Abstract
We investigate the clinical and pathological features related to variations in colorectal tumour apoptosis, proliferation and angiogenesis and the influence of the latter in short-term mortality (2 years); 551 tumour samples from a prospective cohort of patients with colorectal cancer were examined and tumour biology markers were determined as follows: percentage of apoptotic cells, by the terminal deoxynucleotidyl transferase (TdT) dUTP Nick-End Labeling technique; Ki-67 antigen, as a cell proliferation marker and density of microvessels (as a marker of angiogenesis). An increase in the percentage of cellular apoptosis is significantly related to the presence of poorly differentiated tumours, with vascular invasion (p < 0.001). The CD105 angiogenesis marker is not related to any clinical–pathological parameter except that of higher frequency in older patients (p = 0.03). Ki-67 is more frequently expressed in tumours with less nervous invasion (p = 0.05). Neither apoptosis nor angiogenesis present any significant association with short-term survival. The only marker clearly related to 2-year survival is Ki-67, which is shown to be a good prognostic factor in the multivariate analysis (hazard ratio = 0.49; 95% confidence interval = 0.27–0.90). Therefore, in a prospective cohort of colorectal cancer patients, only Ki-67 is a marker of good prognosis in short-term follow-up.
Introduction
With an estimated incidence of 1.4 million cases and an overall mortality rate of 693,900 in 2012, colorectal cancer (CRC) is the third most common diagnosed malignancy and a major public health challenge worldwide. 1
Importantly, despite recent improvements in CRC management, approximately 40%–50% of early diagnosed CRC patients will relapse, which justifies the need to find new biomarkers with a prognostic value. 2
In CRC patients, different histopathological features such as tumour grade, depth of invasion, lymph nodes and the presence of liver metastasis are prognostic factors. 3 However, as these conventional prognostic parameters are not able to predict the outcome of every individual patient, there is still a need to find novel markers to help predict these patients’ outcome and guide therapy decisions more accurately.
Carcinogenesis is fundamentally characterized by alterations in cellular proliferation, apoptosis and formation of blood vessels. Given the evidence of apoptosis’ important role during CRC pathogenesis and progression, the significance of apoptotic rates as potential prognostic marker should be given serious consideration. Nevertheless, little research has been conducted into the prognostic significance of apoptosis in CRC. In this regard, a small number of studies have reported that in some distal colon carcinomas, reduced apoptosis is related to undesirable outcomes and metastasis. 4 Others have suggested that there is an inverse relationship between apoptotic index (AI) and survival.5–7 In a retrospective study presented by Alcaide et al., 8 it was reported for the first time the evidence that high AI is related to a significant decrease in CRC patients’ overall and disease-free survival.
Vascular–endothelial, hepatocyte and platelet-derived growth factors secretion by neoplasms stimulate endothelial migration and proliferation and promote angiogenesis. 9 Neovascularization promotes tumour growth by supplying nutrients and oxygen. 10 CD105 (endoglin), a co-receptor of the transforming growth factor-beta superfamily, has been proposed as a marker of neovascularization in solid malignancies. As sustained angiogenesis is one of the hallmarks of cancer, anti-angiogenic therapies are one of the most promising strategies for cancer treatment. Intratumoral microvessel density (MVD) is an independent prognostic indicator; thus, increased MVD correlates with shorter survival.11–13 However, other investigators have failed to confirm these findings. 14 Another important proliferation marker is Ki-67 antigen. Ki-67 (MKI67, KIA, MIB-1), a nuclear protein with a key role during cell division, can be detected during cell cycle active phases (G1, S, G2 and M) but not during the Gap0 phase. Although Ki-67 measurement has been widely used as a cell proliferation marker, few studies have considered the prognostic role of this protein, many of which report conflicting results.15–19
In view of the dearth of prospective studies of biomarkers that might supply short-term prognostic information, this study was designed to evaluate the effects of angiogenesis (assessed by CD-105), apoptosis (by the terminal deoxynucleotidyl transferase (TdT) dUTP Nick-End Labeling (TUNEL) method) and proliferation (by the Ki-67 antibody) on short-term prognosis and the relations between these and diverse clinical–pathological variables in a large prospective cohort of CRC patients.
