Abstract
Icarisid II, one of the main active components of Herba Epimedii extracts, shows potent antitumor activity in various cancer cell lines, including osteosarcoma cells. However, the anticancer mechanism of icarisid II against osteosarcoma U2OS needs further exploration. This study aims to investigate further antitumor effects of icarisid II on human osteosarcoma cells and elucidate the underlying mechanism. We cultivated human osteosarcoma USO2 cells in vitro using different concentrations of icarisid II (0–30 µM). Cell viability was detected at 24, 48, and 72 h using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide analysis. Cell cycle was tested by flow cytometry after treatment with icarisid II for 48 h. Annexin V–allophycocyanin and 7-aminoactinomycin D staining were conducted to detect cell apoptosis. Quantitative real-time polymerase chain reaction and Western blot assay were performed to measure the levels of genes and proteins related to cell cycle and apoptosis. Results showed that icarisid II significantly inhibited the proliferation and induced apoptosis of human osteosarcoma U2OS cells. The half maximal inhibitory concentration values were 14.44, 11.02, and 7.37 µM at 24, 48, and 72 h, respectively. Cell cycle was arrested in the G2/M phase in vitro. In addition, icarisid II upregulated the expression levels of P21 and CyclinB1 whereas downregulated the expression levels of CyclinD1, CDC2, and P-Cdc25C, which were related to cell cycle arrest in U2OS cells. The cell apoptotic rate increased in a dose-dependent manner after treatment with icarisid II for 48 h. Icarisid II induced apoptosis by upregulating Bax, downregulating Bcl-2, and activating apoptosis-related proteins, including cleaved caspase-3, caspase-7, caspase-9, and poly (ADP-ribose) polymerase. These data indicate that icarisid II exhibits an antiproliferation effect on human osteosarcoma cells and induces apoptosis by activating the caspase family in a time- and dose-dependent manner in vitro. Therefore, icarisid II may be used as a candidate agent for the clinical treatment of osteosarcoma in the future.
Introduction
Osteosarcoma is a common primary bone tumor in children and adolescents. 1 The main treatment methods for osteosarcoma include radical resection and chemoradiotherapy. 2 However, osteosarcoma cells can easily spread through the blood and shows varied clinical behavior in early stage; patients with lung or renal metastases eventually lose response to combined chemotherapy and radiotherapy. 3 In addition, severe toxicities often lead to treatment suspension during chemotherapy. Therefore, new strategies to improve the treatments of advanced tumors in osteosarcoma patients should be explored. 4
In recent years, natural products and medicinal plants have been widely used as anticancer drugs.5,6 Emodin is a natural plant derivative that can induce the apoptosis of human osteosarcoma cells. 7 Icarisid II (IS), a flavonol glycoside with a molecular formula of C27H30O10 was derived from the metabolite of icariin (ICA). ICA is a major component of the traditional Chinese medicinal plant, Epimedium koreanum Nakai, a leguminous plant grown in China, northern part of North Korea, and Japan. 8 Given its wide-range pharmacological effects, Nakai has been used in the treatment of osteoporosis, erectile dysfunction, premature ejaculation, urinary incontinence, joint pain, and irregular menstruation. 9 ICA exhibits many biological activities, such as cardiovascular function enhancement, hormone regulation, immunological function modulation, antiaphrodisiac, antirheumatic, sexual enhancement, immunity improvement, and antiaging. At present, the applications of ICA in disease treatment have been successively reported.10–12 ICA suppresses cell proliferation in human ovarian cancer cells through regulating the anticancer genes by microRNA 21 (miRNA-21). 13 Glioblastoma is also treated with ICA by inhibiting nuclear factor kappa B (NF-κB) activity, 14 and this treatment shows no obvious toxicity or side effects.
