Abstract
The acquisition of a castration-resistant prostate cancer phenotype by prostate cancer cells is the alteration that has the worst prognosis for patients. The aim of this study was to evaluate the role of the microRNAs-23b/-27b as well as the possible CCNG1 target gene in tissue samples from patients with localized prostate cancer that progressed to castration-resistant prostate cancer and in a castration-resistant prostate cancer cell line (PC-3). The microRNAs and target gene expression levels of the surgical specimens were analyzed by quantitative real-time polymerase chain reaction. The prostate cancer cell line, PC-3, was transfected with pre-miR-23b, pre-miR-27b, and their respective controls using Lipofectamine RNAiMAX and exposed or not to flutamide. After transfections, expression levels of both the microRNAs and the gene, CCNG1, were analyzed by quantitative real-time polymerase chain reaction. The apoptosis and cell cycle assays were performed on the mini MUSE cytometer. MicroRNAs-23b/-27b were underexpressed in surgical specimens of prostate cancer; however, their target gene, CCNG1, was overexpressed in 69% of the cases. After transfection with the microRNAs-23b/-27b and flutamide, we observed a reduction in gene expression compared with cells that were treated only with microRNAs or only with flutamide. In the apoptosis assay, we demonstrated cell sensitization following transfection with microRNAs-23b/-27b and potentiation when co-administered with flutamide. The number of cells in apoptosis was almost three times higher with the simultaneous treatments (miR + flutamide) compared with the control (p < 0.05). In the cell cycle assay, only flutamide treatment showed better results; a higher number of cells were found in the G0-G1 phase, and a lower percentage of cells completed the final phase of the cycle (p < 0.05). We conclude that microRNAs-23b/-27b are downexpressed in prostate cancer, and their target gene, CCNG1, is overexpressed. We postulated that microRNAs-23b/-27b sensitize the PC-3 cell line and that after the addition of flutamide in the apoptosis assay, we would observe synergism in the treatments between miR and flutamide. In the cell cycle assay, the use of flutamide was sufficient to decrease the number of cells in mitosis. Therefore, we postulate that microRNAs, along with other drugs, may become very useful therapeutic tools in the treatment of castration-resistant prostate cancer.
Introduction
Prostate cancer (PCa) is the sixth most common type of cancer in the world and the third leading cause of death among men in the United States. 1 The acquisition of a castration-resistant prostate cancer (CRPC) phenotype by PCa cells is the frequent alteration that presents poor prognosis for patients. 2
Antiandrogens are an established alternative treatment in patients with advanced PCa, either alone or in combination with luteinizing hormone-releasing hormone analogs. 3 Flutamide (FLU) was the first non-steroidal antiandrogen widely used to treat patients with advanced PCa. Lower survival rates were observed when FLU was used as a monotherapy. 4
Therapeutic combinations in the field of oncology, especially when we refer to CRPC, are of great interest. microRNAs comprise a group of small molecules of RNAs, with fundamental actions in the regulation of gene expression 5 and are involved in a number of cellular processes, such as survival, apoptosis, differentiation, development, and cell proliferation. 6
In recent decades, microRNAs have received primary attention as upregulated with growth factors and tumor progression, and recently, some authors have suggested the use of microRNAs in the treatment of PCa.7–9
Some microRNAs, small noncoding 18- to 24-nucleotide RNAs, are predicted to regulate the expression of more than 90% of protein encoding genes and have been linked to the process of the PCa hormone-sensitive transition to CRPC.2,10 Evidence indicates that microRNAs also contribute to the initiation, development, and metastasis of various types of cancers. 11
The expressions of miRs-23b/-27b, which are encoded on chromosome 9, are specifically subexpressed in the tissue samples of metastatic PCa compared to benign prostatic tissue.2,10,12 The CCNG1 gene is a potential target gene for miRs-23b/-27b, and this gene is overexpressed in several neoplastic cells, including PCa cell lines. Studies have attempted to demonstrate that CCNG1 plays a role similar to the TP53 gene in repairing DNA damage.13,14 The aim of this study was to evaluate the role of the microRNAs-23b/-27b as well as their possible CCNG1 target gene in CRPC.