Materials and methods
Study design
A prospective, multicentre observational study was conducted, with the participation of seven Spanish national public health system hospitals from two different Autonomous Communities (Andalusia and the Basque Country). The following hospitals took part in the study: Antequera Hospital, Costa del Sol Hospital, University Hospital of Valme, University Hospital Virgen del Rocío, University Hospital Virgen de las Nieves, Galdakao-Usansolo Hospital and Basurto University Hospital.
Inclusion criteria for participation in this study were as follows: Patients diagnosed with new or incidental colon or rectum cancer, recruited during 2010–2012, stages I–IV and surgically treated either as an urgent case or according to schedule.
Data collection
Tissue specimens were obtained from curative surgery and processed by the corresponding Pathological Anatomy Unit. Prior to immunohistochemistry analyses, resected samples were formalin fixed for 48 h (formalin–tissue volume ratio 10:1), dehydrated with ethanol and embedded into paraffin blocks using an automated tissue processor.
A total of 551 tumour samples were analysed to detect markers of tumour biology (percentage of apoptotic cells by the TUNEL technique, Ki-67 antigen as a marker of cell proliferation and MVD as a marker of angiogenesis).
Data collection methods
In order to ensure consistency among centres and reviewers, clinical data from medical records and database were directly collected by qualified reviewers, following a data collection form and an instruction manual. Each patient was included in the study prospectively and consecutively from each of the participating hospitals. CRC diagnosis was based on anatomopathological findings after a biopsy by colonoscopy.
Inclusion criteria
Patients diagnosed with cancer of the colon (>15 cm above the anal margin) or of the rectum (≤15 cm above the anal margin), who underwent initial curative and/or palliative surgery for this condition.
Exclusion criteria
Patients diagnosed with colon or rectal cancer in situ. Patients not operable. Patients who had received prior radiotherapy or chemotherapy were excluded from the survival analysis.
Study variables
Explanatory (independent) variables
The following sociodemographic factors associated with the patient were evaluated: sex, age, family history and type of CRC symptom observed.
Clinical–pathological factors of the tumour
Location of the tumour (rectum or colon); degree of histological differentiation; tumour (T) or lymph node (N) staging, using the tumour–node–metastasis (TNM) clinical staging system; lymphovascular and perineural invasion; the presence of metastasis and state of the serum tumour markers CEA and CA19-9.
Characteristics of the initial treatment
Type of treatment applied is surgery, chemotherapy, radiotherapy, biological treatment or support treatment.
The main
The
Immunohistochemistry
Levels of Ki-67 and CD105 were determined by the streptavidin–biotin–peroxidase enzyme assay. In order to measure Ki-67, we employed MIB1 monoclonal antibody, which allows the detection of Ki-67 nuclear antigen in the formalin-fixed paraffin-embedded tissue sections and whose reactivity is not affected by any delay in fixation. Ki-67 expression was determined as the percentage of tumour cells positively stained with MIB1in each sample, being the nuclear staining the most common criterion of positivity. The median value was used to distinguish between high and low Ki-67 expression.
The results of each case were evaluated independently (blind study with anonymized samples) by two pathologists in a semi-quantitative analysis in which the intensity and percentage of staining were determined. Cases that produced disagreement were re-evaluated by a third pathologist.
Detection of cellular apoptosis using the TUNEL method
To detect apoptotic cells, the 3′-ends of the DNA fragments generated by apoptosis-associated endonucleases were labelled in situ using a commercial apoptosis detection kit (Roche Diagnostics GmbH, Mannheim, Germany). The sections were pre-treated with DNase as a positive control, and the absence of the enzyme was taken as a negative control. Cells are defined as apoptotic if the nuclear area is completely stained. Apoptotic bodies are defined as small globular bodies that are positively marked in the cytoplasm of the tumour cells, whether isolated or in groups; 1000 cells were counted for each sample. The cut-off point used was the median apoptosis value in each tumour series.
