Abstract
Angiogenesis, induced by the vascular endothelial growth factor A through its ligation to the vascular endothelial growth receptor 2, has been described as a crucial point in high-grade glioma development. The aim of this study was to evaluate the influence of VEGFA–2578C/A, −2489C/T, −1154G/A, −634G/C, and −460C/T, and KDR–604T/C, −271G/A, +1192G/A, and +1719A/T single-nucleotide polymorphisms on risk and clinicopathological aspects of high-grade glioma. This case–control study enrolled 205 high-grade glioma patients and 205 controls. Individuals with VEGFA–2578 CC or CA, VEGFA–1154 GG, VEGFA–634 GC or CC, and VEGFA–460 CT or TT genotypes were under 2.56, 1.53, 1.54, and 1.84 increased risks of high-grade glioma, compared to others, respectively. And 1.61, 2.66, 2.52, 2.53, and 2.02 increased risks of high-grade glioma were seen in individuals with VEGFA–2578 CC plus VEGFA–1154 GG, VEGFA–2578 CC or CA plus VEGFA–634 GC or CC, VEGFA–2578 CC or CA plus VEGFA–460 CT or TT, VEGFA–1154 GG or GA plus VEGFA–634 GC or CC, and VEGFA 634 GC or CC plus VEGFA–460 CT or TT combined genotypes, respectively, when compared to others. The “CAGT” haplotype of KDR single-nucleotide polymorphisms was more common in patients with grade IV than in those with grade III tumors, and individuals carrying this haplotype were at 1.76 increased risk of developing grade IV tumors than others. We present, for the first time, preliminary evidence that VEGFA–2578C/A and VEGFA–1154G/A single-nucleotide polymorphisms increases high-grade glioma risk, and “CAGT” haplotype of the KDR gene alters high-grade glioma aggressiveness and risk of grade IV tumors in Brazil.
Introduction
Gliomas account for nearly 70% of primary brain tumors.1,2 Anaplastic astrocytoma (AA) and glioblastoma multiforme (GBM) are classified by World Health Organization (WHO) as grade III and IV central nervous system tumors, respectively, and are also known as high-grade gliomas (HGGs).3,4 HGGs can be diagnosed as a primary or secondary disease, when they originate through the transformation of a low-grade glioma. 5 The prognosis of AA and GBM remains unfavorable, with expected 5-year survival around 23% for AA and median overall survival of 14 months for GBM patients.6,7
The risk factors for developing HGG are still not well understood,8,9 but angiogenesis (AG), induced mainly by the vascular endothelial growth factor A (VEGFA) through its ligation to vascular endothelial growth receptor 2 (KDR),10–14 is a crucial point. The AG is variable in humans and this variability is attributed to functional single-nucleotide polymorphisms (SNPs) in VEGFA and KDR genes.15–18
The allele “C” of VEGFA–2578C/A and allele “G” of VEGFA–1154G/A SNPs were associated with higher VEGFA production compared with the allele “A” in a cohort of ovarian cancer and renal allograft rejection,19,20 respectively, while that the allele “T” of VEGFA–2489C/T was related to higher VEGFA transcription in colon cancer patients. 21 However, the functional impact of VEGFA–634G/C19,22 and −460C/T19,22 SNPs was evaluated in ovarian cancer and breast cancer cell lines and remains uncertain. Concerning KDR SNPs, the “T” and “G” alleles of −604T/C and +1192G/A SNPs were related to higher expression and binding efficiency for VEGFA in coronary disease, respectively, 18 and the “A” allele of −271G/A and “T” allele of +1719A/T SNPs were related to higher KDR expression in a cohort of lung cancer patients 17 and higher binding efficiency of KDR for VEGFA in coronary disease, respectively. 18
A meta-analysis of genome-wide association studies identified specific differences in genetic susceptibility for GBM and non-GBM tumors, but the SNPs enrolled in this study were not cited by the authors. 23 There are only few studies focusing the association of VEGFA24–28 and KDR25,29,30 SNPs with risk of developing HGG, and VEGFA26,27 and KDR29,30 SNPs with clinicopathological aspects of HGG patients. None of these previous studies were conducted exclusively in HGG, and, except for Linhares et al., 26 who evaluated pure Caucasian Europeans, all studies were conducted only in Chinese. To the best of our knowledge, there are no previous studies focusing on the role of VEGFA–1154 GG and HGG risk and clinicopathological aspects.
