Abstract
The aim of this study was to correlate matrix metalloproteinase-2 and matrix metalloproteinase-9 expression with the clinicopathological features and outcome of patients with early gastric cancer and to clinically elucidate more information on the role of matrix metalloproteinase-2 and matrix metalloproteinase-9 protein overexpression with regard to lymph node metastasis of early gastric cancer. The levels of matrix metalloproteinase-2 and matrix metalloproteinase-9 protein expression were assessed by immunohistochemistry. An association was observed between matrix metalloproteinase-2, matrix metalloproteinase-9, and matrix metalloproteinase-2/matrix metalloproteinase-9 overexpression and clinicopathological factors, such as ulceration and lymph node metastasis. Furthermore, matrix metalloproteinase-9 and matrix metalloproteinase-2/matrix metalloproteinase-9 overexpression both were strongly correlated with histological grade. In addition, matrix metalloproteinase-2/matrix metalloproteinase-9 overexpression correlated with deep invasion. Multivariate Cox regression analysis revealed that matrix metalloproteinase-2 and matrix metalloproteinase-9 expression were both independent factors of overall survival in patients with early gastric cancer. In novelty, we found that matrix metalloproteinase-2/matrix metalloproteinase-9 overexpression was an independent indicator of lymph node metastasis in early gastric cancer which will be helpful in clinic to select the appropriate treatment of these patients.
Keywords
Introduction
The incidence of early gastric carcinoma (EGC; confined to the mucosa or submucosa, regardless of the presence or absence of lymph node metastasis) has been increasing steadily. Patients with EGC were associated with an optimal overall survival (OS). In addition, some minimally invasive surgeries such as endoscopic mucosal resection, endoscopic submucosal dissection, laparoscopic gastrectomy, and robotic surgery with or without lymphadenectomy which have improved quality of life and obtained early recovery were proposed as a suitable alternative to conventional open surgery in selective patients with EGC.1–3
However, lymph node metastasis still occurred in EGC, even in patients with mucosa cancer. 4 So, it is dangerous to perform minimally invasive surgery on patients with node-positive EGC.5,6 In addition, a subset of them still experience recurrence and metastasis even after R0 gastrectomy with lymphadenectomy. Therefore, it is important to identify a sensitive biomarker to predict lymph node metastasis in patients with EGC.
Matrix metalloproteinases (MMPs) comprise a large family of zinc- and calcium-dependent proteolytic enzymes that degrade, and remodeling of extracellular matrix (ECM), such as collagen, laminin, fibronectin, elastin, and the protein core of proteoglycans, plays an important role in tumor growth, progression, and metastasis.7,8
Matrix metalloproteinase-2 (MMP-2, 72-kDa gelatinase, type IV collagenase) and matrix metalloproteinase-9 (MMP-9, 92-kDa gelatinase, called type IV collagenases) were key members of the MMPs family.9,10 MMP-2 and MMP-9 highly were expressed in ovarian cancer, cervical cancer, hepatocellular, head and neck and thyroid carcinomas, squamous cell carcinomas, and gastric cancer and were closely associated with tumor stage, lymphatic invasion, lymph node metastasis, and recurrence.7,11,12
However, the expression of MMP-2 and MMP-9 in EGC and the relationship between lymph node metastasis and the expression of MMP-2 or MMP-9 were still unknown. So, this study was designed to examine the expression of MMP-2 and MMP-9 in patients with EGC and evaluate the relationship of their expression with clinical significances.
Materials and methods
Patients
The clinicopathological data and specimens (n = 204) were obtained from the Affiliated Cancer Hospital of Zhengzhou University between March 2008 and March 2011. After being obtained, the samples were immediately formalin-fixed and paraffin-embedded for long-term preservation. All patients were histologically proved to be EGC (mucosa or submucosa) and underwent radical gastrectomy with lymphadenectomy (D1 lymphadenectomy or D2 lymphadenectomy). Patients with history of gastrectomy or other malignancy, distant metastasis, peritoneal dissemination, and chemotherapy or radiotherapy before surgery and those who died during periopertaive (30 days after surgery) period were excluded.