MVD (angiogenesis)
Endothelial cells were stained with CD105 to evaluate the density of intratumour microvessels. For each case, a histologic section was immunostained and the microvessels were counted by two investigators (who were blinded to diagnosis and to the immunohistochemical markers). Microvessels were only counted in intratumour ‘hotspots’ and were identified using high magnification (400×) for precise quantification. The median value observed was used to distinguish between high and low densities of intratumour microvessels.
Statistical analysis
A descriptive analysis was performed, using measures of central tendency, dispersion and position for the quantitative variables. A bivariate analysis was then performed to evaluate the differences in distribution of the independent variables with respect to dichotomized tumour markers according to the median values obtained. Student’s t test was used for the quantitative variables and the chi-square test for the qualitative ones. Kaplan–Meier survival analysis was performed to evaluate overall survival according to tumour markers, assessing mean survival rates with a 95% confidence interval (CI). Finally, Cox regression analysis was performed to evaluate markers previously found to be significant in the Kaplan–Meier analysis, adjusting for tumour staging, with the hazard ratio being described by the corresponding 95% CI. In each of these analyses, the level of statistical significance was p < 0.05. All analyses were performed with SPSS v15 statistical software.
Ethics
Patients were informed of the study objectives, invited to voluntarily participate and were included in the study sequentially. All of them signed a written informed consent to participate in the study. The Institutional Review Boards of the participating hospitals approved this project, in particular the Regional Committee of Clinical Trials of Andalucía (Sevilla, Spain)
Results
A total of 551 samples were evaluated. Valid samples for each of the studied markers were 457 for TUNEL, 487 for CD105 and 541 for Ki-67.
There were no differences in the two biological parameters – Ki-67 and the AI – concerning colon or rectal location, age, gender or past history of colorectal or other neoplasias (Table 1). However, CD105 was observed more frequently in older patients and in the colon, although in the last case without reaching statistical significance (p = 0.03 and p = 0.06, respectively). In addition, contrary to Ki-67 and CD105, increased apoptosis was significantly related to high-grade carcinomas and vascular invasion (p = 0.001 in both cases) and was related to the presence of lymph node metastasis, although the relation was not statistically significant (p = 0.06). Figure 1 shows three examples of tumours presenting positive expression for all the biological markers studied.
Distribution of sociodemographic and clinical characteristics according to the biological marker.

SD: standard deviation; N: number of valid observations; TUNEL: terminal deoxynucleotidyl transferase (TdT) dUTP Nick-End Labeling.
Valid/missing cases: 474/77 (TUNEL), 487/64 (CD105) and 541/10 (Ki-67).
Cut-off values: 1.4 (TUNEL), 10.66 (CD105) and 29 (Ki-67).
