Abstract
The limited efficacy of conventional therapies for pancreatic ductal adenocarcinoma has led to the growing interest for identifying potential antigenic targets for immunotherapy. Placenta-specific 1 (PLAC1) is a new member of cancer-testis antigens with restricted expression in normal tissues. Ectopic activation of PLAC1 has been found in different types of cancers, but its role in pancreatic ductal adenocarcinoma remains unknown. This study evaluated the protein expression of PLAC1 and its clinical significance in pancreatic ductal adenocarcinoma. We examined PLAC1 expression in 93 pancreatic ductal adenocarcinoma samples by immunohistochemistry. The expression of PLAC1 was detected in 41 (44.1%) patients. Among patients’ clinicopathological characteristics, PLAC1 expression was only significantly correlated with tumor differentiation (p = 0.028). Univariate analysis revealed that PLAC1 expression (p = 0.016) and tumor differentiation (p = 0.003) were significantly correlated with poor survival in the whole cohort. Subgroup analysis showed that PLAC1 expression was an independent prognostic biomarker in the perineural invasion positive subgroup (p < 0.05). This study demonstrated that the protein expression of PLAC1 was significantly associated with decreased overall survival in patients with pancreatic ductal adenocarcinoma, indicating that it was a valuable prognostic marker for pancreatic ductal adenocarcinoma and might be a potential target for immunotherapy.
Keywords
Introduction
Pancreatic ductal adenocarcinoma (PDAC), which comprises more than 90% of pancreatic cancer, is a highly lethal malignant tumor, whose death rates almost match incidence. 1 Though being the 12 most commonly diagnosed cancer worldwide, PDAC is the sixth leading cause of cancer-related death in China with a 5-year survival rate of less than 5%.2–4 Due to the lack of early symptoms and aggressive behavior of the decease, PDAC is often at an advanced stage when diagnosed, with only 15%–20% of patients being eligible for curative resection. 5 Furthermore, compared to other cancers, it shows marked resistance to chemotherapy and radiotherapy, leading to the exceptionally poor prognosis of the decease. Therefore, novel therapeutic strategies such as tumor-specific immunotherapy for PDAC are highly required to impact pancreatic cancer treatment and to extend and save PDAC patients’ lives.
Although great efforts have been made to identify tumor antigens in PDAC, many of these antigens are not only overexpressed in cancer cells but also can be found in benign cells, which restricts their application as antigenic targets for tumor-specific immunotherapy. One promising method which can overcome the restriction is to identify tumor-specific antigens, which are barely or limitedly expressed in somatic tissues. Placenta-specific 1 (PLAC1), as one of the cancer-testis (CT) antigens, is a novel tumor marker whose expression is limited predominantly to the placenta.6,7 Relatively less amount of PLAC1 expression have been discovered in cerebellum and testis, while no expression of PLAC1 is found in other human somatic tissues. 7 The PLAC1 gene is localized on the X chromosome (Xq26.3), the region where CT genes frequently present. 8 As demonstrated by topology analysis, PLAC1 is a transmembrane protein, indicating that it may be targeted to the membranous compartment.9,10 Though the function of PLAC1 remains poorly understood, available evidences indicate that it is involved in embryonic and fetal development.11,12 In recent years, escaping transcriptional repression and ectopic activation of PLAC1 have been found in various tumors including lung, 7 breast, 13 gastric, 14 liver, 15 colorectal, 16 ovarian, 17 and prostate cancers 18 and cancer cell lines. 7 In vitro, knockdown of PLAC1 in the breast cell lines led to the inhibition of malignant phenotypes, such as proliferation, invasion, and migration. 13 The clinical correlation of PLAC1 expression was found in different cancer types, especially in gastrointestinal cancers. Liu et al. 19 demonstrated that the nuclear staining of PLAC1 was correlated with tumor grade, lymph node metastasis, and tumor–node–metastasis (TNM) stage in colorectal carcinoma. For gastric cancer, PLAC1 expression was inversely associated with patient survival. 14 Additionally, the specific immune response against PLAC1 has been reported in hepatocellular, 15 colon, 20 and gastric 21 cancer patients.
