Abstract
A role for matrix metalloproteinase polymorphisms in breast cancer development and progression was proposed, but with inconclusive results. We assessed the relation of matrix metalloproteinase-2 variants with breast cancer and related phenotypes in Tunisians. This case-control retrospective study involved 430 women with breast cancer and 498 healthy controls. Genotyping of matrix metalloproteinase-2 rs243866, rs243865, rs243864, and rs2285053 was analyzed by allelic exclusion. The minor allele frequency of rs2285053 was significantly lower in women with breast cancer cases as compared to control women; minor allele frequencies of the remaining single-nucleotide polymorphisms were similar between cases and control women. The distribution of rs243865 and rs2285053 genotypes was significantly different between breast cancer patients and control subjects. This persisted when key covariates were controlled for. None of the matrix metalloproteinase-2 variants were associated with estrogen receptor positivity, progesterone receptor positivity, or with double estrogen receptor–progesterone receptor positivity in breast cancer patients. Matrix metalloproteinase-2 rs243866, rs243865, and rs243864 were positively associated with menstrual irregularity and histological type, while rs243866 and rs2285053 were negatively associated with menarche and nodal status. In addition, rs2285053 was negatively associated with triple negativity, tumor size, distance metastasis, molecular type, and chemotherapy. Haploview analysis revealed high linkage disequilibrium between matrix metalloproteinase-2 variants. Four-locus Haploview analysis identified haplotypes G
Introduction
Breast cancer is a highly prevalent malignancy in females and a leading cause of cancer-related mortality in women worldwide (https://gco.iarc.fr/). 1 In Tunisia, the prevalence of breast cancer is estimated at 15.6% and is responsible for 8.4% of all cancer-related deaths among females (https://gco.iarc.fr/). Despite its national significance as a major health problem, 2 available screening programs for breast cancer in Tunisia remain inadequate.2,3 Early screening of breast cancer is an important determinant of disease prognosis and in reducing breast cancer-related mortality.4,5 Current screening of breast cancer relies on mammography, ultrasonography, and genetic screening.6,7 However, these screening tests suffer from limited sensitivity, 8 risk of false-positive/negative results, 6 and limited or no availability in remote areas.9,10
Several studies evaluated new biomarkers of breast cancer.7,11,12 Blood-based biomarkers13,14 and genome-wide association15,16 were explored for possible diagnostic and prognostic roles, and several loci linked to the pathogenesis of breast cancer were identified.15,17 These included matrix metalloproteinase (MMP) gene variants, an enzyme family which controls tumor invasion and metastasis. 18 Particular attention was given to the gelatinase family, in particular, matrix metalloproteinase-2 (MMP2), which act by degrading collagen type IV. As it is an extracellular matrix (ECM) component, 19 this results in the loss of cellular structural support and thus destabilization of the basal membrane, an essential step for the spread of cancer.19,20
Altered MMP2 activity resulting from the presence of specific MMP2 variants was implicated in the destruction of ECM 21 and disruption of basement membrane barriers.18,22 Insofar as MMP2 is overexpressed in breast cancer,20–23 genetic variation in MMP2 gene affecting its expression was shown to contribute to cancer susceptibility.24–26 Few studies evaluated the (likely) link between MMP2 promoter gene variants with altered breast cancer susceptibility and were reported for different populations, but with mixed outcome.25–29 A recent Tunisian study involving 251 breast cancer cases documented a protective effect of rs243865 (promoter) variant in breast cancer development. 30 Future studies involving larger number of subjects, and testing additional variants, are needed to confirm a role for MMP2 as at-risk breast cancer locus.
Several studies proposed that genetic polymorphism in MMP2 promoter region control MMP2 expression23,31 and that sustained increased MMP2 levels might render the carriers more susceptible and aggressive to tumorigenesis. In this context, we examined the relationship between breast cancer susceptibility and the presence of MMP2 (promoter) variants: rs243864 (−790 G/T), rs243865 (−1306 C/T), rs243866 (−1575 G/A), and rs2285053 (−735 C/T) as potential risk biomarkers of breast cancer.
Subjects and methods
Study subjects
A case-control retrospective study involved 430 women with breast cancer (age: 45.6 ± 9.3 years) and 498 cancer-free women serving as control (age: 46.8 ± 11.1 years). Cases and controls were recruited from the outpatient surgery and oncology services of Salah Azaiez Institute (Tunis, Tunisia) between June 2018 and October 2018. Control women, who comprised hospital staff or volunteer women, were free of personal or family history of breast cancer and were similar in self-declared ethnic origin to patients. Diagnosis of breast cancer was consistent with the guidelines of the American Cancer Society (www.cancer.org). This included mammography and testing of breast biopsies for confirmation of breast cancer; these were for done all patients.