Methods
Patients
This study was performed using 44 samples of surgical specimens from patients with clinically localized PCa who underwent radical prostatectomy, with a mean age of 63.64 years and a mean follow-up time of 94.32 months. All 44 patients had at least one episode of biochemical relapse and started hormone treatment. Of the 44 patients, only nine responded to hormone therapy for at least 1 year, and 35 patients were resistant to hormone therapy in less than 1 year of treatment and showed disease recurrence. The control group consisted of 10 patients who had a confirmed diagnosis of benign prostatic hyperplasia (BPH).
Patients were classified according to their clinical-pathological factors and correlated with the expression of microRNAs-23b/-27b and their possible target gene, CCNG1. For further analysis, the expression was correlated with the pathological stage that was defined as confined (pT2) and not confined (pT3) organ; the preoperative prostate specific antigen (PSA) was divided into <10 and ≥10 ng/mL and with the new categorization of the International Society of Urological Pathology (ISUP), where the subgroups were divided into ISUP-1, ISUP 2-4, and ISUP-5. According to the response to hormone therapy, the patients were classified to have castration-sensitive tumors, and those who did not respond to hormone therapy were classified as castration-resistant.
This study was approved by the Institutional Board of Ethics (CAPPesq, Hospital das Clinicas, School of Medicine, University of Sao Paulo, no. 085/15).
Cell culture
The PC-3 prostate cancer cell line was obtained from ATCC and grown in MEM medium (Life Technologies, Waltham, MA, EUA), supplemented with 10% fetal bovine serum (FBS) and 1% antibiotic/antimycotic solution (Sigma Co., St. Louis, MO, USA) at 37°C in an atmosphere of 5% CO2.
Transfection
The cells were seeded in 24-well plates at a concentration of 1 × 105/well for miRNA transfection. The reverse transfections into the PC-3 cell lines were carried out in Opti-MEM I using Lipofectamine RNAiMAX (Thermo Fisher Scientific, Waltham, MA, EUA) according to the manufacturer’s protocol. Synthetic miR-23b (Assay ID MC10711-Ambion), miR-27b (Assay ID MC10750-Ambion), and their respective scrambled controls, control miR (ctrl miR; catalog number AM17110), were used at a final concentration of 50 nM. All PC-3 cell line assays were performed in triplicate.
Exposure of PC-3 cells to FLU
The PC3 cell line was cultured as previously described, and FLU was used at a concentration of 100 μM/mL 15 in OPTI-MEM I (31985-062, Thermo Fisher Scientific) for 48 h; for the control of the experiment, we added only Opti-MEM I.
Extraction of microRNAs and RNAs and quantitative real-time polymerase chain reaction
The microRNAs and RNA extractions were performed using the mirVana kit (Ambion, Austin, TX, USA) according to the manufacturer’s instructions. MicroRNAs and RNA concentrations were determined by 260/280 nm absorbance using a Nanodrop® ND-1000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, EUA). Complementary DNA (cDNA) from total RNA was generated using a High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, CA, USA). Reactions were incubated at 25°C for 10 min, followed by 37°C for 120 min and 85°C for 5 min. cDNA from miRNAs was generated using a TaqMan miRNA Reverse Transcription kit (Applied Biosystems, Foster City, CA). Reactions were incubated at 16°C for 30 min, 42°C for 30 min, and 85°C for 30 min.
Expression levels of miR-23b, miR-27b, CCNG1, and AR were analyzed by quantitative real-time polymerase chain reaction (qRT-PCR) using the ABI 7500 Fast Real-Time PCR System (Applied Biosystems). Target sequences were amplified in a 10-μL reaction mixture containing 5 μL of TaqMan Universal PCR Master Mix, 0.5 μL of TaqMan Gene Expression Assays for CCNG1 and AR, and 0.5 μL of TaqMan miRNA expression assays for miR-23b and miR-27b, 1 μL of cDNA, and 3.5 μL of DNase-free water. The PCR cycling conditions were 2 min at 50°C, 10 min at 95°C, and then 40 cycles of 15 s at 95°C and 1 min at 60°C. Beta-2 microglobulin was used as an endogenous control in the analysis of CCNG1 and AR expression. RNU48 was used as an endogenous control for miR-23b and miR-27b.
The relative expression was calculated by the ΔΔCT method. 16 The fold change in gene expression was calculated as 2–ΔΔCT.
