This is the first study to investigate the effect of vitamin D receptor (VDR) gene single-nucleotide polymorphisms on the clinicopathologic features of papillary thyroid cancer in Turkey. A total of 165 patients with papillary thyroid cancer and 172 controls were included in this case–control study. VDR gene single-nucleotide polymorphisms FokI (rs2228570), BsmI (rs1544410), ApaI (rs7975232), and TaqI (rs731236) were evaluated using reverse-transcription polymerase chain reaction. VDR gene polymorphisms BsmI, ApaI, and TaqI did not differ between the papillary thyroid cancer group and control group (p > 0.05, each). BsmI, ApaI, and TaqI were not associated with papillary thyroid cancer risk. The VDR gene FokI CT/TT genotype was associated with an increased papillary thyroid cancer risk (CT vs CC: odds ratio = 1.71, 95% confidence interval = 1.15–2.76, p = 0.028; TT vs CC: odds ratio = 2.44, 95% confidence interval = 1.29–4.62, p = 0.005; CT/TT vs CC: odds ratio = 1.88, 95% confidence interval = 1.20–2.96, p = 0.006; CT/CC vs TT: odds ratio = 1.80, 95% confidence interval = 1.05–3.20, p = 0.041). VDR gene polymorphisms were not in linkage disequilibrium. The FokI TT genotype was associated with having T3 and T4, stage III/IV, extra-thyroidal invasion. The FokI CT/TT or TT genotype was associated with developing N1 status, multifocality, tumor size ≥10 mm, and treatment with radioiodine therapy. Persistence/recurrence did not differ between the FokI genotypes. Carriers of the FokI T allele were at an increased risk of more advanced tumor-node-metastasis stage, greater tumor size, multifocality, and extra-thyroidal invasion of papillary thyroid cancer compared with the CC genotype. VDR gene FokI T allele and TT genotype correlated with aggressiveness of papillary thyroid cancer; thus, FokI could be useful as a poor prognostic factor to assess the high risk of papillary thyroid cancer.
Introduction
Thyroid cancer is a relatively rare neoplasm that affects an estimated 14.9 people per 100,000. It is the most common endocrine cancer and constituted predominantly of its papillary subtype. Thyroid cancer accounts for <2% of all cancers in males and 1%–1.5% in females with a female-to-male ratio of 3:1 in almost all ethnic groups.1,2 Genetic variation contributes to the development of thyroid cancer; however, the pathogenesis of thyroid cancer is not yet known exactly.2,3 Molecular biomarkers have not been identified for the diagnosis and prognosis of thyroid cancer.
4
In addition to the effect on calcium and bone hemostasis, 1,25-diydroxyvitamin D3 (1,25(OH)2D3), the biologically active form of vitamin D, has many extra-skeletal effects such as anti-proliferative, anti-apoptotic, and pro-differentiation properties.5–7 Active vitamin D shows its biologic actions by tightly binding to the intracellular vitamin D receptor (VDR), a hormone-regulated transcription factor. VDR is a member of the nuclear hormone receptor family of transacting transcriptional regulatory factors, including the steroid hormone receptors, thyroid hormone receptors, retinoic acid receptors, and retinoid-X receptors. The VDR gene is located on chromosome 12q13.1 and is composed of five promoters, eight coding exons, and six untranslated exons.5–8 Its promoter region is able to generate multiple transcripts. During activation, VDR forms a heterodimer with the related retinoid-X receptor and regulates the expression of many target genes because it binds to the vitamin D response elements in the chromatin region. Active vitamin D regulates gene transcription by binding to the VDR. It shows anti-tumoral effect by stimulating differentiation and apoptosis.
9
It reduces cellular proliferation, inflammation, angiogenesis, and invasion. VDR regulates other metabolic pathways such as immune response and cancer signaling.5–10 These are considered as VDR-mediated signaling pathways. Studies have shown that VDR gene polymorphisms are found in patients with cancer.
11
Bsml (A>G, rs1544410), ApaI (A>C, rs7975232), TaqI (T>C, rs731236), and FokI (C>T, rs2228570) are human VDR gene polymorphisms. VDR BsmI, ApaI, and FokI gene polymorphisms have shown to increase the risk of having breast cancer.
12
VDR FokI gene polymorphism increases the susceptibility for prostate cancer, BsmI polymorphism does so for malignant melanoma, and TaqI increases the risk for renal cell carcinoma.