Though the expression of PLAC1 has been observed in many cancers, the role of it in PDAC has not yet been reported. Therefore, our study evaluated PLAC1 expression in 93 patients with PDAC by immunohistochemical staining and analyzed the correlation of PLAC1 expression between patient survival and clinicopathological characteristics of patients with PDAC1.
Materials and methods
Patients and tissue specimens
Paraffin-embedded samples were obtained from 93 PDAC patients who underwent tumor resection at Fuzhou Dongfang Hospital and Xijing Hospital from 2005 to 2014. Patients were included in this study when preoperative clinical data were complete and follow-up information was available. Patients who did not survive more than 2 months after the operation were excluded to eliminate the interference of perioperative death. Additionally, none of the patients had any history of anticancer treatments prior to surgery. Follow-up period ranged from 1 month to 5 years after operation, and the latest date of follow-up was 5 March 2016. TNM stage was defined in accordance with the seventh edition of American Joint Committee on Cancer (AJCC) staging manual. Ethical permission for our study was granted by the Ethics Council of Fuzhou Dongfang Hospital and Xijing Hospital.
Immunohistochemistry
In all, 93 paraffin-embedded PDAC specimens were subjected to PLAC1 immunohistochemistry. Human placenta and benign pancreas served as positive and negative specimens, respectively. Paraffin tissue blocks were sectioned into 4 µm slides and then incubated overnight at 37°C. After that, tissue slices were deparaffinized with xylol and then rehydrated through decreasing concentrations of alcohol. After antigen retrieval (slices soaked in citrate buffer, PH 6, and subjected to high pressure cooking for 20 min at 120°C), the slides were treated with 3% H2O2 for endogenous peroxide blocking. Subsequently, sections were incubated with anti-PLAC1 polyclonal antibody (1:100 dilutions; no. ab131630, Abcam, USA) overnight at 4°C, followed by the immunohistochemical EliVision method (kit-0016; Fuzhou Maixin Biotech. Co., Ltd., China). After diaminobenzidine staining, sections were finally counterstained with hematoxylin, rehydrated, and mounted.
Immunostaining evaluation and scoring
Immunohistochemistry assessment of PLAC1 expression was carried out blindly by two experienced pathologists, and all samples were scored independently by the semi-quantitative scoring method. Three independent microscopic fields (100×) were selected randomly for each sample, and all the tumor cells within each microscopic field were counted. The final score of each sample was determined by the average score of the three microscopic fields. Each slide was scored based on the staining intensity and percentage of stained cells (staining intensity: 0 = no staining, 1 = weak staining, 2 = moderate staining, 3 = strong staining; percentage of stained cells: score 0: <1%, score 1: 1%–24%, score 2: 25%–50%, score 3: >50%). Then, we multiplied the score of staining intensity by that of the percentage of stained cells as staining index: score <3 was considered as negative and ≥3 as positive.
Statistical analysis
Analyses of statistics were done with SPSS 19.0 software. To evaluate the correlations between PLAC1 expression and patients’ clinicopathological characteristics, Fisher’s exact text and χ2 test were applied. Kaplan–Meier curves were depicted to reflect the overall survival (OS) and the differences between the two curves were analyzed by log-rank test. Univariate and multivariate Cox regression analyses were performed to assess the prognostic significance of PLAC1 expression. The p value below 0.05 was regarded as statistically significant.
Results
Expression of PLAC1 protein in PDAC tissues and control specimens
In total, 93 tumor samples were evaluated for the expression of PLAC1 protein. Immunohistochemical analysis of PDAC specimens demonstrated that PLAC1 heterogeneously localized to the cytoplasm of the cancer cells (Figure 1). Among the 93 PDAC specimens, immunostaining for PLAC1 protein was detected in the cancer tissues of 41 (44.1%) cases, whereas no staining was present in adjacent non-cancer tissues. As for the control slides, cytoplasmic and microvillus membranous staining was observed in differentiated syncytiotrophoblast of human placenta, while the antibody exhibited no staining in normal pancreatic tissues.
