Abstract
The exchange of metabolites between mitochondria and cytosol occurs through pores formed by voltage-dependent anion channel proteins. Voltage-dependent anion channels appear to be master regulators of mitochondrial bioenergetics and the intracellular flow of energy. Deregulation of voltage-dependent anion channels expression is thought to be related to mitochondrial dysfunction in cancer. The aim of this study was to investigate the mRNA and protein expression levels of VDAC1, VDAC2, and VDAC3 in relation to clinicopathological characteristics of endometrial cancer as well as the prognostic significance of voltage-dependent anion channels expression for overall survival. VDAC1 and VDAC3 expressions were significantly higher in cancer compared to normal tissues. Kaplan–Meier analysis indicated that high expression of all
Introduction
Tumor cells are characterized by enhanced glycolysis and suppression of mitochondrial metabolism. Mitochondrial dysfunction contributes to the adaptation of cancer cells to uncontrolled divisions.1,2 The flux of respiratory substrates into mitochondria and the release of mitochondrial adenosine triphosphate (ATP) to the cytosol occur through voltage-dependent anion channels (VDACs) which are the most abundant proteins in the mitochondrial outer membrane. In eukaryotic cells, three isoforms of VDAC have been identified: VDAC1, VDAC2, and VDAC3. Each of these isoforms is encoded by separate gene to produce proteins of about 30 kDa. VDACs share approximately 70% of amino acid sequence identity; however, each isoform displays different expression levels and has distinct protein partners.3,4 VDACs can interact with pro- and anti-apoptotic members of Bcl-2 family, thus participating in the cytochrome c release. 5 However, VDACs do not play equal roles in the activation of apoptosis. Association of VDAC1 with Bax and Bak promotes rapid cytochrome c release into the cytosol and the activation of apoptotic cascade. Conversely, VDAC2 seems to play an anti-apoptotic role by sequestering the pro-apoptotic Bak protein. 6 Little is known of VDAC3 function; however, this isoform is important for proper mitochondrial membrane potential. 7 It is suggested that while VDAC1 and VDAC2 play mainly pore-forming role, VDAC3 is mostly involved in the regulation of other proteins function. 5 Based on their role in pore formation and metabolic exchange between mitochondria and cytosol, VDACs seem to be master regulators of mitochondrial bioenergetics and the intracellular flow of energy.7–9 Recent studies have shown that the closing of the VDAC channels by free tubulin limits the influx of metabolites into mitochondria and limits ATP production. The suppression of mitochondrial metabolism caused by VDAC–tubulin interaction favor the biosynthesis of new macromolecules during the growth stages and promotes the Warburg phenotype. 10 Furthermore, VDACs participate in the adaptation of cells to anaerobic glycolysis through their interaction with hexokinases (HK). The HK/VDAC association gives HK easy access to ATP and promotes the conversion of pyruvate to lactic acid, thus reprograming energy metabolism and providing a growth advantage to cancer cells.11,12
VDAC porins play a significant role in cellular energy homeostasis and apoptosis, which makes them promising anti-cancer targets; however, their expression in cancer needs more research. To date, no alterations of VDAC expression have been reported in endometrial tumors. Endometrial cancer is a multifactorial disease; however,
Materials and methods
Reagents and antibodies
Chemicals were purchased from Sigma-Aldrich (St. Louis, MO, USA) except as noted. The used antibodies: mouse monoclonal anti-VDAC1 (sc-390996) and mouse monoclonal anti-β-actin (sc-47778) were from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). The polyclonal goat anti-VDAC2 (ab37985), polyclonal rabbit anti-VDAC3 (ab130561), and secondary donkey polyclonal anti-goat immunoglobulin G (IgG)-horseradish peroxidase (HRP) antibodies (ab6885) were from Abcam (Cambridge, UK). Secondary mouse anti-rabbit (#7074) as well as goat anti mouse (#7076) IgG-HRP antibodies were obtained from Cell Signaling Technology, Inc. (Beverly, MA, USA).
