Abstract
Ten-eleven translocation proteins are α-ketoglutarate-dependent dioxygenases involved in the conversion of 5-methylcytosines (5-mC) to 5-hydroxymethylcytosine (5-hmC), 5-formylcytosine, and 5-carboxylcytosine that play a significant role in DNA demethylation. Deregulation of TET genes expression and changes in the level of 5-hmC are thought to be associated with the onset and progression of several types of cancer, but there are no such data related to endometrial cancer. The aim of the work was to investigate the messenger RNA expression levels of TET1, TET2, and TET3 in relation to clinicopathological characteristics of endometrial cancer as well as the correlation between expression of TET genes and the level of 5-hmC/5-mC. The prognostic significance of TETs expression for overall survival was established. We found that TET1 and TET2 messenger RNA expression was lower and TET3 was higher in cancers compared to normal tissues. Positive correlation between 5-hmC and the relative expression of TET1 and TET2 was found, but no correlation was observed in the case of TET3. Decreased expression of TET1 and TET2 was significantly associated with increased lymph node metastasis and International Federation of Gynecology and Obstetrics stage. Kaplan–Meier analysis indicated that low TET1 expression predicted poor overall survival (p = 0.038). Multivariate analysis identified the TET1 expression in endometrial cancer as an independent prognostic factor. Our results suggest that decreased expression of TET1 correlates with tumor progression and may serve as a potential prognostic biomarker in endometrial cancer.
Introduction
Tumor cells are characterized by significant changes in the DNA methylation pattern as compared to normal cells. In tumor cells, a global hypomethylation of the genome is observed. At the same time, an increased level of methylation (hypermethylation) of CpG islands in the promoter sequences of certain genes can be noticed. The global hypomethylation can lead to activation of proto-oncogenes and to reduced genomic stability through an increased activation of transposons, which are normally silenced by methylation. In turn, the hypermethylation of CpG islands in promoter regions of tumor suppressor genes and DNA repair genes can contribute to carcinogenesis by inhibiting the expression of these genes.1–3 A specific DNA methylation pattern is determined not only by the attachment of methyl group to cytosine but also as a result of the process of DNA demethylation.4,5 Ten-eleven translocation (TET) protein family plays a significant role in changing the pattern of DNA methylation, through participation in DNA demethylation. 6 In human, three TET proteins, named TET1, TET2, and TET3, have been identified. 7 These proteins are iron(II)- and 2-ketoglutarate-dependent dioxygenases. TET proteins catalyze the conversion of the 5-methylcytosine into 5-hydroxymethylcytosine (5-hmC), and further to 5-formylcytosine (5-fC) and 5-carboxylcytosine (5-caC), which initiates the process of DNA demethylation.8–10 To date, a number of studies confirmed the important role of TET proteins in the DNA demethylation and their relationship with changes in DNA methylation pattern in tumors. TET proteins seem to be also involved in the regulation of other epigenetic modifications, that is, the modification of histones by interacting with specific proteins responsible for these modifications and their recruitment to chromatin. 11 Reduced expression of TET at the gene and protein levels as well as changes in the level of 5-hmC is thought to be associated with the development and progression of several types of cancer, but there are no such data related to endometrial cancer.
Endometrial cancer is the third most frequent gynecologic malignancy worldwide. 12 Endometrial cancer is a multifactorial disease, and the molecular mechanism underlying its development and progression is poorly understood. However, it is suggested that several aberration in genes expression may be involved. 13
The aim of our study was to analyze the messenger RNA (mRNA) expression levels of TET1, TET2, and TET3 and their correlation with 5-hmC/5-mC levels, clinical features, and patient survival. Our results demonstrated that especially decreased expression of TET1 correlates with tumor progression, and it is an unfavorable prognostic indicator for patients with endometrial cancer.
Materials and methods
Patients and tissue samples
Samples of endometrial cancer were obtained from 131 patients who underwent surgery in the Department of Gynecological Oncology Copernicus Memorial Hospital (Łódź, Poland) from February 2009 to March 2016. Samples of normal endometrial tissues were obtained from 51 patients who had undergone hysterectomy due to leiomyoma or a uterus prolapse. Tissue samples after tumor resection were immediately placed in RNAlater (Ambion, USA). Samples were subsequently stored at −80°C until RNA and DNA extraction.
