Abstract
SPAG9 is a novel tumor associated antigen, expressed in variety of malignancies. However, its role in ovarian cancer remains unexplored. SPAG9 expression was validated in ovarian cancer cells by real time PCR and Western blot. SPAG9 involvement in cell cycle, DNA damage, apoptosis, paclitaxel sensitivity and epithelial- mesenchymal transition (EMT) was investigated employing RNA interference approach. Combinatorial effect of SPAG9 ablation and paclitaxel treatment was evaluated in in vitro. Quantitative PCR and Western blot analysis revealed SPAG9 expression in A10, SKOV-3 and Caov3 compared to normal ovarian epithelial cells. SPAG9 ablation resulted in reduced cellular proliferation, colony forming ability and enhanced cytotoxicity of chemotherapeutic agent paclitaxel. Effect of ablation of SPAG9 on cell cycle revealed S phase arrest and showed decreased expression of CDK1, CDK2, CDK4, CDK6, cyclin B1, cyclin D1, cyclin E and increased expression of tumor suppressor p21. Ablation of SPAG9 also resulted in increased apoptosis with increased expression of various pro- apoptotic molecules including BAD, BID, PUMA, caspase 3, caspase 7, caspase 8 and cytochrome C. Decreased expression of mesenchymal markers and increased expression of epithelial markers was found in SPAG9 ablated cells. Combinatorial effect of SPAG9 ablation and paclitaxel treatment was evaluated in in vitro assays which showed that ablation of SPAG9 resulted in increased paclitaxel sensitivity and caused enhanced cell death. In vivo ovarian cancer xenograft studies showed that ablation of SPAG9 resulted in significant reduction in tumor growth. Present study revealed therapeutic potential of SPAG9 in ovarian cancer.
Introduction
Epithelial ovarian cancer (EOC) is one of the most lethal cancers and the second leading cause of death among gynecological cancers. 1 In developing countries the mortality rate is even higher due to lack of medical infrastructure and limited awareness. 2 Cancer testis (CT) antigens are a unique family of tumor specific antigens which are aberrantly expressed in several different histologic tumor types and have been shown to be associated with tumor growth and survival. 3 Previously, we have documented that sperm associated antigen 9 (SPAG9), a CT antigen, could be a promising molecule for early detection and diagnosis of various cancers including cervix, 4 breast, 5 chronic myeloid leukemia, 6 bladder transitional cell carcinoma 7 and renal cell carcinoma. 8 We also documented SPAG9 expression at mRNA and protein level in ovarian cancer specimens, irrespective of histotypes and disease stages. 9 Recently, it has been reported that SPAG9 plays an important role in various molecular pathways including cell cycle, apoptosis and epithelial mesenchymal transition (EMT) in triple negative breast cancer. 10 However, till date the involvement of SPAG9 at molecular level in ovarian cancer is not yet explored.
In present study we analyzed the role of SPAG9 in various molecular pathways involved in tumor progression which may be of clinical relevance for targeting ovarian cancer for better cancer management. Plasmid-based small hairpin RNA (shRNA) approach was used to ablate SPAG9 in ovarian cancer cells to investigate its effect on various malignant properties of ovarian cancer cells. SPAG9 ablated ovarian cancer cells showed increased DNA damage, S phase cell cycle arrest, caspase mediated cell death and decreased EMT. Also, combinatorial effect of gene silencing and paclitaxel was examined which revealed enhanced effect on cell death. In addition, in vivo ovarian cancer xenograft studies showed reduced volume and tumor growth in SPAG9 shRNA treated mice. Hence, for the first time we have put forth evidence that SPAG9 expression is important for cancer progression and is associated with cell cycle, apoptosis and EMT pathways in EOC.
Methods
Cell lines and cell culture
Three ovarian cancer cell lines A10, SKOV-3 and Caov3 were used in this study. Among these serous papillary cystadenocarcinoma A10 cell line was generously provided by Dr. Kunle Odunsi (Roswell Park Cancer Institute, Buffalo, NY). Ovary adenocarcinoma cell lines Caov3 and SKOV-3 were purchased from American Type Culture Collection (ATCC, Manassas, USA). All cell lines were grown in recommended culture media. The cell lines were revived and mycoplasma contamination was checked using mycoplasma PCR detection kit (Applied Biological Materials Inc., Richmond, Canada) before using for experiments. Human normal ovarian epithelial cells were purchased and maintained according to manufacturer’s directions (Cell Biologics Incorporation., Chicago IL).
Antibodies
Western blot and immunohistochemistry analysis were carried out using following antibodies; anti-SPAG9 antibody was raised in rat, mouse anti-proliferating cell nuclear antigen (PCNA), mouse anti-calnexin (endoplasmic reticulum maker), mouse anti-GM130 (Golgi body marker) and mouse anti-lamin A/C (nuclear envelope marker) were purchased from Santa Cruz Biotechnology, USA. Horseradish peroxidase-conjugated anti-rat IgG, FITC-conjugated anti-rat IgG, and Texas Red-conjugated anti-mouse IgG were procured from Jackson ImmunoResearch Laboratories, West Grove, PA, USA. Mouse anti-beta actin, anti-MTCO2 (mitochondrial marker), mouse anti-E-cadherin, mouse anti-N-cadherin, mouse anti-P-cadherin, mouse anti-SLUG, mouse anti-TWIST, rabbit anti-Vimentin, mouse anti-Caspase 3, mouse anti-CDK1, rabbit anti-CDK2 were procured from Abcam, Cambridge, UK. Mouse anti-BCL-2-associated death promoter (BAD), mouse anti-BCL-2-associated X Protein (BAX), rabbit anti-BID, rabbit anti-Bcl-xL, mouse anti-cytochrome-C, rabbit anti-p53 upregulated modulator of apoptosis (PUMA), mouse anti-Caspase 7, mouse anti-Caspase 8, Cyclin-dependent kinases (CDKs): mouse anti-CDK4 and mouse anti-CDK6, mouse anti-Cyclin B1, mouse anti-Cyclin D1, mouse anti-Cyclin E, mouse anti-cyclin-dependent kinase inhibitor (CKI) and mouse anti-p21 were procured from Santa Cruz Biotechnology. Mouse anti-B-cell lymphoma 2 (BCL-2) and rabbit anti-induced myeloid leukemia cell differentiation protein (Mcl-1) was procured from Cell Signaling Technology, USA.
