Abstract
Toll-like receptors play an essential role in our innate immune system and are a focus of interest in contemporary cancer research. Thus far, Toll-like receptors have shown promising prognostic value in carcinomas of the oral cavity, colon, and ovaries, but the prognostic role of Toll-like receptors in pancreatic ductal adenocarcinoma has not been established. We set out to investigate whether Toll-like receptor expression could serve in prognostic evaluation in pancreatic ductal adenocarcinoma, as well. Our study comprised 154 consecutive stage I–III pancreatic ductal adenocarcinoma patients surgically treated at Helsinki University Hospital between 2002 and 2011. Patients who received neoadjuvant therapy were excluded. Tissue microarrays and immunohistochemistry allowed assessment of the expression of Toll-like receptor 2 and Toll-like receptor 4 in pancreatic ductal adenocarcinoma tissue, and we matched staining results against clinicopathological parameters using Fisher’s test. For survival analysis, we used the Kaplan–Meier method and the log-rank test, and the Cox regression proportional hazard model for univariate and multivariate analyses. The hazard ratios were calculated for disease-specific overall survival. Strong Toll-like receptor 2 expression was observable in 51 (34%) patients and strong Toll-like receptor 4 in 50 (33%) patients. Overall, neither marker showed any direct coeffect on survival. However, strong Toll-like receptor 2 expression predicted better survival when tumor size was less than 30 mm (hazard ratio = 0.30; 95% confidence interval = 0.13–0.69; p = 0.005), and strong Toll-like receptor 4 expression predicted better survival in patients with lymph-node-negative disease (hazard ratio = 0.21; 95% confidence interval = 0.07–0.65; p = 0.006). In conclusion, we found strong Toll-like receptor 2 and Toll-like receptor 4 expressions to be independent factors of better prognosis in pancreatic ductal adenocarcinoma patients with stage I–II disease.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is a notably aggressive disease and a common cause of cancer death. 1 The only curative treatment is radical surgery combined with oncological treatment. Yet PDAC is notorious in its tendency to metastasize early on, and only approximately 15%–20% of patients have resectable disease.2,3 The 5-year survival rate is less than 4%, 4 and even those who undergo radical intent surgery have a 5-year survival of only approximately 20%. 5
A well-known risk factor for cancer development is chronic local inflammation, presumably due to local release of anti-apoptotic, angiogenic, and growth-stimulating factors. 6 However, the role of inflammation is complex, as it exhibits also antitumor properties. 7
Toll-like receptors (TLRs) take part in first-line immune defense: they occur in sentinel immune cells and epithelia. Their main purpose is to activate local inflammation response and to enhance adaptive immunity and cell proliferation. They work through MyD88 proteins, which induce NF-κB activation and ultimately cell proliferation. 8 They are activated by unfamiliar molecules, either particles of microbial origin or endogenic components released in tissue damage.
TLRs are also overexpressed in some carcinomas, which have led to their exploration as prognostic markers and targets for oncological treatment. TLRs have been studied in numerous carcinomas, with strong expression usually being associated with worse prognosis.9–12 TLRs 2 and 4 have also been studied in PDAC, but no prognostic value has been noted thus far.13,14
Our aim was to explore the prognostic roles of TLR2 and TLR4 in PDAC. These receptors are among those linked to bacterial infections and are two of the most studied TLRs in relation to cancer. 15 Our hypothesis was that TLR2 and TLR4 expressions show prognostic value in PDAC patients.
Materials and methods
The data came from consecutive surgically treated PDAC patients who underwent surgery in 2002–2011 at the Department of Surgery, Helsinki University Hospital, with 21 receiving neoadjuvant therapy excluded. Those seven rediagnosed with stage-IV metastatic disease were likewise excluded. Four patients were excluded on the grounds of insufficient data. This left us with 154 PDAC patient samples to analyze. Survival data and cause of death came from the Finnish Population Registry, from Statistics Finland, and from hospital records.