Tumor sites were equally divided into upper, middle, and lower third section of the stomach. Histological grades were classified as G1 (well differentiated), G2 (moderately differentiated), G3 (poorly differentiated), and G4 (undifferentiated, signet ring cell carcinoma, and mucinous adenocarcinoma). Recurrence was classified as locoregional recurrence (remnant gastric and anastomotic recurrence and regional lymph node recurrence), peritoneal dissemination, and distant recurrence. Tumor grade and stage were classified according to the seventh edition of the American Joint Committee on Cancer/International Union Against Cancer (AJCC/UICC) tumor–node–metastasis (TNM) classification. 13 This study was approved by the Ethics Committee of the Affiliated Cancer Hospital of Zhengzhou University.
Follow-up
The follow-up was performed every 6 months with physical examination, blood tests, chest radiographs, abdominal ultrasonography, computed tomography, and serum carcinoembryonic antigen (CEA) and carbohydrate antigen 199 (CA199) levels. The follow-up lasted until death or the cutoff date of 31 May 2016. The median follow-up time was 62 months, with a range of 15 months to 88 months. OS was defined as the time interval in months between completion of therapy and death of this tumor disease.
Immunohistochemistry
Immunohistochemistry of MMP-2 and MMP-9 was carried out on each tissue specimen using the Two-Step IHC Detection Reagent Kit (Zhongshan Golden Bridge Biotechnology Co. Ltd., Beijing, China) according to the manufacturer’ s instruction. Tissue sections were dewaxed in xylene and rehydrated through graded alcohol concentrations using standard procedures. After washing in phosphate-buffered saline (PBS; thrice for 5 min), sections were then incubated 10 min with 3% H2O2 to quench endogenous peroxidase activity. The slides were pretreated with a 0.01 mol/L citrate buffer (pH 6.0) and autoclaved for 3 min, and then, slides were incubated with MMP-2 (1:200 dilution; Abcam, USA) and MMP-9 (1:200 dilution; Abcam) overnight at 4°C. The sections were washed in PBS and incubated with secondary antibodies at room temperature for 20 min, followed by incubation with 3,3′-diaminobenzidine tetrahydrochloride (Dako, Hamburg, Germany) and then counterstained with hematoxylin. For negative controls, isotype control immunoglobulin G was used.
All the tissue sections were analyzed by two independent pathologists experienced in evaluating immunohistochemistry and who were blinded to the clinicopathological data. Based on the number of positive tumor cells, the staining was scored as follows: “0” if 0% of the tumor cells were positively stained and “1” if 1%–10%, “2” if 11%–50%, “3” if 51%–70%, and “4” if ⩾71% of the cells were positive. The staining intensity was scored as follows: score 0 (negative staining), 1 (weak staining), 2 (moderate staining), and score 3 (intense staining) in at least five different high-power fields. The final score was based on the sum of the staining extent and intensity scores. A final staining score less than 4 was considered to be low expression, and a score equal or more than 4 was defined as high expression. For statistical analysis, we defined MMP-2/MMP-9 high expression as cases with both MMP-2 and MMP-9 high expression. Also, we defined MMP-2/MMP-9 low expression as cases with MMP-2 low and MMP-9 high, MMP-2 high and MMP-9 low, or both MMP-2 and MMP-9 low expression.
Statistical analysis
The association between MMP-2 and MMP-9 expression and clinicopathological variables was tested using a Chi-square test. OS and disease-free survival (DFS) were evaluated using the Kaplan–Meier method, and the differences among the levels of possible prognostic factors were compared using the log-rank test in the univariate analyses. The Cox proportional hazards model was used to estimate the independent prognostic factors for survival. Univariate and multivariate logistic regressions with covariate adjustment were used to assess the association between protein overexpression and lymph node metastasis. A two-sided p < 0.05 was considered statistically significant, and all statistical analyses were performed with the SPSS 16.0 software (SPSS, Chicago, IL, USA).
Results
Clinicopathological characteristics of patients with EGC
The clinicopathological characteristics of patients with EGC were shown in Table 1. The median age was 56 years (ranged from 21 to 75 years). Most of the patients were male (68.4%), confined to submucosa (69.6%), and poorly differentiated (65.9%) and had negative lymph node (75.5%) and more than 15 lymph node dissection (68.6%).
Demographic characteristics of patients with early gastric cancer.

