Abstract
The vascular endothelial growth factor regulates angiogenesis that is increased in glioma.
Keywords
Introduction
Gliomas are the most frequent primary malignant central nervous system tumors.1–3 These tumors present a multifactorial etiology, being the result of interactions between environmental exposure and genetic susceptibility. 4 The only proven exogenous environmental cause of glioma is ionizing radiation.5,6 Perhaps other environmental causes are involved but remain to be proven. Accordingly, the genetic polymorphisms most frequently studied in the context of brain tumors are located in genes that might influence susceptibility to these tumors in concert with environmental exposures, such as those involved in DNA repair, cell cycle regulation, or immune response.7–12 In addition, few genome-wide association studies have focused on gliomas identifying five risk loci for glioma mostly related to telomerase.13–17
Common pathways in glioma biology include growth factor receptor tyrosine kinases and their signaling pathways. Polymorphisms on the epidermal growth factor (EGF) and its receptor, EGFR, have been identified as risk factors for glioma.17–19 In addition, vascular endothelial growth factor (VEGF) is the principal angiogenic factor in both embryonic development and tumor growth and is highly up-regulated in glioblastoma, and its receptors are over-expressed in tumor vessels.
20
Importantly, the anti-angiogenic drug bevacizumab, a humanized monoclonal immunoglobulin (Ig) G1 antibody that binds to and inhibits the biological activity of human VEGFA, is currently being explored in different clinical trials involving glioma and has been used in combination with chemotherapy for the treatment of recurrent glioblastoma,20–24 which resulted in improved response rates and extended progression-free survival (PFS). Recent studies have shown that some single nucleotide polymorphisms (SNPs) of VEGF/VEGF-related genes could contribute to the modulation of serum VEGF levels, possibly influencing risk and prognosis of solid tumors, including glioma.25–27 Moreover,
The von Willebrand factor (VWF) is an essential component of hemostasis at sites of vascular injury.
30
The
Given the important role of VEGF signaling in gliomas and the influence tumor risk and prognosis of particular VEGF/VEGFR polymorphisms in different tumor types, we investigated the relevance of 11 genetic polymorphisms in the 6p12 (VEGFA) and 6p21 (VWF) loci on glioma risk and patient survival.
Materials and methods
Study population
In this case–control study, we enrolled 126 glioma samples from Hospital S. João, Porto, and Hospital de Braga, Braga, Portugal. Tumors were classified according to the World Health Organization (WHO) criteria (Table 1). 1 All patients above 18 years of age, with diagnosis of glioma, treated between 2009 and 2010, and that by informed consent accepted to participate in the study, were included. The patients with different diagnosis or that refused to sign the informed consent were excluded. Complete treatment data were retrieved for 90 patients. Of those, 80 patients underwent surgery, 9 stereotactic biopsy, 77 radiotherapy, 71 chemotherapy (65 underwent Stupp protocol, 35 7 surgery only, 8 surgery+radiotherapy, 5 surgery+radiotherapy+chemotherapy (other than temozolomide), 2 surgery+chemotherapy, and 2 biopsy only), and one additional patient refused treatment. The 180 control samples were selected from cancer-free blood donors at Hospital de Braga, to match for gender and adjust for age in the statistical analysis (Table 1). All patients and controls had a Caucasian background and come from the same region in the north of Portugal. The follow-up information was obtained by consulting clinical records. The control group had no follow-up information. Signed informed consent was obtained from each participant. Local institutional ethics committees of Hospital S. João and Hospital de Braga approved the procedures followed in this study.
Clinico-pathological features of gliomas and controls.

WHO: World Health Organization; n.a.: not applicable; SD: standard deviation; M: male; F: female; OS: overall survival; PFS: progression-free survival.
Genotyping
DNA was extracted from the leukocytes of blood samples using the commercially available Citogene® Blood Kit according to the manufacturer’s recommendations. Genotyping of the allele-specific primer extension products, which were generated from amplified DNA sequences, was performed using the Sequenom MassARRAY iPLEX Gold platform (Sequenom, San Diego, CA) at the Instituto Gulbenkian de Ciência (IGC), Lisbon, Portugal. Primers were designed using MassARRAY Assay Design 3.1 software (Sequenom), and genotyping was performed in a blinded manner regarding sample status (i.e. case or control subjects). The genotyping quality was assessed by duplicate analysis of 10% of the samples, which demonstrated a 100% agreement rate.
