Abstract
Expression of regenerating islet-derived protein 4 (REG4), a secretory protein involved in cell differentiation and proliferation, is upregulated in inflammatory bowel diseases and in many gastrointestinal malignancies. The prognostic significance of its expression in pancreatic ductal adenocarcinoma is unknown. Our aim was to investigate tumor tissue and serum REG4 expression in pancreatic ductal adenocarcinoma patients. We also evaluated as a control the diagnostic value of serum REG4 level in patients with chronic pancreatitis. Immunohistochemical expression of REG4 was evaluated in 154 surgical specimens and serum REG4 level in 130 samples from pancreatic ductal adenocarcinoma patients treated at Helsinki University Hospital, Finland, in 2000–2011. REG4 tissue and serum expression was assessed in relation to clinicopathological parameters and patient survival. A chronic pancreatitis control group comprised 34 patients who underwent pancreatic resection because of suspicion of malignancy. Significant survival differences were detectable in subgroups: in tumor stages IA–IIA, high serum REG4 level predicted worse survival (p=0.046). In patients with grade I tumor, positive tissue REG4 expression predicted better survival (p=0.006). In multivariate analysis, neither tissue nor serum REG4 expression was independent prognostic factors. Serum REG4 levels were higher in pancreatic ductal adenocarcinoma than in chronic pancreatitis (p=0.002), with diagnostic sensitivity of 45% and specificity of 91%. In logistic regression analysis, a multivariate model with REG4, CA19-9, and age provided sensitivity of 82% and specificity of 79%. REG4 tissue expression is a prognostic marker in subgroups of pancreatic ductal adenocarcinoma patients. Serum REG4 level might be useful in differential diagnosis between pancreatic ductal adenocarcinoma and chronic pancreatitis.
Keywords
Introduction
With over 40,000 annual deaths in the United States, and with a devastating overall 5-year survival under 5%, 1 pancreatic ductal adenocarcinoma (PDAC) is one of the most lethal malignancies. Radical surgical resection with or without chemotherapy is the only treatment for cure but is possible in only 20% of patients. 2 Despite extensive research on potential biomarkers, since CA19-9, none has deserved a role in clinical praxis. Identification of new biomarkers is imperative to predict PDAC patient outcome more precisely and to advance our knowledge of the molecular mechanisms behind this disease.3,4
REG4 in cancer
The regenerating islet-derived (REG) proteins are a group of small secretory proteins involved in regulation of cell regeneration and proliferation.5,6 Among the four REG families (I–IV), REG4 is the most recently discovered member. In 2001, it was identified and isolated from a complementary DNA (cDNA) library of ulcerative colitis tissue. 7 REG4 is physiologically expressed in the colon and small intestine, where it is highly expressed in enteroendocrine cells,8,9 but not in the pancreatic islets.
Upregulation of REG4 expression occurs in inflammatory bowel diseases (IBD),7,8 but its expression is also increased in many gastrointestinal malignancies such as colorectal, 10 gastric, 11 or pancreatic cancer. 12 Evidence exists that REG4 is expressed more strongly in colorectal tumors and normal small intestine than in normal colorectal tissue. 13 Zhang et al. 14 proposed that overexpression of REG4 may be an early event in colorectal carcinogenesis, showing that REG4 expression was higher in dysplastic adenomas than in normal colorectal mucosa. REG4 is expected to play a role also in gastric carcinogenesis. 15
REG4 can serve as a diagnostic serum marker in colorectal cancer (CRC) with liver metastasis, but it does not detect early tumor stages. 10 Serum levels of REG4 are significantly higher in patients with gastric cancer than in healthy individuals at a sensitivity of 36%–73%, higher than that of CEA or CA19-9.11,16 High serum REG4 level can differentiate mucinous ovarian cancer from other ovarian cancer subtypes and may prove useful in follow-up of this disease. 17
REG4 in pancreatic cancer
Several reports show REG4 expression in pancreatic cancer cells to be increased above the level in normal pancreatic cells, and REG4 to promote invasiveness and proliferation of cancer cells.12,18,19 Moreover, REG4-expressing pancreatic tumors tend to grow larger, whereas knockdown of REG4 expression leads to shrinkage of the tumor or to impaired cancer cell growth in vivo and in vitro.12,20 More intense resistance to radiation and chemotherapy (gemcitabine) occurs in pancreatic cancer cell models in vivo and in vitro along with REG4 expression.20,21 Recently, Li et al. 22 demonstrated that tissue REG1A/B expression correlates with prognosis in PDAC, but did not report on the prognostic value of tissue REG4 expression. To date, no studies reveal the prognostic value of REG4 expression in PDAC.