IS, the predominant bioactive form of ICA, possesses strong biological activities against inflammation, osteoporosis, and hypoxia and improves erectile dysfunction. 15 Modern pharmacological studies have demonstrated that IS potentially suppresses tumor growth by inducing the apoptosis and inhibiting the proliferation of different cancer cells, including acute myeloid leukemia, prostate cancer, osteosarcoma, multiple myeloma, and melanoma. Recent studies on the functions of IS for cancers treatment 16 have reported that IS can induce apoptosis in human hepatoblastoma HepG2 cells by mediating the functions of the mitochondrion and lysosome. 17 Studies on the relationship between IS and osteosarcoma cells found that IS can inhibit the proliferation of human osteosarcoma MG-63 and Saos-2 cells by downregulating the epidermal growth factor receptor (EGFR) downstream signaling PI3K/Akt, Raf/MEK (mitogen-activated protein kinase kinase)/ERK (extracellular signal–regulated kinase) and mechanistic target of rapamycin (mTOR) pathways.18,19 The PI3K/Akt pathway is also involved in the ICA-induced cytotoxicity enhancement of doxorubicin in osteosarcoma. 20 However, the mechanisms underlying the anticancer effect of IS warrant further exploration.
In this study, we investigated the cytotoxic effects of IS on human osteosarcoma U2OS cells. Results showed that IS induced the apoptosis of osteosarcoma cells by collapsing the mitochondrial membrane potential, reducing Bcl-2, upregulating Bax, and activating caspase-3, caspase-7, caspase-9, and poly (ADP-ribose) polymerase (PARP). In addition, IS significantly inhibited the proliferation of U2OS cells by arresting the cells in the G2/M phase.
Materials and methods
Cell culture and reagents
Human osteosarcoma U2OS cells were purchased from the Cell Bank of Shanghai Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences (Shanghai, PR China). They were cultured in RPMI-1640 medium (Gibco, Life Technologies, USA) supplemented with 10% heat-inactivated fetal bovine serum (Sijiqing, Hangzhou, PR China) and 1% penicillin/streptomycin. The cells were cultured at 37°C in a humidified atmosphere of 5% CO2, and conventional digestion was carried out when 80% confluence was reached. The medium was usually changed three times a week. IS was purchased from Sigma (St. Louis, MO, USA).
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
Logarithmic phase cells of U2OS (5–10 × 103/mL) were grown on 96-well plates and treated with various concentrations of IS (0, 1.875, 3.125, 3.75, 6.25, 7.5, 12.5, 15, 25, 30, and 50 µM) for 24, 48, or 72 h at 37°C in a humidified atmosphere of 5% CO2. Each concentration gradient was performed in quintuplicate and repeated three times. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; 20 µL, 5 mg/mL) was added to each well. After incubation for 48 h, 150 µL of dimethyl sulfoxide (DMSO) was added to each well. The absorbance was obtained at 570 nm using a microplate reader (Spectramax Plus 384; Molecular Devices, Sunnyvale, CA, USA). The cytotoxic activity was expressed as the half maximal inhibitory concentration (IC50) value.
Cell cycle assay
U2OS cells were incubated with various concentrations of IS (0, 5, 10, and 20 µM) for 48 h, washed three times with phosphate-buffered saline (PBS), centrifuged at 1000g for 5 min, and then fixed with 75% ethanol on ice. The cells were incubated again for 30 min at 37°C in a staining buffer containing RNase A and propidium iodide (Beyotime, Nantong, PR China). DNA content analysis was carried out using a BD Accuri C6 flow cytometer (Becton & Dickinson Co., Miami, FL, USA). The experiment was repeated three times.
Cell apoptosis detection
U2OS cells were treated with various concentrations of IS (0, 10, 15, 20, 25, and 30 µM) for 48 h, washed three times with PBS, and then centrifuged at 1000g for 5 min. After removing the supernatant, 500 µL of 1× Binding Buffer was added to the cells. Annexin V–allophycocyanin (APC; 5 µL) and 7-aminoactinomycin D (7-AAD; 5 µL) were added to the cells and then incubated for 15 min in the dark. The cell apoptotic rate was determined by flow cytometry (Becton & Dickinson Co.) within an hour.