Real-time polymerase chain reaction (qPCR)
SPAG9 mRNA expression was quantified using real-time polymerase chain reaction (qPCR) in all three ovarian cancer cell lines as described earlier. 10 Reaction was carried out using SPAG9 and β-actin specific primer sets (Supplementary Table 1). Subsequently, SPAG9 gene expression level was normalized using β-actin as endogenous control. Experiments were performed three independent times in triplicates.
Western blotting and fluorescence activated cell sorting (FACS)
SPAG9 protein expression was examined in all three ovarian cancer cell lines by performing Western blot analysis as described previously. 10 Flow cytometric analysis was performed using FACS Verse flow cytometer (BD Biosciences, San Jose, CA) and data was analyzed using FlowJo software (Treestar, Ashland, OR) as described previously. 10 Experiments were performed three independent times in triplicates.
SPAG9 protein localization by indirect immunofluorescence (IIF)
SPAG9 protein localization in different cell organelles of ovarian cancer cells was examined using IIF as described previously. 10 Images were captured using Carl Zeiss LSM 510 Meta confocal microscope (Zeiss, Oberkochen, Germany). For co-localization studies antibodies used were: endoplasmic reticulum (Calnexin; Sc70481), mitochondria (MTCO2; ab3298), Golgi bodies (GM130; Sc55591) and nuclear envelope (Lamin a/c; Sc7292). Experiments were performed three independent times in triplicates.
Plasmid-based shRNA gene silencing
To study the role of SPAG9 at molecular level in ovarian cancer cells, transient transfection was carried out in A10 and SKOV-3 cells by two shRNA constructs (shRNA1 or shRNA2) for SPAG9 with scrambled shRNA (NCshRNA) as described earlier. 10 The transfection was carried out with 6 μg of shRNA plasmid using Lipofectamine (Invitrogen, Thermo Fisher Scientific, MA, USA) in optiMEM media (Gibco, Thermo Fisher Scientific, MA, USA). Cells were harvested 48-h post transfection for Western blot analysis. Experiments were performed three independent times in triplicates.
Cellular proliferation and cell viability assay
Cellular proliferation and cell viability was assessed in A10 and SKOV-3 cells transfected with SPAG9 shRNA1 or shRNA2 or in combination with paclitaxel (0.002 µM for A10 and 0.003 µM for SKOV-3 cells) seeded in triplicate in 24 well plate for cellular proliferation assay and 96 well plate for cell viability assay. NC shRNA or only paclitaxel treatment was used as control. Tetrazolium dye MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] was used as described previously 10 and absorbance was recorded at wavelength of 490 nm. Experiments were performed three independent times in triplicates.
Measurement of synergistic effect
Synergistic, additive or antagonistic effect of SPAG9 shRNA and paclitaxel in different combinations was assessed using the median-effect analysis method, as previously described. 11 The SPAG9 shRNA and paclitaxel were combined in a fixed ratio of doses. Combination index (CI) values of interactions between SPAG9 shRNA and paclitaxel were assessed using CompuSyn 1.01 software (ComboSyn, Inc., Paramus, NJ, USA). Data were expressed as CI values, with definition for additive (CI=1), synergism (CI<1) and antagonism (CI>1) in drug combination. 11 Our aim was to achieve maximal effect of combination treatment on ovarian cancer cells. Therefore, a mean CI value was calculated from data points with fraction affected (Fa) >0.5. A Fa<0.5 indicated less growth inhibition and more cellular proliferation. Hence Fa<0.5 was not considered in our experiments. All experiments were done in triplicates over three independent times.
Colony formation assay
Colony formation assay was performed as described previously. 10 Colonies were fixed, stained and counted using Nikon Eclipse E 400 microscope (Nikon, Fukok, Japan). Experiments were performed three independent times in triplicates.
Propidium iodide (PI) staining for cell cycle analysis
Cell cycle analysis was done in A10 and SKOV-3 cells transfected with NCshRNA, or SPAG9 shRNA1 or shRNA2 as described earlier. 10 The acquisition and analysis were done using BD-FACS VERSE (BD Biosciences, CA, USA). Experiments were performed three independent times in triplicates.
Annexin V staining
Annexin V staining was done in A10 and SKOV-3 cells transfected with both SPAG9 shRNA (shRNA1 or shRNA2) or NCshRNA as described earlier. 10 Staining was done with annexin V-FITC kit as per manufacturer’s protocol. The analysis of samples was done with BD-FACS VERSE (BD Biosciences, CA, USA). Experiments were performed three independent times in triplicates.
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay
NC shRNA, SPAG9 shRNA1 or shRNA2 transfected A10 and SKOV-3 cells were assessed for DNA damage by TUNEL assay as described earlier. 10 The cells were analyzed at 488 nm using BD-FACS VERSE (BD Biosciences, CA, USA). Experiments were performed three independent times in triplicates.
M30 assay
M30 assay was carried out in A10 and SKOV-3 cells transfected with SPAG9 shRNA (shRNA1 or shRNA2) or NCshRNA as described earlier. 10 Cells were fixed and probed with M30 antibody (Roche Diagnostics, GmBH, Mannheim, Germany). For detection, anti-mouse FITC-conjugated antibody (Jackson ImmunoResearch Laboratories, Baltimore, USA) was used. Data was acquired using BD-FACS CALIBUR (BD Biosciences, California, USA). Experiments were performed three independent times in triplicates.