Patients and samples
The analyzed specimens of endometrial cancer were obtained from 116 patients who underwent surgery in the Department of Gynecological Oncology Copernicus memorial Hospital (Łódź, Poland) between February 2009 and March 2017. Samples of normal endometrial tissues were obtained from 41 patients who had undergone hysterectomy due to leiomyoma or a uterus prolapsed. Samples were immediately frozen after resection and stored at −80°C until needed. The histological diagnosis report of each patient was obtained from an experienced pathologist. All samples of cancerous tissue were evaluated in terms of histological grade and type according to the World Health Organization (WHO) classification, tumor stage according to the International Federation of Gynecology and Obstetrics (FIGO) criteria, the ability of cancer cells to metastasis to lymph nodes and infiltration into muscularis, and clinicopathological characteristics of specimens are presented in Table 1. The follow-up period was defined as the interval from the date of surgery to the date of death or the last follow-up, that is, March 2019. The studies were performed with the approval of the Bioethical Commission of the University of Lodz (17/KBBN-UŁ/I/2017).
Characteristics of patients and endometrial cancer samples.

FIGO: International Federation of Gynecology and Obstetrics.
RNA extraction and cDNA synthesis
RNA from normal and cancer tissue was isolated using the total RNA isolation kit (A&A Biotechnology, Gdynia, Poland) according to the manufacturer’s instructions and quantified spectrophotometrically. The isolation of RNA from endometrial lesions was performed followed by overnight tissue sample incubation with Proteinase K enzyme at 37°C. First-strand cDNAs were obtained by the reverse transcription of 1 μg of total RNA using a High-Capacity cDNA Reverse Transcription kit (Applied Biosystems, Foster City, CA, USA) following the manufacturer’s protocol. The reverse transcription polymerase chain reaction (RT-PCR) was carried out in a GeneAMP 2400 Thermal Cycler (Perkin-Elmer, USA) according to the following thermal profile: 10 min at 25°C, 120 min at 37°C, and 5 min at 85°C.
Quantitative real-time PCR analysis
The TaqMan Gene Expression Assay (Applied Biosystems) was used to analyze the expression of the
Protein samples preparation
Endometrial specimens (200 mg of wet tissue sample) were minced into small pieces using scissors and trimmed of visible fat, ligaments, and connective tissue. The minced fragments were washed twice with phosphate buffered saline (PBS) supplemented with 10 mM ethylenediaminetetraacetic acid (EDTA) and then resuspended in 2 mL of ice-cold PBS supplemented with 10 mM EDTA and 0.05% trypsin for 30 min. After the incubation, tissue scraps had been spun down at 200 × g for 5 min and the supernatants had been discarded. The obtained pellet was resuspended in ice-cold radioimmunoprecipitation assay (RIPA) buffer (50 mM Tris-HCl–150 mM NaCl–1% Triton X-100%–0.5% Na-deoxycholate (DOC)–0.1% sodium dodecyl sulfate (SDS)–2 mM EDTA (pH 7.4)) with 1 mM phenylmethylsulfonyl fluoride (PMSF) to inhibit protease activity. The efficiency of homogenization was monitored by phase-contrast light microscopy. The supernatants obtained after centrifugation of the tissue lysates at 10,000 × g at 4°C for 10 min were collected and saved for further analysis. Tissue lysates and cellular fractions were mixed with Laemmli buffer and then analyzed by electrophoresis.
Western blotting
Protein samples (40 μg protein/lane) from 41 normal and 107 endometrial cancer specimens were separated by 10% SDS-PAGE and electrotransferred onto Immobilon-P® transfer membranes. The quality of the transfer was verified by Ponceau S staining prior to blocking the membrane. The blots were incubated for 2 h at room temperature with primary antibodies. After having been washed three times with TBS-T (0.1% Tween-20 in Tris-buffered saline (TBS)), the blots were incubated for 1 h with horseradish peroxidase-labeled secondary antibodies. Proteins were visualized on an X-ray film by an enhanced chemiluminescence method using SuperSignalTM West Pico PLUS Chemiluminescent Substrate (Thermo ScientificTM). For loading control, the blots were reprobed with anti-β-actin antibody following a stripping protocol. Gel-Pro Analyzer software version 3.0 (Media Cybernetics, Inc., Bethesda, MD, USA) was used to quantify integrated optical density (IOD) of the protein bands. The levels of VDAC expression in endometrial specimens were normalized by the reference sample (0.2 µg of primary antibody) resolved by electrophoresis that made the comparison of intensity of the bands from different membranes possible. The results of the VDAC proteins expression analyses are presented as a relative protein level which is the ratio of the normalized IOD of the bands corresponding to VDAC in each sample and the normalized IOD of β-actin in the same specimens.