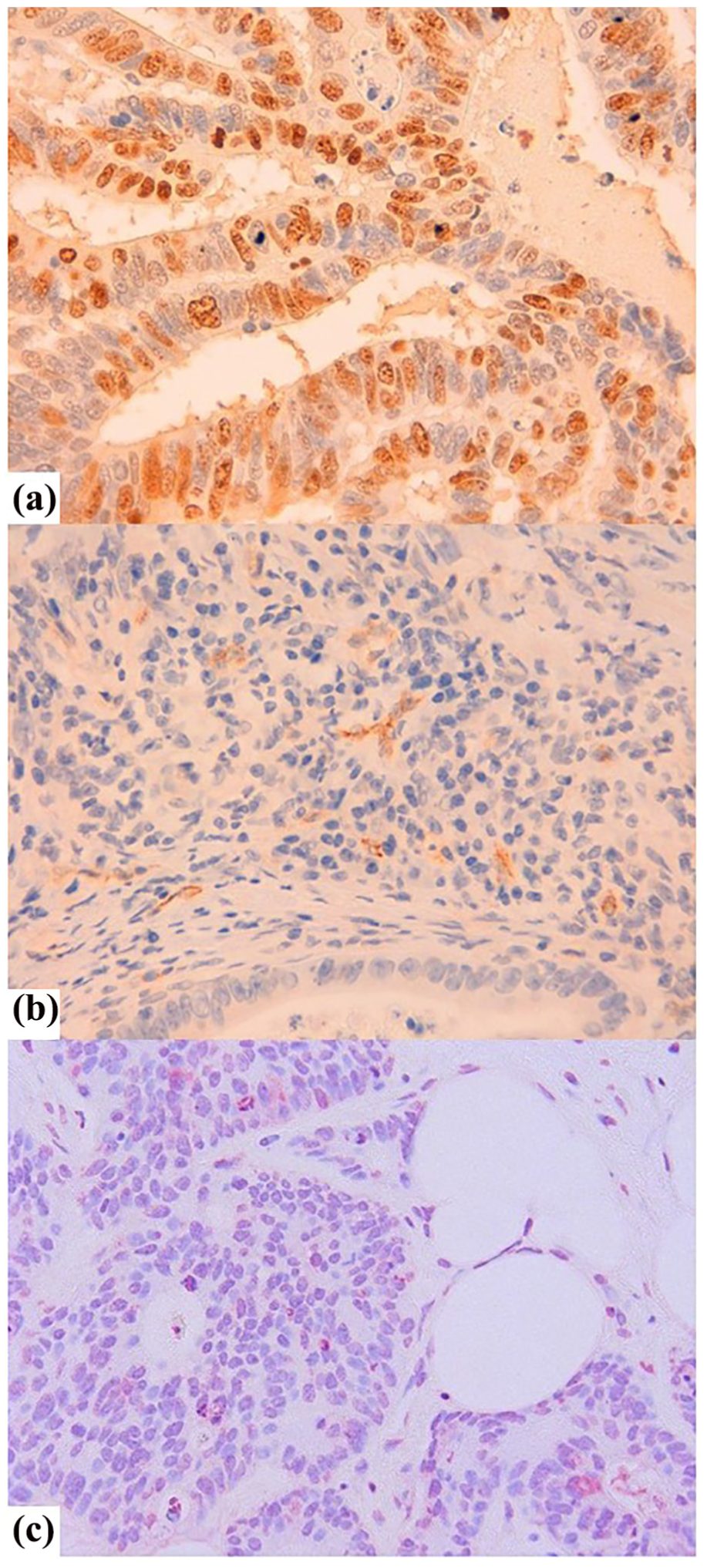
Three examples of tumours presenting positive expression of the biological markers used in this study. (a) Numerous nuclei stained for Ki-67 (400×). (b) Intraneoplastic vascular proliferation. Staining with CD105 (200×). (c) Fragmented nuclei positive for the TUNEL technique are observed in a poorly differentiated carcinoma (200×).
With respect to the clinical serum tumour marker CEA, no relationship was found. However, Ki-67-positive tumours were less frequent with increased levels of CA 19-9 (p = 0.09), while Ki-67 was less frequent when neural invasion took place (p = 0.056) although in both cases without reaching statistical significance (Table 1).
To our knowledge, this study is the first to analyse the possible association between the presence of symptoms and alterations in proliferation, apoptosis or angiogenesis. In fact, no differences were found in this respect between asymptomatic and symptomatic tumours (Table 1). Neither was any relationship observed when each symptom was considered separately (data not shown). Furthermore, no clear relationship was observed between the different biological factors investigated, except that more new vessels were formed in tumours with a high expression of Ki-67 (p = 0.09).
