Abstract
YM155, a small molecule inhibitor of survivin, has been studied in many tumors. It has been shown that YM155 inhibited oral squamous cell carcinoma through promoting apoptosis and autophagy and inhibiting proliferation. It was found that YM155 also inhibited the oral squamous cell carcinoma–mediated angiogenesis through the inactivation of the mammalian target of rapamycin pathway. Rapamycin, a mammalian target of rapamycin inhibitor, played an important role in the proliferation and angiogenesis of oral squamous cell carcinoma cell lines. In our study, cell proliferation assay, transwell assay, tube formation assay, and western blot assay were used to investigate the synergistic effect of rapamycin on YM155 in oral squamous cell carcinoma. Either in vitro or in vivo, rapamycin and YM155 exerted a synergistic effect on the inhibition of survivin and vascular endothelial growth factor through mammalian target of rapamycin pathway. Overall, our results revealed that low-dose rapamycin strongly promoted the sensitivity of oral squamous cell carcinoma cell lines to YM155.
Introduction
Oral squamous cell carcinoma (OSCC) is the most common malignancy in the oral and maxillofacial region. 1 Traditional chemotherapy drugs (such as methotrexate, 5-fluorouracil, and cisplatin) have strong side effects including bone marrow depression and alimentary canal reaction. Drug resistance may occur when those drugs are used frequently.2–4
Survivin, a member of inhibitor of apoptosis protein (IAP), is overexpressed in most human cancers. Survivin is associated with the progression, metastasis, and angiogenesis of tumors.5,6 Therefore, it has been considered as a new target for cancer therapy.7,8 YM155, also named as sepantronium bromide, is an imidazolium derivative which can suppress the transcription of survivin. 9 Moreover, it has been shown that YM155 inhibited the proliferation of many cancers.10–12 In OSCC, YM155 can suppress its growth by promoting apoptosis and autophagy and inhibiting proliferation.13,14
Rapamycin, an immunosuppressive agent, was used widely to avoid immunological rejection in the organ transplantation.15,16 Recently, it was revealed that rapamycin could suppress tumor growth through anti-angiogenesis and promote cell death by inhibiting the mammalian target of rapamycin (mTOR) pathway.17–19 However, it cannot inhibit cell proliferation ability in OSCC cell lines in vitro.20,21
Our aim is to investigate the effect of low-dose rapamycin on the sensitivity of OSCC cell lines to YM155. The results of cell proliferation assay, transwell assay, tube formation assay, and western blot assay in OSCC showed that rapamycin promoted the anti-proliferation ability and enhanced the anti-angiogenesis ability of YM155. Either in OSCC cell lines or in nude mice models, rapamycin and YM155 exerted a synergistic effect on the inhibition of survivin and vascular endothelial growth factor (VEGF) through mTOR pathway.
Materials and methods
Cell culture and conditioned medium collection
The OSCC cell lines (including SCC9, SCC15, SCC25, and CAL27; American Type Culture Collection (ATCC), USA) were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; Gibco, Shanghai, China) containing 10% fetal bovine serum (FBS; Gibco, Shanghai, China), 100 U/mL penicillin (Gibco, Shanghai, China), and 100 µg/mL streptomycin (Gibco, Shanghai, China). The human umbilical vein endothelial cells (HUVECs; ScienCell, USA) were maintained in Endothelial Cell Medium (ECM; Gibco, Shanghai, China) with 5% FBS, 1% endothelial cell growth supplement (ECGS; Gibco, Shanghai, China), 100 U/mL penicillin, and 100 µg/mL streptomycin. Cells were cultured in an incubator with an atmosphere of 5% CO2 at 37°C. The OSCC conditioned media were collected as described previously. 22
Cell proliferation test
The cell proliferation ability was examined by Cell Counting Kit-8 assay. First, the OSCC cells were seeded on a 96-well microplate (Corning Life Sciences, USA) with a final volume of 200 µL. After 12 h, different concentrations of rapamycin (Selleck Chemicals, USA) and YM155 (Selleck Chemicals) were added and cells were incubated for another 72 h. Then, Cell Counting Kit-8 (CCK-8) was added and cells were incubated in the incubator for 40 min. The optical density (OD) was measured by Elx800 Reader (BioTek Instruments, Inc., USA) at 450 nm. The combination index was at last calculated by Chou–Talalay Formula. 23 Subsequently, SCC25, CAL27, and HUVECs were seeded on a 96-well plate. Different concentrations of YM155 and 100 nM rapamycin were added to the SCC25 and CAL27, while different conditioned media were added to HUVECs. The cells were then incubated for several hours and the OD per well was measured as before.