Quantitative real-time polymerase chain reaction
Total RNA was isolated from U2OS cells using an EASYspin Plus tissue or cell RNA Extraction Kit (Aidlab Biotechnologies Co. Ltd, China). RNA was quantified by measuring absorption at 260 nm, and 1 µg RNA was reverse transcribed to complementary DNA (cDNA). The thermal cycling conditions included the following: an initial denaturation at 95°C for 5 min; 40 cycles of denaturation (10 s at 95°C), annealing (15 s at 60°C), and extension (15 s at 72°C with a single fluorescence measurement); a melting curve program (60–95°C with a 0.11°C/s heat increase and continuous fluorescence measurement); and a cooling step at 40°C. The data were normalized to the expression of β-actin. The results are expressed as fold changes. The real-time polymerase chain reaction (RT-PCR) primers used in this study are listed in Table 1.
Primers and annealing temperatures used for qRT-PCR.

qRT-PCR: quantitative real-time polymerase chain reaction.
Western blot assay
Total proteins were extracted from U2OS cells. Equal amounts of protein (40 µg) were added to 8%–12% sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis and then transferred directly onto polyvinylidene difluoride membranes (Bio-Rad Laboratories, Hercules, CA, USA). The membranes were incubated in 5% skimmed milk for 2 h; blotted with primary antibodies specific for Bax, Bcl-2, cleaved caspase-3, cleaved caspase-7, cleaved caspase-9, cleaved PARP, CDC2, Cdc25C, P-Cdc2, P-Cdc25C, and CyclinB1 overnight at 4°C; and then probed with secondary isotype-specific antibodies tagged with horseradish peroxidase (Cell Signaling Technology, USA). The bound immuno-complexes were detected using a ChemiDOC™ XRS+ system (Bio-Rad Laboratories).
Statistical analysis
The results were given as the mean ± standard error of the mean (SEM). One-way analysis of variance (ANOVA) was used to compare three or more groups. The Student’s t test was used to compare variables between two groups. Statistical analysis was performed using GraphPad Prism (Version 5.0; GraphPad Software, San Diego, CA, USA). Statistical tests were two-tailed, and p < 0.05 was regarded as statistically significant.
Results
IS inhibited the proliferation of U2OS cells
Human osteosarcoma U2OS cells that were treated with different concentrations of IS (1.875, 3.125, 3.75, 6.25, 7.5, 12.5, 25, 30, and 50 µM) for 24, 48, or 72 h were measured by MTT assay. The IC50 values of U2OS cells were 14.44 µM at 24 h, 11.02 µM at 48 h, and 7.37 µM at 72 h, respectively. Cell viability decreased significantly in a time- and dose-dependent manner (Figure 1).

Effects of IS on cell viability. The viability of human osteosarcoma U2OS cells decreased in a time- and dose-dependent manner. Each point was calculated in comparison with the control group. The experiments were carried out three times, and the values were presented as mean ± SEM.
IS triggered cell cycle arrest in U2OS cells
The cell cycle of U2OS cells exposed to different concentrations of IS (0–20 µM) was analyzed by flow cytometry to determine how IS inhibits cell proliferation in human osteosarcoma. Treatment with IS at 10 and 20 µM significantly decreased the number of cells in G1 phase and increased the number of cells arrested in the G2/M phase (as shown in Figure 2). These results suggest that IS can inhibit cell growth by blocking the division of osteosarcoma cells.

IS triggered cell cycle arrest in U2OS cells. (a) U2OS cells were treated with various concentrations of IS (0, 5, 10, and 20 µM) for 48 h. The peaks in the figure represent the G0/G1, S, and G2/M phases in the cell cycle. (b) Histogram showing the percentages of cell numbers in the cell cycle. The experiments were measured with a flow cytometer for three times. Data are presented as mean ± SEM (*p < 0.05; **p < 0.01; ***p < 0.001 in comparison with the control group).