Statistical analysis
To improve statistical conclusions, the obtained results were evaluated by interquartile range (IQR) analysis, then outliers were eliminated. Differences in expression levels of VDAC1, VDAC2, and VDAC3 in studied groups were analyzed using the Kruskal–Wallis test with the post-hoc Dunn test. The comparison of the groups was done using the non-parametric Mann–Whitney U test. The Spearman test was applied to estimate the correlations between
Results
Expression of VDAC1, VDAC2 , and VDAC3 genes in endometrial cancer
The mRNA levels of

((a)–(c)) Expression of VDAC1, VDAC2, and VDAC3 mean mRNA measured by real-time PCR in control and endometrial cancer samples; ((d)–(f)) a comparison between subgroups with different FIGO stages; ((g)–(i)) histological differentiation grade; ((j)–(l)) lymph node metastasis status; and ((m)–(o)) myometrial invasion. The results are shown as number of each VDAC mRNA copy per 1000 copies of HPRT1 mRNA (reference gene).
VDAC porin levels in endometrial cancer
The expression levels of VDAC porins in endometrial specimens were determined using Western blotting method. Representative blots are shown in Figure 2. Bands corresponding to VDAC porins were analyzed using densitometric analysis. The obtained values were normalized to the expression of β-actin in the same sample and presented as a relative expression. The results of densitometric analysis are shown as mean estimates obtained from all analyzed samples. Analysis showed that the mean protein expression of VDAC1 was significantly higher in cancer samples than in normal tissue samples (Figure 2(a)). Similarly, the VDAC3 level was significantly higher in tumors, especially those classified as FIGO stages I and II than in non-tumoral tissue (Figure 2(c) and (f)). Densitometric analysis revealed that the mean protein expression of VDAC1 was significantly higher in samples of cancers in which the infiltration included more than a half the thickness of the muscle (≥1/2) than cancers in which the infiltration did not exceed half the thickness of muscle (<1/2) (Figure 2(j)). The results indicate that the expression of VDAC3 strongly decreases in tumor samples that exhibit the ability to metastasize to regional lymph nodes compared to non-metastatic cancers (Figure 2(o)). There was no significant association (

((a)–(c)) Expression of VDAC1, VDAC2, and VDAC3 proteins in control and endometrial cancer samples; ((d)–(f)) a comparison between subgroups with different FIGO stage; ((g)–(i)) histological differentiation grade; ((j)–(l)) metastasis to lymph nodes, and ((m)–(o)) myometrial invasion. Samples containing 40 μg of protein were resolved by SDS-PAGE and transferred onto membranes for immunodetection. The intensity of bands corresponding to VDACs was analyzed by densitometry and integrated optical density (IOD) was normalized by β-actin level and reference sample (see Methods for details). The results are presented as graphs associated with representative Western blots of VDAC detection.