Surgical tissue samples were stored fixed in formalin and were embedded in paraffin at the Department of Pathology, Helsinki University Hospital. These archived samples were deparaffinized in xylene and rehydrated with a decreasing concentration of ethanol. Subsequently, they were stained with hematoxylin and eosin. All tumor samples were re-evaluated by experienced pathologists to ensure correct PDAC diagnosis.
Multipunch tissue microarray blocks (TMAs) were prepared from the tissue samples. First, representative regions were chosen and marked on the hematoxylin–eosin stained slides, and with their guidance, 1.0 mm cores were cut from the paraffin blocks with a semiautomatic tissue microarrayer (Tissue Arrayer 1, Beecher Instruments Inc., Silver Spring, MD, USA). Six cores were taken from each patient to optimize TMA representativeness.
The TMA blocks were cut into 4 μm sections and then deparaffinized and rehydrated. The slides were then treated in Tris-HCL (pH 8.5) buffer for 20 min at 98°C and incubated at room temperature for 18 h in the appropriate antibody solution. For TLR2, rabbit polyclonal antibody TLR2 (H-175): sc-10739 (Santa Cruz Biotechnology, Santa Cruz, CA, USA; 1:50) and for TLR4, rabbit polyclonal antibody TLR4 (H-80): sc-10741 (Santa Cruz Biotechnology; 1:50) were used.
Expression of TLR2 and TLR4 in PDAC tissue was assessed by the intensity of the staining and scored on a scale from 0 to 3, where 0 indicated negative staining, and 1, 2, and 3 indicated mild, moderate, and strong staining, respectively. Staining intensity was evaluated by two independent observers (M.A.L. and J.K.H.) and in cases with different scores, discussion led to consensus. The initial interobserver agreement was evaluated using Cohen’s kappa coefficient.
Staining results were matched against clinicopathological parameters with Fisher’s test. We applied the Kaplan–Meier method and log-rank test for survival analysis. The Cox regression proportional hazard model served for univariate and multivariate analyses, and the hazard ratios (HRs) were calculated for disease-specific overall survival. The Cox model assumption of constant HR over time was tested by including time-dependent covariates separately for each variable. The inclusion of covariates was based on clinical relevance. The HRs for TLR4 were analyzed during two time periods from 0 to 3.75 years and later on, by dividing the time axis to meet the Cox model assumption. Exact 95% confidence intervals (CIs) were calculated for survival rates. Spearman’s rho correlation coefficient served to assess the correlation between TLR scores and clinical parameters. Due to the low number of TLR-negative patients, TLR scores 0 and 1 were combined for statistical purposes for both TLR2 and TLR4. If a patient had different scores for different samples, the highest was the choice for that patient. Statistical analyses were performed with SPSS version 24.0 (IBM SPSS Statistics, version 24.0; SPSS, Inc., Chicago, IL, USA, an IBM Company), and with SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
This study was approved by the Helsinki University Hospital Surgical Ethics Committee (document number HUS 226/E6/06, additional petition TMK02 § 66/2013) and the Finnish National Supervisory Authority for Welfare and Health (document number 1004/06.01.03.01/2012).
Results
We obtained successful TLR2 scores from 153 patients and TLR4 scores from 152 patients. Both TLR2 and TLR4 were stained evenly in the cytoplasm with no distinctive membranous staining. In most samples, healthy pancreatic epithelium remained negative, although in some samples even normal pancreatic ductal epithelium showed positivity. In two TLR4 samples, healthy epithelium was positive, whereas cancerous tissue was not, but with no distinctive nuclear staining (Figure 1). Before reaching unanimity, the initial interobserver agreement was κ = 0.48 for TLR2 and κ = 0.52 for TLR4.

Toll-like receptor 2 (TLR2) and Toll-like receptor 4 (TLR4) staining. Immunopositivity scoring: For TLR2, (a–d) show negative, mild, moderate, and strong staining. For TLR4, (e–h) show negative, mild, moderate, and strong staining.