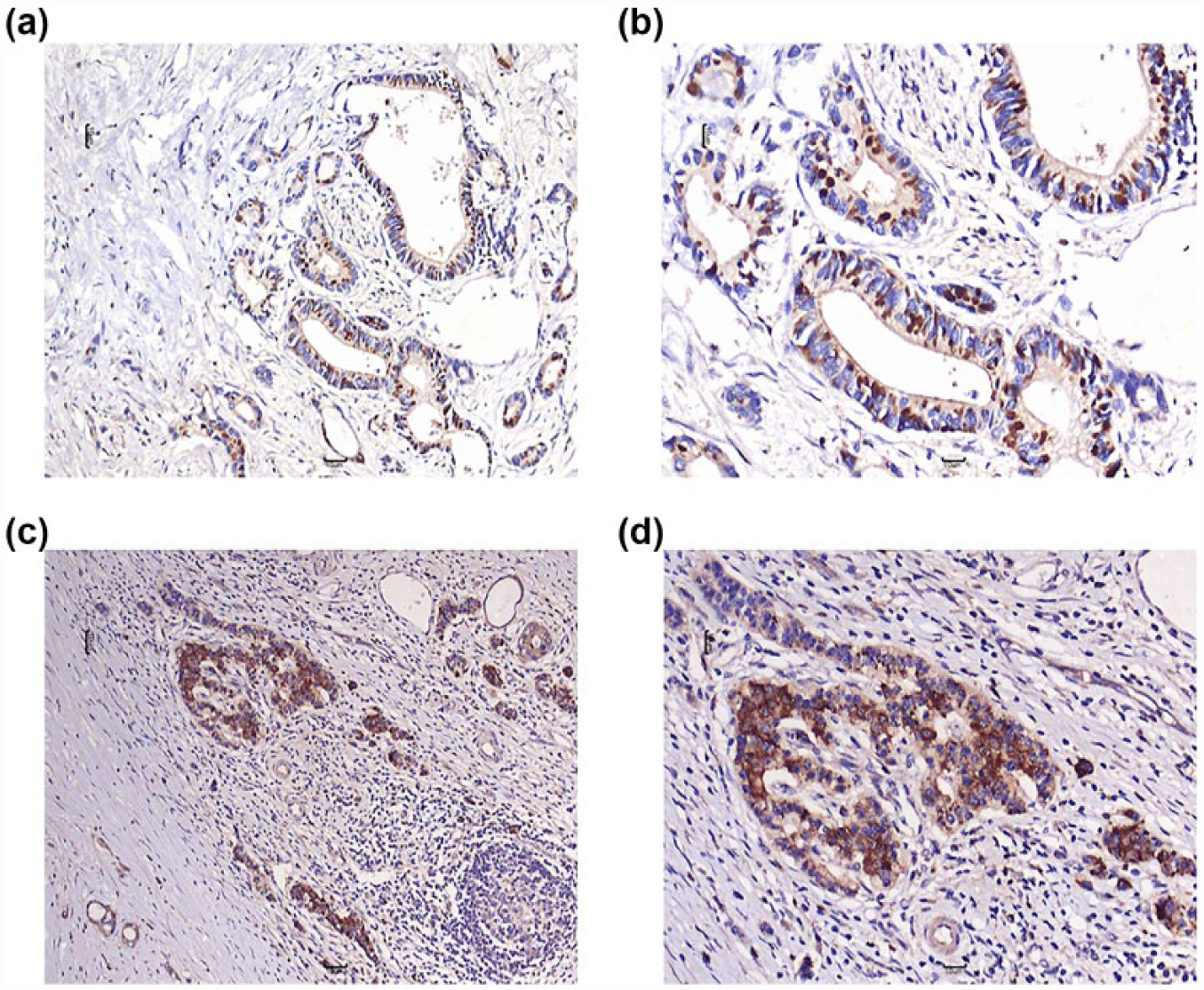
The protein expression of PLAC1 in PDAC tissues by immunohistochemistry. (a and b) Expression of PLAC1 in well-moderately differentiated PDAC (staining intensity: 3 = strong staining; (a) 100× and (b) 200×). (c and d) Expression of PLAC1 in poorly differentiated PDAC (staining intensity: 3 = strong staining; (c) 100× and (d) 200×). PLAC1 protein localized to the cytoplasm of the cancer cells and no staining was present in adjacent non-cancer tissues.
