Abstract
Paraoxonase 1 plays an important role in protection from oxidative stress and also decomposes homocysteine thiolactone, the toxic metabolite of homocysteine. A limited number of reports evaluated the role of paraoxonase 1 in women affected by female genital tract neoplasms, including endometrial cancer. This study aimed to analyze the paraoxonase activity in the group of endometrial cancer patients (n = 48) who underwent primary surgery and to compare the data available with a well-matched control group (n = 30). Due to the role of paraoxonase 1 in the metabolism of homocysteine (Hcy) thiolactone, the amount of Hcy-thiolactone as well as total serum Hcy concentrations was also measured. Serum paraoxonase 1 activity toward synthetic substrates, paraoxon and phenyl acetate, in the study group was significantly lower compared to the control one. The mean paraoxonase 1 activity toward homocysteine thiolactone tended to be lower in the endometrial cancer group but this difference was not significant. There was no relationship between endometrial cancer and Q192R polymorphism of
Introduction
Endometrial cancer (EC) is the sixth most common female neoplasm, the second most common gynecologic cancer, and the most common gynecological malignancy in the Western world.1,2 In 2013, 5706 cases were detected in Poland and 1243 women died of the neoplasm. Unfortunately, a number of new cases increased substantially during the last decades in Poland, a trend being reported worldwide. 3 Incidentally, the ECs may also develop in young women or even in adolescents. 4 Most of ECs is diagnosed at an early clinical stage of the disease and they are usually associated with a favorable patients’ outcome.1,2
Oxidative stress plays a crucial role in the pathophysiology of many human common disorders, including cardiovascular disease, type 2 diabetes mellitus, inflammatory bowel disease as well as Parkinson disease.5,6 The human organism possesses a variety of mechanisms protecting against oxidative products, including the family of paraoxonase (PON) enzymes.
7
The
PON1 is a 43–45 kDa calcium-dependent enzyme with paraoxon-, phenyl acetate- and lactone-hydrolyzing activities.9,10 PON1 is synthesized and secreted by the liver and is associated with high-density lipoprotein (HDL). 8 This enzyme possesses anti-oxidative properties and protects low-density lipoprotein (LDL) from oxidation, breaks down oxidized LDL into non-harmful products, attenuates oxidative stress in serum, macrophages, inhibits macrophage cholesterol biosynthesis rate as well as stimulates HDL-mediated cholesterol efflux. Interestingly, PON1 hydrolyzes several organophosphorus insecticides and nerve agents being responsible for protection against xenobiotic toxicity. 5 The enzyme metabolizes many substances, including glucocorticoids, statin adducts, toxic organophosphorous compounds, L-homocysteine thiolactone, and bacterial quorum-sensing factors. Finally, numerous cardiovascular and antidiabetic pharmacological agents, dietary macronutrients, lifestyle factors, and antioxidant supplements affect PON1 expression and enzyme activity levels.7,9,10
In the literature, PON1 has been investigated in selected human neoplasms, reporting lower enzyme activity in the patients with lung, breast, colorectal, esophageal, or bladder cancer compared to the well-matched control groups, in general.11–14 In a study of Samra et al., 15 low serum PON/arylesterase activity as well as high lipid peroxide levels was reported in all cancer patients. However, there are also opposite results published.16,17 A limited number of data, which assessed the role of PON1 in women affected by female genital tract neoplasms, has been reported.18–20 Based on PubMed® database, the only one study has been published yet assessing the role of PON1 activity in 20 EC women from Turkey. 19
This study was undertaken to assess serum PON1 activity in the group of EC women who underwent primary surgery and to compare the data available with a well-matched control group. Due to the role of PON1 in the metabolism of homocysteine (Hcy) thiolactone, the amount of Hcy-thiolactone as well as total serum Hcy concentrations was also measured.
Methods
Patients
The study group consisted of 48 ECs patients who underwent primary surgery at the IInd Department of Gynecology, Medical University of Lublin, Lublin, Poland, between 2012 and 2016. Clinico-pathological features of the study group are depicted at Table 1. Control group consisted of 30 women operated due to non-cancer disorders (pelvic organ prolapse, urinary stress incontinence) at the IInd Department of Gynecology, Medical University of Lublin, Lublin, Poland.
Clinical and pathological features of 48 EC patients.