MMP: matrix metalloproteinase.
Relationship between clinicopathological variables and MMP-2 and MMP-9 overexpression in EGCs
The expression of MMP-2 and MMP-9 was explored with immunohistochemical method. The representative images of MMP-2 and MMP-9 protein in EGC tissues were displayed in Figure 1. The MMP-2 and MMP-9 were both predominately expressed within the cytoplasm of tumor cells of EGC.
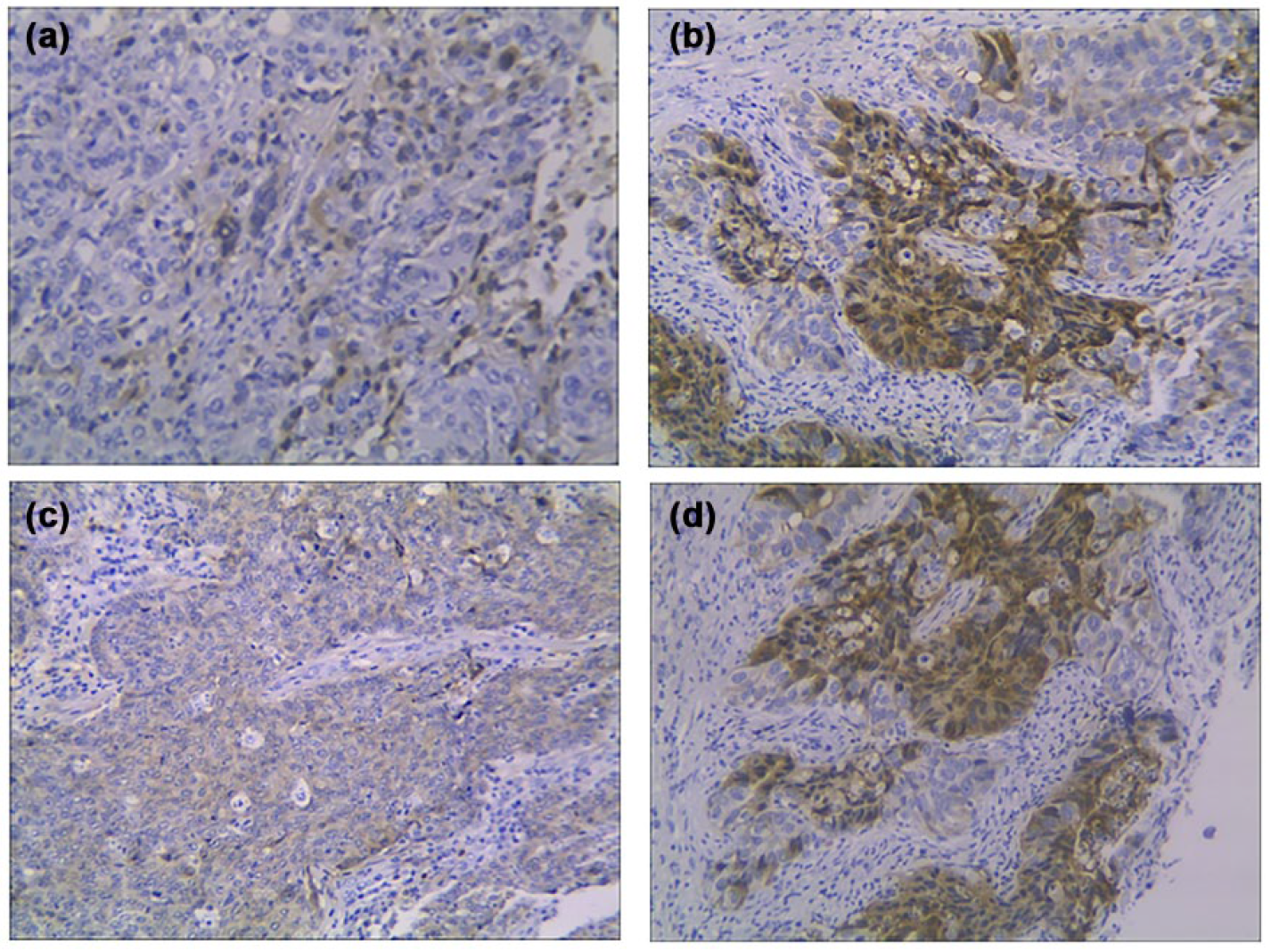
Representative images of MMP-2 and MMP-9 protein expression in early gastric cancer tissues by immunohistochemistry. (a) Low expression of MMP-2. (b) High expression of MMP-2. (c) Low expression of MMP-9. (d) High expression of MMP-9 (original magnification, 200×).