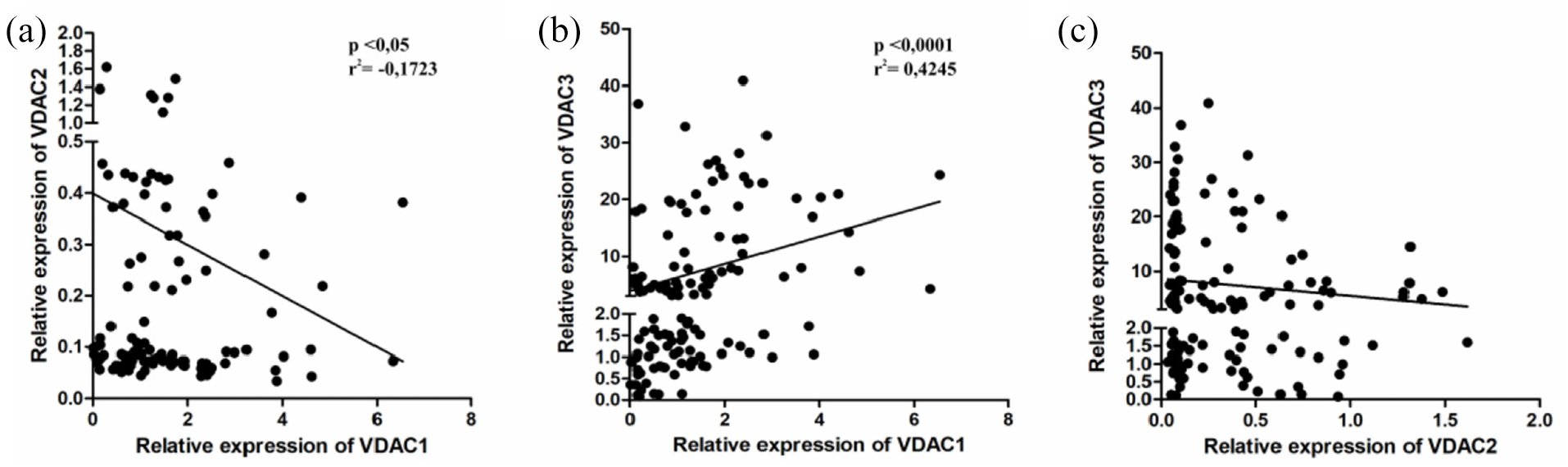
Correlation between VDAC proteins expression levels in endometrial cancer samples performed by Spearman rank correlation analysis. VDAC1 level was significantly associated with (a) VDAC2 (r = −0.172,
VDAC expression in endometrial cancer of diabetic patients
VDAC porins regulate cellular energy metabolism. The primary source of energy for cancer cells is glucose; therefore, we suppose that long-term alterations of blood glucose concentration may impact on VDAC levels in tumors. One of the most common chronic diseases characterized by hyperglycemia is diabetes mellitus (DM). Type 2 DM (T2DM) is well-established as a significant risk factor for various types of cancer including endometrial cancer.
17
To find out whether T2DM status in patients is associated with VDAC expression in endometrial tumors, we analyzed VDAC expression in groups of T2DM positive and negative tumors in the first stage of development according to the FIGO classification. No significant changes were found in

Expression of VDAC1, VDAC2, and VDAC3 on the ((a)–(c)) mRNA and ((d)–(f)) protein levels in endometrial tumors from diabetic and non-diabetic patients. The representative results of VDAC Western blot detection are shown in the figure. The intensity of bands corresponding to VDACs in all samples was analyzed by densitometry and integrated optical density (IOD) was normalized by β-actin level and reference sample (see Methods for details). The results of this analysis are shown as graphs representing mean ± SEM. All of analyzed samples were corresponded to first stage of endometrial cancer according to FIGO classification without invasion to lymph node (see Results for more details). DM−: without type 2 diabetes mellitus and DM+: with type 2 diabetes mellitus.
VDAC status and endometrial cancer prognosis
To further understand the significance of VDAC1, VDAC2, and VDAC3 expression in endometrial cancer, patients were divided into two groups based on the dichotomized scores of survival period. Overall survival was defined as the interval from the date of surgery to an endometrial cancer-related death. The endometrial cancer patients who underwent surgery were classified into high and low VDAC expression subgroups using their median expression value as the cutoff point. The Kaplan–Meier method combined with log-rank analysis was used to estimate the predictive effect of VDAC expression in overall survival. The analysis showed that groups of patients with high expression each of three

Kaplan–Meier curves showing overall survival for endometrial cancer patients with low and high levels of VDAC mRNA and protein expression. The cutoff values were established to be the median of VDAC expression level. The survival curves were compared between two groups: high (≥median value) and low (<median value) expression. Patients in the high VDAC expression group had a significantly shorter overall survival than those in low VDAC expression group.