IS changed the levels of antiproliferation genes and proteins in U2OS cells
The genes (CyclinD1 and P21) that regulated cell cycle were detected by quantitative RT-PCR (qRT-PCR). The results were shown in Figure 3. Treatment with IS at 24 µM significantly downregulated the messenger RNA (mRNA) expression of CyclinD1 (p < 0.01). By contrast, the expression of the P21 gene was upregulated with increasing IS concentrations. Significant differences in the mRNA expression of P21 were noted after treatment with 9, 12, 16, and 24 µM IS. Meanwhile, the proteins related to cell cycle were also measured by Western blot analysis (Figure 4). IS upregulated the expression level of CyclinB1 whereas downregulated the expression levels of CDC2 and P-Cdc25C, which were related to cell cycle arrest in U2OS cells. Significant differences in the protein expression of CyclinB1 were observed between the groups treated with 10, 20, or 30 µM IS and the control group. The exact results were also found in the expression of protein P-Cdc25C in comparison with the control group. A significant difference in the protein expression of CDC2 was observed between the groups treated with 20 and 30 µM IS. However, the expression of P-Cdc2 did not significantly differ when compared with the control group.
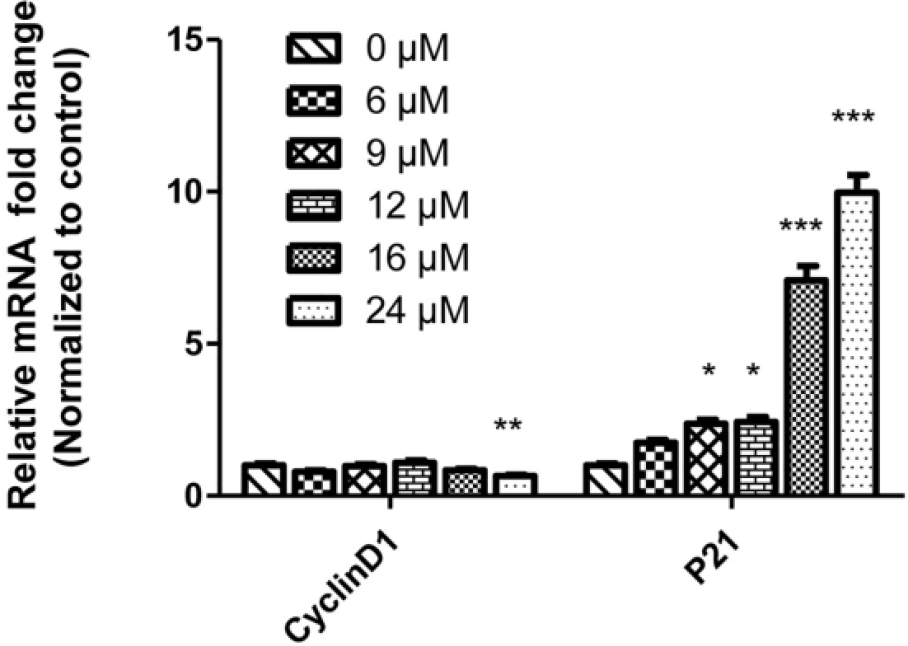
IS regulated mRNA expression levels of CyclinD1 and P21 in U2OS cells. After treatment with various concentrations of IS (0, 6, 9, 12, 16, and 24 µM) for 48 h, the mRNA expression levels of CyclinD1 and P21 mRNA were determined by real-time PCR. The relative expression level of mRNA was normalized to the control group (IS = 0 µM). The experiments were measured by real-time polymerase chain reaction for three times. Data are presented as mean ± SEM (*p < 0.05; **p < 0.01; ***p < 0.001 in comparison with the control group).