11
The FokI polymorphism was associated with an increased ovarian cancer risk.
13
FokI T/T genotype was associated with higher progression rate in head and neck squamous cell carcinoma.
14
VDR BsmI polymorphism was a risk factor for colorectal and skin cancer in a Caucasian population and the ApaI polymorphism was a risk factor for basal cell cancer in an Asian population. The TaqI polymorphism was a risk factor for oral, breast, and basal cell cancer, and the FokI polymorphism was a risk factor for ovarian and skin cancer.
15
However, to date, the association between the VDR gene polymorphism and thyroid cancer has only been investigated in two studies. The VDR ApaI CC and FokI TT polymorphisms were associated with decreased follicular thyroid cancer but haplotype Tabf was associated with an increased risk for follicular thyroid cancer.
10
The VDR gene polymorphism was not associated with differentiated thyroid cancer.
16
This study aimed to investigate the relation between the VDR gene polymorphism and the clinical features and prognostic significance of patients with papillary thyroid cancer (PTC) in a Turkish population. This is the first study to investigate the effects of VDR gene polymorphisms on the clinical features of PTC including cancer staging, outcomes, pathologic findings, and prognostic factors.
Patients and methods
Study population
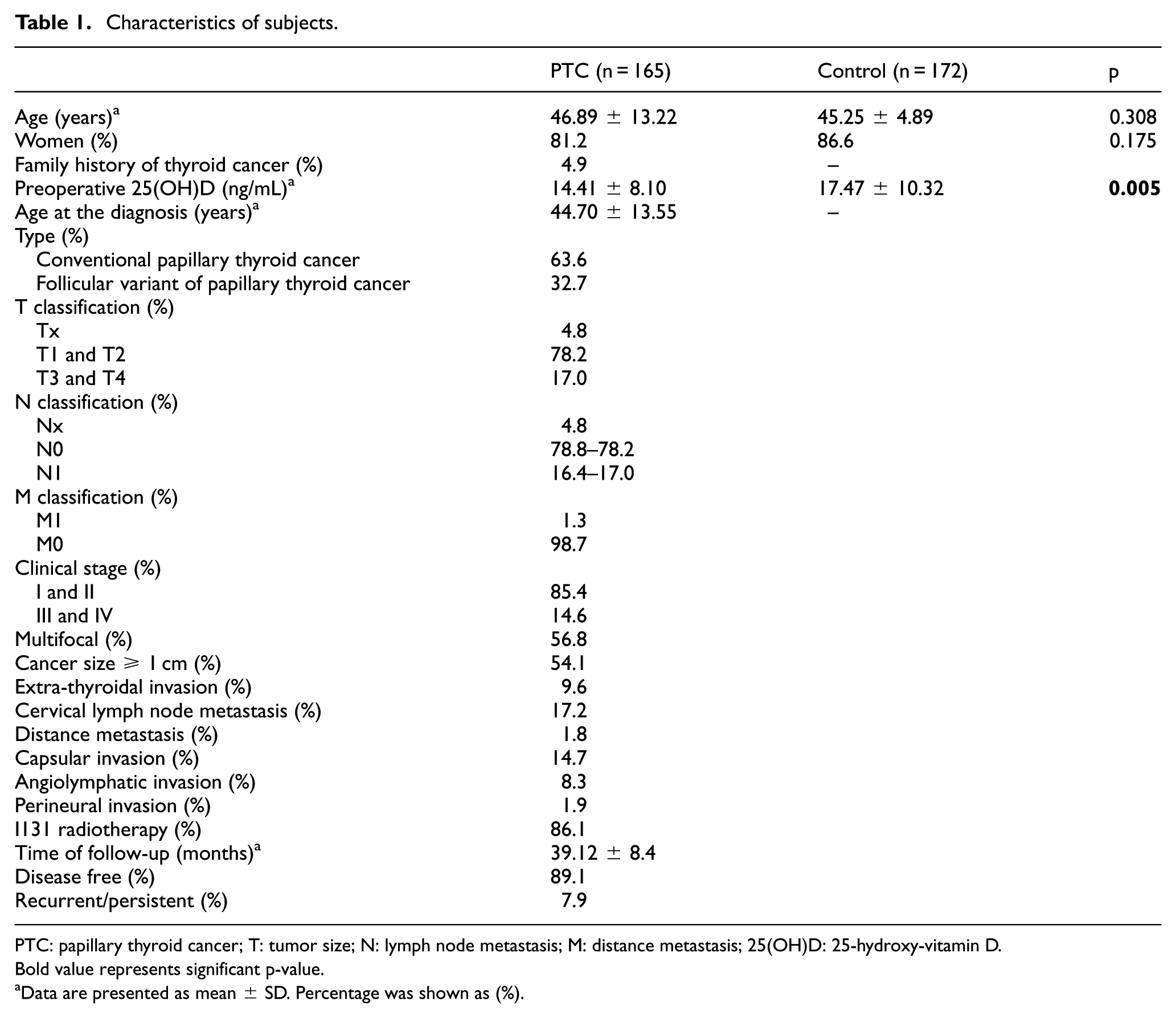
A total of 165 patients with PTC (mean age: 46.89 ± 13.22 years) and 172 control subjects (mean age: 45.25 ± 4.89 years) were enrolled in this case-controlled study. The subjects with PTC were recruited among patients who were treated at the Department of Endocrinology and Metabolism at Diskapi Teaching and Research Hospital, Ankara, Turkey, between 2011 and 2015. Patients were followed up for a mean of 39.12 ± 8.4 months. Subjects with anaplastic carcinoma, follicular carcinoma, and nodular hyperplasia were excluded. PTC was confirmed by pathology. Controls with thyroid disease, cancer, autoimmune disease, and severe disease or family history of thyroid carcinoma were excluded. The histopathologic features of thyroid carcinoma were assessed according to tumor size (T), tumor-node-metastasis (TNM) cancer staging, uni-/multi-focal tumor, extra-thyroidal invasion, lymph node metastasis (N), distance metastasis (M), and angiolymphatic invasion. The clinical features of subjects with thyroid carcinoma were assessed according to treatment with/without radioactive iodine treatment. TNM cancer staging and outcomes (disease-free and recurrent/persistent) were assessed according to the American Thyroid Association (ATA) guideline.
17