VEGF measurement
After being exposed to different concentrations of YM155 and rapamycin for 12 h, the cells were washed twice and incubated with serum-deprived DMEM for another 24 h. Finally, the release of VEGF in the supernatants was measured by enzyme-linked immunosorbent assay (ELISA; R&D Systems, USA), according to the manufacturer’s instructions.
Wound-healing assay
HUVECs were seeded on six-well plates (Corning Life Sciences) and grown to approximately 100% confluence. Scratches were generated with a sterile 200 µL pipette tip and then different conditioned media were added. Cell migration toward the wounds was monitored at 0 and 12 h with a phase-contrast microscope (Olympus, Tokyo, Japan).
Transwell assay
The migration ability of HUVECs was also examined with Transwell Boyden chamber (Corning Life Sciences). HUVECs in 200 µL of serum-deprived DMEM were seeded on the upper chamber, while 800 µL conditioned media were filled in the lower chamber as a chemo induction. After 12 h of incubation, the cells stayed in the upper surface of the membrane were wiped off with a cotton swab. The migrated cells were fixed with 4% methanol, stained with 0.1% crystal violet, washed three times, and examined under a bright-field microscope (Olympus, Tokyo, Japan).
Tube formation assay
Growth factor–reduced Matrigel (BD Biosciences, USA) was melted overnight at 4°C and then diluted to 10 mg/mL with cold serum-deprived DMEM. The 96-well plates were coated with 40 µL of Matrigel per well and incubated for 1 h, and then, the HUVECs in different conditioned media were seeded on the Matrigel and incubated for 24 h. The capillary-like structures were then photographed and the length of the tube was analyzed by Image-Pro Plus Software (Media Cybernetics, USA).
Chick chorioallantoic membrane assay
As previous studies described, 24 a sterile rubber ring was placed on the chorioallantoic membrane of a 10-day-old embryo, then approximately 50 µL of conditioned medium was added in the ring cycle. After incubation for another 3 days, the area in the rubber ring cycle was photographed. In total, 8–10 eggs were used for each group, and two observers were invited to count the newly formed vessels.
Matrigel plug assay
The angiogenesis ability in vivo was examined by Matrigel plug assay as described previously. 25 A volume of 0.8 mL of Matrigel was mixed with 0.2 mL of cooled conditioned medium on the ice and injected into the back of the nude mice. A week later, the skin of nude mice was opened to completely expose Matrigel plug. The hemoglobin level was measured by Drabkin reagent kit 525 (Sigma-Aldrich, USA), and Matrigel plugs were photographed.
Nude mice xenografts
In total, 6–8 weeks male BALB/c nude mice were purchased from the Experimental Animal Center in Guangdong. Guidelines for the Use and Care of Small Laboratory Animals were followed strictly and the studies were approved by the Ethics Committee of Peking University Shenzhen Hospital. CAL27 cells were injected subcutaneously into the flank. Two weeks later, the nude mice were randomly divided into three groups (n = 7/group) and treated with different drugs. YM155 (5 mg/kg) was injected intraperitoneally constantly per day in the single drug group. A volume of 5 mg/kg of YM155 or 1 mg/kg of rapamycin was injected every other day alternatively in the drug combination group, while 200 µL of 0.9% saline was injected in the control group. The size of the tumors and the weight of the mice were measured every other day. Two weeks later, the tumors were carefully removed from the nude mice and photographed and then placed into radioimmunoprecipitation assay (RIPA) buffer with protease and phosphatase inhibitors for western blot analysis, or embedded in paraffin for immunohistochemical analysis.
Western blot analysis
Total protein of the cell lines or xenograft tissues was extracted, separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), and transferred to polyvinylidene difluoride membranes. The membranes were incubated with the primary antibodies at 4°C overnight followed by horseradish peroxidase–conjugated secondary antibody at room temperature for another 1 h. The expression of related proteins was assessed with enhanced chemiluminescence (ECL) reagents (Pierce, MA, USA), and densitometry of the protein bands were quantified by Image J software (National Institutes of Health, Maryland, USA). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal control. Primary antibodies for GAPDH, phospho-mTOR (Ser-2448), mTOR, phospho-S6 (Ser-235/236), S6, VEGF, and survivin were purchased from Cell Signaling Technology (USA).