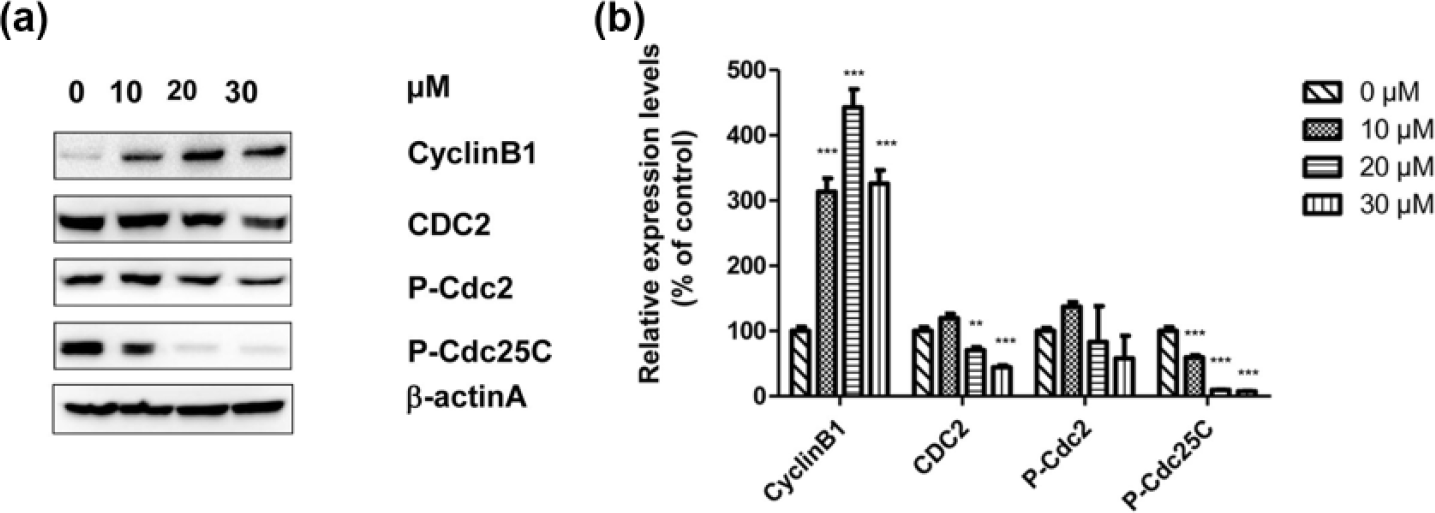
IS regulated the expression of proteins related to cell cycle in U2OS cells. (a) U2OS cells were treated with different concentrations of IS (0, 10, 20, and 30 µM) for 48 h, and then the expression levels of related proteins (CyclinB1, CDC2, P-Cdc2, and P-Cdc25C) in the cell cycle were measured by Western blot analysis, with β-actin serving as the loading control. (b) Histogram showing the relative expression levels of proteins after normalizing to the levels of β-actin. Representative data from three independent experiments are shown. Data are presented as mean ± SEM (*p < 0.05; **p < 0.01; ***p < 0.001 in comparison with the control group).
IS induced the apoptosis of U2OS cells
Human osteosarcoma U2OS cells were treated with IS (0, 10, 15, 20, 25, and 30 µM) for 48 h. The rates of cell apoptosis were assayed in Figure 5. The apoptosis rate increased in a dose-dependent manner. Treatment with 15, 20, and 25 µM IS induced obvious apoptosis compared with the control group. Moreover, when the concentration of IS was 30 µM, the rate of cell apoptosis reached 97%. The main apoptotic pathway proteins caspase and Bcl-2 family were detected by qRT-PCR and Western blot analysis. The results were shown in Figures 3 and 6. The mRNA expression of Bax was upregulated with increasing concentrations of IS, and a significant difference was observed when the cells were treated with 6 µM IS compared with the control group. The mRNA expression of Bcl-2 was downregulated after treatment with IS at 24 µM (Figure 6).

IS induced apoptosis of U2OS cells. (a) U2OS cells were treated with different concentrations of IS (0, 10, 15, 20, 25, and 30 µM) for 48 h. Annexin V–APC and 7-AAD staining were conducted to detect cell apoptosis. (b) Histogram showing the percentages of apoptotic cells at different concentrations of IS. Experiments were carried out for three times. Data are presented as mean ± SEM (*p < 0.05; **p < 0.01; ***p < 0.001 in comparison with the control group).