Statistical analysis
Independent samples
Results
Our cohort of samples included 126 glioma samples (WHO grade II–IV) and 180 cancer-free controls (Table 1). All studied variants were in HWE, apart from the polymorphism rs3025035, for which it was not possible to calculate due to the absence of the homozygous recessive genotype.
Glioma risk assessment
In order to explore the associations between
We could successfully include on the multivariate logistic analysis 107 cases and 143 controls. The frequencies of each genotype in controls and cases are presented in Table 2 for
Multivariate logistic regression analysis of associations between
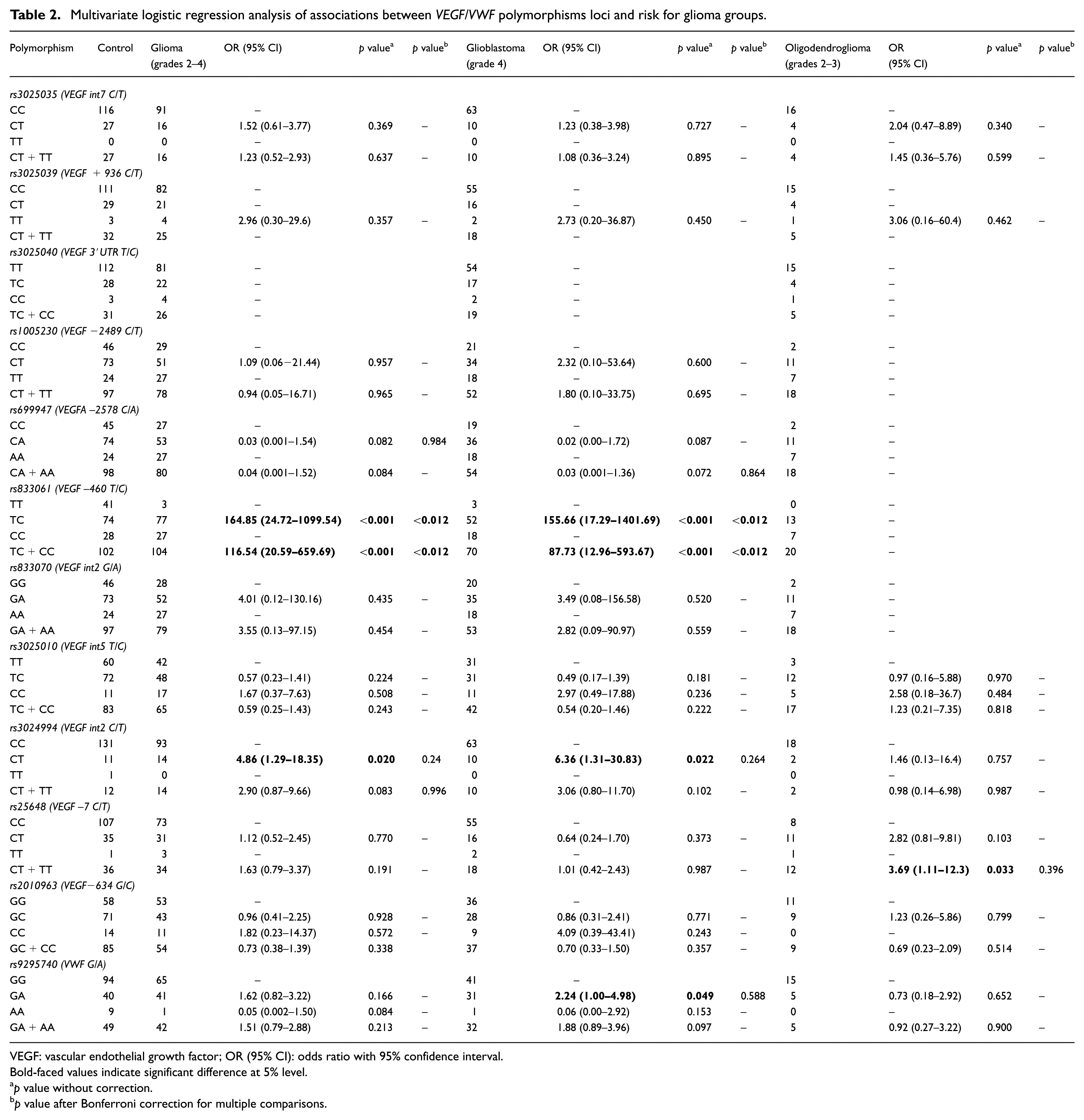
VEGF: vascular endothelial growth factor; OR (95% CI): odds ratio with 95% confidence interval.