Strong TLR2 expression was observable in 51 (34%) patients and strong TLR4 expression in 50 (33%) patients. Expression of neither marker associated directly with patient survival when all data were analyzed.
However, survival advantage of strong TLR2 and TLR4 expressions was clearly noticeable during the first few years after surgery, in different subpopulations. Among patients with small (≤30 mm) tumors, the cumulative survival of those with negative or mild TLR2 expression at 1 year after surgery was 42% (95% CI = 15%–67%), whereas among those with moderate TLR2 expression, it was 81% (95% CI = 64%–90%), and with strong TLR2 expression, 82% (95% CI = 59%–93%); Sidak-corrected log rank for strong versus negative or mild TLR2 expression p = 0.032. Likewise, among patients with lymph-node-negative disease, survival at 1 year after surgery with negative or mild TLR4 expression was 50% (95% CI = 15%–78%), with moderate TLR4 expression 84% (95% CI = 63%–94%), and with strong TLR4 expression 93% (95% CI = 61%–99%); Sidak-corrected log-rank for strong versus negative or mild TLR4 expression p = 0.11 (Figure 2).
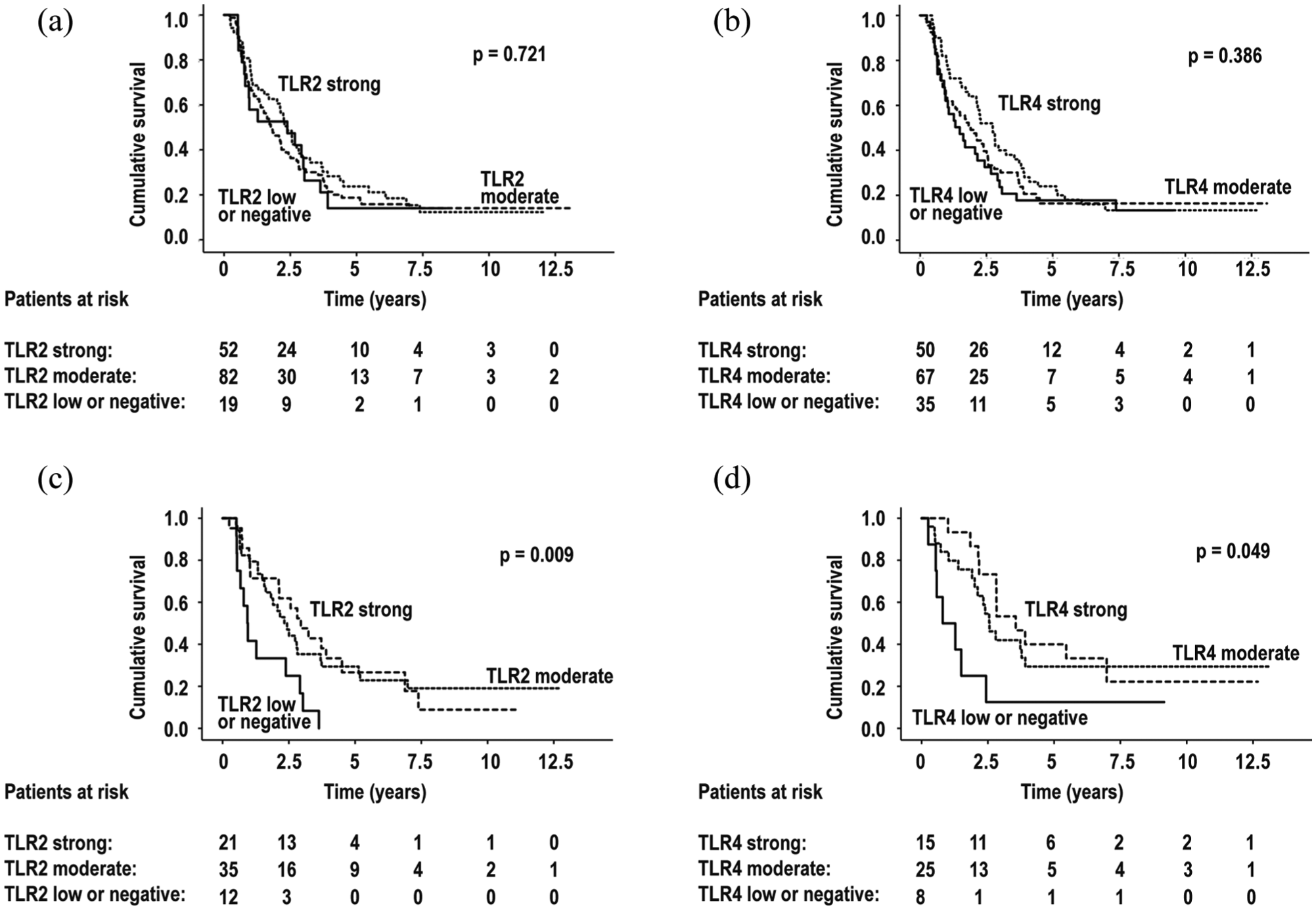
Survival in all patients, grouped by (a) Toll-like receptor 2 (TLR2) expression and (b) Toll-like receptor 4 (TLR4) expression. (c) Survival in patients with small tumor size (≤30 mm), grouped by TLR2 expression. (d) Survival with lymph-node-negative disease, grouped by TLR4 expression.
No significant survival differences between different TLR2 groups emerged when patients with tumor size >30 mm were added to the analysis, as neither with TLR4 groups when patients with lymph-node-positive disease were included. A statistically significant weak correlation appeared between TLR2 and TLR4 expressions, ρ = 0.28, p = 0.001, but no correlation between TLR expression and age, gender (Tables 1 and 2), or C-reactive protein (CRP) level emerged (for TLR2 and CRP ρ = 0.071, p = 0.424; for TLR4 and CRP ρ = 0.111, p = 0.213).