Table 2 shows survival segmented by biological marker and by tumour staging. Overall survival at 2 years of follow-up was significantly decreased in patients with a low number of Ki-67-positive cells (Figure 2). This prognostic information persisted in the multivariate analysis (Table 3). However, there were no differences in survival concerning the percentages of CD105 and TUNEL positive cells.
Survival segmented by levels of markers and tumour staging.

CI: confidence interval; N: number of valid observations; TUNEL: terminal deoxynucleotidyl transferase (TdT) dUTP Nick-End Labeling.
Total valid cases: 402 (TUNEL), 396 (CD105) and 444 (Ki-67).
Cut-off values: 1.4 (TUNEL), 10.66 (CD105) and 29 (Ki-67).
Bold values represent p < 0.05.

Survival of patients grouped according to Ki-67 expression. Survival of patients with primary tumours lacking Ki-67 expression (thick line) is significantly shorter than that of patients with tumours presenting Ki-67 expression (thin line).
Survival assessed through the multivariate Cox regression model.

CI: confidence interval.
n evaluated = 432.
Bold values represent p < 0.05.
We also considered the question of prognostic information from these three markers in patients treated with chemotherapy after surgery. However, no such information was obtained (Figure 3).

Survival of patients undergoing chemotherapy after surgical resection grouped according to Ki-67 expression. No differences were found between Ki-67-positive tumours (thick line) and those negatives (thin line).
Discussion
This article considers the biological aspects of CRC, seeking to establish novel markers providing short-term prognostic information. To do so, we analyse the fundamental processes of proliferation, apoptosis and angiogenesis in the evolution of tumours in a prospective cohort of CRC patients. 20
Defects in the response to apoptosis are implicated not only in pathological aspects of CRC but also in its resistance to ionizing radiation and chemotherapeutic drugs. It has been demonstrated in a previous retrospective series with long follow-up that apoptotic cells, evaluated by the TUNEL assay, are more frequently present in samples from CRC patients with a worse prognosis than in those with a good one. 8 Other studies have also reported this inverse relationship between AI and survival.5–7 The varying or conflicting results reported by such studies, as in the case of the prognostic significance of cleaved caspase-3,21–23 has led to the investigation of other markers to clarify the relationship between tumoral cell apoptosis and patients’ survival.21–23 In this regard, it is worth to highlight the inverse relationship reported between AI and survival, when apoptosis is measured by the M30 antibody,24,25 as AI may reveal a physiological effort to eliminate neoplastic cells with genetic alterations such as DNA mutations or mitotic errors. In other words, if mutated neoplastic cells that can inhibit programmed cell death are the only ones to survive and continue proliferating, then apoptosis would allow the survival of the most aggressive cancer cells for tumour formation by selection. However, it is interesting to examine whether this prognostic information is valid for the first 2 years of follow-up, which is when the majority of events occur. In our series, although tumours with elevated AI were more frequent in patients with a short survival, the association did not reach statistical significance (p = 0.1). Therefore, apoptosis does not represent a clear marker with prognostic information, at least in short-term follow-up.
Concerning measurement methods, although apoptosis can be detected by other methods, TUNEL is the approach most commonly adopted and is considered the reference standard. 26 Moreover, it also presents a very good correlation with the M30 antibody. 27
Although a previous study by Kim et al. 28 in rectal cancer has found that apoptosis is associated with lymphatic invasion, other studies show opposite results. 29 In this regard, even though we did not reach statistical significance (p = 0.06) in our prospective series, we found a statistically significant association between apoptosis and poorly differentiated tumours and tumours with vascular invasion, which is in line with a previous study evidencing that apoptosis is greater in advanced than in earlier stages. 8
In our series, proliferation, measured by the Ki-67 antigen, was independently related to survival. In this respect, Anjomshoaa et al. 30 also reported an inverse association between colon cancer progression and proliferative tumour activity. Moreover, Adams et al. 17 found Ki-67 to be predictive of improved survival during the first 18 months following diagnosis, while Salminen et al. 31 observed that proliferative activity in rectal cancer, as measured by Ki-67, was associated with increased survival compared to patients with low Ki-67. Similar results were obtained recently by Melling et al. 19 It is very important to understand the mechanisms underlying this inverse relationship, as proliferation could play a more significant role at the beginning than at the end of the neoplastic process.