Bold-faced values indicate significant difference at 5% level.
We found a significant association between the VEGF SNPs heterozygous rs833061 and risk for glioma and glioblastoma (OR = 164.85, 95% CI = 24.72–1099.54 and OR = 155.66, 95% CI = 17.29–1401.69, respectively,
Regarding the
No statistically significant associations were found between the VEGF SNPs rs3025035, rs3025039, rs3025040, rs1005230, rs699947, rs833070, rs3025010, and rs2010963 variants and risk for glioma, glioblastoma, or oligodendroglioma (Table 2; all
Glioma prognostic assessment
In a subset of glioma patients, follow-up data were also available (
Multivariate Cox proportional hazard model analysis of associations between
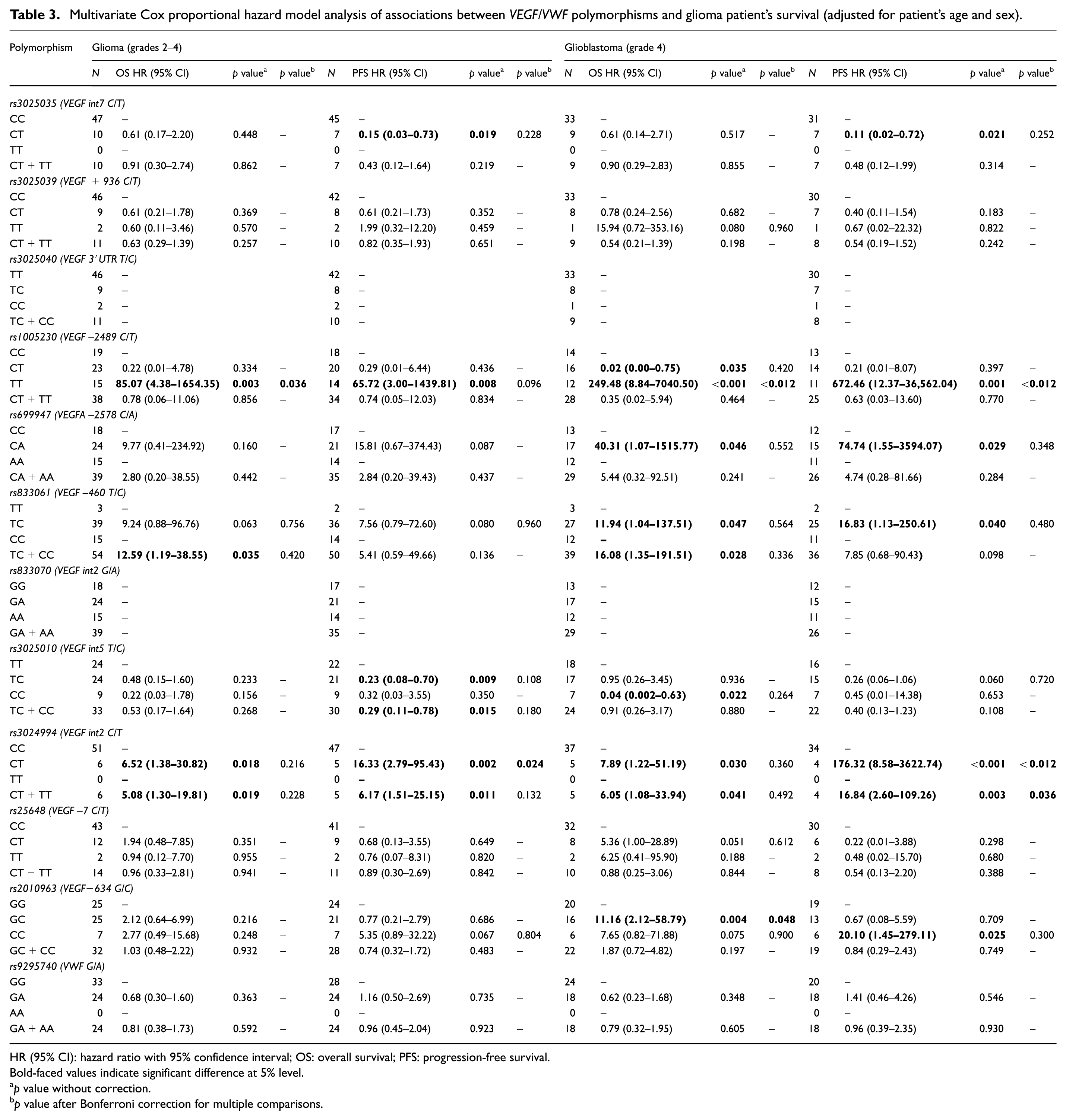
HR (95% CI): hazard ratio with 95% confidence interval; OS: overall survival; PFS: progression-free survival.