Correlation between PLAC1 expression and clinicopathological factors of patients with PDAC
All 93 patients were classified into two groups according to PLAC1 expression. Clinicopathological data of PLAC1-positive and negative patients were compared. The association between PLAC1 protein expression and clinicopathological factors of PDAC patients, including age, gender, TNM stage, tumor grade and size, T classification, lymph node metastasis, surgical margin, tumor location, and perineural invasion (PNI), was analyzed by chi-square test (Table 1). As shown in Table 1, PLAC1 expression was correlated with tumor grade (p = 0.028). The percentage of PLAC1-positive patients was 58.3% in poorly differentiated tumor group, compared with only 35.1% in the well-moderately differentiated tumor group. No correlation between PLAC1 protein expression and other clinicopathological prognostic parameters was found.
Association between PLAC1 protein expression and clinicopathological factors in patients with PDAC.

TNM: tumor–node–metastasis; PNI: perineural invasion.
Fisher’s exact text.
Significance of bold value is P < 0.05.
Association between PLAC1 expression and prognosis of PDAC patients
During the follow-up time, 87 patients had died of this cancer. To determine whether PLAC1 protein expression is correlated with the OS in these groups, Kaplan–Meier analysis was used. The Kaplan–Meier curves revealed that there was a significant difference between PLAC1-positive and -negative patients in the OS (p = 0.016, Figure 2). The median survival of PLAC1-negative patients was 15.1 months, while it was only 9.0 months for the patients with PLAC1-positive expression (Table 2). The OS time in the PLAC1-positive group was greatly shorter than that in the PLAC1-negative group, which indicated that PLAC1-positive patients had a poorer prognosis than PLAC1-negative patients.

Kaplan–Meier analysis for post-operative survival of PDAC patients: the OS of PLAC1-positive patients was shorter than that of PLAC1-negative patients.
Univariate and multivariate analyses.

TNM: tumor–node–metastasis; PNI: perineural invasion; HR: hazard ratio; CI: confidence interval.
Log-rank test; #Cox regression test.
Significance of bold value is P < 0.05.
According to univariate analysis, PLAC1 expression and tumor differentiation were significantly correlated with poor survival (p = 0.016 and p = 0.003, respectively). By multivariate analysis, only tumor differentiation was an independent prognostic indicator for poor survival (hazard ratio (HR) = 2.357, 95% confidence interval (CI): 1.310–4.241, p = 0.004).
Influences of PLAC1 protein expression on OS in PDAC subgroups
Patients were divided into 20 subgroups, according to 10 clinicopathological factors evaluated in our study. On univariate analysis, PLAC1 expression was a significant prognostic indicator for patients’ survival in five subgroups, that is, male patients, patients >58 years, T1–T2, PNI positive, and pancreatic head (p < 0.05, Figure 3). Multivariate analysis showed that PLAC1 expression was an independent prognostic indicator in the PNI-positive subgroup (p < 0.05, Table 3), while it was not in other four subgroups.

Univariate analyses of PLAC1 expression in five subgroups of PDAC patients: (a) male patients (p = 0.042), (b) patients with pancreatic head (p = 0.037), (c) patients with PNI > 4 cm (p = 0.008), (d) T1–T2 tumors (p = 0.046), and (e) patients ≥58 years (p = 0.015).
Multivariate Cox regression analyses of the prognostic relevance of PLAC1 expression in PDAC subgroups in which the poor overall survival of patients was significantly correlated with PLAC1 expression (identified by the log-rank test).
