Abstract
Variable association of transforming growth factor beta 1 (TGFβ1) in breast cancer (BC) pathogenesis was documented, and the contribution of specific TGFB1 polymorphisms to the progression of BC and associated features remains poorly understood. We investigated the contribution of TGFB1 rs1800469, rs1800470, rs1800471, and rs1800472 variants and 4-locus TGFB1 haplotypes on BC susceptibility, and pathological presentation of BC subtypes. Study subjects comprised 430 female BC cases, and 498 cancer-free control women. BC-associated pathological parameters were also evaluated for correlation with TGFB1 variants. Results obtained showed that the minor allele frequency (MAF) of rs1800471 (+74G>C) was higher seen in BC cases than in control subjects, and was associated with increased risk of BC. Significant differences in rs1800471 and rs1800469 (−509C>T) genotype distribution were noted between BC cases and controls, which persisted after controlling for key covariates. TGFB1 rs1800472 was positively, while rs1800470 was negatively associated with triple negativity, while rs1800470 positively correlated with menarche, but negatively with tumor size and molecular type, and rs1800469 correlated positively with menstrual irregularity, distant metastasis, nodal status, and hormonotherapy. Heterogeneity in LD pattern was noted between the tested TGFB1 variants. Four-locus (rs1800472-rs1800471-rs1800470-rs1800469) Haploview analysis identified haplotype
Introduction
Breast cancer (BC) is the most prevalent cancer among women, with more than 2.08-million new cases of BC identified globally in 2018. 1 BC is the leading cancer in women in Tunisia, with reported high incidence (15.61%) and mortality (8.43%) rates, and currently ranks as the second cause of female mortality in Tunisia. 1 The pathogenesis and progression of BC requires participation of modifiable factors such as lifestyle, treatment with xenobiotics, and environmental factors, and nonmodifiable factors which include age, racial, and genetic background. 1 Current screening tools include radiological (mammography), ultrasonography, genetic (BRCA), and biochemical techniques.2,3 This prompts search for novel diagnostic and prognostic biomarkers, with sufficient sensitivity and/or specificity.
Earlier studies identified common germline variants associated with altered BC susceptibility.4,5 Of these, transforming growth factor beta 1 (TGFβ1), a pleiotropic anti-inflammatory cytokine secreted by cancerous and non-cancerous cells, was extensively studied for its role in modulating the risk of BC.6,7 TGFβ1 is the most abundant member of the TGFβ family of growth factors which also comprises TGFβ2 and TGFβ3, and is expressed by endothelial cells, connective tissues, and hematopoietic cells. 8 TGFβ1 plays a crucial role in embryogenesis and development of mammary glands, 9 and in many physiological and disease states, largely by controlling cellular differentiation and cell cycle progression.10,11 Paradoxically, TGFβ1 has a dual role in cancer.11,12 It was described to act as tumor suppressor in early carcinogenesis by inducing apoptosis or inhibiting cell growth, while promoting tumorigenicity and metastasis through suppression of local immunosurveillance and supporting tumor progression in advanced stages of cancer.13,14
Located on chromosome 19 (19q13.1), TGFB1 gene contains several functional and non-functional polymorphisms, some of which were shown to contribute to altered BC susceptibility.7,15–18 Of these, rs1800469 (–1347C>T) promoter, rs1800470 (+29T>C) codon 10, and rs1800471 (+74G>C) signal peptide sequence variants were widely studied in BC.15,19–22 However, the association of TGFB1 variants with BC remains controversial,12,15,17 and apparently varies in different ethnic groups.12,23,24 No association25,26 and weak/moderate association25,27 were reported. These apparently contradictory findings were explained by ethnicity-related variations, along with the likely interactions with related and distant variants.
We previously demonstrated the association of genetic variants in progesterone receptor (PR), 28 estrogen receptor (ER), 29 and NF-κB gene 30 with altered risk of BC. Given the central role of TGFβ1 as inflammatory mediator in modulating processes associated with BC, and as TGFβ1 signaling is linked with NF-κB signaling, 31 here we analyze the contribution of genetic variation in TGFB1 to BC susceptibility in Arab-speaking Tunisian women.