Cell migration, invasion and wound healing assay
Role of SPAG9 in migration and invasion abilities of ovarian cancer cells was investigated as described previously. 12 For wound healing assay A10 and SKOV-3 cells transfected with SPAG9 shRNA (shRNA1 or shRNA2) or NCshRNA were seeded in 35mm culture plates. At full confluency a scratch was made in cell monolayer. Images were captured at regular intervals (0h, 12h, 24h and 48h) as described previously. 12 Experiments were performed three independent times in triplicates.
Effect of SPAG9 ablation on the growth of ovarian cancer xenograft
In vivo effect of SPAG9 ablation was studied in athymic nude mice after obtaining approval from Institute animal ethical committee (IAEC No. 360/14). For xenograft establishment epithelial ovarian cancer cells (A10 cells; 5 × 106 cells per mice) were injected subcutaneously and processed further as described earlier. 10 Briefly, tumor growth experiments of human tumor xenograft of ovarian cancer A10 cells were carried out in 16 athymic nude mice. When the tumor size reached between 50mm3-100mm3, animals were divided in two groups (control n=8 mice and experimental, n=8 mice). SPAG9 shRNA plasmid (50μg) was injected intra-tumorly, followed by injections of 25μg SPAG9 shRNA plasmid thrice weekly for 7 weeks. Animals were euthanized and tumors were dissected for Immuno-histochemical studies.
Immunohistochemistry (IHC)
Immunohistochemistry (IHC) was carried out to detect SPAG9 protein expression and other molecules involved in different pathways as described earlier. 10 Tumor tissue sections were probed with various antibodies (anti-SPAG9, anti-PCNA; Cell cycle molecules: anti-Cyclin B1, anti-Cyclin D1, anti-CDK4, anti-CDK6 and anti-p21; Apoptosis molecules: anti-BID, anti-BAX, anti-Bcl-xL, anti-BCL2, anti-Mcl-1, anti-caspase 3, anti-caspase 8 and anti-cytochrome C; EMT molecules: anti-N-cadherin, anti-SLUG, anti-TWIST, anti-Vimentin and anti-E-cadherin). Images were obtained using Nikon Eclipse E400 microscope (Nikon, Fukuoka, Japan).
Statistical analysis
For all in vitro and in vivo assays statistical analysis was carried out using statistical software package version 20.0 (SPSS Inc., Chicago, USA). Two tailed student’s t-test was performed to check significance of P value. Data are expressed as mean±standard error of the mean of three independent experiments in triplicates in in-vitro assays. A P-value of less than 0.05 was considered statistically significant.
Results
SPAG9 is expressed in ovarian cancer cells
SPAG9 mRNA expression was detected by real time PCR (qPCR) in A10, Caov3 and SKOV-3 ovarian cancer cells. We observed higher expression of SPAG9 in A10 (2.7 fold), Caov3 (1.7 fold) and SKOV-3 cells (3.2 fold) relative to normal ovarian cells (Figure 1(a)). SPAG9 protein expression was validated by Western blotting in ovarian cancer cells (Figure 1(b)). Subsequently, FACS analysis revealed surface localization of SPAG9 protein in A10 (97.2%) and in SKOV-3 (94.2%) cells (Figure 1(c)). In addition, we also found cytoplasmic localization of SPAG9 in ovarian cancer cells. SPAG9 co-localization was also observed with endoplasmic reticulum, golgi bodies and mitochondria (Figure 1(d)). However, SPAG9 did not co-localize with nuclear envelope (Figure 1(d)).

SPAG9 gene and protein expression in ovarian cancer cell lines. (a) Real time PCR shows higher SPAG9 mRNA expression in A10, SKOV-3 and Caov3 ovarian cancer cells relative to normal ovarian cells (b) Western blot shows SPAG9 protein expression in A10, SKOV-3 and Caov3 ovarian cancer cells. β actin was used as endogenous loading control. (c) Flow cytometric analysis of surface expression of SPAG9 protein in A10 and SKOV-3 cells indicates higher SPAG9 positive A10 (97.2%) and SKOV-3 (94.2%) cells as compared to control cells (12%) and (7%) respectively. (d) IIF depicts SPAG9 cytoplasmic localization (green color) in ovarian cancer A10 and SKOV-3 cells. SPAG9 co-localization (yellow- orange staining) with endoplasmic reticulum (ER), Golgi bodies, mitochondria and nuclear envelope. However, SPAG9 was not localized with nuclear envelope. Nuclear staining is performed using DAPI. (Original magnification: ×630, objective: ×63).