EC: endometrial cancer; FIGO: Fédération internationale de gynécologie et d’obstétrique; G1: well-differentiated; G2: moderately differentiated; G3: poorly differentiated; LVSI: lymph-vascular space invasion.
The study protocol was approved by the Independent Ethics Committee of the Medical University of Lublin, Lublin, Poland, and all women enrolled gave their informed consent before the blood samples were collected. Patients who previously received chemotherapy, radiotherapy, or hormonotherapy were excluded. The cancer diagnosis was based on histopathological examination of the material collected during the dilatation and curettage procedure or during the Pipelle® sampling. Patients were analyzed in terms of age at the diagnosis, parity, body mass index (BMI), Fédération internationale de gynécologie et d’obstétrique (FIGO) staging, histological type and grading, myometrial infiltration, lymph node metastasis, cervical involvement, and coexistence of endometrial hyperplasia with cancer.
The treatment protocol consisted of total abdominal hysterectomy, salpingo-oophorectomy, and lymph node dissection (if necessary). The surgery was performed by a highly experienced gynecologist–oncologist. Surgical staging was performed by operating gynecologist–oncologist based on FIGO staging system, while the material was reviewed by a gynecologic pathologist according to the World Health Organization (WHO) classification. After surgery, all patients were subjected to the Oncology Department, Medical University of Lublin, Lublin, Poland where the further oncological treatment was performed, if necessary. Blood samples were collected from the cubital vein just before surgery, separated from the cells by centrifugation 5000×
PON1 activity
PON1 activity toward paraoxon was determined by measuring the initial rate of p-nitrophenol formation in the assay mixture containing 2.0 mM paraoxon and 2.0 nM CaCl2 in 800 μL of 100 mM Tris–HCl buffer (pH 8.0) at a temperature of 37°C. 21 Paraoxon stock solution was prepared in the air extraction fume hood by the operator wearing face mask and nitrile gloves to prevent from accidental contact or inhalation. After the assay, waste glass, tubes, and pipettes were treated with 1 M NaOH to accelerate paraoxon hydrolysis. For the assay, 20 mL of serum was added and absorbance was read at 412 nm for 2 min. The blank sample containing assay mixture without serum was assayed simultaneously to correct for spontaneous substrate hydrolysis. Increase in absorbance of the blank sample was subtracted from the increase in absorbance of the test sample and enzyme activity was calculated from E412 of p-nitrophenol (18,290 M−1 cm−1) and was expressed in U/mL; one unit of PON1 hydrolyzes 1 nmol of paraoxon/min.
PON1 activity toward phenyl acetate (arylesterase activity) was determined by measuring the initial rate of phenol formation, monitored at 270 nm, in the assay mixture (3 mL) containing 2 mM phenyl acetate, 2 mM CaCl2, and 10 μL serum in 100 mM Tris–HCl (pH 8.0). Increase in absorbance at 270 nm was monitored for 3 min and the activity was calculated from
PON1 activity toward Hcy-thiolactone was determined by measuring the amount of hydrogen ions generated during thiolactone hydrolysis to homocysteine in the assay mixture (800 μL) containing 5 mM substrate, 1 mM CaCl2, 0.0005% bovine serum albumin, 0.004% phenyl red, and 20 μL of serum in 5 mM HEPES buffer (pH 7.0). 21 The absorbance was monitored for 4 min at 412 nm, and the results were expressed in nmol min−1 mL−1. The blank sample containing incubation mixture without serum was simultaneously assayed to correct for spontaneous substrate breakdown. PON1 activity toward Hcy-thiolactone was calculated from the standard curve prepared by titrating the assay medium with the different HCl concentrations.