Considering the paucity of information on this issue, the differences in carcinogenesis of low-grade gliomas and HGG, 31 and that the impact of SNPs in the tumor might depend on the ethnic origin of population, the aim of this study was to determine whether VEGFA–2578C/A, −2489C/T, −1154G/A, −634G/C, −460C/T, and KDR–604T/C, −271G/A, +1192G/A, +1719A/T affect the risk and clinicopathological aspects of HGG, represented by age, gender, and grade of tumor in an ethnically diverse and mixed population from Brazil.
Materials and methods
Patients
This case–control study enrolled adult patients diagnosed with HGG at the State University of Campinas and at the University of São Paulo, between 2000 and 2015. All patients were diagnosed with HGG by histological evaluation based on the WHO criteria. 3 The patients with different diagnosis or that refused to participate in the study were excluded from the analyses. The follow-up information was obtained by consulting clinical records of patients. The control group was obtained from blood donors of the State University of Campinas attended during the same period of time. Signed informed consent was obtained from each participant of the study. All procedures were carried out according to Helsinki Declaration and have been approved by the local ethics committee (process 21998214.3.0000.5404).
Methods
Genomic DNA of individuals were extracted from peripheral blood samples using a vacuum system in 10-mL tubes containing heparin. DNA from patients and controls peripheral blood samples were analyzed by real-time polymerase chain reaction (RT-PCR), using TaqMan® SNP Genotyping Assays from Applied Biosystems® (catalog 4351379; Thermo Fisher Scientific, Foster City, CA, USA), and nine SNPs were included for the following analysis: five SNPs in the VEGFA, −2578C/A (rs699947), −2489C/T (rs1005230), −1154G/A (rs1570360), −634G/C (rs2010963), and −460C/T (rs833061), and four SNPs in the KDR, −604T/C (rs2071559), −271G/A (rs7667298), +1192G/A (rs2305948), and +1719A/T (rs1870377). All reactions were performed according to the manufacturer’s instructions, and positive and negative controls were included in all experiments. 15% of genotype determinations were carried out twice in independent experiments with 100% of concordance.
Statistical analysis
Hardy–Weinberg equilibrium (HWE) was evaluated using the chi-square (χ2) goodness-of-fit test. The Haploview 4.2 software (www.broad.mit.edu/mpg/haploview) was used to tag all common haplotypes and their frequencies, and VEGF and KDR haplotypes with frequency higher than 10% were selected for the analyses. The linkage disequilibrium (LD) was measured by the disequilibrium coefficient (D′), and LD significance was considered at a D′ ≥ 80%. The statistical significance of differences between groups was calculated by the χ2 or Fisher exact test. Conditional analysis was used to obtain age- and gender-adjusted crude odds ratios (ORs) with 95% confidence intervals (CIs). SNP genotypes in patients and controls were analyzed according to the functional role of the alleles, considering the genotypes or alleles associated with lower VEGF or KDR production or lower binding efficiency as references in analyses; since the roles of the “G” and “C” alleles of VEGFA–634G/C and the “C” and “T” alleles of VEGFA–460C/T in VEGFA production are controversial, all isolated genotypes and alleles were initially tested in risk of tumor. After that, VEGFA–634G/C and VEGFA–460C/T genotypes associated with a decreased risk of HGG in the study were used as references in further analyses. Bonferroni adjustment was used to correct for multiple testing. A bootstrap analysis was performed as a resampling technique in significant results. 32
All tests were performed using the SPSS 15.0 software (SPSS Inc., Chicago, IL, USA).
Results
We analyzed 205 HGG patients and 205 controls in this study (Table 1). Patients were younger than controls and an excess of males was seen among patients when compared to controls; for this reason, all comparisons between patients and controls were adjusted by age and gender.