Associations between Toll-like receptor 2 (TLR2) immunoexpression and clinicopathological parameters, n = 153.

AJCC: American Joint Committee on Cancer.
p values <0.05 in bold. Correlation evaluated by linear-by-linear association test.
Staging according to AJCC 7th edition.
Associations between Toll-like receptor 4 (TLR4) immunoexpression and clinicopathological parameters, n = 152.

AJCC: American Joint Committee on Cancer.
Correlation evaluated by linear-by-linear association test.
Staging according to AJCC 7th edition.
Multivariate analysis showed that strong (HR = 0.30; 95% CI = 0.13–0.69; p = 0.005) or moderate (HR = 0.42; 95% CI = 0.20–0.88; p = 0.021) TLR2 expression in patients with tumor size 30 mm or less showed an overall survival benefit over patients with low or negative TLR2 expression. In patients with lymph-node-negative disease, strong (HR = 0.21; 95% CI = 0.07–0.65; p = 0.006) and moderate (HR = 0.37; 95% CI = 0.15–0.91; p = 0.030) TLR4 expression provided more survival benefit than low or negative TLR4 expression during the first 3.75 years (Tables 3 and 4). This same survival benefit was also noticeable in multivariate 5-year survival analysis for both TLR2 and TLR4 (see supplementary tables 5–7). All the HRs were calculated for disease-specific overall survival unless otherwise specified.
Univariate and multivariate survival analyses of patients with pancreatic ductal adenocarcinoma according to clinicopathological parameters and TLR2 and TLR4 immunoexpression.