Elevated REG4 serum levels and positive immunohistochemical staining are present in PDAC patients, suggesting that REG4 may serve as a diagnostic marker.12,22,23 Differential diagnosis between PDAC and chronic pancreatitis (CP) can be challenging, especially in patients with pancreatic mass which can prove either benign or malignant. Reliable preoperative diagnosis would be beneficial for patients possibly making unnecessary and extensive surgery avoidable. CA19-9, the standard serum-based marker for diagnosis of PDAC, 24 has, however, diagnostic limitations, since it can be normal in patients with localized disease or high in patients with benign diseases including CP.
In this study, we evaluated the prognostic significance of REG4 tissue expression and serum level in PDAC and also analyzed serum REG4 levels in a control series comprising patients with CP.
Methods
Patients
Of the 188 patients with PDAC who underwent surgery between 2000 and 2011 at the Department of Surgery, Helsinki University Hospital, Finland, we excluded 34: 22 who received neoadjuvant chemotherapy, 8 who were diagnosed with stage IV disease, and 4 in whom stage was not reliably determinable. Median age at surgery was 64 (range: 39–83) years and median follow-up was 2.0 (range 0.2–13.1) years. The control group for serum REG4 analysis comprised 34 patients with histopathologically verified CP who, between 2000 and 2008, underwent pancreatic resection because of suspicion of malignancy. Median age at surgery for this group was 54 (range: 35–74) years. Clinical data came from patient records, survival data from the Finnish Population Registry, and cause of death from Statistics Finland.
Tissue microarray blocks and immunohistochemistry
Formalin-fixed and paraffin-embedded surgical tissue samples came from the archives of the Department of Pathology, Helsinki University Hospital. All samples were re-evaluated by experienced pathologists for confirmation of the histopathological diagnosis of PDAC. To prepare tissue microarray blocks (TMA), representative areas of tumor samples were defined and marked on hematoxylin- and eosin-stained slides. Six 1.0-mm-diameter punches from both the invasive front and from the center part of the tumor were taken from each tumor sample with a semiautomatic tissue microarrayer (Tissue Arrayer 1, Beecher Instruments Inc., Silver Spring, MD, USA). 25
TMA blocks were freshly cut into 4-μm sections. After deparaffinization in xylene and rehydration through a gradually decreasing concentration of ethanol to distilled water, slides were treated in a PreTreatment module (Lab Vision Corp., Fremont, CA, USA) in Tris–HCl (pH 8.5) buffer for 20 min at 98°C for antigen retrieval. The staining procedure was performed in an Autostainer 480 (Lab Vision) by the Dako REAL EnVision Detection system, Peroxidase/DAB+, Rabbit/Mouse (Dako, Glostrup, Denmark). Tissues were incubated with an in-house monoclonal antibody (diluted to 1:50=50 μg/mL) for 1 h at room temperature (RT). The REG4 antibody is described in detail elsewhere. 26 Samples of colon tissue and normal lymph node served as positive controls in each staining series.
Scoring of samples
REG4 cytoplasmic expression in tumor cells was scored either as negative or positive, when staining was visible
Immunostaining was scored independently by K.S. and J.H., and the investigators were blinded to clinical data and outcome. Differences in scoring were discussed until consensus. The highest score of each patient was regarded as representative for analysis.
ELISA
The REG4 sandwich ELISA assays were performed using the Human REG4 ELISA Pair Set (SEK11186, Sino Biological Inc., Beijing, China) according to the manufacturer’s instructions. Briefly, the primary anti-REG4 antibody (2 μg/mL in citrate-buffered saline (CBS) buffer containing 0.05 M Na2CO3, 0.05M NaHCO3, pH 9.6) was immobilized to a 96-well plate overnight at +4°C, after which the wells were blocked with 1% BSA in 0.05% Tris-buffered saline with Tween (TBST) for 1 h at RT. Serum samples of the 130 patients with pancreatic carcinoma and the 34 CP controls were diluted 1:10 in sample buffer (0.1% BSA in 0.05% TBST) and incubated in duplicate wells (100 µL per well) for 2 h at RT. The horseradish peroxidase (HRP)-conjugated secondary anti-REG4 antibody (0.5 μg/mL in 0.5% BSA in 0.05% TBST) was allowed to bind for 1 h at RT, after which TMB substrate solution was added and allowed to react for 20 min at RT. The color reaction was stopped with 1 N H2SO4, and the absorbance (450 nm) was measured with Victor 1420 Multilabel Counter (Perkin Elmer, Waltham, MA, USA).