Clinical characteristics of high-grade glioma cases and controls.

SD: standard deviation; NA: not applicable.
Histological grade classified according to the World Health Organization criteria, where grade III included anaplastic astrocytoma and oligoastrocytoma and grade IV included glioblastoma multiforme.
Control and patient samples confirmed HWE at VEGFA −2578C/A (χ2 = 0.83, p = 0.36; χ2 = 1.62, p = 0.20), −2489C/T (χ2 = 0.60, p = 0.43; χ2 = 2.22, p = 0.13), −1154G/A (χ2 = 1.38, p = 0.24; χ2 = 0.58, p = 0.44), −634G/C (χ2 = 0.25, p = 0.61; χ2 = 0.006, p = 0.93), −460C/T (χ2 = 0.001, p = 0.97; χ2 = 3.54, p = 0.06), and KDR–604T/C (χ2 = 0.72, p = 0.39; χ2 = 2.14, p = 0.14), −271G/A (χ2 = 0.59, p = 0.44; χ2 = 2.56, p = 0.10), +1192G/A (χ2 = 0.07, p = 0.79; χ2 = 0.19, p = 0.27), +1719A/T (χ2 = 0.16, p = 0.68; χ2 = 3.54, p = 0.06) loci.
The frequencies of VEGFA–2578 CC or CA, VEGFA–1154 GG, VEGFA–634 GC or CC, and VEGFA–460 CT or TT genotypes were higher in patients than in controls; individuals with these genotypes were under 2.56, 1.53, 1.54, and 1.84 increased risks of HGG, compared to the those with the remaining genotypes, respectively. The alleles “C” of VEGFA–2578C/A, “G” of VEGFA–1154G/A, “C” of VEGFA–634G/C, and “T” of VEGFA–460C/T SNPs were associated with 1.53, 1.39, 1.38, and 1.32 increased risks of HGG, respectively. Only the VEGFA–2489 TT genotype and “T” allele were more common in controls than in patients; individuals with the TT genotype and T allele were under 0.38 and 0.64 decreased risks of HGG, respectively, than those with the CC or CT genotype and “C” allele. Similar frequencies of KDR genotypes were seen in patients and controls; individual with the distinct genotypes of KDR were under similar risks for disease (Table 2).