Of 204 patients, MMP-2, MMP-9, and MMP-2/MMP-9 protein overexpression was highly observed in 96 (47.1%), 103 (50.5%), and 58 (28.4%) patients, respectively. Table 2 displayed the association between clinicopathological variables and MMP-2 and MMP-9 overexpression. Increased MMP-2 expression was significantly associated with ulceration (p < 0.001) and lymph node metastasis (p < 0.001). MMP-9 overexpression was also significantly associated with ulceration (p < 0.001), lymph node metastasis (p = 0.004), and poor histological grade in addition (p < 0.001). Further observation showed that MMP-2/MMP-9 overexpression correlated with poor histological grade (p = 0.015), ulceration (p < 0.001), lymph node metastasis (p < 0.001), and deep invasion (p = 0.025).
The association between MMP-2 and MMP-9 expression and clinicopathological variables in early gastric cancers.

MMP: matrix metalloproteinase.
Chi-square test.
Prognostic significance of MMP-2 and MMP-9 protein expression in EGC patients
Univariate and multivariate analyses were conducted to determine the predictors for OS (Table 3). Univariate survival analysis revealed that histological grade (p = 0.015), tumor size (p < 0.001), depth of invasion (p = 0.013), ulceration (p < 0.001), lymph node metastasis (p < 0.001), number of lymph node dissection (p = 0.019), MMP-2 overexpression (p < 0.001), MMP-9 overexpression (p < 0.001), and MMP-2/MMP-9 expression (p < 0.001) were positively associated with OS in EGC patients. The survival effect of MMP-2, MMP-9, and MMP-2/MMP-9 expression on EGC patients was presented in Figure 2.
Univariate and multivariate survival analysis of overall survival in 204 patients with early gastric cancer.

HR: hazard ratio; CI: confidence interval; MMP: matrix metalloproteinase.
Log-rank test.
Cox regression test.

Kaplan–Meier curves for overall survival (OS) in 204 patients with early gastric cancer. (a) OS curves according to MMP-2 expression status. (b) OS curves according to MMP-9 expression status. (c) OS curves according to MMP-2/MMP-9 expression status. (d) OS curves according to lymph node metastasis status.
The results of the multivariate analysis demonstrated that tumor size (p = 0.001), lymph node metastasis (p = 0.022), number of lymph node dissection (p = 0.003), MMP-2 overexpression (p = 0.011), and MMP-9 expression (p = 0.004) were independent prognostic predictors for patients with EGC. However, there was no significance between MMP-2/MMP-9 overexpression and patient outcome.
Relationship between clinicopathological variables and lymph node metastasis in EGCs
The relationships between lymph node metastasis and clinicopathological variables were shown in Table 4. The lymph node metastasis was significantly associated with poor histological grade (p = 0.031), larger tumor size (p = 0.023), deep invasion (p < 0.001), and number of lymph node dissection (p = 0.001).
The association between lymph node metastasis and clinicopathological variables in early gastric cancers.