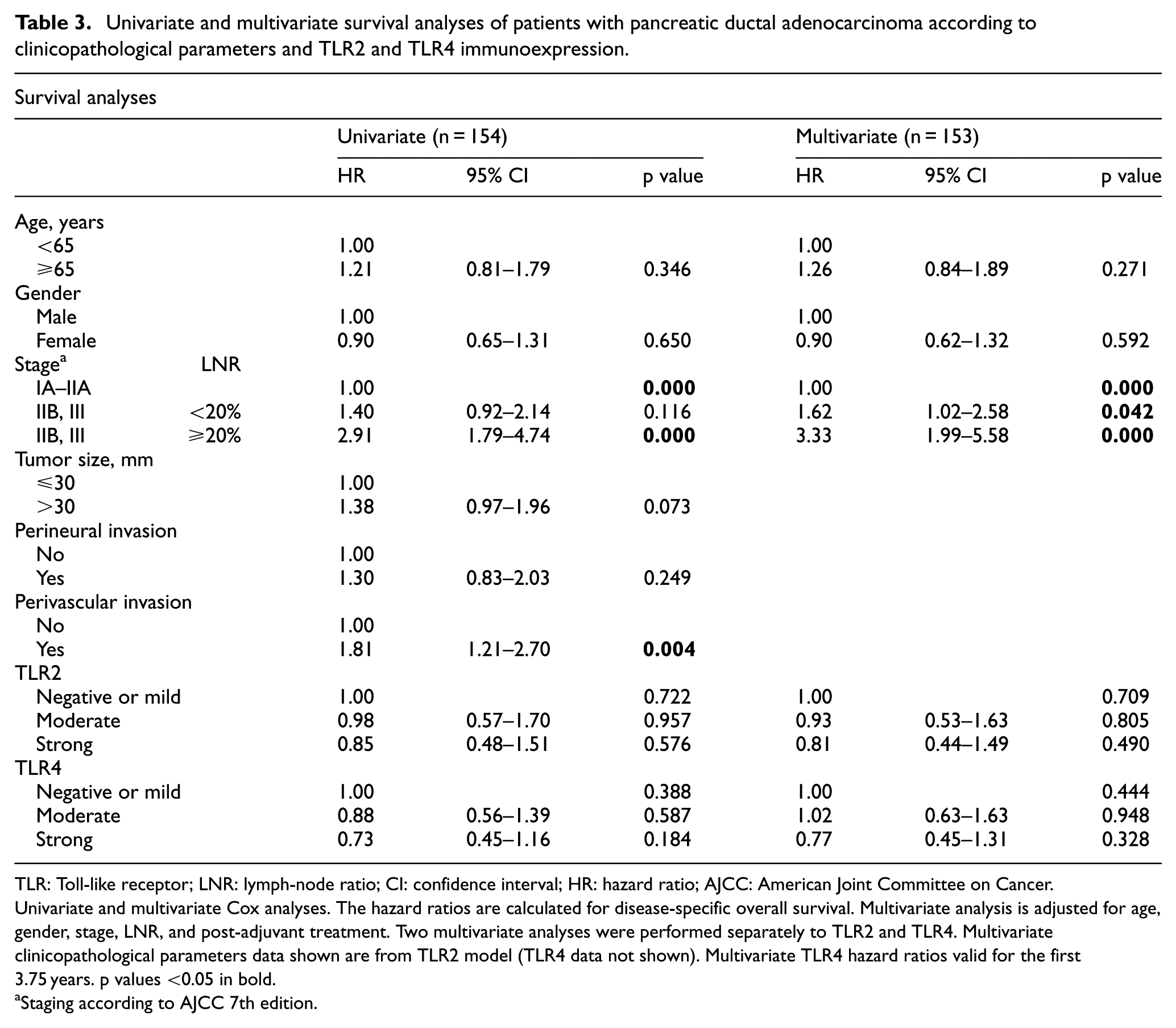
TLR: Toll-like receptor; LNR: lymph-node ratio; CI: confidence interval; HR: hazard ratio; AJCC: American Joint Committee on Cancer.
Univariate and multivariate Cox analyses. The hazard ratios are calculated for disease-specific overall survival. Multivariate analysis is adjusted for age, gender, stage, LNR, and post-adjuvant treatment. Two multivariate analyses were performed separately to TLR2 and TLR4. Multivariate clinicopathological parameters data shown are from TLR2 model (TLR4 data not shown). Multivariate TLR4 hazard ratios valid for the first 3.75 years. p values <0.05 in bold.