PON1 phenotyping
PON1 exhibits Q192R polymorphism (glutamine to arginine substitution) which affects enzyme activity toward some organophosphate compounds; QQ, QR, and RR genotypes correspond to the so-called AA, AB, and BB phenotypes. PON1 phenotyping was performed by the dual substrate methods of Eckerson and collegues.22,23 PON1 activity toward paraoxon was additionally measured in 50 mM glycine buffer (pH 10.5) in the presence of 1 M NaCl. The rate of salt-stimulated PON1 activity toward paraoxon to arylesterase activity was calculated for each sample. Patients with the ratio <2.5, 2.6–7.5, and >7.5 were classified into the AA, AB, and BB phenotypes. 24
Serum total homocysteine and protein N-homocysteinylation
Serum total homocysteine (Hcy) was assayed by enzyme immunoassay using commercially available kit based on the methods proposed by the manufacturer (Axis Shield Diagnostics Ltd, Dundee, UK). In this method, Hcy bound to serum proteins by disulfide bonds is first released by adding dithiotreitol (DTT) and then converted enzymatically to
To measure the amount of homocysteine thiolactone bound to serum proteins (protein N-homocysteinylation), after DTT-induced liberation of disulfide-bound Hcy serum proteins were precipitated with ethanol and hydrolyzed at 110°C in the presence of 6 M HCl. Hydrolyzate was evaporated under N2 and Hcy-thiolactone was converted to Hcy by adding NaOH. Hcy was then assayed by the method described above. 21
Statistical analysis
Statistical analysis was performed with the SPSS 11.5 (SPSS Inc., Chicago, IL, USA). Data were presented as mean ± standard deviation. PON1 activities, Hcy and Hcy-thiolactone concentrations were compared between groups by the Mann–Whitney
Results
There were no significant differences between study group and control group with regard to age, height, BMI, or parity (Table 2).
Comparison of demographic and anthropometric data (mean ± SD) between the cancer and control groups.

SD: standard deviation; EC: endometrial cancer; BMI: body mass index; NS: not significant.
Serum PON1 activities toward paraoxon and phenyl acetate were significantly lower in the cancer group compared to the control (Table 3). The distribution of PON1 AA, AB, and BB phenotypes did not differ between groups (Table 4). Because PON1 activity toward paraoxon is affected by the phenotype (Q192R polymorphism), this activity was also separately calculated in subgroups of patients with specific enzyme phenotypes (Table 5). PON1 activity toward paraoxon activity was lower in cancer than in the control group in patients with either AA or AB phenotypes. The mean PON1 activity toward paraoxon activity in the cancer patients with BB phenotype was also lower than in the control subjects but the difference did not reach the level of significance.
Serum paraoxonase activity, homocysteine, and protein-bound homocysteine thiolactone concentrations in the study and control groups.
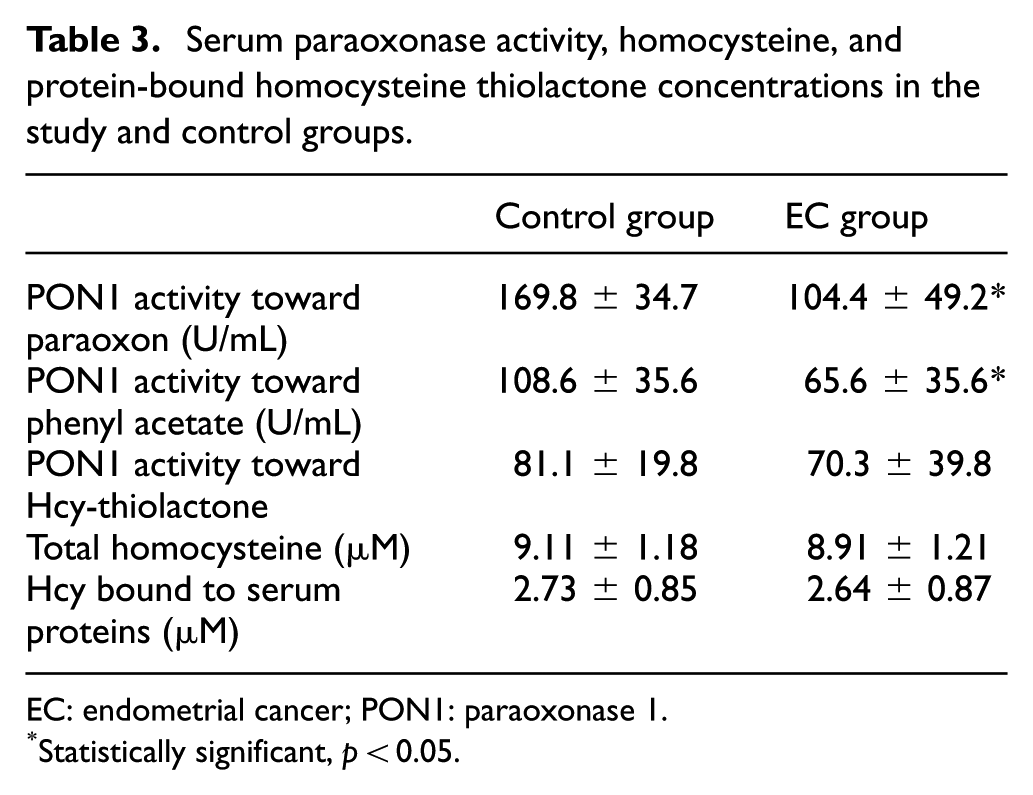
EC: endometrial cancer; PON1: paraoxonase 1.