Subjects and methods
Study subjects
Between June 2017 and November 2018, 430 women with BC and 498 cancer-free control women were recruited into this study from the outpatient oncology and surgery oncology services of Salah Azaïz Institute Hospital (Tunis, Tunisia). All participants consented in writing to be included in the study. Assessment of BC was done according to American Cancer Society guidelines. 3 These included mammography and breast biopsy testing for confirmation of BC; all patients had these tests done. Control women comprised volunteer from the community or women reporting for annual physical checkup, and none had personal or a family history of BC. Control women were matched to BC patients according to age and self-declared origin.
Demographic and clinical biodata were obtained from medical records and through personal interview by the referring physician or senior resident, using a unified structured questionnaire. Collected data included the age at entry of the study, age at primary diagnosis of BC, status of menopause, and metastatic disease at presentation. Histological assessment (disease stage, nuclear grade), ER and PR status, and treatment regimen (surgery, endocrine-based therapy, radiotherapy, chemotherapy) were collected for all BC cases. The study was done per Helsinki II declaration and was approved by the Research & Ethic Committee of Salah Azaïz Institute in Tunis (registration number: ISA/2018/19, granted on 25 June 2017).
Informed consent
Informed consent was obtained from all individual participants included in the study.
TGFB1 genotyping
Four single nucleotide polymorphisms (SNPs) in TGFB1 gene with clinical relevance and minor allele frequency (MAF) ≥4% in control Tunisian subjects were identified, using National Center for Biotechnology Information (NCBI) Entrez Gene SNP Geneview. These comprised the missense variants rs1800472 (+791C>T; position 41341955; assay number C___8708464_20), rs1800471 (+74G>C; position 41352971; assay number C__11464118_30), and rs1800470 (+29T>C; position 41353016; assay number C__22272997_10), and the 5′ near-gene variant rs1800469 (–1347C>T; position 41354391; assay number C___8708473_10). TGFB1 genotyping was performed by assay-on-demand (VIC- and FAM-labeled primers) TaqMan assays (Applied Biosystems, Dubai, UAE). The reaction was performed on StepOnePlus real-time PCR system (Applied Biosystems). The reproducibility of genotyping was ascertained by inclusion of replicate blinded quality control samples; concordance exceeded 99%, and average successful sample and SNP genotyping rate was 98.9%.
Statistical analysis
Statistical analysis was performed on SPSS version 24 (IBM, Armonk, NY). Continuous (quantitative) data which were normally distributed are shown as mean (±SD), while categorical (qualitative) variables are expressed as percent of total. Student’s t-test and Pearson χ2 test were employed in testing differences in means and inter-group significance, respectively. Genetic Power Calculator (http://pngu.mgh.harvard.edu) was employed in calculation of study power, using the following parameters: 430 BC cases and 498 control women, MAF of tested variants (in BC cases), estimated BC prevalence in Tunisia, and genotypic relative risk for heterozygote (1/2), and minor allele homozygous (2/2). Accordingly, the overall power (87.3%) was determined as the average of the power of the four tested variants.
Haploview 4.2 (Broad Institute, Cambridge, MA) was utilized in the evaluation Hardy–Weinberg equilibrium (HWE), and to check for linkage disequilibrium (LD) between TGFB1 variants, and for estimation of haplotype patterns, which were reconstructed by the expectation maximization method. Taking control women as reference group (odds ratio (OR) = 1.00), logistic regression analysis was used in determination of the ORs and 95% confidence intervals (CIs) associated with the risk of BC. Statistical significance set at p < 0.05.
Results
Study subjects
Table 1 presents the demographic and clinical characteristics of study subjects. Significant differences between BC cases and control women were noted in body mass index (BMI; p < 0.001), menarche (p = 0.021), and number of smokers (p = 0.022). In addition, irregular menses (p < 0.001) and previous oral contraceptives use (p < 0.001) were more frequent in BC cases, and breast feeding frequency was lower in BC cases than in control women (p < 0.001). Accordingly, we selected as these as the main covariates that were controlled for in later analysis.