Knockdown of SPAG9 results in reduced cellular proliferation, colony forming ability and enhances cytotoxicity of paclitaxel
To investigate the role of SPAG9 in tumorigenic properties, plasmid based gene silencing approach was used to ablate SPAG9 expression in ovarian cancer cells. Two SPAG9 shRNA targets (shRNA1 or shRNA2) and control scrambled shRNA (NC shRNA) in A10 and SKOV-3 cancer cells were used to ablate SPAG9 expression. Our qPCR data showed that SPAG9 transcript was significantly downregulated with SPAG9 shRNA1 (P<0.01) or shRNA2 (P<0.001) treatment as compared to scrambled NC shRNA (Supplementary Figure S1(a)) in A10 and SKOV-3 cells respectively. Western blot analysis further validated reduced SPAG9 protein expression in both A10 and SKOV-3 cells (Figure 2(a)). In all subsequent in vitro assays, SPAG9 shRNA1 or SPAG9 shRNA2 were used along with scrambled NC shRNA. Effect of SPAG9 ablation on cellular proliferation revealed significant reduction in A10 (45.41%; P<0.001) and SKOV-3 (43.86%; P<0.001) cells with shRNA1 as compared to NC shRNA treatment (Figure 2(b)). Similarly, significant reduction of cellular proliferation in A10 (49.86%; P<0.001) and SKOV-3 (45.09%; P<0.001) cells with shRNA2 as compared to NC shRNA treatment (Figure 2(b)) was observed. Further, paclitaxel treatment on A10 and SKOV-3 cells resulted in significant reduction in cellular proliferation. There was significant reduction in cellular proliferation of A10 (52.78%; P<0.001) and SKOV-3 (41.03%; P<0.001) cells respectively as compared to untreated ovarian cancer cells (Figure 2(b)). We further examined the combined effect of paclitaxel and SPAG9 shRNA on cellular proliferation in A10 and SKOV-3 cells which revealed enhanced chemotherapeutic sensitivity with reduced cellular proliferation. There was significant reduction in cellular proliferation post 72h with paclitaxel and shRNA1 in A10 (60.94%; P<0.001) and SKOV-3 (63.88%; P<0.001) cells as compared to untreated cells. Similarly, we observed significant reduction when treated with paclitaxel and shRNA2 in A10 (60.62%; P<0.001) and SKOV-3 (62.88%; P<0.001) cells as compared to untreated cells. (Figure 2(b)).
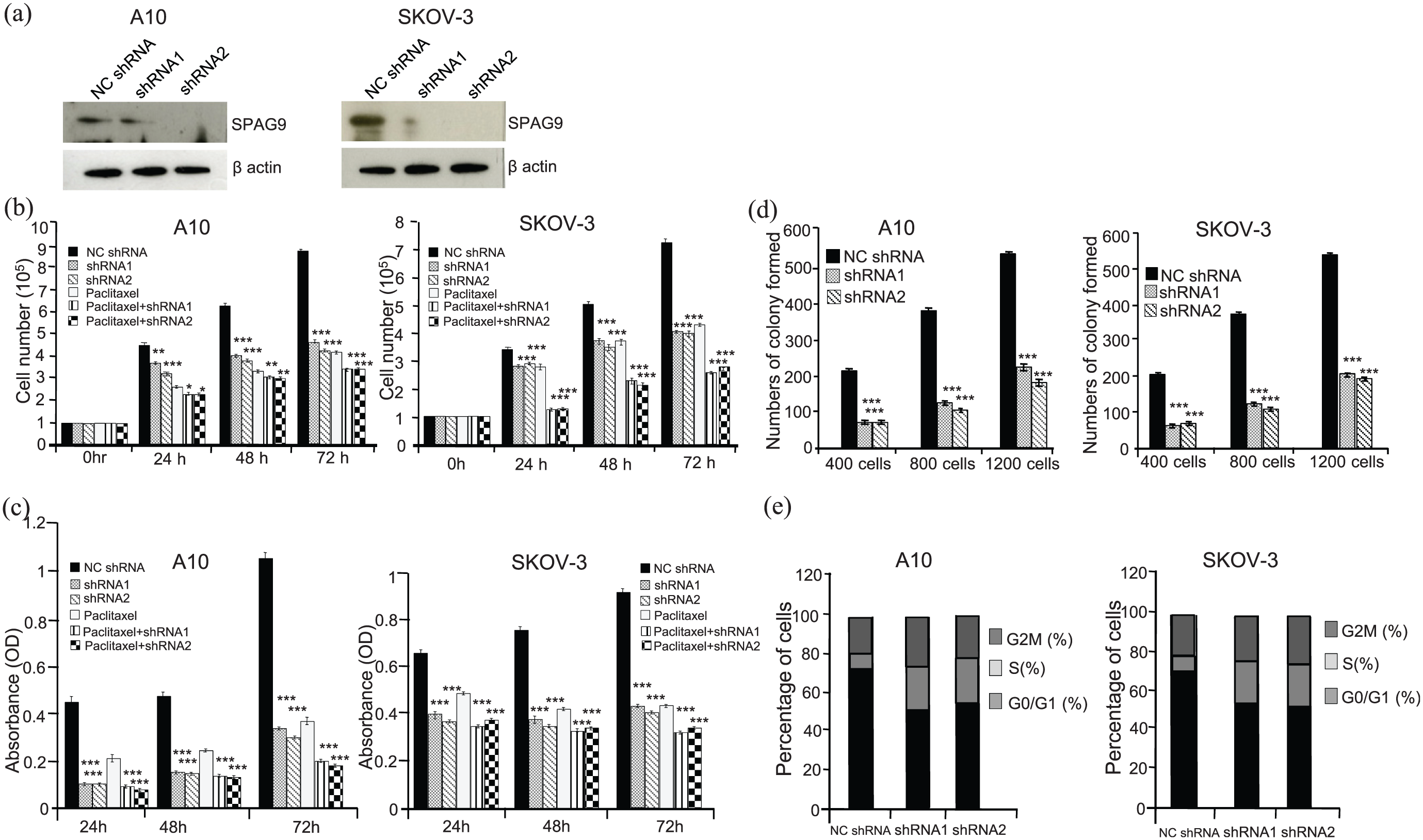
Ablation of SPAG9 results in reduced growth and colony forming ability. (a) Western blot analysis show reduced SPAG9 expression in SPAG9 shRNA (shRNA1 or shRNA2) transfected A10 and SKOV-3 cells as compared to NC shRNA cells. β actin was used as endogenous loading control.(b) Histogram shows significant reduction in cellular proliferation post 24 h, 48 h and 72 h treatment with SPAG9 shRNA1or shRNA2 or paclitaxel alone or combination of paclitaxel with SPAG9 shRNA1 or shRNA2 as compared to NC shRNA transfected cells in A10 and SKOV-3 cells. (c) Cellular viability assay (MTT assay) in A10 and SKOV-3 cells.Histogram depicting significant reduction in number of viable cells post 24 h, 48 h and 72 h treatment with shRNA1 or shRNA2 or paclitaxel alone or combination of paclitaxel with shRNA1 or shRNA2 as compared to NC shRNA. (d) Histogram shows number of colonies formed in A10 and SKOV-3 cells. Significant reduction in colony forming ability was observed in SPAG9 shRNA1 or SPAG9 shRNA2 treated cells as compared NC shRNA cells. (e) Histogram and bar diagram depicts S phase growth arrest in A10 and SKOV-3 cells when transfected with shRNA1 or shRNA2 as compared to NC shRNA. Experiments were performed three independent times in triplicates. Data are represented as mean ± standard error of the mean (*P<0.05; **P<0.01; ***P<0.001).