Staging according to AJCC 7th edition.
Multivariate sub-group analysis of pancreatic ductal adenocarcinoma patients with tumor size ≤30 mm and lymph-node-negativity, according to TLR2 and TLR4 immunoexpression.

TLR: Toll-like receptor; LNR: lymph-node ratio; CI: confidence interval; AJCC: American Joint Committee on Cancer.
Multivariate Cox analysis. Multivariate analysis adjusted for age, gender, stage, LNR, and post-adjuvant treatment. Multivariate TLR4 hazard ratios valid for the first 3.75 years. The hazard ratios are calculated for disease-specific overall survival. p values <0.05 in bold.
Staging according to AJCC 7th edition.
Discussion
We found strong TLR2 and TLR4 expressions independently to indicate better prognosis in PDAC patients with stage I–II disease. To our knowledge, we report for the first time the expression of TLR2 in PDAC. The expression of TLR4 and its association with prognosis in PDAC has been reported earlier in two studies.14,15
In PDAC, TLR4 expression has been associated with more advanced disease and poor patient outcome, and pancreatic TLR4 expression has been higher in cancerous cells than in normal epithelial cells. A positive association between TLR4 expression and tumor size, lymph-node involvement, venous invasion, and pathological stage has also appeared. 14 According to that 2010 report, TLR4 was not identifiable as an independent prognostic factor—its associated features such as tumor size, disease stage, and venous invasion carry a heavy prognostic load themselves, and stage I–II patients were not a focus of that study. The study consisted only of 65 PDAC patients. Our relatively large series of 154 patients allowed us to focus more on stage I–II patients. Our study also differed from that 2010 report in the methods: we used TMA and immunohistochemistry (IHC), whereas they measured TLR4 mRNA by real-time PCR and TLR4 protein immunoexpression. We used the same antibody. In our samples, immunopositivity was primarily cytoplasmic, while in their work, the cell membranes also highlighted. The reason for this difference in staining pattern is unclear.
A recent study did not find any prognostic value for TLR2 and TLR4 in PDAC in 65 resected PDAC patients. 15 They evaluated TLR2 and TLR4 expressions in PDAC by IHC as did we, but their approach was different. They evaluated separately membranous, nuclear, and cytoplasmic staining and the proportion of affected epithelial cancer cells. They used these data to calculate a histoscore for each type of staining. They found no survival benefit for membranous, nuclear, or cytoplasmic staining. The staining patterns in their results were different from ours: they noted cytoplasmic staining in all PDAC cells for both TLR2 and TLR4, and membranous staining for some of the cells (24/65 for TLR2; 25/65 for TLR4). They also noted nuclear staining in their samples (42/65 for TLR2, 5/65 for TLR4). In our results, staining was cytoplasmic with no distinctive membranous or nuclear staining, and we also found PDAC cells unstained in the cytoplasm for TLR2 and TLR4. The difference in staining pattern may result from different antibodies used: theirs were from Abnova, whereas ours were from Santa Cruz. As the patient number in their study was markedly lower than in ours, sub-group analysis was not possible to the same extent as in our work.
Given the setting, it was intriguing to find an association between expression of TLR2 and TLR4 and better prognosis in stage I–II PDAC patients. Most earlier studies have demonstrated a TLR association with poor prognosis in various cancers. In hepatocellular carcinoma, TLR4 expression has not only been linked to metastatic disease,16,17 and in colorectal cancer to poorer prognosis, 11 but interestingly also to non-metastatic disease. 18 In esophageal cancer, the association between TLR4 and prognosis was negative. 19 TLR2 and TLR4 have also been studied in squamous cell carcinoma of the tongue, where both have been linked to invasion. 12 In Merkel cell carcinoma, cytoplasmic TLR2 has been linked to small tumor size, but nuclear TLR2 expression to larger tumors, and nuclear TLR4 expression to older age. However, no prognostic value of TLRs emerged in Merkel cell carcinomas. 20
TLR2 has been studied less widely than TLR4, and apart from its association with small tumor size in Merkel cell carcinoma, to our knowledge, no results similar to ours have appeared. We wish to see further investigations elucidating whether increased TLR2 expression shows any positive prognostic value in patients with diverse cancers of small tumor size. Similarly, to our knowledge, no reports of divergent significance have involved TLR4 expression in patients with lymph-node-negative disease compared to expression in patients who are lymph-node-positive. What would be worthwhile would be to investigate whether similar phenomena occur in other cancers, as well.