Although Fodor et al. 18 reported that Ki-67 proliferation index was not a useful prognostic factor for CRC patients, they also highlighted the need to continue with a further evaluation of this marker as a predictive factor for the additional benefits derived from adjuvant chemotherapy. In this respect, however, our own results do not show that the expression of Ki-67 is related to survival in patients with colorectal carcinomas who have undergone chemotherapy. Finally, Peng et al. 15 in a study of stage II tumours found an association between Ki-67 expression and a worse prognosis. Despite these two studies with different results to ours, the observations made in this work will make a significant contribution to the knowledge on this topic.
Our findings also show that an abnormal result for the CA 19-9 serum marker tends to be associated with lower Ki-67 although without reaching statistical significance (p = 0.09), which might indicate that in more advanced stages of the disease, with a lower proportion of cells positive for Ki-67, there would be a greater presence of the clinical marker CA 19-9.
Our series revealed a positive association between the neoformation of vessels with Ki-67 expression (i.e. the greater the proliferation, the greater the formation of vessels) although without reaching statistical significance (p = 0.09). A similar relationship was found by Zlobec et al. 13
Several studies show contradictory results about the role of angiogenesis in colorectal tumours. Thus, although Bossi et al. 32 and Pietra et al. 33 reported that an increased vascularization was not a valuable prognostic factor for CRC patients, in their study with factor VIII, Saclarides et al. 34 and Frank et al., 35 reported a significant correlation between angiogenesis, recurrence, metastasis and survival. Moreover, Zlobec et al. 13 and Li et al. 36 reported that CD105 was a marker of disease progression. However, our study did not find angiogenesis to be a prognostic indicator in short-term follow-up. Nevertheless, it could be a crucial factor in late phases of the disease, with the progression to a metastatic state. 37
In our study, we targeted cancer vasculature via CD105 instead of other commonly used pan-endothelial markers such as CD31, CD34 or Factor VII as the endoglin antibody particularly binds to the activated endothelial cells implicated in tumour angiogenesis but not to the vascular endothelium in normal tissues. 38
This study demonstrates a very limited value for TUNEL assay and CD-105 as short survival biomarkers for CRC. A wide range of other markers that influence these phenomena in the human body should also be assessed in CRC. In fact, there are numerous factors that can influence these parameters (e.g. cell cycle phase, circadian rhythms and cell types unrelated to the tissue of interest) which have not been considered in this study.
In conclusion, our study demonstrates that Ki-67 labelling is an independent prognostic parameter for survival at short-term follow-up. Nevertheless, further pre-clinical and clinical investigation is required.