None of the subjects (cases and controls) have unrelated comorbidities. Demographic profile and clinical biodata were collected for patients from medical records and interviews using a structured questionnaire by physicians or senior resident. These comprised age at entry into the study, age at first diagnosis of breast cancer, status of menopause, and disease stage at presentation. Histological assessment included stage of disease and nuclear grade, estrogen receptor (ER) and progesterone receptor (PR) status, along with treatment regimen (chemotherapy, surgery, and radiation). The study was done per Helsinki II declaration, and approval was obtained from the Research and Ethics Committee of Salah Azaiez Institute (IRB number: ISA/2018/19); all patients and control subjects provided written informed consent.
MMP2 genotyping
Four single-nucleotide polymorphisms (SNPs) in MMP2 gene, having minor allele frequency (MAF) >10% in Tunisians and clinical relevance, were identified using National Center for Biotechnology Information (NCBI) Gene SNP Geneview (www.ncbi.nlm.nih.gov/projects/SNP/). These comprised rs243866 (context sequence [VIC/FAM]: TAG CTG TGA TGA TCA AGA CAT AAT C
Statistical analysis
Statistical analysis was done on SPSS 24 (IBM, Armonk, NY, USA). Mean (± SD) and percent total were used in presenting continuous and categorical data, respectively. Means differences and inter-group significances were evaluated using Student’s t-test and Pearson χ2 test, respectively. Genetic Power Calculator (http://pngu.mgh.harvard.edu/∼purcell/cgi-bin/cc2k.cgi) was employed for calculating the study power, considering the number of study subjects (430 patients and 498 controls), MAF of the included variants, breast cancer prevalence in Tunisia (estimated), and relative risk for heterozygous (1/2) and minor allele homozygous (2/2) genotypes. The overall power (87.3%) was calculated as the average of included SNPs. Haploview 4.2 (www.broad.mit.edu/mpg/haploview) was used for Hardy–Weinberg equilibrium (HWE) calculation. Allele frequencies established in this study were compared frequencies reported for Caucasians (CEU), African Americans of the American Southwest (ASW), and Yoruban in Ibadan, Nigeria (YRI) in HapMap release #28.
Analyses were done assuming additive genetic effect. Linkage disequilibrium (LD) between any pair of SNPs and haplotype patterns (done by the expectation maximization method) were checked by Haploview 4.2. Among the 16 theoretical MMP2 haplotypes, 4 were found to be common with frequency exceeding 2%, capturing 97.3% of all haplotypes. Calculation of odds ratios (ORs) and 95% confidence intervals (95% CIs) associated with the risk of breast cancer was determined using logistic regression analysis; statistical significance was set at p < 0.05.
Results
Study subjects
Demographic and clinical characteristics of study participants are listed in Table 1. Significant differences between breast cancer patients and control women were noted in body mass index (BMI; p < 0.001), menarche (p = 0.021), and number of smokers (p = 0.022). Higher frequency of women with irregular menses (p < 0.001), past users of oral contraceptives (p < 0.001), and lower frequency of breastfeeding women (p < 0.001) were noted in patients than in control women. As such, these covariates were selected as the main covariates that were controlled for later analysis.
Characteristics of study subjects.