Downregulation of SPAG9 in ovarian cancer cells also inhibited the viability with enhanced cytotoxic effect of chemotherapeutic agent paclitaxel. Our data showed significant reduction in cellular viability in A10 (67%; P<0.001) and SKOV-3 (52.9%; P<0.001) cells with shRNA1 as compared to NC shRNA treatment (Figure 2(c)). Similar, results were obtained with shRNA2 for A10 (70.29%; P<0.001) and SKOV-3 (55.7%; P<0.001) cells as compare to NC shRNA treatment (Figure 2(c)). As expected, paclitaxel treatment resulted in significant reduction in cellular viability in A10 (64.43%; P<0.001) and SKOV-3 (52.53%; P<0.001) cells as compared to NC shRNA alone (Figure 2(c)). We also assessed the IC50 of the paclitaxel which was 1.4 nM in A-10 and 3.2 nM in SKOV-3 ovarian cancer cells respectively. In addition, when cells were treated in combination of paclitaxel with shRNA1, we observed significant reduction in cellular viability and enhanced cell death in A10 (80.6%; P<0.001) and SKOV-3 (65.27%; P<0.001) cells as compared to untreated cells (Figure 2(c)). Similarly, we observed significant reduction in cellular viability and enhanced cell death, in A10 (81.57%; P<0.001) and SKOV-3 (62.52%; P<0.001) cells when treated with paclitaxel and shRNA2 as compared to untreated cells (Figure 2(c)).
The rationale for using the paclitaxel in combination of SPAG9 shRNA was to assess if the effect of combination therapy on ovarian cancer cells is synergistic, additive or antagonistic. Combinatorial effect of treatment with the paclitaxel and SPAG9 shRNA induced increased levels of cell death, as compared with treatment with either paclitaxel or SPAG9 shRNA alone. Our observations suggest the synergistic growth inhibitory effect of SPAG9 shRNA and paclitaxel on the A10 and SKOV- 3 cells (Figure 2(b)). This was further confirmed by using the median-effect analysis method, as previously described. 11 Combination index (CI) values of interactions between SPAG9 shRNA and paclitaxel were assessed using CompuSyn 1.01 software (ComboSyn, Inc., Paramus, NJ, USA) and were found to be <1 indicating synergistic effect of combinatorial therapy in ovarian cancer cells.
Colony-forming ability of ovarian cancer cells was examined, which revealed a significant reduction in number of colonies formed in SPAG9 depleted cells as compared to NC shRNA treated cells. Importantly, the clonogenic potential of A10 cells was significantly reduced when transfected with shRNA1 (46.2%, 71.5% and 64.7%, for 400, 800 and 1200 cells per well respectively; P<0.001) and shRNA2 (48.7%, 71.5% and 70.7%, for 400, 800 and 1200 cells per well respectively;P<0.001) as compared to NC shRNA (Figure 2(d)). Similarly, in SKOV-3, a marked decrease in colony forming potential was observed when transfected with shRNA1 (41.6%, 66.1% and 61.4%, for 400, 800 and 1200 cells per well respectively; P<0.001) and shRNA2 (48.1%, 69.91 and 63.1%, for 400, 800 and 1200 cells per well respectively; P<0.001) as compared to NC shRNA alone (Figure 2(d)).
Role of SPAG9 in cell cycle arrest
Next, we investigated the role of SPAG9 in cell cycle regulation in ovarian cancer cells. Our data showed that knockdown of SPAG9 led to accumulation of A10 cells in S phase with SPAG9 shRNA1 (21.66%; P<0.0001) and SPAG9 shRNA2 (21.03%; P<0.0001) as compared to NC shRNA transfected cells (6.55%). Similarly, ablation of SPAG9 resulted in accumulation of SKOV-3 cells in S phase with SPAG9 shRNA1 (20.87%; P<0.001) and SPAG9 shRNA2 (20.16%; P<0.001) cells as compared to NC shRNA transfected cells (5.96%; Figure 2(e)). Western blot analysis showed a significant decrease in the expression of cyclins (cyclin B1, cyclin D1, cyclin E) and cyclin dependent kinases (CDK1, CDK2, CDK4 and CDK6) in both ovarian cancer cells (A10 and SKOV-3) transfected with SPAG9 shRNA (shRNA1 or shRNA2) as compared to NC shRNA treatment. The cyclin dependent kinase inhibitor protein p21 was also found to be upregulated in SPAG9 ablated A10 and SKOV-3 cells (Figure 3(a)). These data indicate that knockdown of SPAG9 in ovarian cancer cells results in cell cycle arrest and reduced cellular proliferation as a consequence of reduced expression of cyclins and cyclin-dependent kinases.

Ablation of SPAG9 alters expression of cell cycle molecules involved in cellular growth and proliferation in vitro and in vivo. (a) Western blot data reveals down regulation of cyclin B1, cyclin D1, cyclin E, CDK1, CDK2, CDK4 and CDK6 and up regulation of p21 in SPAG9 ablated A10 and SKOV-3 cells. β actin was used as endogenous loading control.(b) Representative images of IHC analysis show reduced immunoreactivity of SPAG9, PCNA, cyclin B1, cyclin D1, CDK 4, CDK 6 and increased immunoreactivity of p21 in SPAG9 shRNA2 treated tumor as compared to NC shRNA treated tumor sections. H&E staining shows cytostructure of the tumor cells. Experiments were performed three independent times in triplicates. Data are represented as mean ± standard error of the mean (*P<0.05; **P<0.01; ***P<0.001). (Original magnification ×200, objective ×20).