All cancer samples were characterized in terms of tumor stage according to the International Federation of Gynecology and Obstetrics (FIGO) criteria, histological grade and type according to World Health Organization (WHO) classification, infiltration into muscularis, the ability of cancer cells to metastasize to lymph nodes, and coexistence of endometrial hyperplasia. Clinicopathological characteristics are summarized in Table 1. The follow-up period was defined as the interval from the date of surgery to the date of death or last follow-up. The latest follow-up was updated in March 2016.
Characteristics of patients and endometrial cancer samples.

RNA extraction and cDNA synthesis
Total RNA from normal and cancer tissue samples was isolated using Trizol® Reagent (Sigma Aldrich, USA) according to manufacturer’s protocol and quantified spectrophotometrically. The reverse transcription of 2 µg of RNA was performed using High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, USA) following manufacturer’s instructions. The reverse transcription polymerase chain reaction (RT-PCR) was performed in a thermocycler GeneAMP 2400 Thermal Cycler (Perkin-Elmer, USA) according to the following thermal profile: 10 min at 25°C, 120 min at 37°C, and 5 min at 85°C.
Quantitative real-time PCR analysis
The relative expression levels of TET1, TET2, and TET3 were analyzed by real-time polymerase chain reaction (PCR) using the TaqMan® Gene Expression Assays (Life Technology, USA) according to manufacturer’s instructions. The assay numbers for studied genes were as follows Hs00286756_m1, Hs00325999_m1, Hs00379125_m1, and Hs02800695_m1 (reference gene: HPRT1). Each PCR reaction was performed in a 10 µL volume which included 1 µL of complementary DNA (cDNA), 3.5 µL of water, 5 µL of 2 × TaqMan Universal PCR MasterMix (Life Technology, USA), and 0.5 µL of TaqMan Gene Expression Assays which consisted of a pair of unlabeled PCR primer and a TaqMan probe with FAM™ dye label on the 5′ end and minor groove binder (MGB) nonfluorescent quencher on the 3′ end. The following PCR program was used: 95°C for 10 min, 40 cycles at 95°C for 15 s, 1 min annealing and extension at 60°C. PCR reactions were carried out using the Mastercycler ep realplex (Eppendorf, Germany). The equation 1000 × 2−ΔCt was applied to calculate the expression of studied genes, where ΔCt = Ct of the target gene − Ct of the reference gene (HPRT1). Results are expressed as number of target gene mRNA copies per 1000 copies of HPRT1 mRNA.
DNA extraction
The phase remaining after isolation of RNA was mixed with ethanol, incubated at room temperature for 5 min, and centrifuged for 10 min at 10,000 × g. Supernatant was removed and 1 mL of 0.1 M citrate solution in 10% ethanol was added to the pellet; probes were incubated for at least 30 min at room temperature, stirring occasionally by inverting the tube, and then centrifuged for 10 min at 10,000 × g. Washing with citrate was done three times. After removing the supernatant, pellet was washed with 75% ethanol and incubated for 20 min at room temperature and centrifuged for 10 min at 12,000 × g. The DNA pellet was dried at 37°C. The precipitate was suspended in an appropriate amount of 8 mM NaOH, incubated at 37°C for 15 min, and vortexed vigorously until complete dissolution of DNA. After last centrifugation at 16,000×g for 10 min, supernatant containing the DNA was transferred to a new tube.
Global 5-mC and 5-hmC measurement
MethylFlash™ Methylated DNA Quantification Kit (Epigentek, USA) and MethylFlash™ Hydroxymethylated DNA Quantification Kit (Epigentek, USA), respectively, were used for the quantification of global 5-mC and 5-hmC in DNA according to manufacturer’s instructions. In both assays, hydroxymethylated and methylated fractions of DNA were detected using capture and detection antibodies through an enzyme-linked immunosorbent assay (ELISA)-like reaction. Absorbance was read in a microplate spectrophotometer at 450 nm. Because of high specificity, these assays are able to distinguish between 5-hmC, 5-mC, and C.
Statistical analysis
Differences in expression levels of TET1, TET2, and TET3 between studied groups were analyzed using non-parametric Kruskal–Wallis test with the post hoc Dunn test. Correlations between TET genes expression and level of 5-hmC/5-mC in DNA were analyzed using Spearman test. Survival curves were calculated using the Kaplan–Meier method and compared by the log-rank test. Univariate and multivariate survival analyses were performed on all characteristics (age, FIGO stage, histological grade, tumor stage, lymph nodal status, and myometrial infiltration) through the Cox proportional hazard regression model. A value of p < 0.05 was considered statistically significant.