Genotypes and alleles of VEGFA and KDR single-nucleotide polymorphisms in high-grade glioma cases and controls.
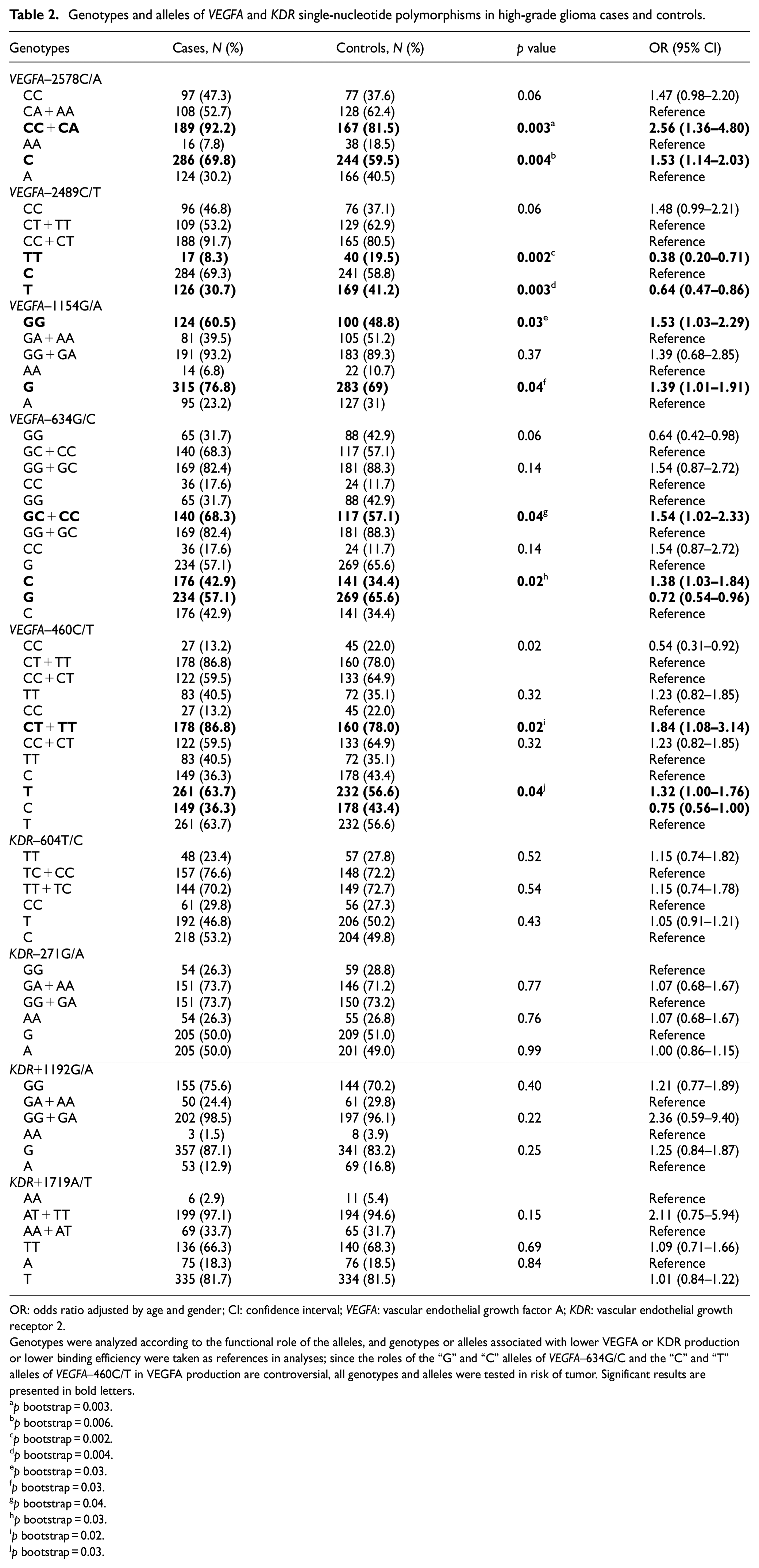
OR: odds ratio adjusted by age and gender; CI: confidence interval; VEGFA: vascular endothelial growth factor A; KDR: vascular endothelial growth receptor 2.
Genotypes were analyzed according to the functional role of the alleles, and genotypes or alleles associated with lower VEGFA or KDR production or lower binding efficiency were taken as references in analyses; since the roles of the “G” and “C” alleles of VEGFA–634G/C and the “C” and “T” alleles of VEGFA–460C/T in VEGFA production are controversial, all genotypes and alleles were tested in risk of tumor. Significant results are presented in bold letters.
p bootstrap = 0.003.
p bootstrap = 0.006.
p bootstrap = 0.002.
p bootstrap = 0.004.
p bootstrap = 0.03.
p bootstrap = 0.03.
p bootstrap = 0.04.
p bootstrap = 0.03.
p bootstrap = 0.02.
p bootstrap = 0.03.
Excesses of VEGFA–2578 CC plus VEGFA–1154 GG, VEGFA–2578 CC or CA plus VEGFA–634 GC or CC, VEGFA–2578 CC or CA plus VEGFA–460 CT or TT, VEGFA–1154 GG or GA plus VEGFA–634 GC or CC, and VEGFA 634 GC or CC plus VEGFA–460 CT or TT combined genotypes were seen in patients when compared to controls; individuals with respective genotypes were under 1.61, 2.66, 2.52, 2.53, and 2.02 increased risks for HGG than others (Table 3). No differences in frequencies of combined genotypes of VEGFA plus KDR and KDR were seen in patients and controls; individuals with the distinct genotypes were under similar risks for HGG (Supplementary Table 1).