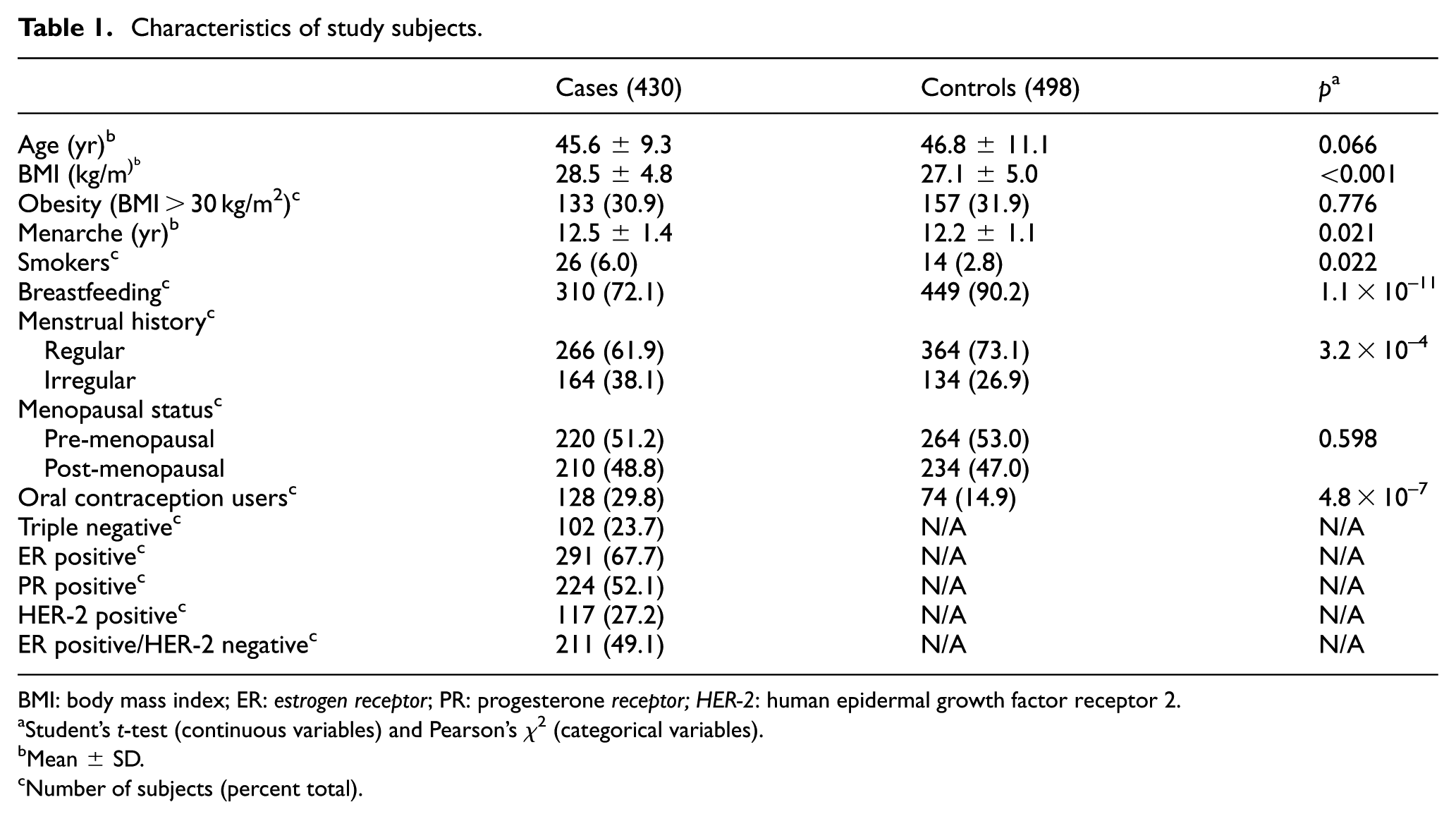
BMI: body mass index; ER: estrogen receptor; PR: progesterone receptor; HER-2: human epidermal growth factor receptor 2.
Student’s t-test (continuous variables) and Pearson’s χ2 (categorical variables).
Mean ± SD.
Number of subjects (percent total).
Association studies
The genotype distribution of the investigated MMP2 SNP was in HWE in study subjects (Table 2). MAFs of the tested MMP2 SNPs in breast cancer patients and control women are presented in Table 2. Compared to control women, MAF of rs2285053 was lower in breast cancer patients (p < 0.001); MAF of the other tested MMP2 SNPs was comparable between breast cancer cases and controls.
Distribution of MMP2 alleles in breast cancer cases and control women.

MMP2: matrix metalloproteinase-2; SNP: single-nucleotide polymorphism; HWE: Hardy–Weinberg equilibrium; OR: odds ratio.
SNP genotyping was done by allelic exclusion method, using VIC- and FAM-labeled primers.
Location on chromosome based on dbSNP build 125.
Major allele > minor allele.
Minor allele number (frequency).
Crude (unadjusted) OR.
The distribution of MMP2 genotypes in breast cancer cases and control women is shown in Table 3. The distribution of rs243865 (p = 0.001) and rs2285053 (p < 0.001) genotypes was significantly different between breast cancer patients when compared to control women. The association of MMP2 rs243865 (p < 0.001) and rs2285053 (p < 0.001) with breast cancer remained significant after controlling for the covariates age, BMI, menses pattern and menarche, breastfeeding, smoking, and use of oral contraceptives.
Association of MMP2 genotypes with breast cancer.