Immunohistochemical assay
The sections of embedded xenograft tissues were incubated in primary antibodies overnight at 4°C. After washing with phosphate-buffered saline (PBS), the sections were then incubated with horseradish peroxidase–conjugated secondary antibodies in the room temperature for 20 min. Then, the antibody-binding rate was detected by diaminobenzidine substrate kit. Photographs were taken by Leica 4000 microscope (Leica, Heidelberg, Germany) and analyzed with Image-Pro Plus Software (National Institutes of Health, Maryland, USA). Primary antibodies for CD31, CD34, and Ki67 were purchased from Santa Cruz Biotechnology (USA).
Statistical analyses
All experiments were carried out in triplicates, and the results were expressed as mean ± standard deviation (SD). Statistical significances between groups were determined by two-tailed Student’s t test. One-way analysis of variance (ANOVA) and Student–Newman–Keul post hoc comparisons were used to analyze multiple comparisons.
Results
YM155 inhibits the proliferation and angiogenesis ability of OSCC cell lines
The proliferation ability of four OSCC cell lines (including SCC9, SCC15, SCC25, and CAL27) were examined by CCK-8 assay after treating with YM155. As shown in Figure 1, the proliferation of all cell lines was inhibited in a dose-dependent manner, and the half maximal inhibitory concentration (IC50) of these cell lines are shown in Table 1. After treating with different concentrations of YM155 for 12 h, SCC25 and CAL27 cells showed no significant changes in the proliferation ability (Figure S1, p > 0.05). It indicated a nontoxic condition for conditioned medium collection. Angiogenesis played an important role in the development of the tumors. 26 Endothelial cell line HUVEC was used to examine the angiogenesis ability induced by tumor cells. The treatment with different conditioned media had no effect on the proliferation of HUVECs (Figure S2, p > 0.05).

The inhibitory effect of rapamycin and YM155 on the proliferation ability of OSCC cell lines. (a) The cell growth curve of the four OSCC cell lines (SCC9, SCC15, SCC25, and CAL27) treated with varied rapamycin doses. (b) The cell growth curve of SCC9 cell lines treated with different doses of YM155 and rapamycin. (c) The cell growth curve of SCC15 cell lines treated with different doses of YM155 and rapamycin. (d) The cell growth curve of SCC25 cell lines treated with different doses of YM155 and rapamycin. (e) The cell growth curve of CAL27 cell lines treated with different doses of YM155 and rapamycin. All data are presented as mean ± SD for three different experiments performed in duplicate (***p < 0.001; **p < 0.01).
The IC50 of YM155 in different cell lines when used alone or used with 100 nM rapamycin.

IC50: half maximal inhibitory concentration.
Because the migration of endothelial cells is necessary in the angiogenesis process, the effect of YM155 on OSCC-induced HUVEC migration was investigated. It revealed that YM155 prevented conditioned medium–induced migration ability of HUVECs (Figure 2(a) and (b), p < 0.01). The endothelial cells can form capillary-like structures spontaneously on normal Matrigel, so growth factor–reduced Matrigel was used to detect OSCC-induced tube formation of HUVECs. As shown in Figure 2(c), the tube formation of HUVECs was significantly inhibited by the conditioned medium pretreatment with YM155 (p < 0.01).