SPAG9 depletion reduced tumor growth in vivo mouse xenograft model
To validate in vitro effect of SPAG9 ablation on cellular proliferation and growth, in vivo A10 ovarian cancer xenograft mice model revealed significant reduction in tumor size and volume in SPAG9 shRNA2 treated tumor as compared to NC shRNA treated group (P<0.001; Supplementary Figure S1(b) and (c)). Further, xenograft tumors were dissected and were processed for SPAG9 expression. Immuno-histochemical analysis showed significant reduction (P<0.0001) in SPAG9 expression in SPAG9 shRNA2 treated group as compared to NC shRNA treated group (Figure 3(b); Supplementary Figure S1(d)). Further, SPAG9 shRNA2 treated mice revealed significant reduced level of proliferating cell nuclear antigen (PCNA) expression (P<0.001) as compared to NC shRNA treated group (Figure 3(b); Supplementary Figure S1(d)). We also observed decreased expression of cell cycle molecules cyclin A2, cyclin B1, cyclin D1, CDK2, CDK4, and CDK6) and increased immunoreactivity of p21 in SPAG9 shRNA2 treated tumors (Figure 3(b)), supporting in vitro observations of effect of ablation of SPAG9 on ovarian cellular growth.
Knockdown of SPAG9 induces apoptosis in ovarian cancer cells
To study the effect of ablation of SPAG9 on apoptosis, we examined phosphatidylserine translocation to the cell surface by annexin-V-Per-CP-Cy5-5-A assay. Flow cytometry analysis revealed marked increase in annexin V staining in A10 cells transfected with SPAG9 shRNA1 (45.6%) or shRNA2 (46.5%) as compared to NC shRNA treated cells (2.3%) (Figure 4(a) and (b)). Similarly, in SKOV-3 cells, annexin V staining was higher in cells transfected with SPAG9 shRNA1 (39.33%), or shRNA2 (39.65%) as compared to NC shRNA treated cells (1%; Figure 4(a) and (b)). In order to examine the onset of apoptosis, DNA fragmentation was assessed by TUNEL assay which showed marked increase in BrdU positive A10 cells transfected with shRNA1 (32%) and shRNA2 (55.18%) as compared to NC shRNA treated cells (0.83%; Figure 4(a) and (b)). Similarly, in SKOV-3 cells, BrdU positive cells were observed with SPAG9 shRNA1 (43.66%) or SPAG9 shRNA2 (53.36%) as compared to NC shRNA treated cells (2%; Figure 4(a) and (b)).

Knockdown of SPAG9 causes DNA damage and initiates apoptosis. (a) Flow cytometric analysis demonstrates the effect of SPAG9 ablation on onset of apoptosis in A10 and SKOV-3 cells by annexin V (phosphotidyl serine expression), TUNEL assay (DNA damage) and M30 assay (caspase activation). (b) Histogram depicts percentage of A10 and SKOV-3 cells with increased phosphotidyl serine expression, increased DNA damage and enhanced caspase activation in SPAG9 shRNA1 or shRNA2 transfected cells as compared to NCshRNA cells. Experiments were performed three independent times in triplicates. Data are represented as mean ± standard error of the mean (*P<0.05; **P<0.01; ***P<0.001).
Further, we investigated onset of apoptosis involving caspase cascade by examining caspase cleavage by M30 assay. We found higher population of M30 positive A10 cells transfected with SPAG9 shRNA1 (37%)or SPAG9 shRNA2(52%) as compared to NC shRNA treated cells (11%; Figure 4(a) and (b)). Similarly, higher population of M30 positive SKOV-3 cells transfected with SPAG9 shRNA1 (45.8%) or SPAG9 shRNA2 (51.2%) was found as compared to NC shRNA treated cells (1%; Figure 4(a) and (b)). Further, pro-apoptotic molecules such as BAD, BID and PUMA showed increased expression in SPAG9 ablated (SPAG9 shRNA1 or SPAG9 shRNA2) A10 and SKOV-3 cells as compare to NC shRNA transfected cells (Figure 5(a)). As expected anti-apoptotic molecules such as Bcl-xL, BCL2 and Mcl-1 were downregulated in SPAG9 ablated (SPAG9 shRNA1 or SPAG9 shRNA2) A10 and SKOV-3 cells (Figure 5(a)). Interestingly, higher expression of caspase 3, caspase 7, caspase 8 and cytochrome C was observed in SPAG9 ablated cells (Figure 5(a)). To validate our in vitro findings, ovarian cancer xenograft treated with SPAG9 shRNA2 were dissected and were probed for molecules involved in apoptosis pathway. IHC analysis revealed increased expression of pro-apoptotic molecules (BID and BAX) and reduced expression of anti-apoptotic molecules (Bcl-xL, BCL2 and Mcl-1), along with increased expression of caspases (caspase 3 and caspase 8) and cytochrome C in SPAG9 shRNA2 treated xenograft as compared to NC shRNA treatment (Figure 5(b)).