Univariate analyses of VDAC expression and clinicopathological factors influencing patients’ survival.
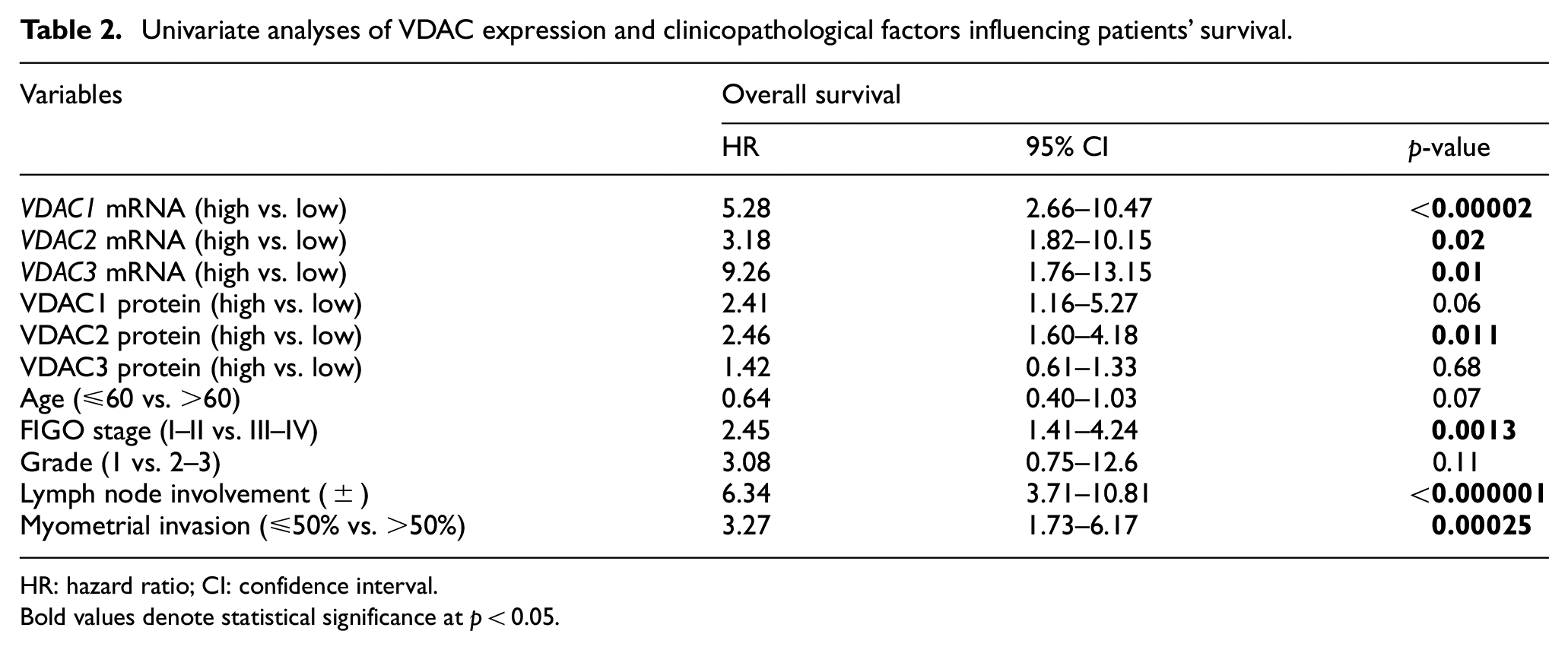
HR: hazard ratio; CI: confidence interval.
Bold values denote statistical significance at
Multivariable Cox regression analyses of VDAC expression and clinicopathological factors influencing patients’ survival.