Results
Expression of TET1, TET2, and TET3 in endometrial cancer
Expression levels of TET1, TET2, and TET3 in samples of endometrial cancers and normal endometrial tissues were evaluated by real-time quantitative PCR analysis with the HPRT1 gene applied as a reference and correlated with clinical and pathomorphological data (Figure 1). Analysis showed that the mean mRNA expressions of TET1 and TET2 were significantly lower in cancer samples than in normal tissues samples. In more advanced tumors, classified as stage III and IV, the expression of TET1 and TET2 was lower than in tumors less advanced, corresponding to the first and second stage according to FIGO classification (Figure 1(a)). There were no significant differences (p > 0.05) in TET1 and TET2 mRNA expression between tumors of different grade (Figure 1(b)). However, TET1 and TET2 expressions were lower in tumors that exhibited the ability to metastasize to regional lymph nodes compared to non-metastatic cancers (Figure 1(c))

Expression of TET1, TET2, and TET3 mean mRNA measured by real-time PCR in control and endometrial cancer samples; (a) a comparison between subgroups with different FIGO stage, (b) histological differentiation grade, and (c) lymph node metastasis status. The results are shown as number of each TET mRNA copies per 1000 copies of HPRT1 mRNA (reference gene). Graphs represent mean ± SEM. The number of cases in each group is shown in parentheses.
In contrast to the results obtained for TET1 and TET2, the relative mRNA expression of the TET3 was higher in the cancer samples compared to normal tissues. There were no significant differences (p > 0.05) in TET3 expression between tumors in different stage of development according to FIGO classification, but TET3 mRNA expression showed a tendency to increase with increasing tumor histological grade (Figure 1(a) and (b)).
Analysis did not reveal significant differences (p > 0.05) in the expression of all three TET genes between samples in which the infiltration did not exceed half the thickness of muscle (<1/2) and those in which infiltration included more than half the thickness of muscle (≥1/2). There was a statistically significant decrease in expression of the TET1 in the tumor samples characterized by coexistence of hyperplasia compared to those in which there was no excessive growth (p < 0.05).
Correlation between 5-mC and 5-hmC levels and TETs expression
The cancer samples showed slightly increased level of 5-mC and decreased level of 5-hmC compared to control samples. However, both 5-mC and 5-hmC levels showed no significant association with clinicopathological parameters such as tumor stage, histological grade, depth of myometrial invasion, hyperplasia coexistence, and lymph node metastasis (data not shown). The percentage of 5-hmC in the total DNA (5-hmC %) was correlated with the relative expression of TET1, TET2 and TET3 in the same sample. Positive correlation between 5-hmC % and the relative expression of TET1 (r = 0.3755) and TET2 (r = 0.3184) was observed, but no correlation was observed in the case of TET3 (Figure 2). There was no statistically significant correlation between expression of TET genes and the level of DNA methylation (data not shown).

Correlation between TETs expression and the level of 5-hydroxymethylcytosine (5-hmC %). Spearman rank correlation analysis was performed to analyze the association of (a) TET1, (b) TET2, and (c) TET3 expression with 5-hmC level in endometrial cancer samples. *5-hmC level was significantly associated only with TET1 (r = 0.375, p < 0.0001) and TET2 (r = 0.318, p < 0.0001) expression levels.
TET1 status and endometrial cancer prognosis
The 66 endometrial cancer patients who underwent surgery before March 2011 were classified into high and low TET1, TET2, or TET3 expression subgroups using their median expression value as the cutoff point. Kaplan–Meier survival analysis was used to estimate the predictive effect of TET1, TET2, and TET3 expressions on overall survival. Overall survival differences in case of TET2 and TET3 were not statistically significant. The Kaplan–Meier analysis showed that patients in the low TET1 expression group had a significantly shorter overall survival than those in the high TET1 group (5-year survival, log-rank test, p < 0.04, Figure 3).