Analysis of apoptosis and cell cycle by flow cytometry
For apoptosis and cell cycle analyses, PC-3 cells were transfected as described above. After 72 h of treatment (24 h of transfection + 48 h of transfection, and exposition with FLU), the cells were marked with Annexin V and Muse™ cell death kits (MCH100105) and Muse™ Cellular Cycle (propidium iodide and RNAse A) (MCH100106) according to the manufacturer’s recommendations. Apoptosis and cell cycle analyses were performed on the Muse® Cell Analyzer (Merck Millipore, Burlington, MA, EUA).
Statistical analysis
To compare the clinical characteristics of patients with PCa, apoptosis, and cell cycle assay, we used the Mann–Whitney test, the Student’s t-test, or analysis of variance (ANOVA). Statistical analysis was performed using SPSS 19.0 for Windows using a significance of p ≤ 0.05.
Results
Expression of microRNAs-23b/-27b and CCNG1 gene in surgical specimens
The expression profile of miRs-23b/-27b in the surgical specimens was downregulated by 90.91% and 88.64%, respectively, when compared to the control. However, the CCNG1 gene was overexpressed in 63.63% of the analyzed samples (Figure 1).
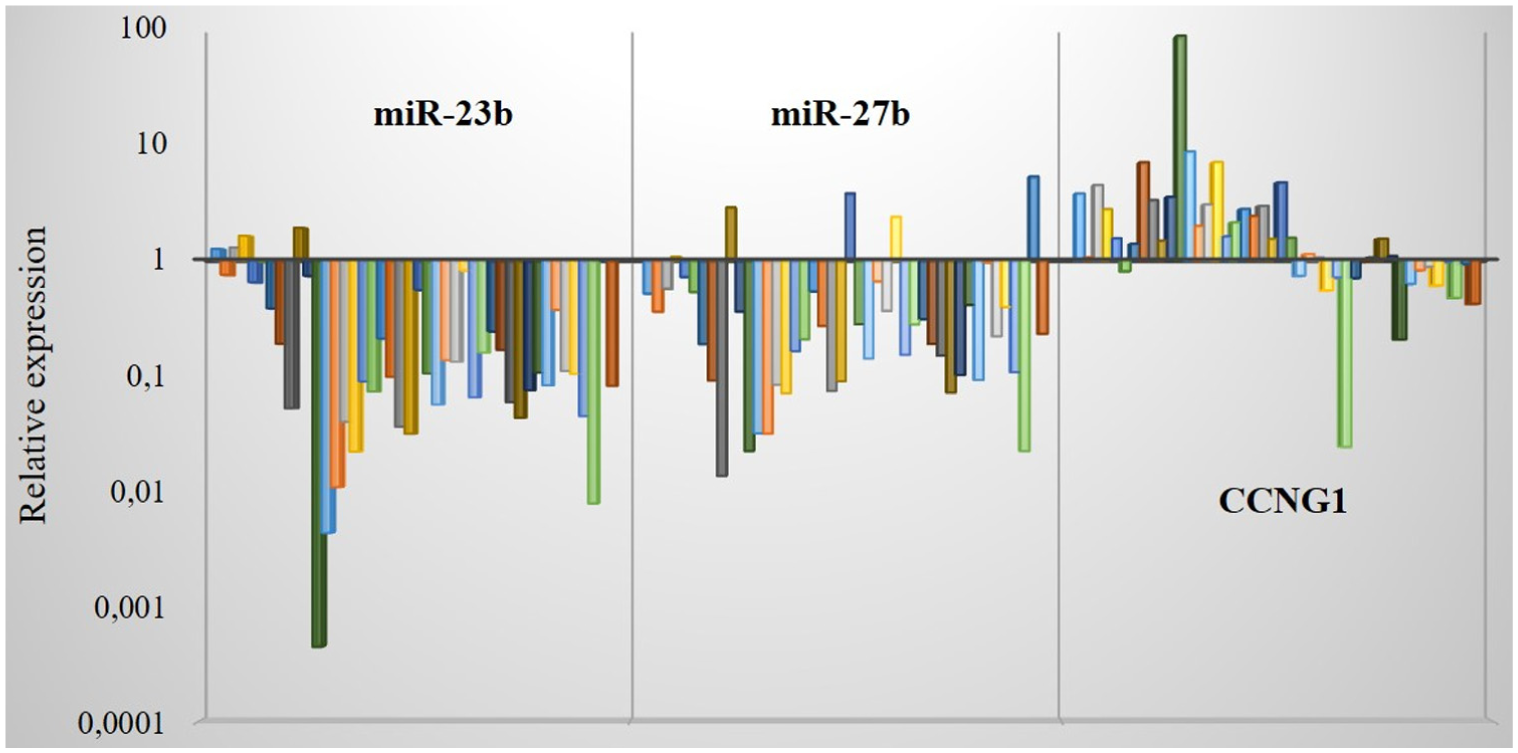
Expression profile of the microRNA-23b, microRNA-27b, and the CCNG1 gene in the tissue samples of PCa evaluated by qRT-PCR (N = 44).