Characteristics of study subjects.
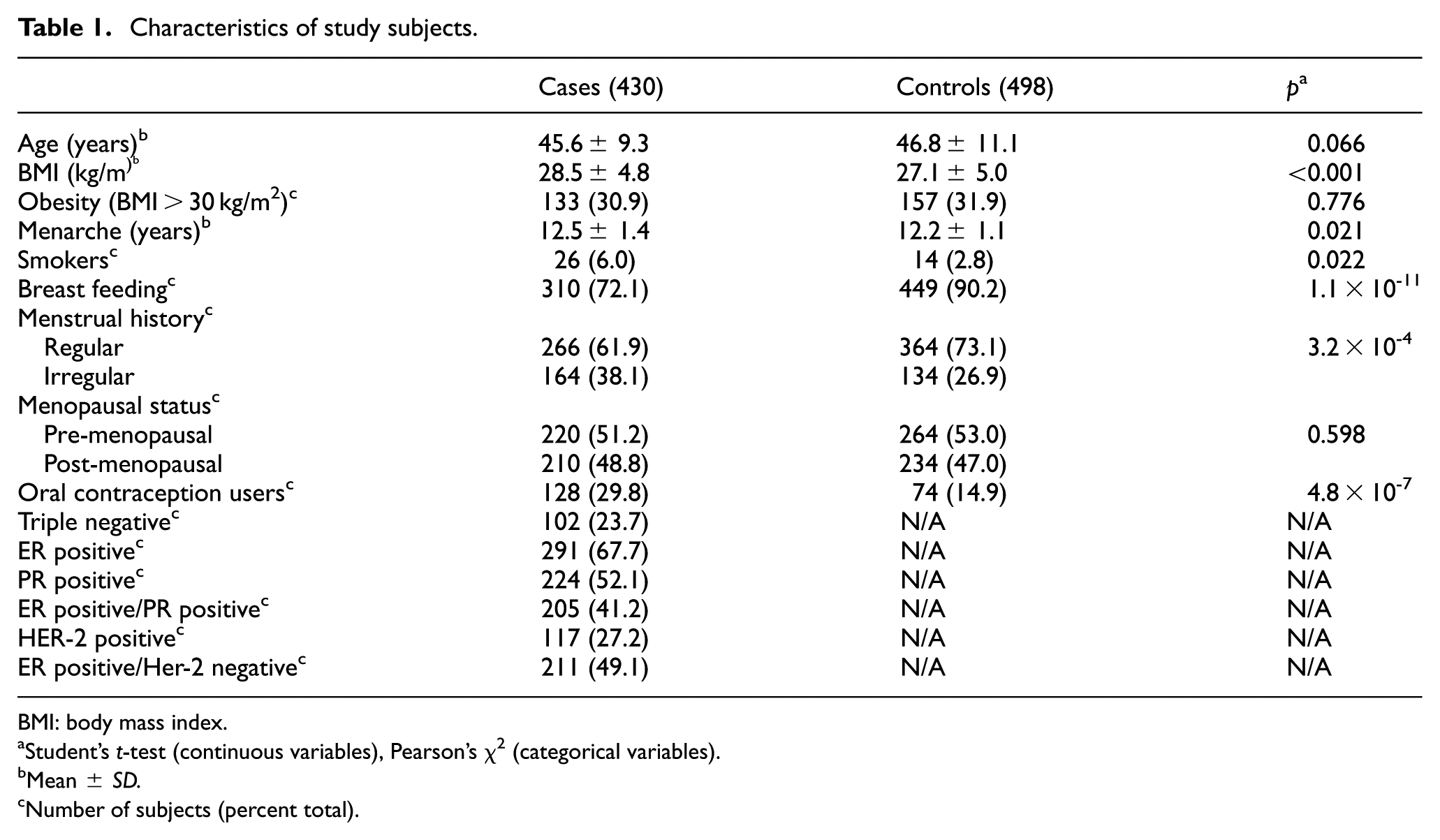
BMI: body mass index.
Student’s t-test (continuous variables), Pearson’s χ2 (categorical variables).