Clinical follow-up was examined in all patients and outcomes were classified according to disease status. Disease-free survival was defined as negative radiologic examination with a serum thyroglobulin value <1 µg/L and undetectable thyroglobulin antibodies (<40 IU/L). Patients without these criteria were defined as having persistent disease. Tumor recurrence was defined as reappearance of tumor after complete cure. Each subject gave written informed consent. The study was approved by the local ethics committee of Diskapi Teaching and Research Hospital (2015/28).
Genotype analysis
Genotyping of VDR gene single-nucleotide polymorphisms (SNPs) FokI (rs2228570), BsmI (rs1544410), ApaI (rs7975232), and TaqI (rs731236) were performed. Genomic DNA was isolated from collected peripheral blood samples of the subjects using a DNA Isolation Kit (Roche Diagnostics, Indianapolis, IN, USA). VDR polymorphisms were evaluated using reverse transcription-polymerase chain reaction (RT-PCR). VDR gene SNPs were separately assessed using a fluorescence-based allele-specific PCR assay, KASPar (KBiosciences, Hoddesdon, UK), performed on a Rotor-Gene Q real-time cycler (Qiagen, Hilden, Germany). Allele discrimination was performed using Rotor-Gene Q software v.2.3.1 (Qiagen, Hilden, Germany). The genotype identification was performed blind without information on the phenotypes of the subjects.
Statistical analysis
Statistical analysis was performed using the SPSS 18.0 (SPSS, Inc.) software. Variables are presented as mean ± standard deviation (SD) or median (min–max), percentages (%), odds ratios (OR), and 95% confidence intervals (CIs). Normality was tested using the Kolmogorov–Smirnov and Shapiro–Wilk W test. SNPs are expressed as allelic frequency (q) or prevalence of genotypes (%). Categorical variables were analyzed using the chi-square test or Fisher’s exact test, where appropriate. Student’s t-test was used for normally distributed continuous variables or log-transformed variables between two groups. The Hardy–Weinberg equilibrium (HWE) at individual loci was assessed using the chi-square test. Multiple logistic regression analysis and Fisher’s exact test were tested using models: dominant (major allele homozygotes vs heterozygotes + minor allele homozygotes), recessive (major allele homozygotes + heterozygotes vs minor allele homozygotes), and codominant (major allele homozygotes vs heterozygote and minor allele homozygotes vs major allele homozygotes). Statistical significance was defined as p value <0.05. Pairwise linkage disequilibrium (LD) and correlation coefficients (r2) were analyzed using the HAPLOVIEW program. We made a variable reflecting all possible combinations of BsmI-ApaI-TaqI genotypes for each SNP.