Bold-faced values indicate significant difference at 5% level.
The rs1005230 homozygous carriers presented a decreased OS and PFS both for glioma (OS: HR = 85.07, 95% CI = 4.38–1654.35,

Survival graphs of significant associations between overall survival of glioblastoma patients and VEGF SNPs. Multivariate Cox proportional hazard model regression with respect to glioblastoma patient survival of (a) rs1005230, hazard ratio (HR) = 249.48; 95% confidence interval (CI) = 8.84–7040.50,
In addition, when only glioblastoma patients were analyzed, we further observed a decreased survival for rs699947 heterozygous (OS: HR = 40.31, 95% CI = 1.07–1515.77,
Discussion
VEGF is the major proangiogenic factor involved in tumor growth and progression, including glioblastoma, being over-expressed in tumor vessels.
20
VEGF–VEGFR signaling pathway stimulates not only angiogenic signals but also the secretion of the VWF from endothelial cells, and VWF was described as having a potential role on angiogenesis.36–38 We have studied the contribution of 11
We found that, in our population, the
The rs3024994 C/T genotype presented an OR = 4.86 (95%CI = 1.29–18.35) for glioma and OR = 6.36 (95% CI = 1.31–30.83) for glioblastoma and also the
Importantly,
The rs1005230 variant homozygous consistently presented a decreased OS and PFS both for glioma and glioblastoma patients. Similarly, the rs3024994 variant heterozygous consistently presented a decreased PFS both for glioma and glioblastoma patients. Also the rs2010963 heterozygous glioblastoma patients had a reduced OS.
In conclusion, our data strongly suggest that different
Footnotes
Acknowledgements
Paulo Linhares and Marta Viana-Pereira contributed equally to this work.
Compliance with ethical standards
This study was approved by “Comissão de tica para a Sade,” stating that it was unanimously approved, with nothing to object. Signed informed consent was obtained from each participant. Local institutional ethics committees of Hospital São João, Portugal and Hospital de Braga, Portugal, approved the procedures followed in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Fundação para a Ciência e Tecnologia (FCT), Portugal (![]() ; grant PTDC/SAU-ONC/115513/2009_FCOMP-01-0124-FEDER-015949 to R.M.R.). This article has been developed under the scope of the project NORTE-01-0145-FEDER-000013, supported by the Northern Portugal Regional Operational Programme (NORTE 2020), under the Portugal 2020 Partnership Agreement, through the European Regional Development Fund (FEDER). M.V.-P. is recipient of an FCT Post-Doctoral fellowship (ref: SFRH/BPD/104290/2014).
; grant PTDC/SAU-ONC/115513/2009_FCOMP-01-0124-FEDER-015949 to R.M.R.). This article has been developed under the scope of the project NORTE-01-0145-FEDER-000013, supported by the Northern Portugal Regional Operational Programme (NORTE 2020), under the Portugal 2020 Partnership Agreement, through the European Regional Development Fund (FEDER). M.V.-P. is recipient of an FCT Post-Doctoral fellowship (ref: SFRH/BPD/104290/2014).