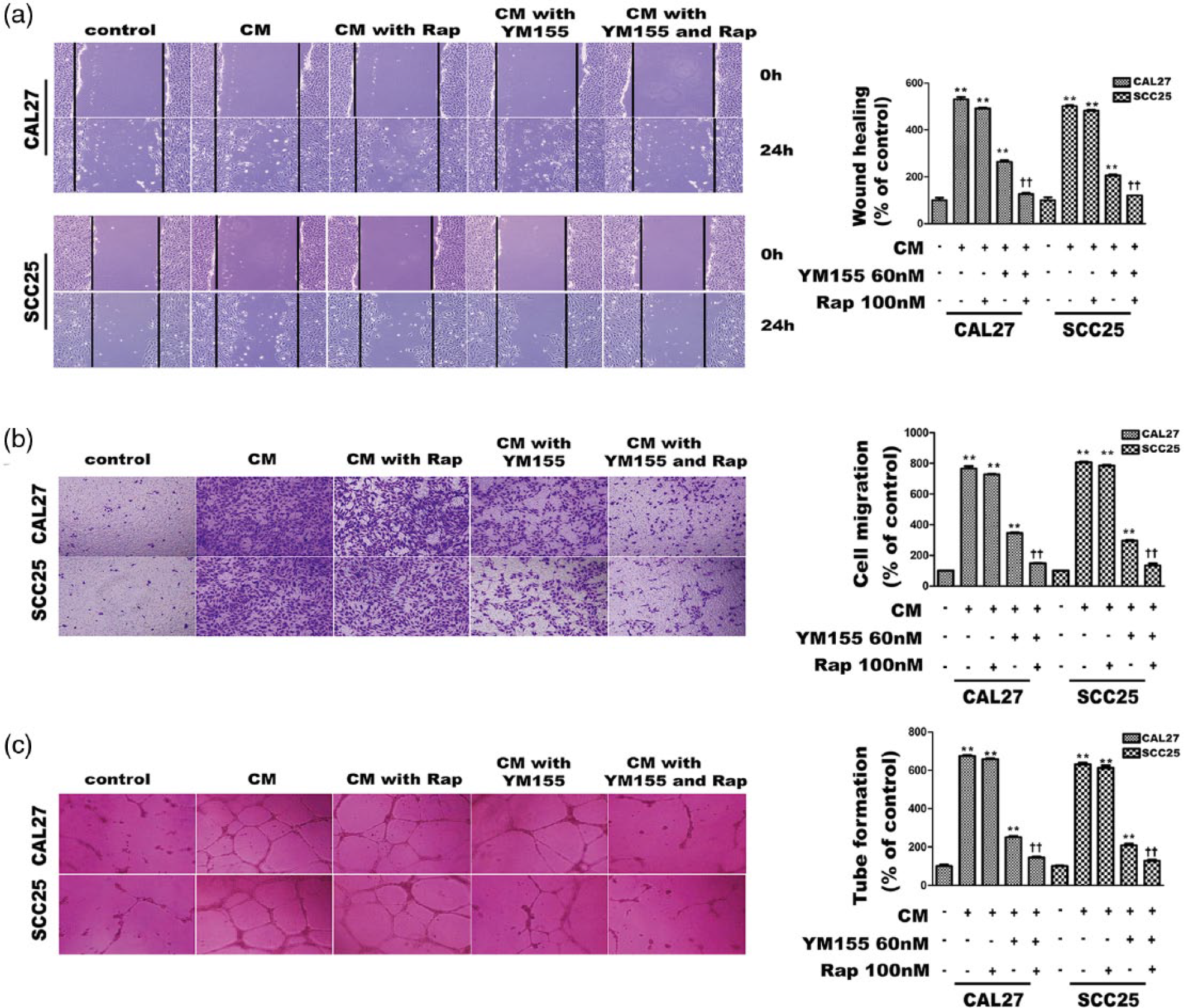
The inhibitory effect of rapamycin and YM155 on the angiogenesis ability of OSCC cell lines. (a) The wound-healing assay was performed to examine the effects of rapamycin and YM155 on the OSCC-induced angiogenesis ability of the HUVECs. (b) Transwell assay was performed to examine the effects of rapamycin and YM155 on the OSCC-induced angiogenesis ability of the HUVECs. (c) Tube formation assay was performed to examine effects of rapamycin and YM155 on the OSCC-induced angiogenesis ability of the HUVECs. In these experiments, 100 nM of rapamycin and 60 nM of YM155 were used. All data are presented as mean ± SD from three different experiments performed in duplicate (**p < 0.01 vs the control group (no drug added); ††p < 0.01 the drug combination group vs the YM155 single group).