Combined genotypes of VEGFA single-nucleotide polymorphisms in high-grade glioma cases and controls.

OR: odds ratio; CI: confidence interval; NA: not applicable; VEGFA: vascular endothelial growth factor A; KDR: vascular endothelial growth receptor 2.
Genotypes were considered according to the functional role of the alleles, and combined genotypes associated with lower VEGFA or KDR production or lower binding efficiency were taken as references in analyses. In the cases of VEGFA–634G/C and −460C/T, GG and CC genotypes were taken as references in analyses, respectively, since GC + CC and CT + TT genotypes were associated with increased risks of high-grade gliomas in this study. Significant results are presented in bold letters.
p bootstrap = 0.04.
p bootstrap = 0.003.
p bootstrap = 0.001.
p bootstrap = 0.03.
p bootstrap = 0.008.
The LD of patients with HGG for VEGFA and KDR SNPs is presented in Figure 1. The CCGCT (64.4%), ATACG (36.1%), CCGGT (68.3%), and ATAGGC (48.3%) haplotypes of the VEGFA–2578C/A, −2489C/T, −1154G/A, −634G/C, and −460C/T SNPs, and the TGGT (65.9%) and CAGT (69.8%) haplotypes of the KDR–604T/C, −271G/A, +1192G/A, and +1719A/T SNPs were considered in further analyses. The ATAGC haplotype of VEGFA was associated with 0.61 decreased risk for HGG compared to other haplotypes (Table 4).

Linkage disequilibrium plot for high-grade glioma patients and VEGFA and KDR single-nucleotide polymorphisms (SNPs). The rs number (top; from left to right) corresponds to the SNP’s name and number; VEGFA–2578C/A (1, rs699947), −2489C/T (2, rs1005230), −460C/T (3, rs833061), −1154G/A (4, rs1570360), −634G/C (5, rs2010963), and KDR+1192G/A (6, rs1870377), +1719A/T (7, rs2305948), −271G/A (8, rs7667298), and −604T/C (9, rs20715599). The measure of LD (D′) among the pairs of SNPs is shown in each square, whereas white represents very low D′ and red represents very high D′.
VEGFA and KDR haplotypes in high-grade glioma cases and controls.