Mean ± SD.
Number of subjects (percent total).
Association studies
MAF of the tested TGFB1 variants in BC cases and cancer-free control women is presented in Table 2. The genotype distributions of the four tested TGFB1 variants were in HWE among study subjects. Higher frequency of rs1800471 minor (“C”) allele (p = 0.003) was seen in BC cases than in control women and was associated with increased risk of BC (OR (95% CI) = 1.84 (1.22–2.78)). MAF of the remaining TGFB1 variants was comparable between BC cases and control subjects.
Distribution of TGFB1 alleles in breast cancer cases and control women.

TGFB1: transforming growth factor beta 1; SNP: single nucleotide polymorphism; HWE: Hardy–Weinberg equilibrium; OR: odds ratio; CI: confidence interval.
Boldface indicates minor allele.
Location on chromosome based on dbSNP build 125.
Minor allele number (frequency).
Table 3 illustrates the distribution of TGFB1 genotypes in BC cases and control women. The distribution of rs1800469 C/T (p = 0.05) and rs1800471 G/C (p = 0.003) genotypes was significantly different between BC cases and control women. This association remained significant after controlling for the main covariates (age, BMI, menarche and menses pattern, previous use of oral contraceptives, smoking, and breast feeding). The overall BC risk associated with a specific TGFB1 genotype was further confirmed by testing the effect of BMI, menarche, smoking, and oral contraceptive use as confounders. Results from Table 4 confirmed the association of rs1800471/+74G>C (p = 2.3 × 10−3; OR (95% CI) = 1.92 (1.25–2.92)) with increased risk of BC, and to a lesser extent rs1800469/–1347C>T (p = 0.048) with decreased risk of BC under the codominant genetic model.
TGFB1 genotype frequencies.

SNP: single nucleotide polymorphism.
Genotypes were coded as “1” = major allele and “2” = minor allele. The corresponding genotypes were C/C, C/T, and T/T for rs1800472; G/G, G/C, and C/C for rs1800471; T/T, T/C, and C/C for rs1800470; and C/C, C/T, and T/C for rs1800469.
Chi-square test.
Number of subjects (frequency).
Effects of TGFB1 SNP genotypes on the risk of CRC according to the different genetic models.

TGFB1: transforming growth factor beta 1; SNP: single nucleotide polymorphism; CRC: colorectal cancer; OR: odds ratio; CI: confidence interval; BMI: body mass index.
Boldface indicates statistically significant differences.
Models controlled for BMI, menarche, smoking, and oral contraceptive use.
Association of TGFB1 genotypes with BC features
The association of the tested TGFB1 variants with BC features was investigated. Results from Table 5 show that rs1800472 was positively associated with triple negativity (ρ = 0.165; p = 0.001), molecular type (ρ = 0.143; p = 0.001), ER/HER-2 status (ρ = 0.132; p = 0.005), and menarche (ρ = 0.960; p = 0.002). On the other hand, rs1800470 was positively associated with menarche (ρ = 0.100, p = 0.015), but negatively with triple negativity (ρ = –0.127; p = 0.018), tumor size (ρ = –0.119, p = 0.003), nodal status (ρ = –0.094, p = 0.038), and ER/HER-2 status (ρ = –0.073, p = 0.022). Furthermore, rs1800469 correlated positively with menstrual irregularity (ρ = 0.190, p < 0.001), menarche (ρ = 0.108; p = 0.010), distant metastasis (ρ = 0.184, p < 0.001), nodal status (ρ = 0.158, p = 0.001), molecular type (ρ = 0.194, p = 0.007), ER/HER-2 status (ρ = 0.121, p = 0.032), and hormonotherapy (ρ = 0.127, p = 0.011).
Matrix of correlation between TGFB1 variants and breast cancer feature and outcome.