Rapamycin enhances the anti-proliferation ability of YM155
Though Rapamycin exerted growth inhibition in preclinical and clinical trials in many cancers, it was found that rapamycin cannot inhibit the proliferation of certain cancer cell lines. 20 It has been revealed that rapamycin showed no significant effect in the proliferation of four OSCC cell lines even with high concentration (Figure 1(a)). However, low dose of rapamycin may have synergistic effects with YM155, which plays important roles in the proliferation ability of the OSCC cell lines (Figure 1(b)–(e), p < 0.01). The combination index of different dose of YM155 with 100 nM rapamycin showed strong synergistic effect (Table 2), and the IC50 of the OSCC cell lines decreased prominently after administration of both the drugs (Table 1).
The combination index of different doses of YM155 with 100 nM rapamycin.

Rapamycin promotes the inhibition of OSCC-induced angiogenesis by YM155
Rapamycin was considered as an angiogenesis inhibitor in many tumors. A series of assays were carried out to verify the pro-inhibition ability of low-dose rapamycin on YM155 in OSCC-induced angiogenesis. HUVECs were treated with conditioned media collected respectively from the SCC25 and CAL27 treated with YM155 and rapamycin. As shown in Figure 2, the migration ability and the tube formation of HUVECs were significantly decreased in the drug combination group.
The OSCC-induced angiogenesis ability in vivo was examined by the chick chorioallantoic membrane (CAM) assay and Matrigel plug assay. Rapamycin in combination with YM155 reversed the effect of OSCC conditioned medium on new microvessel formation in the the CAM assay (Figure 3(a), p < 0.01). In the Matrigel plug assay, the hemoglobin contents in the plugs were much less in the drug combination group (Figure 3(b), p < 0.01). It revealed that rapamycin promoted the anti-angiogenesis ability of YM155 in OSCC.

Effects of rapamycin and YM155 on the OSCC-induced angiogenesis ability. (a) The CAM assays as well as Matrigel plug assay were performed to examine the effects of rapamycin and YM155 on the OSCC-induced angiogenesis ability in vivo. The eggs and the nude mice were divided into five groups as before: Control group, conditioned medium without YM155 and rapamycin group, conditioned medium with YM155 single group, conditioned medium with rapamycin single group, conditioned medium with rapamycin and YM155 group. A volume of 100 nM of rapamycin and 60 nM of YM155 was used. (b) All data are presented as mean ± SD from three different experiments performed in duplicate and then analyzed by Graphpad (**p < 0.01 vs the control group (no drug added); ††p < 0.01 the drug combination group vs the YM155 single group).
Rapamycin promotes the downregulation of VEGF and survivin through mTOR signaling pathway
VEGF is one of the growth factors associated with angiogenesis, while survivin is closely associated with the proliferation of tumor cells. 27 After treating with different concentrations of YM155, the expression of survivin and VEGF in OSCC cell lines was significantly downregulated (Figure 4(a) and (b), p < 0.05). So, YM155 may prevent the OSCC cell line proliferation and angiogenesis ability via inhibiting survivin expression and VEGF production.