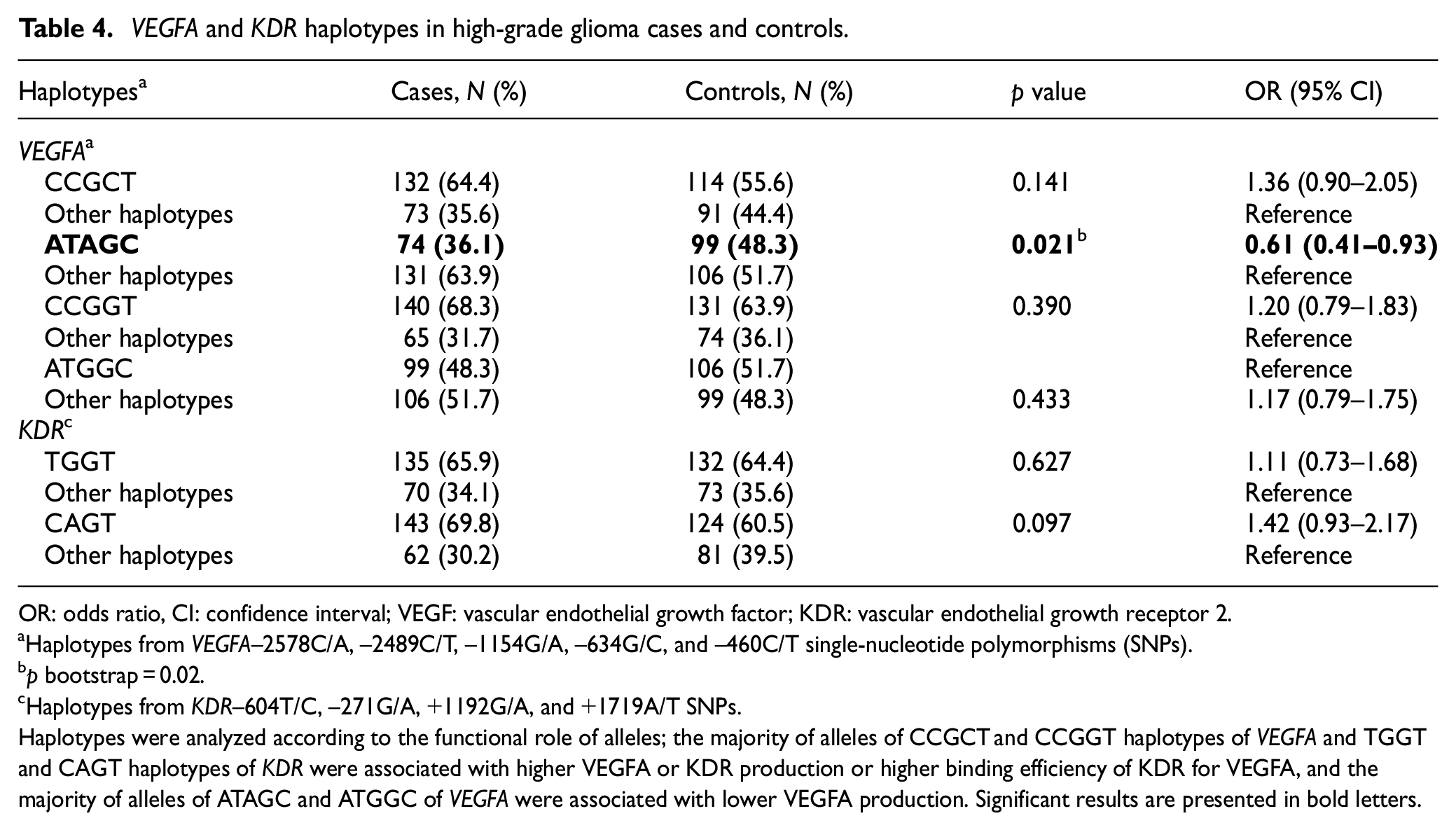
OR: odds ratio, CI: confidence interval; VEGF: vascular endothelial growth factor; KDR: vascular endothelial growth receptor 2.
Haplotypes from VEGFA–2578C/A, −2489C/T, −1154G/A, −634G/C, and −460C/T single-nucleotide polymorphisms (SNPs).
p bootstrap = 0.02.
Haplotypes from KDR–604T/C, −271G/A, +1192G/A, and +1719A/T SNPs.
Haplotypes were analyzed according to the functional role of alleles; the majority of alleles of CCGCT and CCGGT haplotypes of VEGFA and TGGT and CAGT haplotypes of KDR were associated with higher VEGFA or KDR production or higher binding efficiency of KDR for VEGFA, and the majority of alleles of ATAGC and ATGGC of VEGFA were associated with lower VEGFA production. Significant results are presented in bold letters.
Similar frequencies of the VEGFA and KDR isolated (Supplementary Table 2) and combined (Supplementary Table 3) genotypes were seen in patients stratified by age, gender, and histological grade. The KDR CAGT haplotype was more common in patients with grade IV tumors than in those with grade III tumors (Table 5), even after Bonferroni correction. The KDR CAGT haplotype was also more common in patients with grade IV tumors than in controls; individuals carrying the KDR CAGT haplotype were under 1.76 (95% CI: 1.10–2.81) increased risk for grade IV tumors than others (p = 0.01, p bootstrap = 0.02).
Frequencies of VEGFA and KDR haplotypes in high-grade glioma cases stratified by clinicopathological characteristics.

VEGF: vascular endothelial growth factor; KDR: vascular endothelial growth receptor 2.
Histological grade classified according to World Health Organization criteria, where grade III included anaplastic astrocytoma and oligoastrocytoma and grade IV included glioblastoma multiforme.
Haplotypes from VEGF–2578C/A, −2489C/T, −1154G/A, −634G/C, and −460C/T and from KDR–604T/C, −271G/A, +1192G/A, and +1719A/T single-nucleotide polymorphisms.
p bootstrap = 0.02.