MMP2: matrix metalloproteinase-2; SNP: single-nucleotide polymorphism; BMI: body mass index.
Genotypes were coded as per “1” = major allele and “2” = minor allele.
Number of subjects (percent total).
p values adjusted for age, BMI, menses pattern and menarche, breastfeeding, smoking, and use of oral contraceptives.
Association of MMP2 genotypes with breast cancer features
We tested the possible association of MMP2 genotypes with breast cancer according to ER and PR status. Of the included breast cancer patients, 291 (67.7%) were ER positive and 224 (52.1%) were PR positive. Data from Table 4 demonstrate that none of the tested MMP2 variants were associated with ER positivity or PR positivity in breast cancer patients. Furthermore, none of the tested MMP2 variants were associated with double ER–PR positivity in breast cancer patients.
Association of MMP2 variants with risk of breast cancer according to ER and PR status.

MMP2: matrix metalloproteinase-2; ER: estrogen receptor; PR: progesterone receptor; ANOVA: analysis of variance.
ER and PR positivity were determined by immunocytochemistry of formalin-fixed and paraffin-embedded breast tissue sections; specimens were considered positive for ER and PR if ≥1% of tumor cells showed a positive nuclear staining.
Two-way ANOVA.
Alleles were designated as “1” (major allele) and “2” (minor allele).
We investigated the possible association of the tested MMP2 variants with breast cancer features. Results from Table 5 show that rs243866, rs243865, and rs243864 were positively associated with menstrual irregularity and histological type, while rs243866 and rs2285053 were negatively associated with menarche and nodal status. In addition, rs2285053 was negatively associated with triple negativity (p = 0.014), tumor size (p = 0.002), distance metastasis (p = 0.001), molecular type (p = 0.024), and outcome of chemotherapy (p < 0.001).
Matrix of correlation between MMP2 variants and breast cancer features and outcome.

MMP2: matrix metalloproteinase-2; HER-2: human epidermal growth factor receptor 2.
Bold values indicate statistically significant differences.
Spearman’s correlation coefficient.
Haploview analysis
High LD was noted between MMP2 variants (Figure 1), and four-locus haplotypes were determined based on the MAF of individual variants and LD pattern between them. Of the potential 16 haplotypes, 4 were assigned as common (>2% of total), capturing 98.6% (patients) and 96.3% (controls) of all haplotypes. Haploview analysis identified haplotypes G

Haploview analysis of the typed MMP2 variants.
MMP2 four-locus haplotypes in breast cancer cases and control women.

MMP2: matrix metalloproteinase-2; aOR: adjusted odds ratio; BMI: body mass index.
four-locus MMP2 haplotype consists of 243866, rs243865, rs243864, and rs2285053 alleles; minor alleles are designated as underlined.
Haplotype frequencies.
Covariates that adjusted for were age, BMI, menses pattern and menarche, breastfeeding, smoking, and use of oral contraceptives.
Discussion
Breast cancer is the leading cause of cancer-related deaths worldwide. A number of studies have demonstrated increased expression of MMP2 in breast cancer,21,32,33 and genetic functional variants in MMP2 gene were associated with altered breast cancer susceptibility.26,27,29,34 In this regard, a link between MMP2 polymorphisms and the risk of breast cancer was reported for several populations.26,27,30,35 Our study confirmed the association of rs243865 and rs2285053 MMP2 SNP with the presence and the aggressiveness of breast cancer.
