermore, the oxidative stress produces deleterious effects by initiating lipid peroxidation, and reduction in GSH level resulted from oxidative stress and/or intoxication of DMBA. 27
Many studies recorded that patients with leukemia have higher levels of MDA 28 and a decrease in GSH level following carcinogen (DMBA) administration; which may be due to the utilization of antioxidants to scavenge the free radicals. 29
Malignant proliferation of leukemic cells is supported by a cytokine produced partially by the leukemic cells themselves. 30 Some of these are produced and secreted by the leukemic cells themselves.
DMBA mediates the carcinogenic process through chronic inflammation induction and over production of ROS. 31 In this regard, this study suggested that DMBA induced inflammation and cellular damage by increasing the levels of pro-inflammatory cytokines (Il-6 and TGF-β).
Dysregulation of TGF-β can give rise to the development of various hematologic disorders and malignancies, and the increase can lead to hematopoietic failure (myelodysplasia). TGF-β can potentiate myelomagenesis by stimulating secretion of IL-6. 32 In hematopoietic malignancies, leukemic cells secrete high levels of TGF-β1 that decrease the growth of normal hematopoietic progenitors and increase leukemic cells. 33 TGF-β1 is beneficial either to the normal HSC development or leukemic stem cells (LSCs).
IL-6 is a Th2-type cytokine affects a variety of inflammations, metabolic processes, and carcinogenesis produced by cells of the innate and adaptive immune system 34 and plays a major role in hematopoiesis 35 and its elevation in patients with malignancies, suggesting its involvement in leukemogenesis. 36
In this study, the intravenous treatment of DMBA-treated group with MSCs modulate the release of pro-inflammatory cytokines in serum after the release of anti-inflammatory cytokines in serum to balance the responses between inflammatory and anti-inflammatory and to balance inappropriate immune system disorders. 37 Treatment with MSCs basically suppressed the production of inflammatory cytokines (TNF-a, IL-1b and IL-6) predicting its possible anti-inflammatory effect. 38 Therefore, the therapeutic effects of MSCs may be an effective anti-inflammatory cell-based therapy.
Chemokines are low molecular weight proteins expressed in an inflammatory region and engage WBCs to the site of infection. Chemokine stromal cell-derived factor-1 (SDF-1 α, also termed CXCL-12) and its major receptor CXCR-4, is expressed and secreted by several organs tissue in the body, especially BM. CXCR-4/CXCL-12 axis influences many aspects of hematopoietic stem and progenitor cell (HSPC) and MSCs biology including migration, retention within stem cell niches, proliferation, and calmness. 39 It maintains a quiescent stem cell pool residing at the endosteum, whereby these cells are induced into the cell cycle to allow re-establishment of a depleted marrow cavity resulting in recovery of a damaged BM environment. 40
In agreement with the results of Georgiou et al., 41 the present study revealed that the injection of DMBA downregulates the expression of CXCR-4, which may be due to over production of ROS and reduction of BM cellularity, 39 which is confirmed by histopathology of BM. The deregulation of CXCR-4 is potentially associated with the reduced HSC cell density observed in the BM cavity as a result of the disruption of CXCL-12/CXCR-4 interaction. 42 Elevated ROS levels have been reported to limit the lifespan of HSCs, cause deleterious consequences on progenitor growth and hematopoietic reconstitution capacity through an activation of the p38 MAPK pathway, and increased DNA double-strand breaks and apoptosis leading to marked reduction in HSC repopulating potential, leading finally to exhaustion of the HSC pool. 39
Conversely, treatment with MSC upregulates the expression of CXCR-4 in DMBA group suggesting that MSCs allow progenitor cells homing back to the BM microenvironment, quiescence, and survival of HSCs and regulation of the size to re-establish the continually depleted BM cavity. 43 In other studies, 44 the increased expression of CXCR-4 significantly enhance migration and the paracrine properties of MSCs in vitro or enhanced the colonization of MSCs in damaged tissue 44 and the absence of CXCR-4 expression, and decrease homing efficiency of MSCs toward damaged tissue, which results in a poor curative effect of MSC-mediated repair. 45
The intracellular signaling pathway from activated receptors to PI3K and its downstream targets AKT, and mechanistic target of mTOR is very frequently deregulated by genetic and epigenetic mechanisms in human cancer (leukemia and lymphoma). 46 Activation of PI3K leads to AKT activation, which signals to various downstream substrates including GSK-3, FOXO, mTOR, Bad, and NF-kB and modulates diverse cell processes including survival, proliferation, apoptosis, and autophagy. 47 Meanwhile, sustained activation of the PI3K/AKT pathway accompanied with deletion of PTEN has been identified in different types of leukemia. 6
Targeting the PI3K/AKT/mTOR pathway may have pro-apoptotic and anti-proliferative effects on hematological malignancies through simultaneous targeting of Bcl-2/Bcl-xL pathways represent an effective anti-leukemic strategy via down-regulation of Mcl-1 and up-regulation/activation of Bak, Bax, Bim, and Bad. 48
Many appearances properties of MSCs that include anti-apoptosis, growth factor production, neuroprotection, anti-fibrosis, and chemo-attraction provide a broad spectrum for its using in disease therapies. 49 MSCs also appear to have intrinsic anti-tumor properties by arrest malignant cells at G0/G1 which reduced proliferation accompanied by an increase in cancer cell apoptosis. 50 Many studies confirm the anti-tumor therapeutic effect of MSCs. MSCs led to the inhibition of tumor growth, and increased survival of rats with glioma 51 led to the reduction in growth and metastases of a breast cancer model with the increase of apoptotic markers 52 and inhibit the growth of Kaposi’s sarcoma (KS) in a mouse model due to cell–cell contact-induced inhibition of AKT activity within KS cells. 53 The possible mechanisms for tumor inhibition include (1) promotion of cancer cell cycle arrest and apoptosis; (2) inhibition of AKT activity; (3) down-regulation of nuclear factor kB (NF-kB); and (4) down-regulation of the Wnt pathway by the release of the soluble inhibitor Dickkopf1 (DKK1), both of which reduce cancer cell proliferation. 54 On the contrary, MSCs also have the ability to downregulate Wnt and AKT signaling pathways, having an anti-tumorogenic function. 55 In addition, Dasari et al. 56 reported that MSCs can suppress tumor growth via inhibition of proliferation-related signaling pathways (AKT, PI3K, and Wnt pathways), inhibition of cell cycle progression, down-regulation of XIAP (X-linked inhibitor of apoptosis protein), and suppression of angiogenesis.
Conclusion
These results showed that MSCs treatment have efficient anti-inflammatory, mobilization to sites of tissue injury, inhibit proliferation, and induce apoptosis in leukemic rats and regulate PI3K/AKT/mTOR signaling. These mechanisms may elucidate the MSCs therapeutic potential in leukemogenesis treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