Footnotes
Appendix 1
The REDISSEC CARESS-CCR (Results and Health Services Research in Colorectal Cancer) group
Jose María Quintana Lopez1, Marisa Baré Mañas2, Maximino Redondo Bautista3, Eduardo Briones Pérez de la Blanca4, Nerea Fernández de Larrea Baz5, Cristina Sarasqueta Eizaguirre6, Antonio Escobar Martínez7, Francisco Rivas Ruiz8, Maria M. Morales-Suárez-Varela9, Juan Antonio Blasco Amaro10, Isabel del Cura González11, Inmaculada Arostegui Madariaga12, Amaia Bilbao González7, Nerea González Hernández1, Susana García-Gutiérrez1, Iratxe Lafuente Guerrero1, Urko Aguirre Larracoechea1, Miren Orive Calzada1, Josune Martin Corral1, Ane Antón-Ladislao1, Núria Torá13, Marina Pont13, María Purificación Martínez del Prado14, Alberto Loizate Totorikaguena15, Ignacio Zabalza Estévez16, José Errasti Alustiza17, Antonio Z Gimeno García18, Santiago Lázaro Aramburu19, Mercé Comas Serrano20, Jose María Enríquez Navascues21, Carlos Placer Galán21, Amaia Perales22, Iñaki Urkidi Valmaña23, Jose María Erro Azkárate24, Enrique Cormenzana Lizarribar25, Adelaida Lacasta Muñoa26, Pep Piera Pibernat26, Elena Campano Cuevas27, Ana Isabel Sotelo Gómez28, Segundo Gómez-Abril29, F. Medina-Cano30, Julia Alcaide31, Arturo Del Rey-Moreno32, Manuel Jesús Alcántara33, Rafael Campo34, Alex Casalots35, Carles Pericay36, Maria José Gil37, Miquel Pera37, Pablo Collera38, Josep Alfons Espinàs39, Mercedes Martínez40, Mireia Espallargues41, Caridad Almazán42, Paula Dujovne Lindenbaum43, José María Fernández-Cebrián43, Rocío Anula Fernández44, Julio Ángel Mayol Martínez44, Ramón Cantero Cid45, Héctor Guadalajara Labajo46, María Heras Garceau46, Damián García Olmo46, Mariel Morey Montalvo47, Javier Mar48.
1Unidad de Investigación. Hospital Galdakao-Usansolo, Galdakao-Bizkaia/Red de Investigación en Servicios de Salud en Enfermedades Crónicas–REDISSEC.
2Epidemiologia Clínica y Cribado de Cáncer, Corporació Sanitaria Parc Taulí, Sabadell/Red de Investigación en Servicios de Salud en Enfermedades Crónicas–REDISSEC.
3Servicio de Laboratorio. Hospital Costa del Sol, Málaga/REDISSEC.
4Unidad de Epidemiología. Distrito Sevilla, Servicio Andaluz de Salud.
5Centro Nacional de Epidemiología, Instituto de Salud Carlos III, Madrid/REDISSEC.
6Unidad de Investigación. Hospital Universitario Donostia/Instituto de Investigación Sanitaria Biodonostia, Donostia -REDISSEC.
7Unidad de Investigación. Hospital Universitario Basurto, Bilbao/REDISSEC.
8Servicio de Epidemiología. Hospital Costa del Sol, Málaga -REDISSEC.
9Department of Preventive Medicine and Public Health, University of Valencia/CIBER de Epidemiología y Salud Pública (CIBERESP)/CSISP-FISABIO, Valencia.
10Unidad de Evaluación de Tecnologías Sanitarias, Agencia Laín Entralgo, Madrid.
11Unidad Apoyo a Docencia-Investigación. Dirección Técnica Docencia e Investigación. Gerencia Adjunta Planificación. Gerencia de Atención Primaria de la Consejería de Sanidad de la Comunidad Autónoma de Madrid.
12Departamento de Matemática Aplicada, Estadística e Investigación Operativa, UPV–REDISSEC.
13Epidemiologia Clínica y Cribado de Cancer, Corporació Sanitaria Parc Taulí, Sabadell/REDISSEC.
14Servicio de Oncología. Hospital Universitario Basurto, Bilbao.
15Servicio de Cirugía General. Hospital Universitario Basurto, Bilbao.
16Servicio de Anatomía Patológica. Hospital Galdakao-Usansolo, Galdakao.
17Servicio de Cirugía General. Hospital Universitario Araba, Vitoria-Gasteiz.
18Servicio de Gastroenterología. Hospital Universitario de Canarias, La Laguna.
19Servicio de Cirugía General. Hospital Galdakao-Usansolo, Galdakao.
20IMAS-Hospital del Mar, Barcelona.
21Servicio de Cirugía General y Digestiva. Hospital Universitario Donostia.
22Instituto de Investigación Sanitaria Biodonostia, Donostia.