Results
Age (46.89 ± 13.22 vs 45.25 ± 4.89 years) and sex (women, 81.2% vs 86.8%) did not differ between the patients with PTC and controls (p > 0.05; Table 1). 25(OH)D3 levels were lower in patients with PTC than in controls (14.41 ± 8.10 vs 17.47 ± 10.32 ng/mL, p = 0.005). Four polymorphisms of the VDR gene were HWE in the control group. Minor allele frequency and HWE are shown in Table 2. Distributions of VDR gene polymorphisms are shown in Table 3. Frequency of VDR gene ApaI rs7975232, TaqI rs731236, and BsmI rs1544410 polymorphisms did not differ between patients with PTC and controls in a codominant model and dominant model and recessive model (p > 0.05, each). VDR gene polymorphisms ApaI, TaqI, and BsmI were not associated with PTC. Frequency of VDR gene FokI rs2228570 increased in PTC patients than in control (p < 0.05). Haploview analysis shows that VDR gene polymorphisms are not in LD. Compared to control, FokI TT genotype increased in patients with PTC in a recessive model (TT vs CT/CC, OR = 1.80, 95% CI = 1.05–3.20, p = 0.041) and FokI CT/TT genotype increased in patients with PTC in a dominant model (CT/TT vs CC, OR = 1.88, 95% CI = 1.20–2.96, p = 0.006). Compared to control, FokI CT and FokI TT genotypes increased in patients with PTC in a codominant model (CT vs CC, OR = 1.71, 95% CI = 1.15–2.76, p = 0.028 and TT vs CC, OR = 2.44, 95% CI = 1.29–4.62, p = 0.005).
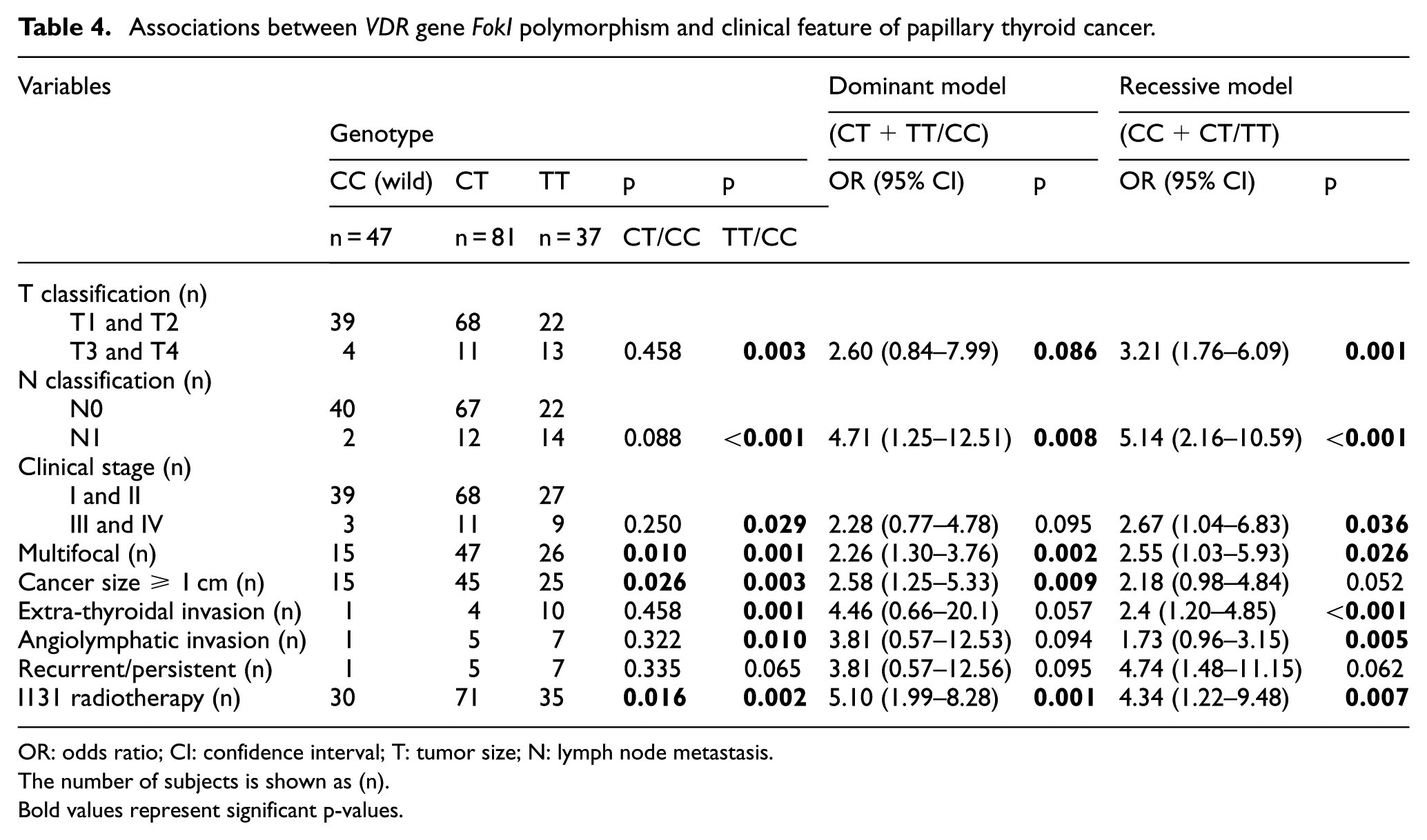
Association between VDR gene FokI rs2228570 and clinical features of PTC patients are shown in Table 4. Patients carrying FokI TT genotype increased risk for developing T3 and T4 PTC compared to patients carrying FokI CC genotype (TT vs CC: OR = 2.71, 95% CI = 1.13–6.53, p = 0.003) and C allele and CC genotype (TT vs CT/CC: OR = 3.21, 95% CI = 1.76–6.09, p = 0.001, recessive model). The FokI T allele and TT genotype increased risk for developing N1 PTC in wild comparison, dominant and recessive model (TT vs CC: OR = 5.16, 95% CI: 1.31–10.42; p < 0.001 and CT/TT vs CC: OR = 4.71, 95% CI: 1.25–12.51, p = 0.008; and TT vs CT/CC: OR = 5.14, 95% CI: 2.16–10.59; p < 0.001, respectively). Patients carrying the FokI TT genotype had increased risk for developing stage III/IV PTC compared with patients carrying the CC genotype (TT vs CC: OR = 4.33, 95% CI: 1.07–10.74; p = 0.029) and C allele and CC genotype (TT vs CT/CC: OR = 2.67, 95% CI: 1.04–6.83; p = 0.036, recessive model). FokI T allele and TT genotype increased risk of multifocal tumor in wild, heterozygote comparison, dominant and recessive model (TT vs CC: OR = 5.20, 95% CI = 1.93–11.94; p = 0.001; CT vs CC: OR = 2.72, 95% CI = 1.25–5.93, p = 0.010; CT/TT vs CC: OR = 2.26, 95% CI = 1.30–3.76, p = 0.002; TT vs CT/CC: OR = 2.55, 95% CI = 1.03–5.93, p = 0.026; respectively). The FokI T allele and TT genotype were more frequent in patients with PTC with tumors ≥10 mm in size compared with tumors <10 mm in size (TT vs CC: OR = 4.09, 95% CI = 1.58–10.52, p = 0.003; CT vs CC: OR = 2.38, 95% CI = 1.10–5.15, p = 0.026; CT/TT vs CC: OR = 2.58, 95% CI = 1.25–5.33, dominant model). The FokI TT genotype was more common in patients with PTC with extra-thyroidal invasion compared with those without extra-thyroidal invasion (TT vs CC: OR = 15.26, 95% CI = 1.90–30.98; TT vs CT/CC: OR = 2.4, 95% CI = 1.20–4.85, p < 0.001, recessive model). The FokI TT genotype and T allele were more frequent in patients with PTC treated with radioiodine therapy compared with those without radioiodine therapy (TT vs CT: OR = 3.15, 95% CI = 1.20–8.27, p = 0.016; TT vs CC: OR = 7.2, 95% CI = 1.71–21.34, p = 0.002; CT/TT vs CC: OR = 5.10, 95% CI = 1.99–8.28, dominant model; TT vs CT/CC: OR = 4.34, 95% CI = 1.22–9.48, p = 0.007, recessive model). Patients with recurrent/persistent disease and disease-free status did not differ between the FokI genotypes (p > 0.05). The VDR gene polymorphisms ApaI, BsmI, and TaqI were not related to pathologic features (multifocality, capsule invasion, extra-thyroidal invasion, TNM-staging, tumor size, lymph node metastasis), radioiodine therapy, and outcome.