Haplotypes were considered according to the functional role of the alleles; the majority of alleles of CCGCT and CCGGT haplotypes of VEGFA and TGGT and CAGT haplotypes of KDR were associated with higher VEGFA or KDR production or higher binding efficiency of KDR for VEGFA, and the majority of alleles of ATAGC and ATGGC of VEGFA were associated with lower VEGFA production. Significant results are presented in bold letters. Bonferroni adjustment was used to correct for multiple testing (p = 0.017).
Discussion
HGGs are characterized by a marked increase in blood vessel formation, and therefore AG is a crucial point for tumor growth and progression in the brain. 33 It is also already well established that VEGFA and KDR are essential for endothelial proliferation, migration, and organization into functional vessels 10 and, consequently, for HGG development. Since the ability to produce these proteins is variable in humans, 10 we analyzed in this study whether SNPs in VEGFA and KDR genes alter the risk of HGG and clinicopathological features of the tumor.
We initially found an increased HGG risk in carriers of VEGF–2578 CC or CA genotype.
To the best of our knowledge, there is just one previous study focusing on the role of this SNP in risk of HGG. 26 Linhares et al. did not demonstrate the association of VEGF–2578C/A with HGG in a significantly smaller population of GBM (N = 84), which can be the reason for this result disparity, since the controls’ allele frequencies were similar in both populations. In fact, the association of −2578 CC or CA genotypes and increased risk of HGG was expected, since the “C” allele has been related to higher VEGFA production 19 and a higher risk of other tumors, such as lymphoma and multiple myeloma.34,35 Thus, individuals harboring the “C” allele of VEGFA–2578C/A may produce more blood vessels than those with the AA genotype and may have a higher risk of HGG as a consequence.
The VEGFA–1154 GG was associated with an increased risk of HGG in this study. There are previous reports focusing on the association of the VEGFA–1154G/A SNP with an increased risk of multiple myeloma 34 and oral squamous cell carcinoma, 36 but its role in HGG origin is unknown. Thus, this new finding might be due to the well-described functional role of VEGFA–1154 “G” allele increasing VEGFA expression,19,37 which may predispose individuals to the tumors.
We found that individuals harboring VEGFA–634 GC or CC and VEGFA–460 CT or TT were at an increased risk of developing HGG. Regarding VEGFA–634G/C, our finding is in accordance with those previously reported from China, also demonstrating that the “C” allele was associated with an increased all-grade glioma risk, including HGG.24,27,28 Linhares et al. 26 also reported an increased risk of gliomas, grades II–IV in patients exhibiting the “T” allele from VEGFA 460C/T in Caucasians from Portugal. Knowing that the roles of “G” and “C” alleles of VEGFA–634G/C and “C” and “T” alleles of VEGFA–460C/T in VEGFA production are uncertain in the literature,19,22 the results of this study and of those described above suggest that the “C” allele of VEGFA–634G/C and “T” allele of VEGFA–460C/T determine more VEGFA production, more blood vessel formation, and more susceptibility to HGG.
Surprisingly, we found for the first time a protective effect of VEGFA–2489 TT in HGG. This finding was not expected by us since the “C” allele of the VEGFA–2489C/T was previously associated with repressed VEGFA transcription and defective AG in gastric cells. 38 However, the authors showed that the “C” allele had no influence on ovarian cell AG in the same study, 38 culminating in the fragility of this finding. Considering the paucity of information regarding the functional role of the “T” allele of VEGFA–2489C/T SNP, we hypothesize that the “T” allele might be related to poor AG. Worse response to anti-angiogenic agents was seen in HGG patients with VEGFA–2489 TT genotype; the finding was attributed to a lower expression of VEGFA and reduced blood vessel formation, 39 and supports our hypothesis.