23Servicio de Cirugía General y Digestiva. Hospital de Mendaro.
24Servicio de Cirugía General y Digestiva. Hospital de Zumarraga.
25Servicio de Cirugía General y Digestiva. Hospital del Bidasoa.
26Servicio de Oncología Médica. Hospital Universitario Donostia.
27Instituto de Biomedicina de Sevilla. Hospital Universitario Virgen del Rocío, Sevilla.
28Servicio de Cirugía. Hospital Universitario Virgen de Valme, Sevilla.
29Servicio de Cirugía General y Aparato Digestivo. Hospital Dr. Pesset, Valencia.
30Servicio de Cirugía General y Aparato Digestivo. Agencia Sanitaria Costa del Sol, Marbella.
31Servicio de Oncología M_edica. Agencia Sanitaria Costa del Sol, Marbella.
32Servicio de Cirugía. Hospital de Antequera.
33Coloproctology Unit, General and Digestive Surgery Service, Corporació Sanitaria Parc Taulí, Sabadell.
34Digestive Diseases Department, Corporació Sanitaria Parc Taulí, Sabadell.
35Pathology Service, Corporació Sanitaria Parc Taulí, Sabadell.
36Medical Oncology Department, Corporació Sanitaria Parc Taulí, Sabadell/REDISSEC.
37General and Digestive Surgery Service, Parc de Salut Mar, Barcelona.
38General and Digestive Surgery Service, Althaia- Xarxa Assistencial Universitaria, Manresa.
39Catalonian Cancer Strategy Unit, Department of Health, Institut Catalá d’Oncología.
40Medical Oncology Department, Institut Catal_a d’Oncología.
41Agency for Health Quality and Assessment of Catalonia eAquAS- and REDISSEC.
42Agency for Health Quality and Assessment of Catalonia eAQuAS-, CIBER de Epidemiología y Salud Pública eCIBERESP.
43Servicio de Cirugía General y del Aparato Digestivo, Hospital Universitario Fundación Alcorcón, Madrid.
44Servicio de Cirugía General y Aparato Digestivo, Hospital Universitario Clínico San Carlos, Madrid.
45Servicio Cirugía General y del Aparato Digestivo, Hospital Universitario Infanta Sofía, San Sebastián de los Reyes, Madrid.
46Servicio de Cirugía General y del Aparato Digestivo, Hospital Universitario La Paz, Madrid.
47REDISSEC. Unidad de Apoyo a la Investigaci_on, Gerencia Asistencial de Atención Primaria de la Comunidad de Madrid, Madrid.
48REDISSEC. Unidad de Gesti_on Sanitaria. Hospital del Alto Deba. Mondragon-Arrasate. Spain.
Acknowledgements
We thank Maria del Carmen Delgado for her excellent technical assistance. M.R. and J.M.Q. contributed to experimental design and conception. M.R., C.A., T.T., M.G.A. and F.R.R. contributed to writing of the manuscript. C.A., T.T., M.M.M.S.-V., A.R., A.E. and J.M.Q. contributed to data collection. R.C.M.G., I.Z., M.S.C., J.J.B.M. and R.G.M. contributed to histopathological evaluation. C.A. and T.P. contributed to evaluation of microvessel density. R.F. and A.M.B. contributed to evaluation of cellular apoptosis. C.A., I.R. and A.M.B. contributed to evaluation of proliferation. F.R.R. contributed to statistical analysis. M.R. supervised the study and revised the manuscript. All authors read and approved the final manuscript. The members in the REDISSEC CARESS-CCR (Results and Health Services Research in Colorectal Cancer) group are listed in ![]() .
.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by grants from REDISSEC (RD12/0001/0010 and RD16/0001/0006), Instituto de Salud Carlos III (PI 09/00910, PI13/00013, PI 18/01181) and the European Regional Development Fund.
Informed consent
Informed consent was obtained from all patients and the study was approved by the ethics committee at our institution.