Ablation of SPAG9 protein alters molecules involved in apoptosis process. (a) Western blot shows down regulation of anti-apoptotic molecule Bcl-xL, BCL2 & Mcl-1 and up regulation of pro-apoptotic molecules BAD, BID, PUMA and caspases (caspase 3, caspase 7 and caspase 8) and cytochrome-C in cells treated with SPAG9 shRNA (shRNA1 or shRNA2) as compared to to NC shRNA treated A10 and SKOV-3 cells. β actin was used as endogenous loading control. (b) Representative IHC images show changes in molecules involved in apoptosis pathway in mice xenograft. Increased immunoreactivity of pro-apoptotic molecules (BID and BAX) and reduced expression of anti- apoptotic molecules (Bcl-xL, BCL2 and Mcl-1), along with increased expression of caspases (caspase 3 and caspase 8) and cytochrome C was observed in SPAG9 shRNA2 treated xenograft as compared to NC shRNA treated mice. Experiments were performed three independent times in triplicates. Data are represented as mean ± standard error of the mean (*P<0.05; **P<0.01; ***P<0.001). (Original magnification ×400, objective ×40).
Knockdown of SPAG9 inhibited migration and invasion abilities
We further examined the effect of ablation of SPAG9 on migratory and invasive properties of A10 and SKOV-3 cells by transwell membrane assays. Knockdown of SPAG9 lead to significant reduction in migration ability of A10 cells treated with SPAG9 shRNA1 (47.6%; P<0.001) or SPAG9 shRNA2 (43.5%; P<0.001) as compared to NC shRNA transfected cells (Figure 6(a)). Invasive abilities of A10 cells were also significantly reduced when transfected with SPAG9 shRNA1 (47.3%; P<0.001) or SPAG9 shRNA2 (52.1%; P<0.001) as compared to NC shRNA transfected cells (Figure 6(b)). Similarly, SKOV-3 cells also showed significant reduction in migration ability with SPAG9 shRNA1 (41.47%; P<0.001) or shRNA2 (46.82%; P<0.001) transfection as compared to NC shRNA transfected cells (Figure 6(a)). In addition, invasion ability was also significantly reduced in SKOV-3 cells transfected with SPAG9 shRNA1 (54.13%; P<0.001) or shRNA2 (58.86%; P<0.001) as compared to NC shRNA transfected cells (Figure 6(b)).

Knockdown of SPAG9 inhibits cellular motility in A10 and SKOV-3 ovarian cancer cells. (a) Phase contrast images show significant difference in migratory abilities through insert membrane in SPAG9 shRNA1 or SPAG9 shRNA2 transfected A10 and SKOV-3 cells as compared to NC shRNA cells. Histogram depicts significant reduction in number of migratory A10 and SKOV-3 cells. (b) Phase contrast images show significant difference in invasive potential through insert membrane and matrigel in SPAG9 shRNA1 or SPAG9 shRNA2 transfected A10 and SKOV-3 cells as compared to NC shRNA cells. Histogram depicts significant reduction in number of invaded A10 and SKOV-3 cells. (c) Western blot analysis shows enhanced expression of E-cadherin and decreased expression of N-cadherin, P-cadherin, SLUG, TWIST and Vimentin in SPAG9 ablated A10 and SKOV-3 cells. β actin was used as endogenous loading control. (d) Representative images of mice tumor sections show decreased N-cadherin, SLUG, TWIST and Vimentin expression and increased E-cadherin expression in SPAG9 shRNA2 treated tumors as compared to NC shRNA treatment. Experiments were performed three independent times in triplicates. Data are represented as mean ± standard error of the mean (*P<0.05; **P<0.01; ***P<0.001). (Original magnification ×400, objective ×40).
Subsequently, we examined EMT markers in SPAG9 ablated cells by Western blotting. Down regulation of N-cadherin, P-cadherin, SLUG, TWIST and Vimentin was observed (Figure 6(c)), whereas, increased expression of E-cadherin was found. Similar results were observed by IHC analysis in SPAG9 shRNA treated tumor (Figure 6(d)). In addition, wound healing assay also indicated reduced cellular motility in SPAG9 ablated cells as compared to NC shRNA transfected cells (Supplementary Figure S1(e)). Thus our results show that SPAG9 plays an important role in cellular migration and invasion potential of ovarian cancer cells.
Discussion
Sperm associated antigen 9 (SPAG9) belongs to a unique family of cancer testis (CT) antigens and has been reported in variety of cancers. 3 Earlier, we reported SPAG9 expression at mRNA and protein level in ovarian cancer patient’s tissue specimen. In addition, we also showed humoral response against SPAG9 in various histotypes of ovarian cancer patients. 9 Recently, SPAG9 has also been shown to generate strong humoral response in hepatocellular carcinoma and lung cancer patients, 13 indicating its therapeutic potential as a tumor-specific biomarker for cancer management. In present investigation, our data show that ablation of SPAG9 in ovarian cancer cells resulted in increased DNA damage, cell cycle arrest, apoptosis, reduction in cellular migration and invasion ability in in vitro and reduced ovarian tumor growth in human xenograft mouse model.
Cancers have the ability to develop resistance to various chemotherapeutic regimes, which is a complex phenomenon and necessitates further research for better treatment options. Among the gynaecological cancers, ovarian cancer has high mortality rate due to late diagnosis in advanced clinical stages, spread within the peritoneal cavity and resistance to chemotherapy. 14 Ovarian cancer has been reported to be chemo-sensitive during early stages of the disease, however, becomes chemo-resistant in advanced stages. 15 Therefore, there is an urgent need to develop novel and effective therapeutic options for ovarian cancer management. In the present study, we demonstrated that combinatorial effect of paclitaxel and SPAG9 shRNA resulted in enhanced reduction in cell viability and cellular proliferation in ovarian cancer cells. Our median-effect analysis 11 using CompuSyn 1.01 software suggests the synergistic growth inhibitory effect of combinatorial effect of paclitaxel and SPAG9 shRNA on the A10 and SKOV-3 cells. Interestingly, our study is in corroboration with an earlier study wherein knockdown of CDK11 enhanced the cytotoxic effect of paclitaxel in ovarian cancer cells. 16 Therefore, our data suggests that ablation of SPAG9 results in enhanced efficacy of paclitaxel cytotoxicity and causes cell death in ovarian cancer cells. The underlying molecular mechanisms for increased efficacy of paclitaxel cytotoxicity in SPAG9 ablated ovarian cell warrants further studies.