Statistical analysis
Associations between REG4 expression and clinicopathological parameters were performed by Fisher’s exact test or the linear-by-linear association test. Survival analysis was with the Kaplan–Meier method, and the log-rank test served to compare groups. A Cox regression proportional hazard model was created for univariate and multivariate survival analysis adjusted for age, gender, stage, metastasized lymph-node ratio (LNR) ≥/<20% (cut-off ≥/<20%), grade, and postoperative adjuvant therapy. LNR at 20% is widely used in the literature, since it is a strong prognostic factor for patient outcome.27,28 As stage and LNR are internally correlated, a combination variable was made for multivariate analyses. The Cox model assumption of constant hazard ratios over time was tested by including time-dependent covariate separately for each testable variable at a time. A time-dependent correlation factor for grade was included in the models, since the hazard ratio for grade was not constant over time. Interaction terms were considered.
For comparison of PDAC and CP, the Mann–Whitney U test was used for determining the significance of the difference in serum REG4 levels. The serum REG4 level cut-off point for assessing survival of PDAC was determined by the higher quarter of interquartile range (IQR) of CP patients (Table 4). PDAC patients were dichotomized into a low (<4.10 ng/mL) and a high group (≥4.10 ng/mL) according to serum REG4 level. Spearman correlation served for assessing correlations between laboratory parameters. Receiver operating characteristic (ROC) curves were constructed, and the area under the curve (AUC) values determined to evaluate different tumor markers. Optimal cut-off values were obtained by maximizing Yonden’s index. Multivariate logistic regression analysis allowed discovery of independent risk factors for PDAC.
We also studied correlations between both tissue and serum REG4 expression and CRP, CA19-9, and CEA values by Spearman correlation. We have published the results of the prognostic values of CRP, CA19-9, and CEA in our PDAC patient series. 29