We found a strong association between VEGFA–2578 CC plus VEGFA–1154 GG, VEGFA–2578 CC or CA plus VEGFA–634 GC or CC, VEGFA–2578 CC or CA plus VEGFA–460 CT or TT, VEGFA–1154 GG or GA plus VEGFA–634 GC or CC, and VEGFA 634 GC or CC plus VEGFA–460 CT or TT combined genotypes in our individuals regarding HGG risk, showing the high relevance of this combined analysis, at least among VEGFA SNPs, and with a possible synergistic effect. To the best of our knowledge, all of these combined associations in HGG risk are presented for the first time in our study.
The VEGFA haplotype indicated a protective effect of ATAGC haplotype, which corroborates and reinforces all of our findings, considering the total lack of risk-increasing alleles in this haplotype composition. This finding was first seen in this study.
We found no association of isolated or combined KDR SNPs in HGG risk. There are only two studies focusing on this association in gliomas, both conducted in Chinese population, and none exclusively in HGG. Associations between the “T” allele of KDR+1719A/T 25 and the “C” allele of KDR–604T/C25,29,30 have been related to glioma risk, although Chen et al. 29 did not demonstrate this association in the GBM subgroup. Since the Brazilian population is heterogeneous, mixed, and composed of Amerindians and European, Asian, and African immigrants,40,41 that Chinese population is homogeneous, and that significant differences in frequencies of KDR+1719 TT genotype (68.3% vs 40.7%; p < 0.0001) 25 and the KDR–604 CC genotype (8.8% vs 27.3%, p < 0.0001) 30 were found in our sample and in Chinese samples, ethnic differences might be the explanation for the disparity of results found in our study and in those conducted in China. The “A” allele of KDR+1192G/A was also previously related to glioma risk,25,29 but this finding is not in accordance with the described functional role of this allele in the reduction of binding efficiency to VEGFA. 18
Finally, we found for the first time a clinically relevant result showing that the CAGT haplotype of the KDR–604T/C −271G/A, +1192G/A, and +1719A/T SNPs was more common in patients with grade IV than grade III tumors, even after Bonferroni correction for multiple testing, denoting greater tumor aggressiveness. Moreover, we also found that the KDR CAGT haplotype was associated with an increased risk of grade IV HGG. Thus, we hypothesize that this haplotype enhances the AG pathway to work faster and with a higher efficacy, consequently resulting in HGG formation and aggressiveness. In fact, the “A,”“G,” and “T” alleles of the KDR–271G/A, +1192G/A, and +1719A/T SNPs were previously associated with higher binding efficiency or higher KDR transcription, respectively, 18 which may favor tumor aggressiveness.12,14
We in fact believe that our results are clinically relevant; however, we are aware that our study has limitations, such as a relatively small sample size and lack of functional studies focusing on the roles of the SNPs in HGG AG. In addition, the WHO classification of 2016 is the current one, and the major milestone was the incorporation of molecular factors for the classification of glial tumors. 4 This is a limitation of our study because we conducted most part of the analysis prior to the new classification and also because we do not have that molecular assessment in our material.
In conclusion, our data suggest that inherited abnormalities in blood vessel formation, related to VEGFA and KDR SNPs, influence the risk and aggressiveness of HGG in an ethnically diverse population from southeastern Brazil. We describe for the first time that SNPs of VEGFA–2578C/A, VEGFA–1154G/A, and CAGT haplotype of KDR–604T/C, −271G/A, +1192G/A, and +1719A/T SNPs increase HGG risk and/or aggressiveness.
Supplemental Material
Supplementary_tables – Supplemental material for Associations of VEGFA and KDR single-nucleotide polymorphisms and increased risk and aggressiveness of high-grade gliomas
Supplemental material, Supplementary_tables for Associations of VEGFA and KDR single-nucleotide polymorphisms and increased risk and aggressiveness of high-grade gliomas by Vivian Castro Antunes Vasconcelos, Gustavo Jacob Lourenço, Angelo Borsarelli Carvalho Brito, Victor Leal Vasconcelos, Marcos Vinicius Calfat Maldaun, Helder Tedeschi, Suely Kaue Nagashi Marie, Sueli Mieko Oba Shinjo and Carmen Silvia Passos Lima in Tumor Biology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