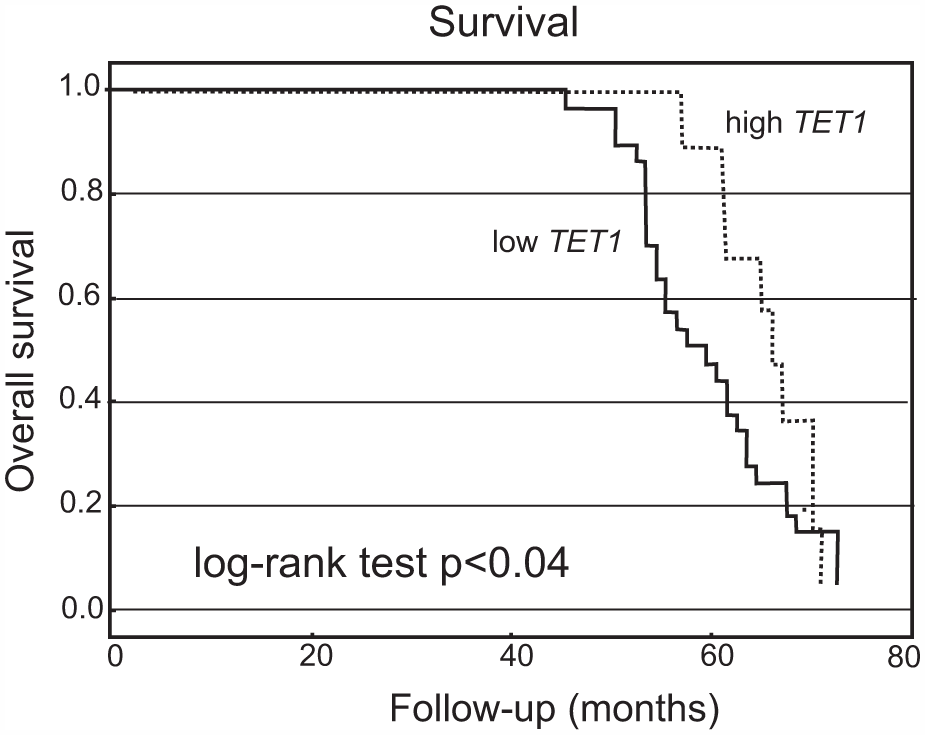
Kaplan–Meier curve showing overall survival for endometrial cancer patients with low and high levels of TET1 mRNA expression. The cutoff value was established to be the median of the TET1 mRNA level. The survival curve was compared between two groups: high (≥ median value) and low (<median value) expression. Patients in the low TET1 expression group had a significantly shorter overall survival than those in the high TET1 expression group. N = 66 (number of cases).
To assess whether TET1 expression represents an independent prognostic indicator in endometrial cancer, the effect of each variable on survival was determined by the Cox regression analysis. Univariate analyses revealed that TET1 expression, FIGO stage, grade, tumor size, and nodal status were significantly associated with overall survival (Table 2). The variables that significantly correlated with survival in the univariate analysis were further assessed by multivariate analysis. The results of the multivariate analysis confirmed that TET1 expression was an independent prognostic factor for overall survival with endometrial cancer (hazard ratio: 0.509, 95% confidence interval (95% CI) = 0.127–0.779, p = 0.038) (Table 2).
Univariate and multivariate analyses of TET1 expression and clinicopathological factors influencing patients’ survival.