IS regulated the mRNA expression levels of Bcl-2 and Bax in U2OS cells. After treatment with various concentrations of IS (0, 6, 9, 12, 16, and 24 µM) for 48 h, the mRNA expression levels of Bcl-2 and Bax were determined by real-time PCR. The relative expression level of mRNA was normalized to the control. The experiments were measured by real-time polymerase chain reaction for three times. Data are presented as mean ± SEM (*p < 0.05; **p < 0.01; ***p < 0.001 in comparison with the control group).
The proteins related to cell apoptosis were also measured by Western blot analysis (Figure 7). The expression levels of apoptosis-related proteins, including cleaved caspase-3, cleaved caspase-7, cleaved caspase-9, cleaved PARP, and Bax, were higher when the cells were exposed to 20 or 30 µM IS compared with the control. In addition, IS decreased the protein expression of Bcl-2, and a statistic difference was observed when the cells were exposed to 30 µM IS.

IS changed the expression levels of proteins related to apoptosis in U2OS cells. (a) U2OS cells were treated with different concentrations of IS (0, 10, 20, and 30 µM) for 48 h, and then the expression levels of related proteins (cleaved casase-3, cleaved casase-7, cleaved casase-9, cleaved PARP, Bax, and Bcl-2) in cell apoptosis were measured by Western blot, with β-actin serving as the loading control. (b) Histogram showing the relative expression levels of proteins after normalizing to the levels of β-actin. Representative data from three independent experiments are shown. Data are presented as mean ± SEM (*p < 0.05; **p < 0.01; ***p < 0.001 in comparison with the control group).
Discussion
Osteosarcoma seriously affects the healthy growth of young people worldwide. Therefore, novel strategies for osteosarcoma treatment should be explored. In this study, we cultivated USO2 cells in vitro with IS to investigate the anticancer mechanism of IS in human osteosarcoma cells. Our results showed that IS exhibited the antiproliferation effect in U2OS cells by regulating CyclinD1 and P21 and induced apoptosis by activating the caspase family in a time- and dose-dependent manner.
Cell cycle is a process that starts at the end of cell division and terminates at the start of next cell division. It consists of four stages, namely, gap 1 (G1), synthesis phase (S), gap 2 (G2), and mitosis (M). DNA replication is in the S phase and the cell prepares itself for cell division in G2 phase. The M phase is a progress of cell division wherein one cell divides into two and the genetic material is distributed equally to the daughter cells. Cell division has specific checkpoints (G1/S and G2/M) that monitor the entire progress. 21 Disorders in the cell cycle often lead to the occurrence or development of tumor. 22 In this study, we found that cell cycle was captured in the G2/M stage after treating with increasing concentrations of IS. The same mechanism was also found in other antitumor agents. Zhang et al. 23 found that ICA treatment causes G2/M arrest and apoptosis in RAW 264.7 osteoclastic cells. MDA-MB-231 breast cancer cells are arrested at the G2/M checkpoint by the tumor suppressor gene RPRM. 24 Apoptosis of glioma cells is induced by TJY-16 through G2/M cell cycle arrest. 25 In addition, IS induces cell cycle arrest at the G0/G1 and G2/M transitions in human melanoma cells in a dose-dependent manner. 26 In this study, treatment with IS at 10 and 20 µM significantly decreased the number of cells in the G0/G1 phase. At present, which cell cycle phase in cancer cells is arrested by antitumor agents is unclear. Further validation needs to be carried out in the future.