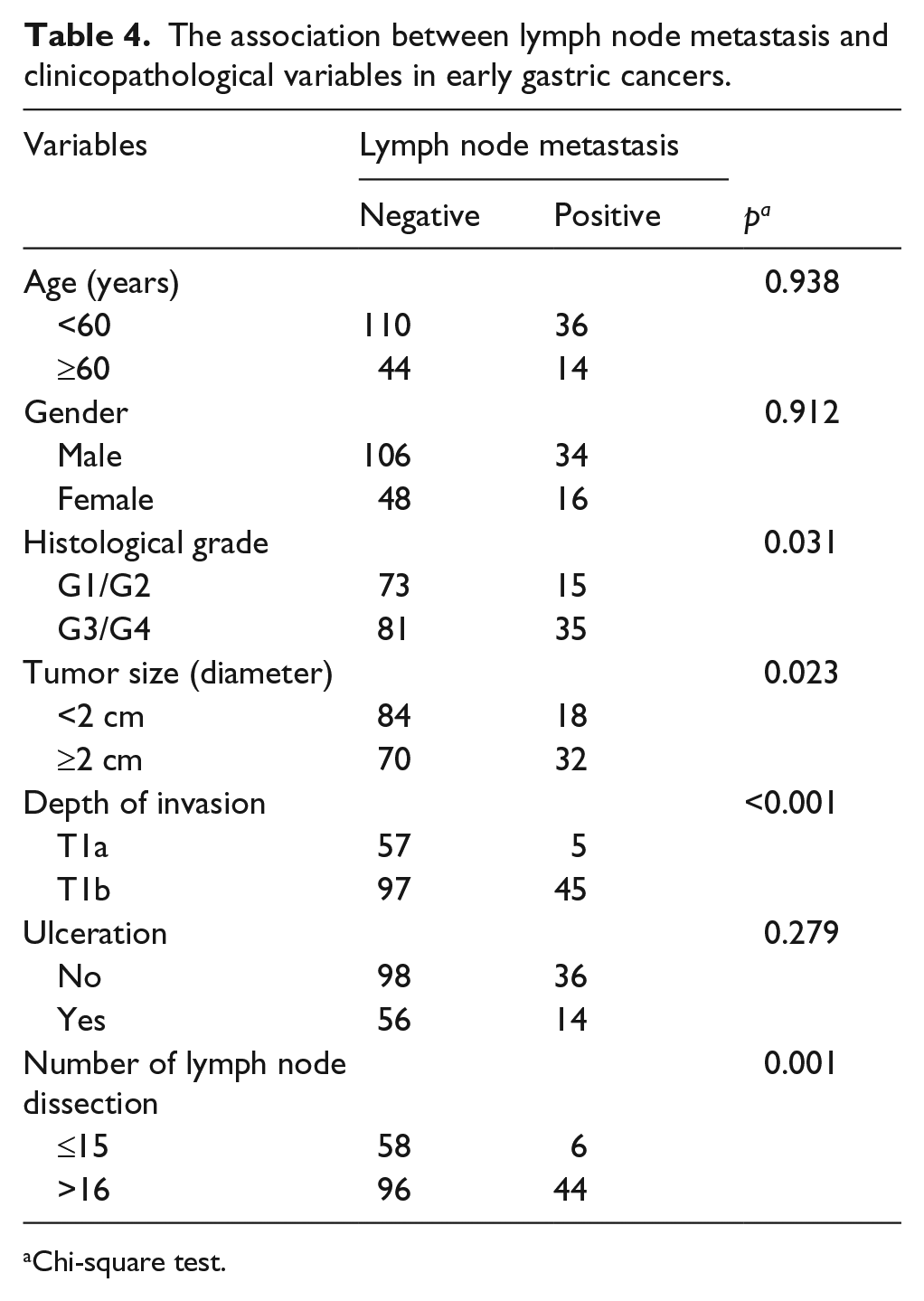
Chi-square test.
The associations between MMP-2 and MMP-9 expression and lymph node metastasis in EGCs were displayed in Figure 3. Cases with MMP-2 protein high expression account for 38.9% and 72.0% in cases with negative and positive lymph nodes, respectively. Also, cases with MMP-9 protein high expression account for 46.1% and 64.0% in cases with negative and positive lymph nodes, respectively. In addition, cases with MMP-2/MMP-9 protein high expression account for 20.8% and 52.0% in cases with negative and positive lymph nodes, respectively.

Associations between MMP-2 and MMP-9 expression and lymph node metastasis (LN-M) in early gastric cancers. (a) LN-M is illustrated according to MMP-2 expression. (b) LN-M is illustrated according to MMP-9 expression. (c) LN-M is illustrated according to MMP-2/MMP-9 expression.