When we correlated the expression of the microRNAs with response to hormone therapy, we did not observe any significant differences for either microRNAs or the CCNG1 gene (Table 1).
Correlation of gene expression and microRNAs between clinical and pathological characteristics.
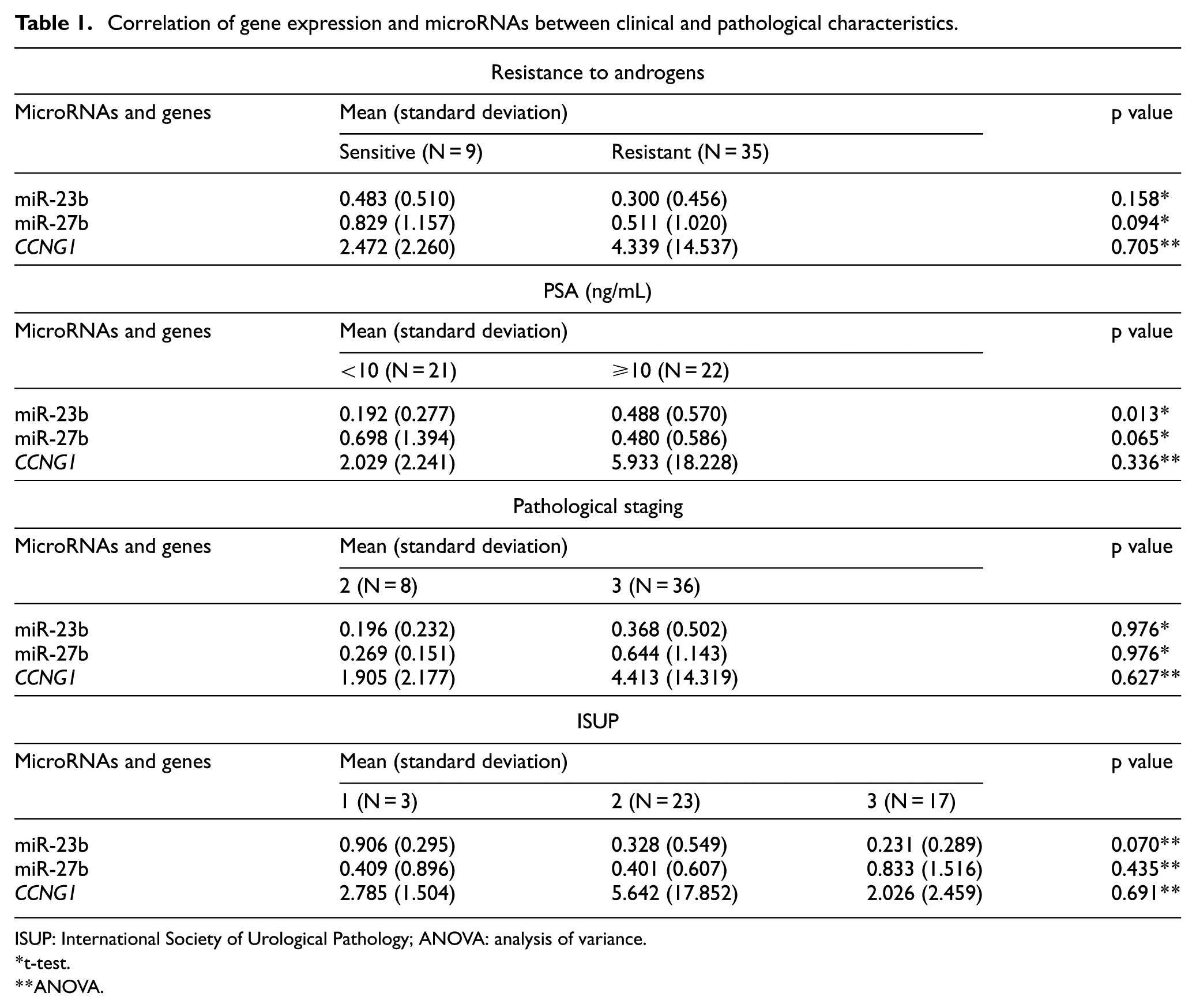
ISUP: International Society of Urological Pathology; ANOVA: analysis of variance.