A p value of <0.05 was considered significant, and all tests were two-sided. Statistical analyses were carried out with SPSS version 22.0 (IBM SPSS Statistics, version 22.0 for Mac; SPSS, Inc., Chicago, IL, USA, an IBM Company).
Results
REG4 is expressed in tumor cells
Positive REG4 expression was cytoplasmic with a granular distribution in tumor cells when present. Expression was often located in apical cell goblets with no nuclear expression evident. Staining could be evaluated in 153 (99.4%) cases: 110 (71.9%) were scored as negative and 43 (28.1%) as positive (Figure 1).

Tissue REG4 expression staining pattern in pancreatic ductal adenocarcinoma (PDAC): (a) negative tissue REG4 expression in PDAC and (b) positive tissue REG4 expression in PDAC. Original magnification ×400.
Tissue REG4 expression is associated with tumor histological grade
Tissue REG4 expression was associated with tumor histological grade (p=0.025). Patients with positive tissue REG4 expression more often had well-differentiated tumors than patients with negative expression (Table 1). Serum REG4 levels showed no significant associations with clinicopathological parameters (Table 2).
Association of tissue REG4 expression and clinicopathological parameters.
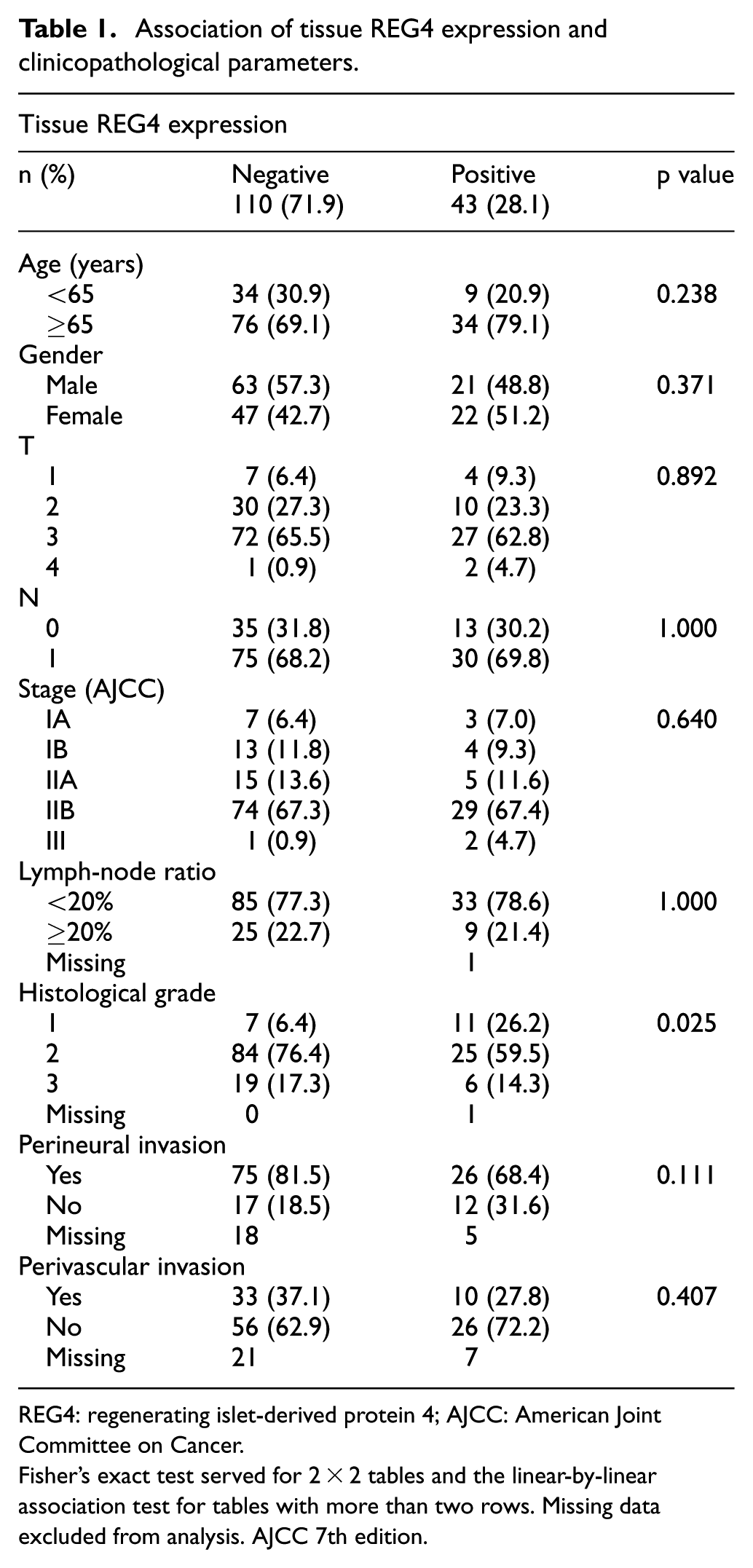
REG4: regenerating islet-derived protein 4; AJCC: American Joint Committee on Cancer.
Fisher’s exact test served for 2 × 2 tables and the linear-by-linear association test for tables with more than two rows. Missing data excluded from analysis. AJCC 7th edition.
Association of serum REG4 levels and clinicopathological parameters.
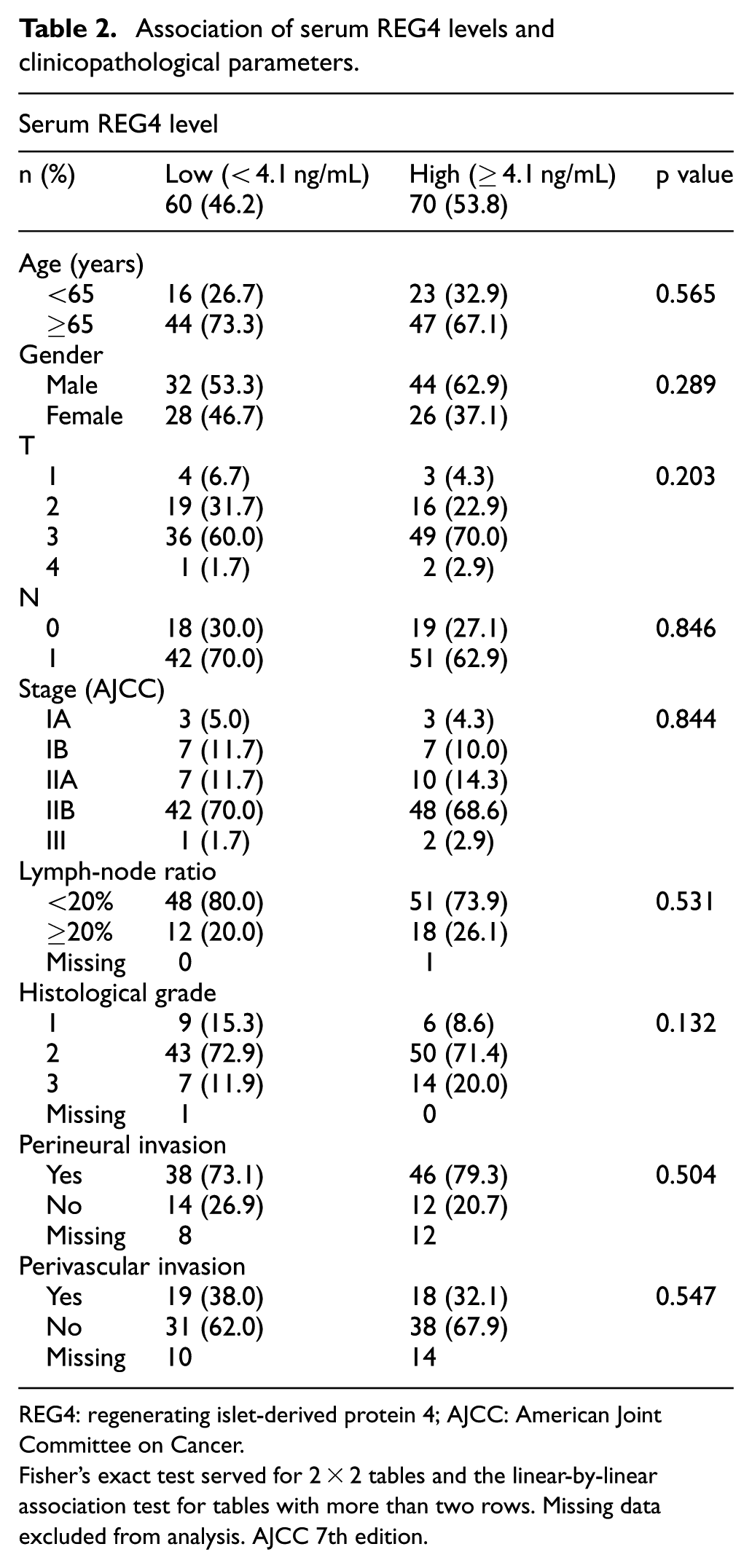
REG4: regenerating islet-derived protein 4; AJCC: American Joint Committee on Cancer.
Fisher’s exact test served for 2 × 2 tables and the linear-by-linear association test for tables with more than two rows. Missing data excluded from analysis. AJCC 7th edition.
Significant survival differences are identified in subgroups of patients