At diagnosis, only 15%–20% of diagnosed PDACs are operable,2,3 which skews our patient pool. From most patients, no histological specimen or biopsy is available for screening of tissue biomarkers. In addition, our positive findings concerned only those fortunate patients with local disease and are not necessarily applicable to those with more advanced, non-operable disease. Nevertheless, we hope that these results may be of help in developing specific, post-operative treatment options for patients with various cancer-related immunological profiles, small tumors, and those with lymph-node-negative disease.
TLRs initiate and strengthen the local inflammation response, which may help create a tumor-promoting microenvironment through anti-apoptotic, angiogenic, and growth-stimulating factors. 6 Cell debris from tumor-induced necrosis can activate TLRs, leading to a self-perpetuating cycle resulting in increased necrosis and inflammation, thus encouraging tumor progression and metastasis. This process could help explain the negative association between strong TLR expression and cancer survival in general, but our findings in PDAC are contradictory.
One reason for our findings may be the exceptional aggressiveness of PDAC—we only had a negligible number of truly TLR-negative PDAC patients, which we combined with score-1 patients for statistical purposes. If negative TLR2 or TLR4 expression has a positive association with PDAC prognosis, it may not have come to light in our material. Another possible explanation is that the circumstances of stage I–II PDAC simply favor more the antitumor effects of TLRs.
7
A notable clinical illustration of the antitumor effects of TLR2 and TLR4 is
The TMA technique permits the use of a larger number of patients. Its most notable downside is that only certain areas of the tumor can be evaluated, allowing sampling error. To counter this effect, we took six cores from various parts of the tumor, both from its invasive front and its center, thus minimizing potential error. IHC evaluation is subjective by nature. Our interobserver agreement was not ideal, and this may influence our results. To minimize this, we re-evaluated and discussed all differing scores after independent evaluation.
Considering the low incidence of operable PDAC, our patient number is quite large. We excluded patients receiving neoadjuvant therapy on the premise that it may alter the tumor microenvironment and skew results. Many of our patients were treated earlier in the 2000s, when neoadjuvant therapy was less common, which left us with a unique opportunity to include inflammation markers in tumor tissue unaffected by chemo- or radiotherapy.
In conclusion, here we show that in PDAC, TLR2 and TLR4 expressions correlate with better prognosis in patients with small tumor size and lymph-node-negative disease. Further investigation needs to validate these results.
Supplemental Material
Supplementary_tables – Supplemental material for Toll-like receptor 2 and Toll-like receptor 4 predict favorable prognosis in local pancreatic cancer
Supplemental material, Supplementary_tables for Toll-like receptor 2 and Toll-like receptor 4 predict favorable prognosis in local pancreatic cancer by Mira A Lanki, Hanna E Seppänen, Harri K Mustonen, Camilla Böckelman, Anne T Juuti, Jaana K Hagström and Caj H Haglund in Tumor Biology
Footnotes
Acknowledgements
We thank the laboratory staff and our research assistant Elina Aspiala for their contribution to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Finnish State Subsidy Fund, the Finnish National Research Foundation, the Finnish Cancer Foundation, the Sigrid Jusélius Foundation, the Mary and Georg C. Ehrnrooths Foundation, Medicinska Understödsföreningen Liv och Hälsa, and Finska Läkaresällskapet.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