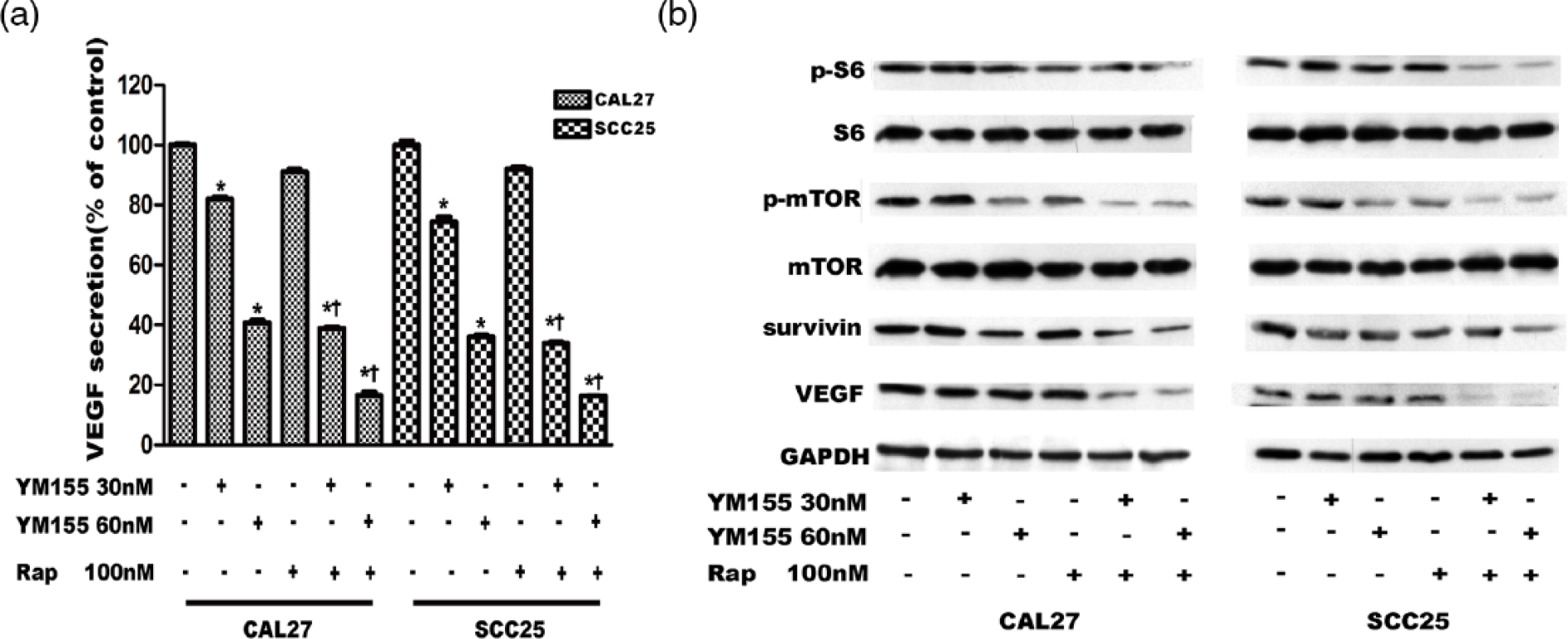
Rapamycin promotes the downregulation of VEGF and survivin of YM155 in OSCC cell lines through the inhibition of the phosphorylation of mTOR pathway. (a) The secretion level of VEGF in OSCC cell lines was examined by ELISAs after they were treated with 30 nM or 60 nM YM155 and 100 nM rapamycin for 12 h. (b) The expression levels of p-mTOR (Ser-2448) and p-S6 (Ser-235/236) and their corresponding total protein and the expression levels of survivin and VEGF were determined by western blot analysis. All data are presented as mean ± SD from three different experiments performed in duplicate (*p < 0.05 vs the control group (no drug added); †p < 0.05 the drug combination group vs the YM155 single group with the same dose of YM155).
Since mTOR pathway is closely related with the angiogenesis ability,28,29 it has been shown that YM155 significantly suppressed the phosphorylation of mTOR and S6 (Figure 4(b)). As a specific mTOR inhibitor, rapamycin was then used in combination with YM155. However, rapamycin cannot affect the expression of survivin in OSCC cell lines. But it can downregulate VEGF production. When low-dose rapamycin was used with YM155, the expression of survivin and VEGF was decreased obviously and so was the phosphorylation of mTOR and S6 (Figure 4(b)). Together, rapamycin may enhance the anti-angiogenesis and anti-proliferation ability of YM155 in OSCC through mTOR pathway.
Rapamycin and YM155 suppress tumor angiogenesis and prevent tumor growth
We also examined the synergistic effect of rapamycin and YM155 in nude mice model. As shown in Figure 5, YM155 alone can inhibit the growth of the CAL27 xenografts. However, the growth suppression was more significant in the drug combination group (p < 0.05). The mice weight curve showed no toxicity in the three groups (Figure 5(c), p > 0.05). The proliferation activity of the xenografts was examined by testing Ki67 expression, while microvessel formation was identified by testing CD34 and CD31 expression in the xenograft tumor samples (Figure 6(a) and (b)). To confirm the mechanisms of the synergistic effects of rapamycin with YM155, the expression of VEGF and survivin and activation of mTOR and S6 in xenograft tumor tissues were also evaluated. The activities of all these factors were lower in the drug combination group than single YM155 group (Figure 6(c)).

Rapamycin promotes the ability of YM155 which can prevent the tumor growth in nude mice model. (a) The xenografts of each group were removed and photographed after 2 weeks. (b) Tumor volumes of each group were calculated every other day. (c) The body weight of the mice was measured every other day and showed no side effects to the treatment. Nude mice were divided into three groups: control group (0.9% saline, 200 µL per day, n = 7), YM155 single group (5 mg/kg YM155 daily, n = 7), and combination group (5 mg/kg YM155 or 1 mg/kg rapamycin every other day, n = 7). All data are presented as mean ± SD (n = 7; *p < 0.05 the drug combination group vs the control group (no drug added); †p < 0.05 the drug combination group vs the YM155 single group with the same dose of YM155).