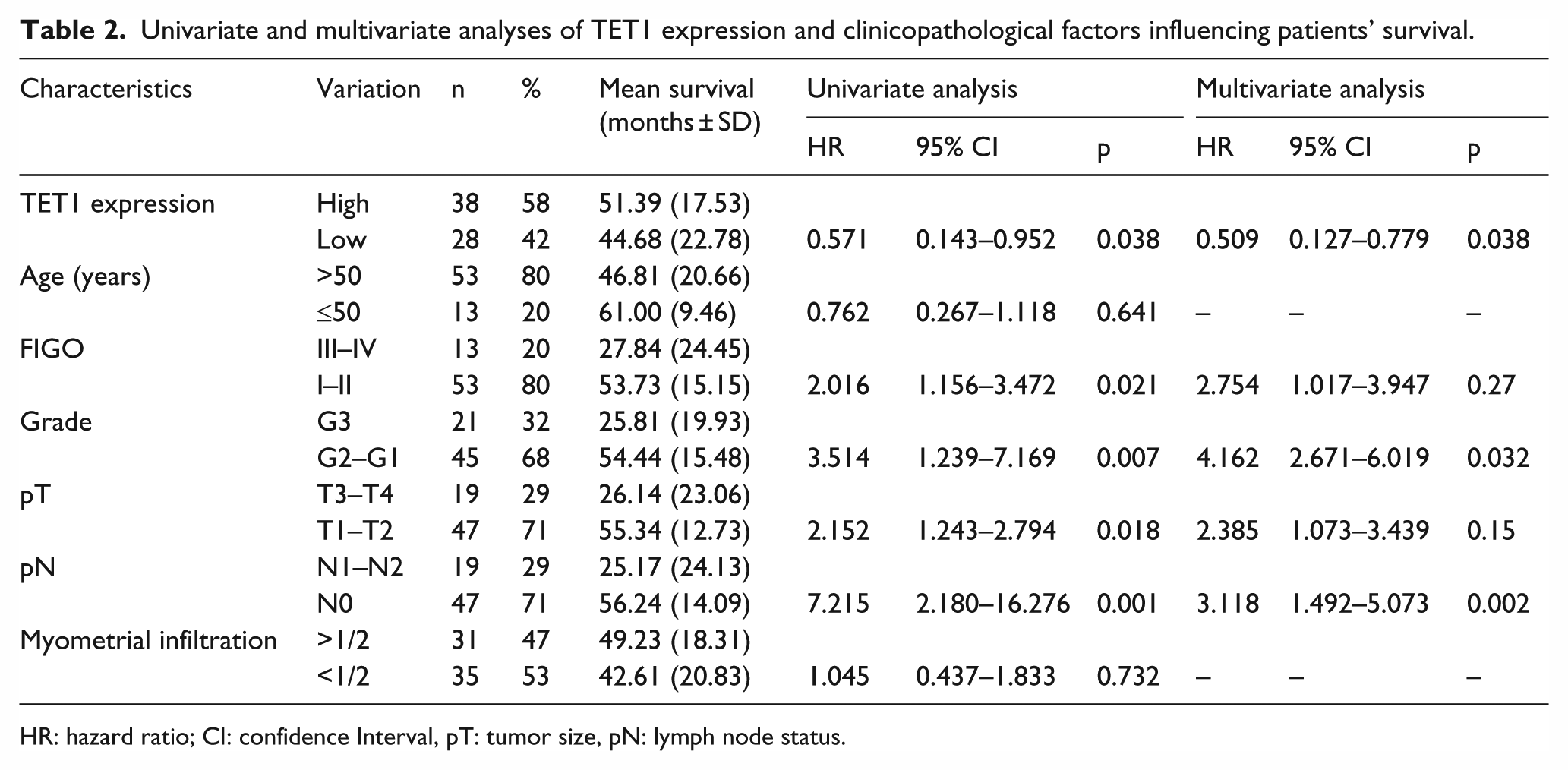
HR: hazard ratio; CI: confidence Interval, pT: tumor size, pN: lymph node status.
Discussion
Reduced 5-hmC levels have been found in numerous solid tumors including glioma, melanoma, breast, colon, liver, lung, prostate, and pancreatic cancers.14–24 It has been found that low 5-hmC levels correlate with poor survival in glioma, melanoma, breast, and liver cancers.14–16,20 The results of many studies suggest that the reduced 5-hmC level is associated with reduced expression of TETs.17,23,20–22,24
According to our knowledge, there are no data concerning the expression of TETs and 5-hmC level in endometrial cancer. Navarro et al. 25 studied expression of TET genes in uterine leiomyomas which are benign smooth muscle tumors of myometrial origin. They found higher expression of TET1 and TET3 on both mRNA and protein levels, but not TET2, in leiomyoma tissue compared to normal myometrial tissue. Navarro et al. 25 demonstrated that the up-regulation of TET1 and TET3 in leiomyoma tissue was associated with increase of 5-hmC level.