t-test.
ANOVA.
Comparing the expression of the microRNAs and the gene with patients’ clinicopathological characteristics, we found statistical significance for miR-23b; interestingly, its highest mean expression was correlated with patients who had PSA ≥10 ng/mL (p = 0.01). For miR-27b, we found a significant marginal value where the highest mean expression was correlated with patients who had PSA <10 ng/mL (p = 0.06; Table 1).
Among the other clinical-pathological characteristics, no significant difference was observed for mean expression of microRNAs and the CCNG1 gene (Table 1).
Expression of AR gene in PC3 cell line
We performed analysis of AR gene expression in the PC3 cell line by qRT-PCR, using as control values from analyses of the AR gene in samples from BPH. We verified that this cell line exhibited a subexpression profile of this gene (0.008 ± 0.001; Figure 2).

AR expression levels in PC3 cell line evaluated by qRT-PCR. The bar graph shows mean and standard deviation values.
Transfection of the PC3 cell line
After transfection with the microRNAs, RNA and microRNAs were extracted, and qRT-PCR was then performed to analyze the expression of each microRNA and its possible target gene (Figure 3).

Overexpression of miR-23b and miR-27b demonstrating the success of transfection in the PC-3 cell line evaluated by qRT-PCR. The bar graphs show mean and standard deviation values.
In this assay, we observed that the microRNAs were overexpressed in PC-3 cells, but the gene, CCNG1, did not demonstrate downexpression, as expected. Interestingly, after transfection analysis of cells that were treated singly with the microRNAs-23b/-27b, FLU, and microRNAs + FLU, we observed that gene expression decreased considerably until it became underexpressed when cells were treated with microRNAs + FLU. In this case, we supposed that the cells were sensitized after the administration of the microRNAs for action of the drug (Figure 4).
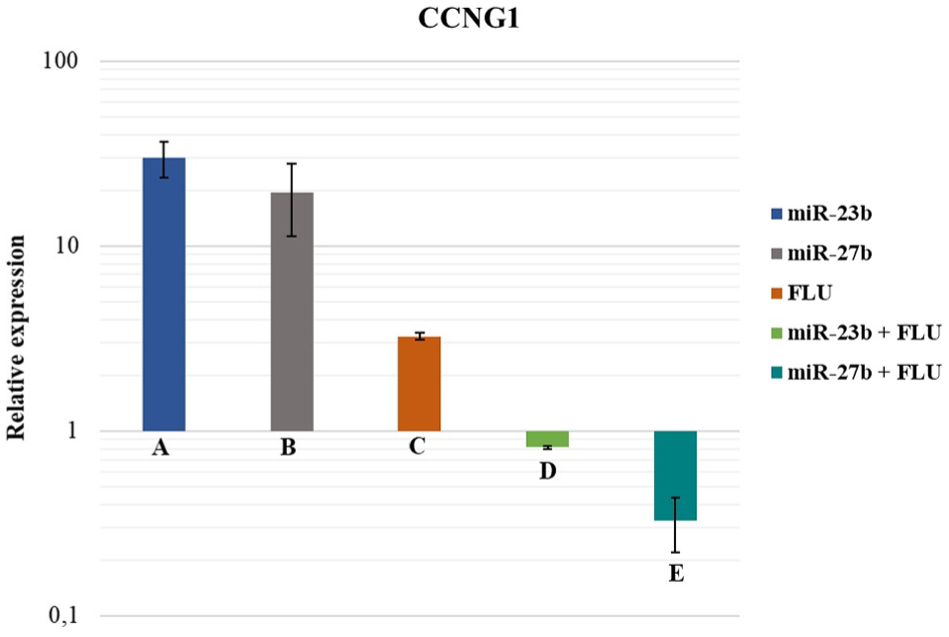
CCNG1 gene expression after transfection and treatments in the PC-3 cell line. Columns A and B represent overexpression of the CCNG1 gene after treatment with miR-23b and miR-27b, respectively. Column C represents CCNG1 gene expression after treatment with FLU. Columns D and E represent CCNG1 gene expression after treatment with miR-23b + FLU and miR-27b + FLU, respectively. The bar graphs show mean and standard deviation values.