Discussion
This is the first study to show the effects of VDR gene polymorphisms on the clinical features of PTC including TNM cancer staging, outcomes, pathologic findings, and prognostic factors in a Turkish population. We found that the frequency of VDR gene ApaI, TaqI, and BsmI polymorphisms did not differ between PTC and controls with no association. The frequency of VDR gene FokI TT genotype and CT heterozygotes were increased in PTC compared with the controls. VDR gene FokI was associated with an increased risk of PTC. Haploview analysis showed that VDR gene polymorphisms were not in LD. Patients with PTC carrying the FokI TT genotype were more likely to have adverse pathologic and prognostic factors including T3 and T4, stage III/IV, and extra-thyroidal invasion. Patients with PTC carrying the FokI CT/TT or TT genotype were more likely to develop N1, multifocal tumor, and tumors ≥10 mm. FokI CT/TT or TT genotype carriers were more likely to be treated with radioiodine therapy. VDR gene FokI was related to aggressiveness of PTC disease; however, it was not correlated with persistence/recurrence. Thus, VDR gene FokI might be used as a biomarker for poor clinicopathologic findings.
In humans, vitamin D shows a supportive effect on thyroid tissue. VDR gene BsmI and TaqI polymorphisms were shown to be associated with decreased risk of autoimmune thyroid disease.
18
VDR gene ApaI, Bsml, and FokI polymorphisms were associated with Graves’ disease in an Asian population, whereas ApaI, Bsml, TaqI, and FokI were not associated with Graves’ disease in Caucasians.
19
TaqI CC and FokI TT were increased in Hashimoto’s thyroiditis in Turkey.
20
Genetic and epigenetic determinants of thyroid cancer may have an effect on 1,25(OH)2D3 signaling. Development and progression of thyroid cancer depends on impaired 1,25(OH)2D3 signaling21,22 which has been confirmed in clinical studies22–26 and thyroid cancer cell lines.
27
Local 1,25(OH)2D3-VDR signaling is decreased in primary thyroid cancer with local lymph node metastasis, and complete loss of signaling is more significant in anaplastic thyroid cancer with distant metastasis.10,22–25 Hence, thyroid cancer progression is characterized by the loss of proteins that play a role in vitamin D signaling or 1,25(OH)2D3 sensitivity.
27
The signaling effect of 1,25(OH)2D3-VDR in thyroid cancer occurs via the local anti-tumoral effect of 1,25(OH)2D3.
21,22 This anti-tumoral effect occurs either directly (via VDR binding) or indirectly (via the interaction between other critical transcriptional regulators or cell signaling systems).21,28,29 Therapeutic use of vitamin D agonists in thyroid cancer requires the presence of VDR. An experimental study showed that VDR FokI CC was associated with vitamin D analog resistance in the treatment of thyroid cancer.
30
Active vitamin D has many anti-cancer features including anti-proliferative, anti-apoptotic, pro-differentiating, and anti-inflammatory effects.5–10 These effects are mediated by VDR. VDR regulates also metabolic pathways such as the immune response and cancer signaling.5–8 VDR is stimulated by p53, a tumor-suppressing gene.
16
An association was found between vitamin D deficiency and thyroid cancer.10,31,32
Epidemiologic studies reported that there was a relationship between VDR gene and breast cancer, prostate cancer, lung cancer, colorectal cancer, and other gastrointestinal cancers.4–8 However, reliable data on thyroid cancer are available only in in vitro experimental studies.10,23,24,27–31VDR gene Apa, Bsml, Taq, and FokI genotypes were not associated with risk of PTC in a German population.
10
In an Iranian population, frequency of VDR gene ApaI, FokI, Bsml, Taq, and Tru9 did not differ between differentiated thyroid cancer and controls, with no relationship.