Statistically significant,
PON1 phenotype distribution in control and EC group.

PON1: paraoxonase 1; EC: endometrial cancer.
PON1 activity toward paraoxon in patients with AA, AB, and BB phenotypes.

PON1: paraoxonase 1; EC: endometrial cancer.
Statistically significant,
PON1 activity toward homocysteine thiolactone activity tended to be lower in cancer patients but the difference was not significant versus control group. Neither total homocysteine nor protein-bound homocysteine thiolactone differed significantly between groups (Table 3).
We analyzed the relationship between clinico-pathological features and PON1 activity in the EC patients. However, no significant differences between serum PON1 activity in subgroups according to FIGO staging, histological type and grading, myometrial infiltration, lymph node metastasis, cervical involvement and coexistence of endometrial hyperplasia with cancer were observed (data not shown).
Discussion
Altered enzymatic antioxidant activity has been associated with the development and progression of various human malignancies, including the lung, breast, colon, gastroesophageal, and rectum.11,12,25,26 Several previous studies have demonstrated that PON1 activity was lower in patients with pancreatic, 26 gastric, 27 lung, 11 ovarian, 18 bladder 28 cancers as well as in those with brain tumors. 29 Samra et al. 15 have demonstrated that PON1 activity toward paraoxon and phenyl acetate as well as PON1 protein concentration is lower in patients with breast, prostate, cervix, or lung cancers as well as in those with non-Hodgkin’s lymphomas than in sex- and age-matched control individuals. Similarly, PON1 activities toward paraoxon and phenyl acetate were reduced in patients with lung, breast, or colorectal cancers. 12 In all these studies, PON1 activity was measured toward synthetic substrates; in most studies, only toward paraoxon. Simultaneous measurement of PON1 activity toward several substrates is important because various PON1 activities are differentially regulated by physiologic and pathologic factors. 30 In particular, because activity toward paraoxon is affected by genetic polymorphism, the interpretation of results is difficult if this polymorphism is not simultaneously assessed. To our knowledge, PON1 activity toward Hcy-thiolactone, one of the natural substrates, has not been assessed in any type of cancer before.
However, little is known about the role of PON1activity/
PON1 Q192R polymorphism affects enzyme’s activity toward some (e.g. paraoxon) but not all (e.g. phenyl acetate) substrates and has been shown to affect the risk of various diseases including uterine leiomyoma 40 and ovarian epithelial carcinoma. 31 In the present study, the distribution of PON1 phenotypes (AA, AB and BB) corresponding to QQ, QR, and RR genotypes, respectively, was similar in cancer and control groups suggesting that Q192R polymorphism is not a risk factor of EC. According to the recent meta-analysis, the relationship between Q192R polymorphism and cancer risk depends on the cancer type and ethnic group. 41 Moreover, although initial studies suggested the strong correlation between Q192R polymorphism and cardiovascular diseases, later studies on large populations as well as meta-analyses of original studies indicate that this association is weak or not observed, and that PON1 concentration/enzymatic activity is more important determinant of the disease than the genotype.42,43 The same seems to apply for EC since PON1 enzymatic activity was reduced in EC patients in each subgroup with specific phenotypes in comparison to the respective phenotype in the control group.
Homocysteine is a non-protein amino-acid which is the intermediate product of methionine to cysteine conversion. Elevated Hcy concentration (hyperhomo–cysteinemia) contributes to the development of atherosclerosis, venous thromboembolism, neural tube defects, liver steatosis, and Alzheimer disease. Increased plasma Hcy concentration is observed in patients with leukemia as well as breast, ovarian, pancreatic, and gastric cancers.44–46 Hyperhomocysteinemia is a marker of folate deficiency, the well-known risk factor of the cancer, and Hcy itself may contribute to cancer growth by inducing oxidative stress and DNA hypomethylation. 44 To our knowledge, blood Hcy concentration in patients with EC has not been reported so far. The present study indicates that Hcy concentration is similar in control and cancer groups indicating that hyperhomocysteinemia is not associated with EC, at least in the early stage of the disease.