CyclinD1, which plays as the main regulatory factor in G1/S, is an end-point biomarker of therapeutic outcome for different treatment modalities in cancer. 27 The inhibitor p21 negative regulates the activity of specific cyclin-cyclin-dependent kinase (CDK) complex, which make function on G1/S transition. 28 The complex of CylinB1 and CDK1 serves a vital function at the checkpoint G2/M. The upstream gene Cdc25C regulates the expression of CylinB1/CDK1. When the phosphatase activity is inhibited in Cdc25C, the CyclinB1–Cdc2 complex becomes dephosphorylated, which prevents G2/M phase transition, thereby terminating the cell cycle. 29 In this study, IS regulated the expression of related genes or proteins in the cell cycle at G1/S and G2/M stages. Treatment of human osteosarcoma U2OS cells with IS upregulated P21 and downregulated CyclinD1 at the G1/S stage. In addition, IS upregulated the expression levels of G2/M cell cycle–related proteins of CyclinB1 and decreased the expression levels of CDC2 and P-Cdc25C. Similar results were also found in other cancers. In gastric cancer, harmaline inhibits the proliferation of tumor cells by upregulating the cell cycle–related proteins of P-Cdc2 and CyclinB and downregulating P-Cdc25C. 30 However, Huang et al. 31 observed that the cell cycle G2/M arrest in colorectal cancer cells can downregulate the expression levels of Cdc2/CyclinB1. Wu et al. 26 found that IS can inhibit the proliferation and induce the cell cycle arrest at the G0/G1 and G2/M transitions of A375 human melanoma cells, consequently inhibiting the expression of related protein of CyclinB1. The different effects of IS may be attributed to the different cell types or properties of antitumor agents.
In general, apoptosis refers to the programmed cell death that some factors make functions on the intracellular genes and related proteins. The mitochondrial pathway consists of the main signal in cell apoptosis.32–34 In the start-up phase, the mitochondria release cytochrome c, which activates the caspase family and then starts cascades in the cytoplasm. 35 To date, at least 15 members of the caspase family have been identified. Caspase-3 initiates cell death, 36 and caspase-9 combined with caspase-6 and -8 may initiate a proteolytic cascade by activating pro-caspases to amplify a death signal. 37 During apoptosis, many regulatory molecules are involved in the cascade in the cytoplasm. Members of the Bcl-2 family are the most common regulatory factors in cell apoptosis; these members include pro-apoptotic proteins (Bad, Bax, and Bid) and antiapoptotic proteins (Bcl-2, BCL-xl, and Mcl-1). 38 Bcl-2 is the well-known antiapoptotic factor that is closely related to tumorigenesis. Moreover, Bax distributed on the outer mitochondrial membrane is expressed more widely than Bcl-2 in tissues and organs. Bcl-2 and Bax are mutual antagonists that play important roles in the regulation of tumor cell apoptosis. In this study, we used Annexin V–APC and 7-AAD staining to detect cell apoptosis and observed that cell apoptosis rate increased with increasing IS concentration. In addition, IS induced apoptosis by upregulating Bax, downregulating Bcl-2, and activating apoptosis-related proteins, including cleaved caspase-3, caspase-7, caspase-9, and PARP. These findings are consistent with the reports that IS induces apoptosis in other tumor cells. Wu et al. 39 found that the IS treatment of melanoma cells increases the numbers of apoptotic cells to 26.3% and upregulates the expression levels of cleaved caspase-3. IS also upregulates the protein expression levels of cleaved caspase-9 and PARP in hepatoblastoma cells. 40 These results provided additional insights for exploring the antiproliferation mechanism of IS in osteosarcoma.
In summary, IS inhibited the proliferation of human U2OS osteosarcoma cells by regulating CyclinD1 and P21, and induced apoptosis by activating the caspase family in a time- and dose-dependent manner. These results not only provide a potential new drug for osteosarcoma treatment but also serve as a solid theoretical basis for the development of the disease.
Footnotes
Acknowledgements
Y.T. and M.X. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from the Guangxi Zhuang Autonomous Region Department of Education project to enhance the capacity of young teachers (No. KY2016YB097) and Guangxi Medical Youth Science Foundation (No. GXMUYSF201510).