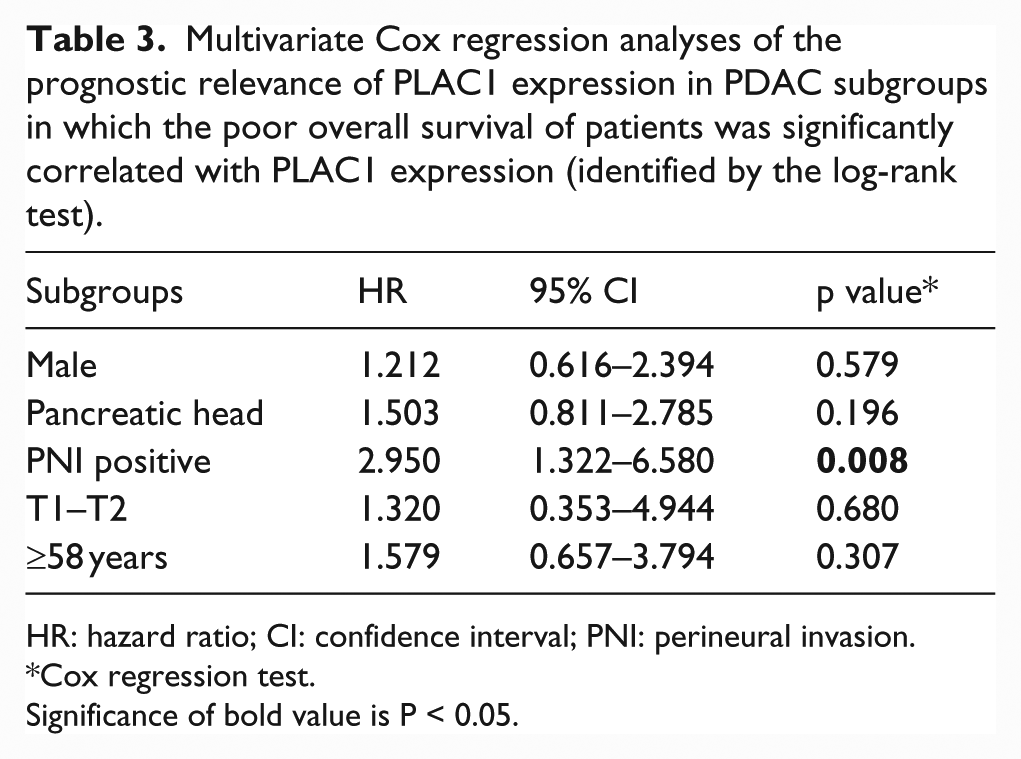
HR: hazard ratio; CI: confidence interval; PNI: perineural invasion.
Cox regression test.
Significance of bold value is P < 0.05.
Discussion
PDAC is a highly lethal neoplasm with extremely low survival rate. Currently, the main treatments for PDAC are surgery, chemotherapy, and radiation therapy. However, because of difficulties of early diagnosis and poor response to conventional forms of chemotherapy and radiotherapy, the overall clinical performance of these treatments is disappointing. Hence, looking for new therapeutic methods for PDAC is of great importance for conquering the disease.
In recent decades, immunotherapy provides new options for PDAC treatment owing to its specific antitumor effects and low non-specific cellular toxicity. Detection of novel biomarkers which can be used as potential targets for tumor-specific immunotherapy is a prerequisite. CT antigens, whose expressions are mostly restricted to testis, placenta, fetal ovary, and a subset of cancers,22,23 are the main category of tumor-specific antigens. They are promising antigens not only for antibody-based immunotherapy but also for tumor vaccines. Since the first CT antigen gene MAGE-1 was identified by van der Bruggen et al., 24 more than 200 CT antigen genes have been found so far. 25 Though immunotherapies based on some CT antigens, such as NY-ESO-1 and MAGE-3, have shown clinical benefits for some cancer patients,26–29 the data of published clinical trials so far cannot support immunotherapy to become the standard treatment for these cancers. Immune selection is a main problem of immune-based therapy for malignancies. One potential method to overcome the obstacle is to develop polyvalent vaccines that target multiple tumor-specific antigens. Therefore, screening to detect more tumor-specific antigens is of great importance. Another important function of tumor-specific antigens is that they can be used as tumor markers to aid the diagnosis of tumor and help to predict the outcomes of cancer patients. Unfortunately, few pancreatic tumor-specific antigens have been detected so far. Therefore, identifying more pancreatic tumor-specific antigens is crucial to the prognosis and treatment of pancreatic cancer.