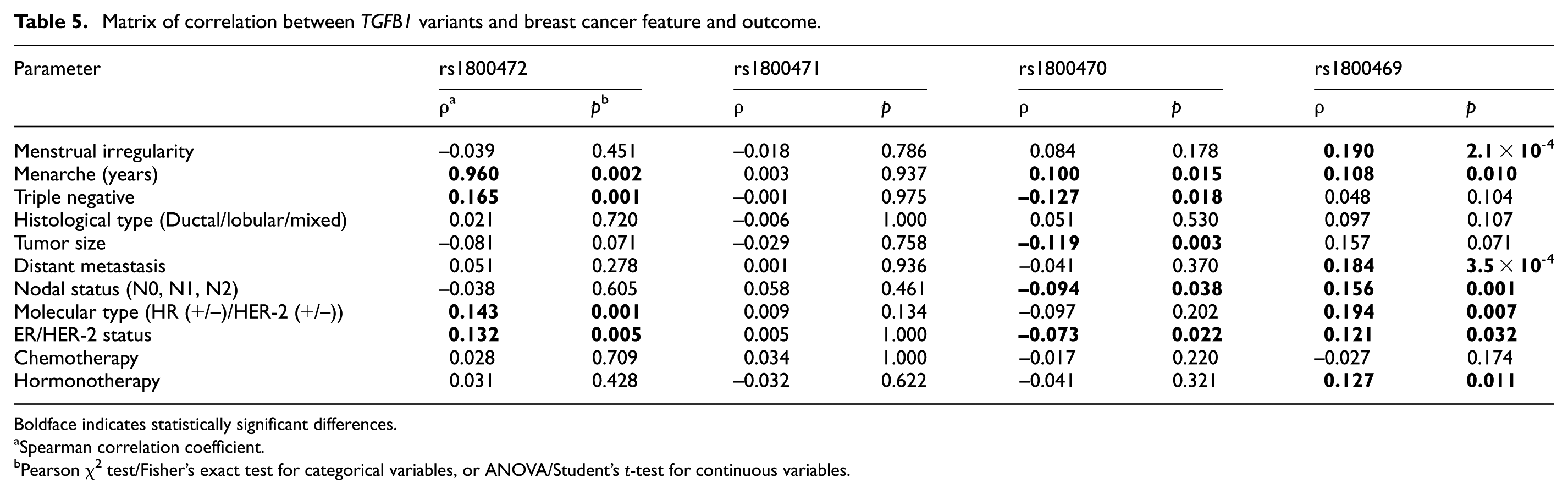
Boldface indicates statistically significant differences.
Spearman correlation coefficient.
Pearson χ2 test/Fisher’s exact test for categorical variables, or ANOVA/Student’s t-test for continuous variables.
Haploview analysis
Haploview analysis revealed mixed LD pattern between the tested TGFB1 variants (Figure 1). Four-locus haplotypes were constructed based on the prevalence of individual TGFB1 SNP and LD pattern. Of the possible 16 TGFB1 haplotypes, 6 were found to be common (frequency > 2%), capturing 98.2% of all possible haplotypes (97.0% in cases and 98.8% in controls). Haploview analysis identified haplotype
Haplotype frequencies across TGFB1 SNPs analyzed.

TGFB1 haplotypes: rs1800472–rs1800471–rs1800470–rs1800469. OR: odds ratio; aOR: adjusted odds ratio; CI: confidence interval.
Underlined indicates minor allele.
Adjusted for BMI, menarche, smoking, and oral contraceptive use.