Apoptosis assay
In the apoptosis assay, when comparing the treatment groups, microRNAs, miR + FLU, and FLU, and correlating with the control, we verified that the group of cells treated with only FLU had a higher rate of apoptosis compared to the groups treated with only microRNA-23b and microRNA-27b. However, this difference was not statistically significant (Table 2).
Apoptosis assay of the PC-3 cell line.

FLU: flutamide; ANOVA: analysis of variance.
ANOVA.
When we analyzed the groups of cells that were treated with miR-23b and miR-27b, and later added FLU, we observed an increase in the mean rate of total apoptosis, when compared to the control and the other treatments (p < 0.05). Thus, demonstrating a possible sensitization of the cells by the microRNAs and potentializing the effect of FLU, we postulate a possible synergism among the treatments.
Cell cycle assay
In the cell cycle assay, the best result was the group treated with FLU only, showing an increase in the percentage of cells in the G0-G1 phase (64.97%), followed by reduction of other phases, S (28.10%) and G2-M (6.86%) (p < 0.05). None of the groups treated with miR + FLU showed sufficient synergism that demonstrated better results when compared to the group of cells treated with FLU only (Table 3).
Cell cycle assay of the PC-3 cell line.

FLU: flutamide; ANOVA: analysis of variance.
ANOVA.
Discussion
The molecular characterization of castration-sensitive prostate cancer and its mechanism of transition to CRPC has been the focus of several researchers. MicroRNAs, which are important regulators of gene expression, have been associated with this process.
In this study, we showed the expression profile of microRNAs-23b/-27b and the CCNG1 gene in tissue samples from patients with localized PCa that progressed or not to CRPC. We also analyzed this profile in in vitro studies and performed apoptosis and cell cycle assays modulating these microRNAs.
The miR-23b/-27b presented downexpression in the majority of PCa cases, when compared to the control group. The downexpression of these miRs was also found in a study by Goto et al. 17 in which 39 patients with aggressive PCa were studied. In this study, the loss of miR-27b was correlated with shorter free survival time. Another study 12 found low expression of these microRNAs-23b/-27b in PCa. Our data are in line with the literature demonstrating the downexpression of 23b/-27b in samples of primary tumors from patients with PCa, and we found a greater downexpression in patients who progressed to CRPC.
We also found correlations between the expression of these microRNAs and some classic prognostic factors of PCa. Although miRs-23b/-27b were underexpressed in the cancer group, there was a higher expression of miR-27b in patients who had PSA <10 ng/mL and in patients who did not progress to the castration-resistant phenotype, although these cases had marginal significance. Considering miR-23b, we observed a higher expression in the group of patients with higher PSA and patients who were in group 1 of the ISUP classification; this difference was marginally significant.
After transfection, the CCNG1 gene showed overexpression. However, this result was not expected because software analyses with TargetScan, 18 PicTar, 19 and miRanda 20 described this gene as a possible target of these microRNAs. Moreover, in vitro studies in liver adenocarcinoma cell lines showed that when there is overexpression of miRs-23b/-27b, a decrease in the levels of CCNG1 occurs. 21 Overexpression of CCNG1 was observed in breast cancer cell lines and PCa, and overexpression of this gene has been correlated with tumor initiation and progression in these tumors. 13 However, our studies showed that after cell sensitization by microRNAs-23b/-27b and the addition of FLU to the PC-3 cell line, the CCNG1 gene was underexpressed. Therefore, we suggest that the cells were sensitized after the administration of the microRNAs for action of the drug.