As a new member of CT antigens, PLAC1 has been regarded as an attractive candidate for vaccine-based immunotherapy because of its restricted expression in normal tissues as well as its ectopic expression in various human malignancies. PLAC1 has been demonstrated to be immunogenic in cancer patients, eliciting both cell-mediated and humoral immune responses. According to the report by Liu et al., of PLAC1 messenger RNA (mRNA) positive colon cancer patients, more than half have shown to have PLAC1-responsive CD8+ and CD4+ T cells. They also detected four new HLA-A2-restricted CD8+ T-cell epitopes derived from PLAC1 in colon cancer.19,20 Additionally, Liu et al. 30 identified a new HLA-A*0201-restricted T-cell epitope, p28, from PLAC1 in breast cancer. Compared with cellular immunity, humoral immune responses of PLAC1 have been investigated more broadly. It has been reported that anti-PLAC1 antibody has been detected in the sera of 6.2% (14/226) of non-small-cell lung carcinoma patients, 7 28.5% (8/28) of gastric cancer patients, 21 3.9% (4/101) of hepatocellular cancer (HCC) patients, 15 and 13.6% (6/44) of colon cancer patients. 20 The high frequency of coordinated specific immune responses makes PLAC1 a very promising target for immunotherapy. Besides, PLAC1 is a useful biomarker for predicting outcomes of some cancer patients. The expression of PLAC1 was correlated with poorer prognosis in gastric carcinoma patients. 14 In the recent report about prostate adenocarcinoma, PLAC1 expression was positively correlated with Gleason score, suggesting that it was a biomarker of poorly differentiated prostate cancer cells. 18
In our study, we selected 93 cases of patients with PDAC and analyzed PLAC1 protein expression by immunohistochemical staining. The results showed that PLAC1 protein was expressed in 44% of our patient cohort, while no staining was observed in the adjacent non-cancer tissues, indicating that it might be a valuable target for immunotherapy of PDAC. Intriguingly, like most CT antigens, PLAC1 protein is expressed heterogeneously in PDAC tissues. There was at times a little clump of cancer cells with strong staining among a background where PLAC1-negative cancer cells were more than 99%. This heterogeneous expression profile implied that the activation of PLAC1 might be epigenetic, for instance, because of the influence of DNA methylation. However, the mechanism still needs further study. We also discovered that PLAC1 expression was correlated with decreased patient survival. Patients whose tumor expressed PLAC1 had a lower survival in comparison with PLAC1-negative patients. According to these results, immunotherapy based on PLAC1 antigen could potentially extend patients’ lives and improve the survival of patients whose tumor expressed PLAC1. In addition, univariate analysis indicated that in the whole cohort, PLAC1 and tumor differentiation were significant prognostic indicators for poor survival. Though only tumor differentiation remained a significant prognostic indicator by multivariate analysis (Table 2), there was a strong relation between PLAC1 protein expression and tumor differentiation. In all, 58.3% of poorly differentiated tumor demonstrated PLAC1 protein expression, whereas only 35.1% of well-moderately differentiated tumors were PLAC1 positive. This suggests that PLAC1 protein may conduce to poor survival through its predominant expression in poorly differentiated tumors. In the research of Koslowski et al., 13 knockdown of PLAC1 was associated with reduced phosphorylation of AKT kinase in breast cell lines, suggesting PLAC1 contributed to some malignant phenotypes via a phosphatidylinositol 3-kinase (PI3K)/AKT-dependent pathway. This may also be the mechanism of PLAC1 expression affecting the survival of PDAC patients. Further studies about the signal mechanisms of PLAC1 in PDAC are needed. Subgroup analyses revealed that PLAC1 expression was a statistical prognosticator of poor OS in five subgroups (male patients, patients >58 years, pancreatic head, PNI positive, and T1–T2). Especially in the PNI-positive subgroup, PLAC1 expression was demonstrated to be an independent prognostic indicator for poor outcomes.
Conclusion
This study demonstrated that the expression of PLAC1, which was limitedly expressed in normal tissues, was significantly correlated with worse survival in patients with PDAC, indicating that it might be a useful marker for identifying PDAC patients with poor prognosis and an attractive target for immunotherapy. However, there are still several limitations in this study. The patients enrolled in our study were all eligible for curative resection, which may not represent the whole patients with PDAC, who often present with advanced stages when surgery resection is no longer possible. PLAC1 may be a valuable target for immunotherapy, so further study about the immunogenicity of PLAC1 in patients with PDAC is also needed.
Footnotes
Acknowledgements
Y.Y. and X.Z. contributed equally to this work.
Compliance with ethical standards
The study was approved by the Ethics Committee of Fuzhou Dongfang Hospital and the Ethics Committee of Xijing Hospital.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Natural Science Foundation of Fujian Province (Nos 2016J01576 and 2016J01586 and 2017J05121).