The immunohistochemical assay and western blot assay of the xenografts. (a) The expression of proliferation-related biomarkers, Ki67, p-S6, and survivin and the expression of angiogenesis-related biomarkers. (b) VEGF, CD31, and CD34 were examined by immunohistochemical analysis of the xenograft tissues. (c) The expression levels of VEGF, survivin, p-mTOR (Ser-2448), and its corresponding total proteins in the xenograft tissues were examined by western blot assay. All the results were examined by triple independent experiments.
Discussion
Traditional chemotherapy drugs used clinically in OSCC (such as 5-fluorouracil, cisplatin, and paclitaxel) have an effect on the proliferation ability of tumor cells. Although most of cancer cells can be killed by these drugs, the rest grow continually for the support of the surrounding blood vessels. Moreover, abnormal vessels can keep the drugs away from the tumor cells. So, tumor microenvironment must be taken into consideration in the drug therapy strategy. Instead of having effect on the proliferation of the tumor, the anti-angiogenesis drugs bind VEGF specifically and prevent it from binding the VEGF receptor. First, they can degrade the existing tumor vasculature and cut off the oxygen or other nutrients required for tumor cell growth. Second, they can normalize the tumor blood vessels and improve the delivery of chemotherapy drugs to the tumor tissue. Finally, they can inhibit angiogenesis, growth, and metastasis of tumors. Though it was showed that YM155 exhibited powerful anti-tumor effect in OSCC, the mechanism of YM155 in tumors was still obscure.13,14 According to our results, YM155 can inhibit OSCC-induced angiogenesis and prevent the proliferation of the OSCC cells.
Previous studies demonstrated that the expression of survivin was much higher in the OSCC.13,30 YM155, a potent survivin inhibitor, has been used as an anti-cancer agent in many tumors, 9 which can suppress the proliferation of the tumor cells and induce tumor cells death.31–33 We examined that YM155 significantly inhibited the proliferation ability of OSCC cell lines in a dose-dependent manner. Moreover, it demonstrated that YM155 also prevented the migration and tube formation of the HUVECs. Many signaling pathways were involved in the angiogenesis process, including Wnt/β-catenin pathways, phosphoinositide 3-kinase (PI3K)/Akt pathways, VEGF/VEGF receptor 2 (VEGFR2) pathways, and so on.34–36 Rapamycin inhibited angiogenesis through mTOR pathway in many tumors.37–39 However, a few studies revealed that rapamycin exerted no significant growth inhibition on OSCC cell lines in vitro.20,21
As a potent survivin inhibitor, YM155 can increase the tumor sensitivity to other anti-cancer agents. YM155 exhibited synergistic effect with ABT-263, an inhibitor of Bcl-2 family member, through downregulating Mcl-1 in various tumors. 40 YM155 enhanced the effect of CDDP in some tumors through the inhibition of survivin.41,42 Moreover, YM155 could reverse the rapamycin resistance of renal carcinoma cells via inhibiting survivin. 43 In our study, low-dose rapamycin (100 nM) was added with different dose of YM155 to enhance anti-angiogenesis ability of OSCC cells. It has been shown that rapamycin not only promoted the anti-angiogenesis ability but also enhanced the anti-proliferation ability of YM155. It was indicated the synergistic effect of rapamycin and YM155 can enhance the inhibition of survivin and VEGF through mTOR pathway.
In conclusion, rapamycin enhanced the anti-angiogenesis and anti-proliferation ability of YM155 in OSCC by promoting the inhibition of survivin and VEGF. The combinatory use of low-dose rapamycin and YM155 may provide a new therapeutic regimen for OSCC.
Footnotes
Acknowledgements
K.-L.L. and Y.-F.W. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Shenzhen Innovation Committee Fundamental Research (No. JCYJ20150403091443286), the Shenzhen Innovation Committee Fundamental Research (No. JCYJ20150403091443303), and the National Natural Science Foundation of China (No. 81572654).