One of the important features of cancer progression is alteration in cell cycle pathway leading to aberrant cellular proliferation. 17 In present study, ablation of SPAG9 severely affected the cell cycle progression and resulted in cell growth arrest in S phase. However, in contrast, our previous studies in triple negative breast cancer cells revealed ablation of SPAG9 resulted in cell growth arrest in G0/G1 phase. 10 The fact that SPAG9 ablation resulted in cell cycle arrest in different stages in various cancer cells may be due to highly heterogeneous nature of cancer cells and hence alter and modulate various key molecules in different molecular pathways. 18 Our in vivo ovarian cancer xenograft immunohistochemical analysis further demonstrated decreased reactivity of cell cycle molecules in SPAG9 ablated ovarian tumor. These findings are consistent with our previous studies where knockdown of SPAG9 in breast cancer cells resulted in alteration of key molecules involved in cell cycle regulation which was confirmed by immunohistochemical analysis in mice xenograft tumor. 10 These findings suggest that SPAG9 plays an important role in cell cycle regulating molecules. Ablation of SPAG9 resulted in inhibiting ovarian cancer cell proliferation and thus may act as a potential therapeutic target in ovarian cancer treatment.
Defects in programmed cell death lead to altered expression of various key molecules due to which majority of the cancer types result in disease progression with increased tumor burden and hence mortality. 19 Knockdown of SPAG9 in ovarian cancer cells affected cellular viability and resulted in enhanced rate of cell death and reduced colony forming ability. Present investigation findings were similar to our previous study where HSP70-2 knockdown resulted in reduced cellular proliferation and colony forming ability in ovarian cancer cells 20 as well as in breast cancer cells. 10 Further in support of our data, yet another study has shown that CT antigen DUSP21 knockdown resulted in reduced colony forming ability in hepatocellular carcinoma cells. 21 Furthermore, SPAG9 ablation lead to caspase mediated DNA damage and cell death. This is in line with our recent study on ablation of AKAP4, revealing increased DNA damage, cell cycle arrest and caspase mediated cell death in ovarian cancer cells. 22 This was further strengthened at molecular level showing increased pro-apoptotic proteins and decreased pro-survival proteins post SPAG9 ablation in ovarian cancer cells. In vivo studies also showed tumor growth reduction due to ablation of SPAG9. These findings were supported by our earlier studies on SPAG9 ablation in breast cancer cells in in vitro as well as in vivo studies. 10 Collectively, our present findings indicated that SPAG9 promotes cellular growth and depletion of SPAG9 causes apoptosis and hence cancer cell death. Thus, SPAG9 could be used as a potential therapeutic target in ovarian cancer.
Metastasis of ovarian cancer cells is the major challenge and is associated with substantial morbidity and mortality. 23 CT antigens are thought to be involved in regulating various aspects of oncogenic process including EMT. 24 Recently, a study has shown that over expression of AKAP4 in oesophageal cancer cells increased metastatic potential of the cancer cells in vitro. 25 Interestingly, our previous study showed that SPAG9 ablation in highly invasive breast cancer cells (MDA-MB231) inhibited the migration and invasion potential of cancer cells. 10 Our present study is in agreement with these findings wherein we found reduced migration and invasion potential of SPAG9 depleted ovarian cancer cells along with inhibited wound healing ability. Interestingly, to further support our findings a recent study has also shown that SPAG9 expression was positively associated with tumor-node-metastasis in hepatocellular carcinoma patients. 26 These findings highlighted the fact that SPAG9 may be involved in regulation of metastasis in ovarian cancer cells.
In conclusion, we showed that SPAG9 expression is involved in regulation of cell cycle, DNA damage, apoptosis, drug sensitivity and EMT. Further, SPAG9 was also shown to be essential for ovarian cancer cell growth and survival in in vivo nude mice model. Our findings indicate that SPAG9 may be a potential therapeutic target in treatment of ovarian cancer.
Supplemental Material
Figure_S1_Supplementary_tub – Supplemental material for Sperm associated antigen 9 (SPAG9) a promising therapeutic target of ovarian carcinoma
Supplemental material, Figure_S1_Supplementary_tub for Sperm associated antigen 9 (SPAG9) a promising therapeutic target of ovarian carcinoma by Nirmala Jagadish, Rukhsar Fatima, Aditi Sharma, Sonika Devi, Vitusha Suri, Vikash Kumar and Anil Suri in Tumor Biology
Supplemental Material
Supplementary_data_TUB – Supplemental material for Sperm associated antigen 9 (SPAG9) a promising therapeutic target of ovarian carcinoma
Supplemental material, Supplementary_data_TUB for Sperm associated antigen 9 (SPAG9) a promising therapeutic target of ovarian carcinoma by Nirmala Jagadish, Rukhsar Fatima, Aditi Sharma, Sonika Devi, Vitusha Suri, Vikash Kumar and Anil Suri in Tumor Biology
Footnotes
Acknowledgements
Nirmala Jagadish, Rukhsar Fatima, Aditi Sharma, Sonika Devi, Vitusha Suri and Vikash Kumar carried out all the experiments, prepared figures and drafted the manuscript. NJ participated in data analysis and interpretation of results. AS designed the study, participated in data analysis and interpretation of results. All authors read and approved the manuscript.
Availability of data and material
Information is included in the material and method section.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by grants from Indo-UK Cancer Research Program (Grant No. BT/IN/UK/NII/2006), Centre for Molecular Medicine (Grant No.BT/PR/14549/MED/14/1291), NII-core funding, Department of Biotechnology, Government of India. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Ethical approval
We have taken the IAEC approval from the IAEC committees for animal studies. This information is included in material and methods section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