Cancer-specific survival (CSS) was not significantly different for PDAC patients with positive or negative REG4 tissue expression (log-rank p=0.496; Figure 2(a)). Five-year CSS in patients with positive REG4 tissue expression was 20.1% (95% confidence interval (CI): 7.8%–32.4%) compared to 19.7% (95% CI: 12.1%–27.3%) for those with negative REG4 tissue expression. By serum REG4 level, CSS did not differ between high and low groups. Five-year CSS for patients with high serum REG4 level was 16.2% (95% CI: 7.4%–25.0%) and with low serum REG4 level was 25.5% (95% CI: 14.1%–36.9%) (log-rank p=0.146; Figure 2(b)).

(a) Cancer-specific survival according to the Kaplan–Meier for tissue REG4 expression. (b) Cancer-specific survival according to the Kaplan–Meier for serum REG4 level. (c) Cancer-specific survival for serum REG4 level in PDAC patients with stages IA–IIA. (d) Cancer-specific survival for tissue REG4 expression in PDAC patients with grade I disease.
When divided by tumor stage, a survival benefit in a subgroup with non-metastasized stage IA–IIA disease was identified by serum REG4 level. Five-year survival in PDAC patients with a high serum REG4 level was 21.3% (95% CI: 2.9%–39.7%) and with a low serum REG4 level 52.9% (95% CI: 29.2%–76.6%) (log-rank p=0.046; Figure 2(c)). Positive tissue REG4 expression predicted better prognosis when compared with negative tissue REG4 expression in PDAC patients with grade 1 disease (5-year CSS 36.4% vs 0.0%; log-rank p=0.006; Figure 2(d)). No such difference was detectable in PDAC patients with grade II–III disease. Unfortunately, we had only 18 grade I patients.
In univariate analysis, neither tissue REG4 expression (hazard ratio (HR)=0.87, 95% CI: 0.59%–1.29%, p=0.496) nor serum REG4 level (HR=1.33, 95% CI: 0.90%–1.95%, p=0.148) associated significantly with survival in PDAC (Table 3). However, positive tissue REG4 expression was a protective factor for survival in PDAC patients with grade I disease as compared to negative expression (HR=0.21, 95% CI: 0.07%–0.70%, p=0.011; data not shown).