Predictive significance of MMP-2 and MMP-9 expression on lymph node metastasis in EGCs
A multivariate logistic regression analysis was performed to evaluate the independent predictive significance of MMP-2, MMP-9, and MMP-2/MMP-9 overexpression for lymph node metastasis (Table 5). The result showed that tumor size (p = 0.011, odds ratio (OR): 2.683, 95% confidence interval (CI): 1.252–5.746), depth of invasion (p = 0.007, OR: 4.246, 95% CI: 1.493–12.071), number of lymph node dissection (p = 0.001, OR: 5.929, 95% CI: 2.168–16.217), and MMP-2/MMP-9 overexpression (p < 0.001, OR: 2.134, 95% CI: 1.456–3.127) were independently associated with lymph node metastasis.
The relationships of MMP-2 and MMP-9 expression with lymph node metastasis by multivariate analysis.

B and SE: the parameter estimator of association coefficient and its standard error; CI: confidence interval; OR: odds ratio.
Multivariate logistic regression text.
Discussion
We analyzed MMP-2 and MMP-9 expression in EGC by immunohistochemistry using 204 surgical specimens. In addition, MMP-2/MMP-9 high expression was significantly associated with ulceration and lymph node metastasis as well as related to depth of invasion. This finding indicates that MMP-2/MMP-9 overexpression may be a pivotal predictor for tumor progression. Moreover, we also demonstrate that MMP-2/MMP-9 overexpression is independently associated with lymph node metastasis. To our knowledge, this study provides the first detailed demonstration of an association of clinicopathological parameters and prognostic significance of MMP-2 together with MMP-9 protein expression in EGCs.
Lymph node metastasis is an independent prognostic factor for patients with EGC, and patients with lymph node metastasis had a significantly poorer OS compared with those without lymph node metastasis.14,15 In this study, lymph node metastasis was an independent factor for predicting prognosis in EGCs. Although deep invasion (submucosa), poor differentiation, larger tumor size, ulceration, and lymphatic involvement were associated with lymph node metastasis in patients with EGC, 13 it is unreliable to define the metastasis of lymph node just based on computed tomography, endoscopic ultrasonography, and the gross appearance of lymph nodes, as 30% of lymph node metastases occurred in nodes less than 3 mm in diameter.16,17 Therefore, it is important to understand the carcinogenesis and progression of EGC.
The formation of lymph node metastasis depends on the ability of malignant cells to enter the blood and lymphatic system and subsequently form tumors in lymph nodes and distant organs. 18 MMP-2 and MMP-9 play an important role in type IV collagen and denatured collagen (key element of basement membranes) degradation, which is often the initial step in carcinoma invasion and metastasis. In addition, high MMP-2 and MMP-9 expression was significantly associated with tumor histological grade, lymph node metastasis, and distant metastasis. 19
MMP-2 and MMP-9 were also detected highly expressed in stromal cells adjacent to the tumor in a variety of malignant tumors and played a critical role in angiogenesis and tumor progression. 10 And the ulceration within the lesion may also facilitate the interchange of lymphatic flow between the mucosa and the submucosa, promoting the development of lymph node metastasis. 19
In this study, we demonstrated significant relationships of MMP-2/MMP-9 expression with poor histological grade, deep invasion, ulceration, and lymph node metastasis. The incidence of lymph node metastasis in patients with MMP-2/MMP-9 overexpression (44.8%) was significantly higher than that with single MMP-2 (37.5%) and MMP-9 (31.1%) overexpression (data not shown). And a multivariate logistic regression analysis revealed that MMP-2/MMP-9 overexpression was an independent indicator for lymph node metastasis instead of MMP-2 or MMP-9 alone expression in EGCs.
Traditionally, we thought that MMPs promoted tumor progression and metastasis through ECM and basement membrane degradation. However, it was found that this process could also be resulted through regulating many important signal pathways that controlled cell growth, inflammation, and angiogenesis or could even work in a nonproteolytic manner. These functions promote MMP counter apoptosis and regulate innate immunity and homeostasis of the extracellular tumor microenvironment.20,21 For the important role of MMPs in tumor progression and regulating signal pathways, it is identified as key biomarkers and treated as treatment target.7,12,20
In conclusion, MMP-2 and MMP-9 protein overexpression could be a predictor for prognosis and lymph node metastasis. In addition, combined evaluation of MMP-2 and MMP-9 expression could be clinically useful to predict lymph node metastasis in patients with EGC. This may be helpful to determine the appropriate treatment of EGC.
Footnotes
Acknowledgements
Y.L. and S.Y. have contributed equally to this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