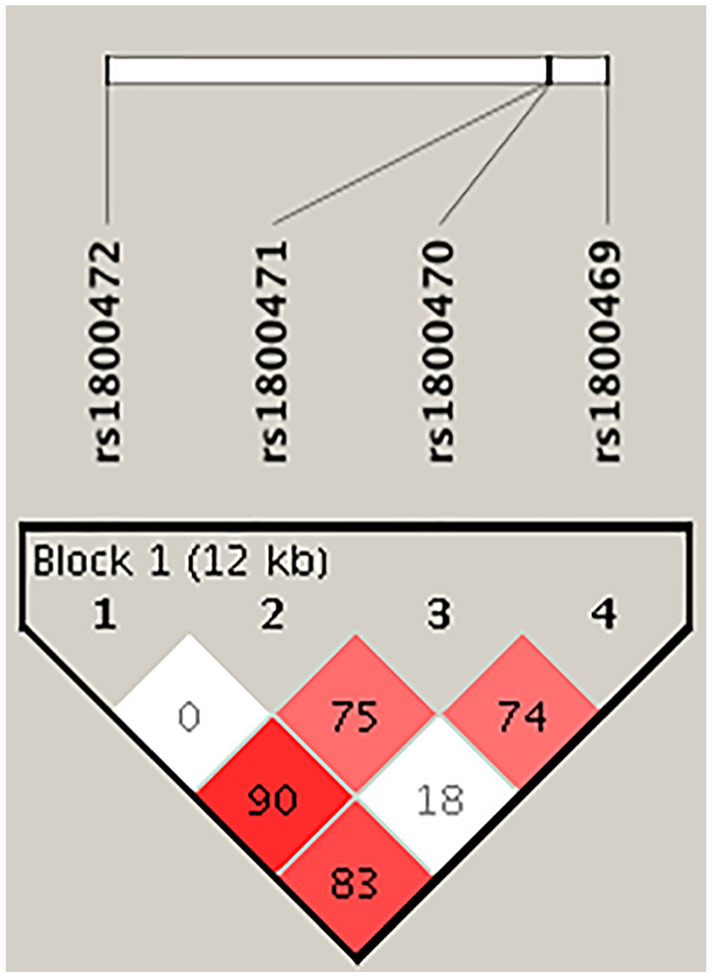
Haploview plot of TGFB1 SNP analyzed. The relative positions of TGB1 SNPs (build 37.3) are displayed, along with the basic gene structure, above the Haploview diagram. The relative LD between pairs of TGFB1 SNPs is color-indicated. This was based on D′, that is, normalized linkage disequilibrium measure or D divided by the theoretical maximum for the observed allele frequencies, multiplied by 100. Values close to zero indicate no LD, while values approaching 100 indicate full LD. The red-colored square represents varying degrees of LD < 1 and LOD (logarithm of odds) > 2 scores; darker shades indicating stronger LD.
Discussion
TGFβ1 plays a central, but paradoxical role in BC development.18,32,33 While it controls cell cycle progression and apoptosis in early carcinogenesis, it induces a state of immunosuppression in advanced malignancies, which promotes aggressive carcinogenesis and metastasis.8,10,11,13 As the secretion of TGFβ1 is genetically determined, we tested the association between common variants in TGFB1 and BC susceptibility. Previous studies documented an association between TGFB1 polymorphisms and susceptibility to BC.15,23,25,33,34 However, controversial and inconclusive associations were also reported.26,35 In the present study, we documented the positive association of TGFB1 rs1800471 and rs1800469 with BC, and identified TGFB1 haplotypes that were negatively (
A case-control study design was employed in investigating the association between the risk of BC and the presence of TGFB1 polymorphisms. The control group consisted of 498 healthy women, who reported no positive family history of BC, and no personal history of any cancer types. This in turn increased the population power in evaluating the role of genetic factors to the risk of BC. These cancer-free control women were matched to BC cases according to age, menopause status, and ethnic origin (only Arabic-Speaking Tunisian women were included). Significant differences between controls and BC cases were noted in the prevalence of smoking, in agreement with findings elsewhere.36,37 Higher prevalence of women with irregular menses, and previous/current users of oral contraceptives were seen in BC cases than in control women. This was reminiscent of findings which documented association of irregular menses38,39 and oral contraceptive use38,40 with increased incidence of BC.