The results of our studies of endometrial cancer similarly to most studies of other malignant tumors showed decreased expression of TET1 and TET2 in cancers compared to control samples. Our finding of higher expression of TET3 mRNA level in cancers than in control stands is in contrast to most previous findings in malignant tumors which showed usually lower mRNA expression of all TET genes. However, there were some exceptions. In diffuse intrinsic pontine glioma, both TET1 and TET3 expressions were increased compared to normal tissue, and in esophageal squamous cell carcinoma, TET3 expression was higher than in matched normal esophageal mucosa.26,27 Our studies showed positive correlation of 5-hmC levels with TET1 and TET2 expression but not TET3. Previous studies of human cancers also showed that TETs were not equally involved in 5-mC hydroxylation. For example, Du et al. 18 found that in gastric cancers the global 5-hmC levels were significantly and positively correlated with TET1 expression, but not with TET2/3. Decreased expression of TETs, but especially TET2, seems to be a key mechanism responsible for reduced level of 5-hmC in melanoma and esophageal squamous cell carcinoma.16,27 The exact function of each TET protein in regulating the 5-hmC level is still unclear. The recent studies suggest that all TETs hydroxylate of 5-mC to 5-hmC, but only TET2 and TET3 are responsible for subsequent removal of 5-hmC in the cytosine demethylation cascade. 28
Our results may suggest that the role of TET3 in endometrial cancer is different from that of TET1 and TET2 and may be independent of its catalytic activity. The three TET proteins share high percentage of sequence homology in the region surrounding the catalytic C-terminal domain, and they have similar enzymatic activity. However, the distinctive regions of each protein suggest that each TET possesses a unique binding affinity to chromatin or interacts with different proteins. Thus, it is possible that each TET can play a specific role depending on cell types and different stages of tumor development. Although most studies were focused on the enzymatic roles of TET family proteins, a few investigations reported a non-catalytic activity of TET proteins. For example, it has been found that TET1 interacts with hypoxia-inducible factors (HIFs) and increases hypoxia responsive gene expression. TET1 also interacts with the SIN3A co-repressor complex leading to transcriptional repression of many TET1-targeted genes.29–31 TET2 and TET3 can interact with O-GlcNAc transferase (OGT), and these interactions promote GlcNAcylation and increase H3K4me3 through the SET1/COMPASS complex. 32 Non-catalytic activities of TETs cause that except tumor-suppressing function they also have tumor-promoting function. Thus, we speculate that TETs may also play a dual function in endometrial tumorigenesis
A variety of mechanisms in human tumors may interfere either directly or indirectly with TETs expression, including TET mutations, upstream regulators of TET genes, and methylation of TET genes. The differences in transcript levels of TETs may dependent also on different microRNA (miRNA) expression. There are several miRNAs that have been proved to affect the TETs transcript levels. Among miRNAs that directly regulate the levels of TETs are miR-29, miR-26, miR-22, miR-101, miR-105, miR-125, and miR767.33–36 Most of these miRNAs affect transcript levels of all TETs. However, some of them showed preferential effect on TET transcripts. Zhang et al. 33 found that the number of binding sites seems to influence the sensitivity of TET transcripts by miR-29 as TET3 has the most predicted sites and is the most significantly regulated TET by miR-29 in human dermal fibroblasts and vascular smooth muscle cells. In melanoma cell lines, it has been shown that miR-767 can function as a regulator of cellular 5-hmC levels via targeting of TET genes, but miR-767 has exhibited a preferential effect on TET1. 37 Although there is are data concerning mechanism of TET’s expression regulation in endometrial cancer, it is known that deregulation of some of these miRNAs, that is, miR-101 and miR-22, are associated with endometrial carcinogenesis.38–40
Our results for the first time suggest that decreased mRNA expression of TET1 correlates with tumor progression and may serve as a potential prognostic biomarker in endometrial cancer. Analysis revealed that lower TET1 expression in tumors significantly predicted poorer overall survival. However, our study included relatively small group of patients, and a well-designed large-scale study is necessary to confirm our results.
In conclusion, our study demonstrates that TET1 and TET2 but not TET3 transcript levels in endometrial cancer tissues are reduced and correlated with 5-hmC levels. TET1 downregulation may be a potential prognostic marker in endometrial cancer. Further studies are needed to explain the significance of individual TETs alteration in onset and progression of endometrial cancer.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
We declare that all experiments were performed in accordance with the current law of Poland. The investigations were approved by the Bioethical Commissions of Medical University of Lodz and University of Lodz.
Funding
This study was supported in part by statutory funds of University of Lodz and by grant from the National Science Centre of Poland UMO-2015/19/N/NZ3/01311.