Cox univariate analysis of relative risk of death from pancreatic ductal adenocarcinoma by REG4 tissue (n=153) and serum (n=130) expression.

REG4: regenerating islet-derived protein 4; HR: hazard ratio; CI: confidence interval; LNR: metastasized lymph-node ratio.
Stage and LNR covariate was formed to cover both in multivariate analysis.
In multivariate analysis adjusted for age, gender, stage, LNR, grade, and postoperative adjuvant chemotherapy, neither tissue REG4 expression nor serum REG4 level predicted survival significantly (HR=0.87, 95% CI: 0.55%–1.35%, p=0.528 and HR=1.18, 95% CI: 0.79%–1.78%, p=0.417, respectively). However, there occurred a significant interaction between grade and tissue REG4 expression. The interaction model also suggests that positive tissue REG4 expression was a protective factor for survival in patients with grade I disease (HR=0.14, 95% CI: 0.03%–0.68%, p=0.015)
Serum REG4 levels are higher in PDAC than in CP
Serum REG4 levels in PDAC were assessed in 130 patients. Median for serum REG4 level in PDAC was 4.90 (range: 1.0–59.1) ng/mL. Serum REG4 levels were significantly higher in PDAC patients than in patients with CP (median 4.90 vs 3.05 ng/mL; p=0.002, Mann–Whitney test; Figure 3 and Table 4). Serum REG4 levels in different stages of PDAC are summarized in Table 4. We found no significant differences in serum REG4 levels between tumor stages (data not shown).

Serum REG4 levels are significantly higher in pancreatic ductal adenocarcinoma (PDAC) than in chronic pancreatitis (CP) (median 4.90 vs 3.05 ng/mL; p=0.002, Mann–Whitney test).
Serum REG4 levels in pancreatic ductal adenocarcinoma and chronic pancreatitis.

REG4: regenerating islet-derived protein 4; IQR: interquartile range; PDAC: pancreatic ductal adenocarcinoma.
Mann–Whitney U test for comparing serum REG4 levels in different stages of PDAC and chronic pancretitis. Serum REG4 levels did not differ significantly between different stages of PDAC (data not shown).
REG4 expression is not correlated with CRP values
No significant correlation existed between tissue REG4 expression and serum REG4 level in PDAC (p=0.289, correlation coefficient (r)=0.094, standard error (SE)=0.100). We also analyzed the correlation in PDAC patients between C-reactive protein (CRP) values and tissue REG4 expression and serum REG4 levels, but no significant correlation was detectable (p=0.885, r=0.013, SE=0.096 and p=0.318, r=0.089, SE=0.088, respectively). In addition, no correlation was detectable between CRP values and serum REG4 levels when divided by tumor histological grade. Serum REG4 level correlated with CEA level (p=0.036, r=0.188, SE=0.088), but not with CA19-9 (p=0.975, r=0.003, SE 0.092). CA19-9 and CEA levels did not differ significantly between low and high serum REG4 level groups (p=0.444, and p=0.097; data not shown).
Serum REG4 increases diagnostic accuracy
ROC analysis was performed to evaluate and compare the diagnostic accuracy of REG4, CA19-9, and CEA. AUC value for REG4 was 0.675 (95% CI: 0.587%–0.763%, p=0.002); for CA19-9, 0.806 (95% CI: 0.737%–0.874%, p<0.001); and for CEA, 0.544 (95% CI: 0.437%–0.650%, p=0.460) (Figure 4). Serum REG4 sensitivity was 45%, and specificity was 91% with optimal cut-off value 5.3 ng/mL. For serum CA19-9, sensitivity of 81% and specificity of 74% were achieved with an optimal cut-off value of 17 kU/L.

Comparison of serum REG4, CA19-9, CEA, and calculated probability of cancer with receiver operating characteristic (ROC) analysis from 130 patients with pancreatic ductal adenocarcinoma; 34 patients with chronic pancreatitis served as controls.
In a multivariate logistic regression model adjusted for age, values for serum REG4 and CA19-9 were significant and independent risk factors for PDAC (Table 5), suggesting that their combination may be able to improve diagnostic accuracy. The ROC curve of the logistic regression model achieved an AUC of 0.867 (95% CI: 0.804%–0.930%, p<0.001), which numerically exceeds REG4 or CA19-9 AUCs alone (Figure 4). At the optimal cut-off, this yields a sensitivity of 85% and specificity of 79%.
Multivariate logistic regression analysis for risk of PDAC.