The polymorphisms included in this study were the missense rs1800472 (+791C>T, T264I), rs1800471 (+74G>C, R25Q), and rs1800470 (+29T>C, P10L), and the 5′ near-gene rs1800469 (–1347C>T) variants.12,18,19,27,34 While MAF and genotype distribution of only rs1800471 was significantly different between BC cases and controls, the genotype distribution of rs1800469 homozygous variant was significantly higher in BC cases. The rs1800469 variant lies within a binding site for YY1 (Yin Yang 1), and the transition of C with T leads to reducing the ligation. 41 While not tested here, functionally carriage of the minor (T) allele was linked with augmented TGF-β1 levels, compared to C/C genotype carriers. 42
TGFB1 rs1800470 is the most extensively studied polymorphism, located in codon 10 (CTG→CCG), resulting in leucine-to-proline substitution, and increased TGFβ1 secretion. 43 The allele and genotype distribution of rs1800470 was not significantly different between BC cases and control women. TGFB1 rs1800470 minor allele was previously reported to increase the risk of BC in Indians, 21 Koreans, 43 and Caucasians, 19 but paradoxically was associated with reduced risk of BC in White US 44 and Indian 6 populations. A recent Iranian study involving 100 BC cases and 100 control women 45 reported no significant difference in MAF between two groups, but demonstrated strong positive association between T/T genotype and BC risk (OR (95% CI) = 2.409 (1.087–5.337)). Furthermore, an earlier meta-analysis demonstrated that T allele from this variant was modestly protective against BC. 20 These apparently conflicting results may be explained by heterogenicity due to sampling variation and background ethnicity.
Compared to rs1800470, rs1800471 (+74G>C) is less studied. This variant is associated with arginine-to-proline substitution in codon 25, 19 and with lower TGFβ1 production. While rs1800471 MAF and genotype distribution was comparable between BC cases and control women, carriage of rs1800471 minor (C) allele correlated positively with menarche, but negatively with triple negativity, tumor size, and hormone receptor (HR) and HER-2 status among BC cases. An earlier Indian study involving 456 BC cases and 239 control women also reported no association between rs1800471 and BC risk. 17 Interestingly, a study on North Indian and South Indian women reported that this variant has a significant protective effect against BC. 21 Additional studies involving additional ethnic groups are needed to confirm or alternatively rule out the association of rs1800471 (+74G>C) with altered risk of BC, or with BC-associated features.
The rs1800472 located at exon 5 codon 263, and the corresponding residue lies near the site where the latency-associated peptide is cleaved from the active peptide, and thus appears to be related to TGFB1 activation. 46 MAF of rs1800472 seen in Tunisians (7.0%) was comparable to frequencies established for Caucasians (7.4%) and Hispanics (7.1%), but was higher than frequencies seen in Pan Europeans (2.5%–5.0%) and Africans (2.2%) (https://www.ncbi.nlm.nih.gov/projects/SNP/snp_ref.cgi?do_not_redirect&rs=rs1800472). While rs1800472 was not associated with BC per se, it was positively linked with triple negative status among BC cases. To the best of our knowledge, this was the first evidence documenting association of this variant with BC-associated features.
Haplotype association analysis revealed that C
Our study had several strengths. First, the number of BC cases and control women was substantial, and thus the study was sufficiently powered. Second, BC cases and control women were matched according to ethnicity (only Arabic-speaking Tunisian women included), which minimizes the problems of ethnic differences inherent in genetic association studies. Another strength of the study is in controlling for number of potential covariates. However, our study has also shortcomings. We could not measure serum TGFβ1 levels in BC cases and control women, which did not allow for addressing genotype–phenotype correlations. Another limitation lies in its retrospective nature, which prompts speculation on cause–effect relationship. The small number of TGFB1 SNPs studied prompts the speculation of the potential association of additional variants with BC and its associated features. Future studies involving other ethnic groups and additional variants are needed to confirm these findings.
Conclusion
TGFB1 constitutes a genetic risk locus of BC and rs1800471 and rs1800469 variants, and CGT
Footnotes
Acknowledgements
The authors wish to thank Ms. Zainab Malalla for her technical assistance.
Author contributions
M.H.-A. and R.M.G. contributed to data entry and drafting of the manuscript. H.B. contributed to patient screening, selection, and referral. A.H. contributed to literature search and data analysis. M.S. contributed to drafting of the manuscript. M.A., M.H., and K.R. contributed to patient screening, selection, and referral. B.Y.-L. contributed to data analysis. W.Y.A., project leader, contributed to statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was done according to Helsinki II declaration and approved by the Research & Ethic Committee of Salah Azaïz Institute (IRB number: ISA/2018/19, granted on 25 June 2017); all participants provided written informed consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