Homocysteine thiolactone is synthesized from Hcy by methionyl-tRNA synthetase which can non-specifically bind Hcy but does not incorporate it into proteins. This mechanism protects from nonspecific incorporation of Hcy to proteins in places occupied by methionine but results in production of highly reactive Hcy-thiolactone. Formation of Hcy-thiolactone is a very attractive mechanism of Hcy toxicity because this derivative is specifically produced from homocysteine but not from other sulfur-containing aminoacids. Hcy-thiolactone spontaneously reacts with ͼ-NH2 residues; the process referred to as protein N-homocysteinylation. Hcy-thiolactone is hydrolyzed to homocysteine by PON1. 47 In the present study, PON1 activity toward Hcy-thiolactone tended to be lower in EC patients than in control individuals but this difference did not reach the level of significance. PON1 activity toward Hcy-thiolactone exhibited high variability evidenced by relatively high standard deviation (SD) values. This high variability could result from low specific activity of PON1 toward Hcy-thiolactone in comparison to its activity toward synthetic substrates. In addition, in contrast to paraoxo- and phenyl acetate-hydrolyzing activities, PON1 activity toward Hcy-thiolactone has to be measured at neutral pH (suboptimal for the enzyme) because Hcy-thiolactone spontaneously decomposes to Hcy at high pH values. High variability and differences in experimental conditions could explain why Hcy-hydrolyzing activity of PON1 did not differ significantly between cancer and control groups.
In the cell culture experiments, low PON1 activity is associated with high Hcy-thiolactone and protein N-homocysteinylation. 47 However, the results of in vivo studies are less convincing. Low PON1 activity and increased protein N-homocysteinylation have been observed in patients with cardiovascular diseases and subjects with homocystynuria. 48 Similarly, high leptin concentration reduced PON1 activity and enhanced N-homocysteinylation of plasma proteins in the rat. 21 In contrast, both Hcy-thiolactone concentration and PON1 activity are increased in vitreous fluid of patients with diabetic retinopathy in comparison to the control group. 49 Hcy-thiolactone concentration is increased in the brain but not in peripheral organs or plasma of PON1−/− mice in comparison to their wild-type littermates. 50 In addition, anti-osteoporosis drugs, bisphosphonates, decrease plasma PON1 activity but had no effect on protein N-homocysteinylation in the rat. 51 In secondary progressive form of multiple sclerosis, plasma PON1 activity is lower than in control subjects but the amount of Hcy-thiolactone attached to plasma proteins is not increased. 52 These data indicate that decrease in PON1 is not necessarily associated with the enhancement of protein N-homocysteinylation. There are at least two other enzymes which metabolize Hcy-thiolactone, bleomycin hydrolase 50 and valacyclovir hydrolase/biphenyl hydrolase-like protein, 53 which could compensate for PON1 deficiency. Nevertheless, because we measured global N-homocysteinylation of all serum proteins, we cannot exclude that N-homocysteinylation of individual proteins in serum and/or tissues was enhanced in the EC group.
This study has some limitations which should be mentioned. PON1 is a HDL-bound enzyme and HDL concentration and composition, in particular apolipoprotein A-I content, have profound effects on enzyme status. Unfortunately, HDL and apolipoproteins were not measured in this study. However, no 54 or even negative correlation between PON1 activity and HDL levels have been observed in some studies suggesting that HDL concentration is not a universal determinant of PON1 concentration/activity. 55 Because we measured PON1 activity toward three different substrates, we believe that the results provide the reliable evidence of PON1 status. In addition, the relationship between PON1 activity and BMI was recently reported in obese women. 56 Nevertheless, in the present study, the mean patients’ BMI was in the overweight but non-obese range and no difference in BMI was observed between control and cancer groups. Therefore, it is unlikely that BMI significantly affected PON1 status.
In conclusion, reduced PON1 activity suggests diminished important antioxidant mechanisms during the development of primary ECs in humans.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