Located on chromosome 16, MMP2 gene contains several polymorphisms, of which (the promoter) rs243865 variant is linked with lower promoter activity, owing to disruption of Sp1-type promoter site (CCACC box). Results shown here revealed a negative association of rs243865 (–1306 C/T) polymorphism with breast cancer, in agreement with a recent Tunisian study involving 210 breast cancer patients and 250 healthy control women, which also documented negative association of the minor (T) allele of rs243865 with breast cancer. 30 Our results were in agreement with Mexican 36 and Chinese 34 studies, which also documented negative association of rs243865 with the risk of breast cancer. Our findings were in apparent disagreement with Swedish 35 and Brazilian 25 studies, which suggested no association between rs243865 and the risk of breast cancer. Functionally, the rs243865 minor (T) allele was associated with disruption of the binding of SP1 binding elements, resulting in decreased promoter activity.26,27,37
Similarly, MMP2 rs2285053 (−735 C/T) was negatively associated with breast cancer, as its minor [T] allele was enriched in breast cancer patients. This was in sharp contrast to Chinese 27 and Iranian 29 studies, which demonstrated that the major [C] allele was positively associated with increased risk of breast cancer. These discrepancies are explained by ethnic differences, selection of cases, and the statistical methods in assessing the association. By disrupting Sp1 binding site, 37 the C-to-T transition at position −735 and −1306 in MMP2 promoter region induces low MMP2 enzyme expression in stromal and neoplastic cells. 31 This explains, in part, the beneficial effect of lower expression of T allele in reducing excessive degradation of fibrillation collagen 29 and other ECM components and thus development of breast cancer.34,38
Neither rs243864 (–790 G/A) nor rs243866 (–1575 G/A) MMP2 variants was associated with breast cancer in Tunisians, as MAF distribution of both variants was comparable between breast cancer cases and control women. A lone Chinese study which investigated the association of 9 promoter (including the 4 tested variants in this study), 19 intron, 1 exon (exon 9), and 7 3’ flanking region (FR) MMP2 variants with breast cancer in 6066 Chinese women also showed lack of association of rs243864 (–790 G/A) and rs243866 (–1575 G/A) MMP2 with altered susceptibility to breast cancer. 27 MMP2 is an estrogenic responsive gene, 39 and many genetic variants of MMP2 (rs243865 and rs243866) are located within an operational ER binding site and reduce the transcriptional response to estrogen. Accordingly, ER-negative tumors, by expressing low MMP2 levels compared to ER-positive tumors, are associated with reduced transcriptional responsiveness to estrogen. 33 Future studies involving additional ethnic groups and larger sample size are needed to confirm, or alternatively rule out, any association of rs243864 or rs243866 with altered breast cancer susceptibility.
Overexpression of MMP2 was shown to be related to invasiveness and metastasis of certain cancers,18,19 including breast cancer.20,23,24,39 In this study, rs2285053 was negatively correlated with triple negative tumors, tumor size, distant metastasis, and lymph nodes. While not tested here, we speculate that MMP2 rs2285053 (–735 C/T) pro-cancer capacity resides in its ability to modulate the degradation of the ECM, as was suggested elsewhere. 31 Furthermore, carriage of MMP2 rs243865 and rs2208553 minor alleles in ER-negative tumors is expected to express low MMP2 activity. Our results are in disagreement with earlier reports, which demonstrated that both MMP2 variants are correlated with ER negativity and/or PR negativity.30,33 To the best of our knowledge, this is the study to analyze the association of rs243864 (–790 G/T) and rs243866 (–1575 G/A) with altered risk of breast cancer. As such, this does not provide for comparison of our results with others.
This study has several strengths, in particular, the relatively large sample size, and thus was sufficiently powered. Additional strengths are in the ethnicity of the studied population (only Tunisian Arabic-speaking cases and controls) and that it investigated the correlation of MMP2 variants with phenotypic aspects of breast cancer. This study had some limitations, which limited the interpretation of the results. It involved a limited number of MMP2 variants, thereby prompting the speculation of additional variants acting as at-risk loci for breast cancer. The retrospective case-control design was another shortcoming, as it failed to address the cause–effect relationship between carriage of the MMP2 variants and the phenotypic presentation of breast cancer and that MMP2 levels as determined by the presence of MMP2 variants were not performed. Additional larger designed studies are needed to better evaluate the effect of these and other likely MMP2 variants on breast cancer risk.
Conclusion
At the genetic level, MMP2 constitutes an at-risk locus of risk of breast cancer, and MMP2 gene variants rs2285053 (–735 C/T) and rs243865 (–1306 C/T) are linked with altered risk of breast cancer development and progression. Of these, rs2285053 (–735 C/T) appears to influence some breast cancer phenotypic features, including metastasis, lymph node involvement, and tumor size. This suggests that analysis of MMP2 variants may constitute potential biomarkers for breast cancer susceptibility and for early identification of individuals at high risk to develop breast cancer.
Footnotes
Acknowledgements
The authors wish to thank Ms Zainab Malalla for her technical assistance.
Author contributions
A.F.H. and R.M.G. contributed to the data entry and drafting of the manuscript. H.B. and A.D. contributed to patient screening, selection, and referral. M.H.-A. contributed to literature search and data analysis. A.M., S.Z., M.H., and K.R. contributed to patient screening, selection, and referral. B.Y.-L. contributed to data analysis. W.Y.A. was the project leader and contributed to the statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was done according to Helsinki II declaration and approved by the Research and Ethics Committee of Salah Azaiez Institute (IRB number: ISA/2018/19); all participants provided written informed consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