16
However, these studies did not investigate the genotype-phenotype correlation such as TNM cancer staging, outcomes, pathologic findings, and prognostic factors. In this study, vitamin D deficiency was found in patients with PTC compared with controls. This study showed that frequency of VDR gene ApaI, TaqI, and BsmI did not differ between patients with PTC and controls. VDR gene ApaI, TaqI, and BsmI were not associated with a risk for PTC. VDR gene FokI rs2228570 showed significant differences between patients with PTC and controls: VDR gene FokI (variant or heterozygotes) compared with wild type (CC) revealed a significant association. VDR gene FokI TT (TT vs CC: OR = 2.44, 95% CI = 1.29–4.62, p = 0.005) and CT heterozygotes (CT vs CC: OR = 1.71, 95% CI = 1.15–2.76, p = 0.028) were associated with an increased PTC risk compared with the controls. Our study suggests that VDR gene FokI may be associated with the development of PTC. Allele frequency distribution showed a significant association of FokI variant allele (T) on susceptibility to PTC. These results revealed that VDR gene FokI might play a critical role in the etiology of the PTC. VDR gene FokI TT genotype was associated with having T3 and T4, stage III/IV, and extra-thyroidal invasion of PTC. FokI CT/TT or TT genotype was associated with developing N1, multifocality, and tumor size ≥10 mm in PTC. Patients carrying FokI CT/TT or TT genotype were mostly treated with radioiodine therapy. The persistence/recurrence rate in PTC did not differ between FokI genotypes. Although VDR gene FokI was associated with more advanced TNM stage, greater diameter of tumor, multifocality, and extra-thyroidal invasion, outcomes in follow-up did not differ. VDR gene FokI might be considered as a risk for poor clinicopathologic features and advanced stage of PTC. VDR gene FokI might be suggested as a poor prognostic factor of PTC. However, the molecular mechanism needs to be identified.
Bsml, ApaI, and TaqI polymorphisms of VDR gene are found in the three primer untranslated region (3′-UTR) and have been shown to be in strong LD.33,34FokI polymorphism was reported as an independent marker of VDR gene because it has not been shown to be in LD with any other VDR polymorphisms.
34
Haploview analysis showed that VDR gene polymorphisms were not in LD. ApaI and BsmI polymorphisms of the VDR gene, both in intron 8, are considered as silent SNPs. These polymorphisms do not change the amino acid sequence of the encoded protein, but they might affect gene expression by modulating stability of messenger RNA (mRNA).
11
TaqI polymorphism is located at codon 352 in exon 9 of the VDR gene. According to the Taq restriction site, products are digested into two or three fragments. TT genotype (absence of restriction site) is related to lower active vitamin D3.4–11 The only locus that has an impact on the structure of VDR protein is the FokI polymorphism, which is located on the 5′ end region of VDR gene. VDR gene FokI polymorphism is functional because it is found in a coding sequence. FokI polymorphism is located in the first ATG starting code of VDR protein. FokI is involved in thymine to cytosine (T/C) substitution at exon 2, the first translation initiation region is removed, and the transcriptional activity of VDR is changed.4–11,16 It alters the ACG codon that is found 10 base pairs upstream from the translation starting codon and leads to generating an additional starting codon. Two different VDR isoforms occur with the transition of allele T to C in ATG. When the initiating translation starts from this alternative site in the thymine variant, it leads to the generation of a longer VDR protein comprising 427 amino acids. The gene is transcribed in normal length if there is a restriction site. Thus, the C/C allele codes the 424-amino acid protein and the T/T allele codes the 427-amino acid protein. The longer VDR protein has low activity in transcription; accordingly, activation is decreased in the target cell.4–11 Arai et al.
35
reported that compared with FokI T/T genotype, FokI C/C showed 1.7-fold greater function in vitamin D-dependent transcriptional activation of a reporter through the regulation of a vitamin D response element. FokI rs2228570 polymorphism is the only VDR gene polymorphism involved in the generation of an altered protein expression.
11
In this study, the control population was consistent with HWE. The small sample size, cross-sectional design, and seasonal change in 25(OH)D3 are limitations of this study.
In conclusion, this study showed that VDR gene FokI might contribute to the susceptibility of PTC risk in the Turkish population. VDR gene FokI correlated with aggressiveness of papillary thyroid cancer but not with outcome of disease. FokI could be useful as supplementary prognostic factor to assess high risk of papillary thyroid cancer and to identify patients who need more aggressive management and follow-up. Further studies in different populations are needed to confirm these results.