The literature presents controversial data regarding the presence of AR in the PC3 cell line; 22 while some authors have already demonstrated that these cells express reduced levels of AR, other authors have shown that they have only nuclear AR or demonstrated absence of AR.23,24 In our study, we found subexpression of AR in the PC3 cell line evaluated by qRT-PCR. In a study by Sica et al., the presence of AR was evaluated by immunohistochemistry assay, presenting strong evidence of AR after labeling the cells with monoclonal antibodies. 25
Although cancer presents very heterogeneous characteristics, all of the malignant tumors, including PCa, acquire a domain of growth beyond the limit imposed by the normal cells. Evidence has shown that resistance to apoptosis is one of the most striking features of most tumors. 26
Analyzing miRs-23b/-27b, we found significant differences for both microRNAs in the apoptosis assay. When we compared the total apoptosis rate, considering the treatments with FLU, miR + FLU, and miR, we found high levels of apoptosis in the groups treated with miR + FLU for both microRNAs when compared to the control.
We observed an increase in the rate of apoptosis in the group treated with miR + FLU, almost three times higher than in the group treated with miRs only, thus suggesting a possible synergistic effect between these microRNAs and FLU. Majid et al. 27 have shown in their studies with PCa lines, PC-3 and DU-145, that the rate of apoptosis increases after transfection with miR-23b; however, a decrease in cell viability was observed.
miR-27b sensitizes cancer cells to a wide spectrum of anticancer drugs in vitro and in vivo; functionally, miR-27b improves the response to anticancer drugs activating the apoptosis-dependent p53. 28 This miR has shown dysregulated levels of ectopic expression in various cancers; some studies have already suggested this microRNA as a possible underlying biomarker for targeted molecular therapy. After the demonstration of synergism of miR-27b with multiple anticancer drugs, prospects for clinical application as a “cocktail” package are being fanned.17,29,30 Our data are consistent with the literature, since after administration of miR-27b with the drug FLU, we found a higher effect for the apoptosis assay in the PC-3 cell line.
Cell cycle regulation is a critical role for cell survival and maintenance, as this process leads to the repair of genetic damage and prevention of exacerbated cell division. The cell cycle analysis has become a very important event in understanding the action of carcinogenic compounds 31 and in cell mitotic mechanisms. 32
We found that administration of the drug FLU was shown to be effective in the cell cycle assay with PC3 cells. After treatment with FLU, the cells were retained in the G0-G1 phase, and consequently, a gradual decrease in cells was observed in the later phases.
The final percentage of cells that were able to complete mitosis was significantly lower when compared with the other groups. In this assay, we could not show any synergistic effects between treatments.
Conclusion
CRPC is considered the major cause of death in patients with PCa; thus, the study of molecular alterations at this castration-resistant stage of the tumor is of paramount importance. Although the correlation with classical prognostic factors of PCa in our study presented only marginal statistical values, higher levels of expression of these microRNAs were observed in favorable clinical characteristics, including patients who remained with the hormone-sensitive phenotype. In addition, miR-23b and miR27b have been shown to sensitize PC-3 cells to FLU action when administered together with this antiandrogen, increasing the apoptosis rate and decreasing the expression of the CCNG1 oncogene. Further studies of microRNA-23b and microRNA-27b are required for better understanding of the role of microRNAs in CRPC.
Footnotes
Acknowledgements
We thank Fernando Bandeira Szulczewski for support with apoptosis assay. We thank Iran Silva for technical support with cell cycle assays. Rubens Park, Gabriela Q Amaral, Denis R Morais, and Ruan CA Pimenta performed the research. Nayara I Viana, Sabrina T Reis, and Ruan CA Pimenta analyzed the data. Ruan CA Pimenta and Sabrina T Reis wrote the paper. Sabrina T Reis, Kátia RM Leite, William C Nahas, Miguel Srougi, and José Pontes Jr designed the research study. Juliana A Camargo and Vanessa R Guimaraes contributed the essential reagents and tools.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was submitted and approved by the Research Ethics Committee of the Medical School of the University of São Paulo under the number 085/15.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundação de Amparo à Pesquida do Estado de São Paulo (FAPESP; grant number: 2015/03582-6).