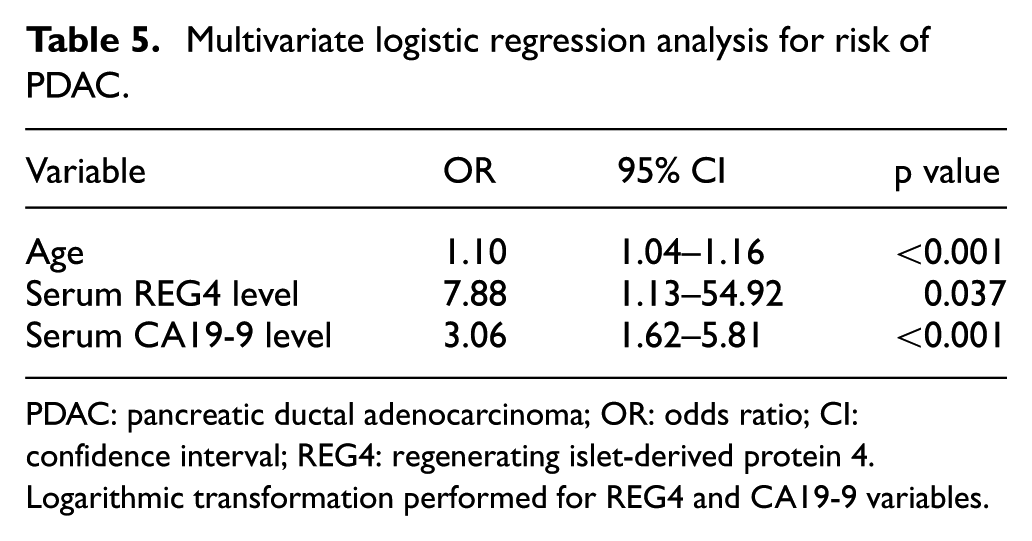
PDAC: pancreatic ductal adenocarcinoma; OR: odds ratio; CI: confidence interval; REG4: regenerating islet-derived protein 4.
Logarithmic transformation performed for REG4 and CA19-9 variables.
Discussion
We demonstrate that REG4 expression could serve as a prognostic factor in PDAC patients with early disease and in those with well-differentiated tumors. We also identified significant differences in serum REG4 levels between PDAC and CP. Both REG4 and CA19-9 provided independent diagnostic information. This suggests that serum REG4 might prove to be a diagnostic marker in PDAC.
To date, no reports show the prognostic significance of REG4 expression in PDAC. Li et al. 22 recently reported that low REG1A/B tissue expression is a marker of poor prognosis in PDAC. However, REG1A/B is normally expressed by the exocrine pancreas, and REG4 is not. 7 Moreover, the REG4 gene is on chromosome 1, whereas other REG genes are on chromosome 2.7,8 One can speculate that the biological functions of these proteins are distinct from each other because they are located on different chromosomes and encoded by different transcription factors.
REG4 has been under study in other gastrointestinal cancers as well. High tissue REG4 expression is a marker of poor prognosis in CRC by immunohistochemistry 30 and by mRNA expression. 31 However, Kaprio et al. 32 showed positive REG4 tissue expression to be a marker of better prognosis in non-mucinous CRC. High tissue REG4 expression predicts poor survival in gastric cancer, 33 in which it can also promote peritoneal metastasis. 34 In gallbladder cancer, positive tissue REG4 expression is associated with favorable prognosis. 35 In our study, 5-year CSS did not differ significantly in PDAC patients with positive or negative tissue REG4 expression, or with high or low serum REG4 level.
An intriguing finding is that patients with early stage (IA–IIA) disease showed better prognosis with lower serum REG4 levels. What animal models and cell cultures demonstrate is that pancreatic tumors tend to grow larger when REG4 expression is introduced.12,18–20 It is possible that negative or low-REG4-expressing pancreatic tumors grow slower and are better differentiated also in humans. We show that positive tissue REG4 expression predicts better prognosis in PDAC patients with histological grade I disease. Li et al. 22 came to the same conclusion for tissue REG1A/B expression. They also showed that higher levels of REG1A/B were associated with lower cancer-cell differentiation grade. The cut-off value of serum REG4 at 4.10 ng/mL for survival estimation was determined to find the optimal value, which both differentiates PDAC from CP and predicts prognosis in PDAC.
In addition to malignancies, REG4 expression is upregulated in inflammatory processes, such as IBD.7,8 In experimental pancreatitis in mice, increased REG4 expression can protect against acinar cell necrosis, 36 suggesting that REG4 may have anti-inflammatory features. We can speculate that in well-differentiated pancreatic tumors, REG4 expression may have a protective function against dedifferentiation. However, we found no correlation between different histological grades, or between REG4 and CRP values. The possible anti-inflammatory role of REG4 needs further study.