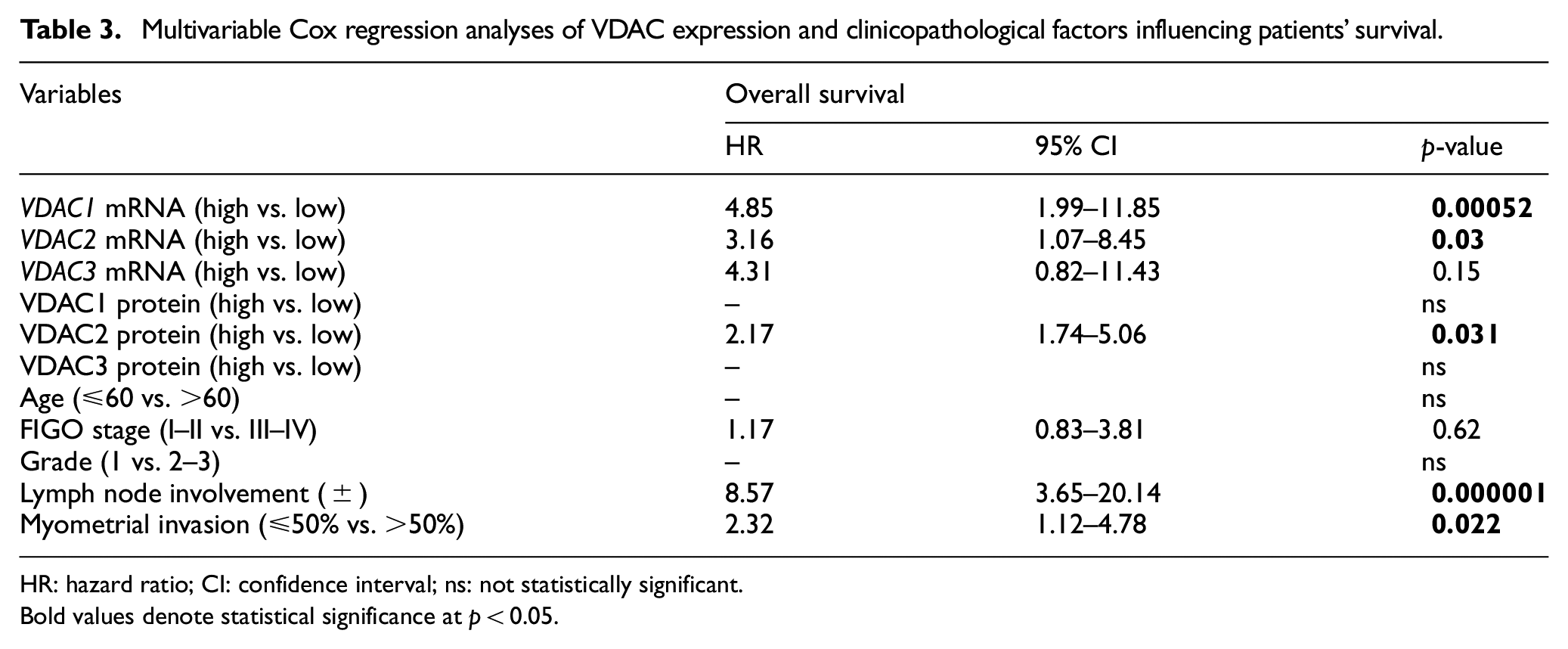
HR: hazard ratio; CI: confidence interval; ns: not statistically significant.
Bold values denote statistical significance at
Discussion
In this study, we demonstrated for the first time that changes in VDAC expression are significantly associated with progression of endometrial cancer and may be a valuable prognostic indicator for patient survival. To date, the expression of VDAC porins has been poorly investigated, but numerous studies using cell lines have already shown that VDACs play an important role in carcinogenesis.18–20
According to published data, the changes in
Most of studies seem to suggest that VDAC1 is a widely and dominantly expressed isoform.16,24–26 Our results indicated that the expression of
Footnotes
Author contributions
All authors read and approval the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The investigations were performed with the approval of the Bioethical Commission of the University of Lodz (17/KBBN-UŁ/I/2017).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