We demonstrate that between PDAC and CP serum REG4 levels differ significantly. All patients with CP had undergone surgery to exclude malignancy, but histopathology of the surgical samples confirmed CP. To our knowledge, this is the first study to report such a significant difference. Li et al. 22 found that REG1A/B functions better as a diagnostic marker in PDAC than does REG4, but REG1A/B levels did not differ significantly between CP and healthy controls. The cut-off level for REG4 in our series was somewhat higher than in other studies. Takehara et al. 12 used a cut-off point of 4.53 ng/mL but reported neither their sensitivity nor specificity. Takayama et al. 23 used a cut-off point of 3.49 ng/mL with 94.0% sensitivity and 64.0% specificity. However, they included also patients with stage IV PDAC, and their comparison of REG4 levels was between levels in PDAC and in healthy subjects. We excluded stage IV patients and compared serum REG4 levels between PDAC and CP, which is a clinically more relevant comparison. This probably explains the differing results between these studies.
In gastric cancer, the usefulness of REG4 as a diagnostic marker has been under study, showing that REG4 seems to function better than does CEA or CA19-9. 16 Lehtinen et al. 17 reported recently that REG4 expression could serve as a diagnostic serum biomarker for differentiating mucinous ovarian cancer from other epithelial ovarian cancer subtypes, and that it could be useful during follow-up. They detected that in mucinous ovarian carcinomas, serum REG4 levels were nearly 40 times higher than in serous ovarian carcinomas before surgery and that serum REG4 levels declined rapidly after surgery. It would be intriguing to study REG4 levels during follow-up of PDAC.
Data collection was comprehensive, but some important clinicopathological parameters were unavailable. The TMA technique allows analysis of large patient cohorts compared to analysis of whole-tissue blocks. Considerably smaller areas of tumors are evaluated by TMA, but the potential sampling error is reduced by taking core samples from different parts of the tumor. Less than 1% of specimens were lost for technical reasons. Because of the extended time-course of data collection, some serum samples were unavailable for serum analyses. The strength of this study is a quite large patient cohort with long follow-up. However, the protective role of positive tissue REG4 expression in grade I PDAC patients was not strong, since the low number of patients in this subgroup limited its significance. The clinical setting was relevant, taking into account that CP patients, because their diagnosis could not be determined by preoperative testing, underwent extensive surgery.
Conclusion
We demonstrate that REG4 expression could prove useful as a prognostic marker in PDAC in subgroups of patients but also serve as a diagnostic serum marker in addition to CA19-9. Validation of REG4 expression as a prognostic marker requires further study.
Footnotes
Acknowledgements
We thank Päivi Peltokangas and Olli-Matti Sirviö for technical assistance and Stig Nordling for histopathological evaluation. Caj Haglund and Hanna Seppänen contributed to experimental design and conception; Hanna Seppänen and Kapo Saukkonen contributed to clinical data collection; Kapo Saukkonen, Jaana Hagsträm, and Laura Lehtinen have contributed to experimentation; Kapo Saukkonen and Harri Mustonen contributed to data analysis; Caj Haglund, Olli Carpen, and Leif C Andersson have contributed to reagent/material/analysis tool preparation; and Kapo Saukkonen, Laura Lehtinen, Olli Carpen, Leif C Andersson, Harri Mustonen, Hanna Seppänen, Caj Haglund have contributed to writing of the manuscript. Hanna Seppänen and Caj Haglund share equal last authorship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was conducted according to the World Medical Association Declaration of Helsinki. It was approved by the Surgical Ethics Committee of Helsinki University Hospital (Dnro HUS 226/ E6/06, extension TMK02 §66 17.4.2013), and the National Supervisory Authority of Welfare and Health (Valvira Dnro 10041/06.01.03.01/2012). Written informed consent was acquired from the participants for their clinical records to be used in this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants to Caj Haglund from the Sigrid Jusélius Foundation and the Cancer Society of Finland, and to Hanna Seppänen from the Mary and Georg C Ehrnrooth Foundation, the Sigrid Jusélius Foundation and the Helsinki University Hospital Research Fund. Kapo Saukkonen was supported by grants from the Mary and Georg C Ehrnrooth Foundation, the Finnish Norwegian Medicine Foundation, the Finnish Medical Foundation, and the Cancer Foundation.
