Abstract
Radioresistance often leads to poor survival in concurrent chemoradiotherapy–treated cervical squamous cell carcinoma, and reliable biomarkers can improve prognosis. We compared the prognostic potential of hemoglobin, absolute neutrophil count, and absolute lymphocyte count with that of squamous cell carcinoma antigen in concurrent chemoradiotherapy–treated squamous cell carcinoma. We analyzed 152 patients with concurrent chemoradiotherapy and high-dose-rate intracavitary brachytherapy–treated cervical squamous cell carcinoma. Hemoglobin, absolute neutrophil count, absolute lymphocyte count, and squamous cell carcinoma antigen were quantitated and correlated with survival, using Cox regression, receiver operating characteristic curve analysis, and Kaplan–Meier plots. Both hemoglobin and absolute lymphocyte count in the second week of concurrent chemoradiotherapy (Hb2 and ALC2) and squamous cell carcinoma antigen in the third week of concurrent chemoradiotherapy (mid-squamous cell carcinoma antigen) correlated significantly with disease-specific survival and progression-free survival. The ratio of high-dose-rate intracavitary brachytherapy dose to total dose (high-dose-rate intracavitary brachytherapy ratio) correlated significantly with progression-free survival. Patients with both low Hb2 (≤11 g/dL) and ALC2 (≤639 cells/µL) showed a lower 5-year disease-specific survival rate than those with high Hb2 and/or ALC2, regardless of mid-squamous cell carcinoma antigen (mid-squamous cell carcinoma antigen: ≤4.7 ng/mL; 5-year disease-specific survival rate: 85.5% vs 94.6%, p = 0.0096, and mid-squamous cell carcinoma antigen: >4.7 ng/mL; 5-year disease-specific survival rate: 43.8% vs 66.7%, p = 0.192). When both Hb2 and ALC2 were low, the low high-dose-rate intracavitary brachytherapy ratio (≤0.43) subgroup displayed significantly lower 5-year disease-specific survival rate compared to the subgroup high high-dose-rate intracavitary brachytherapy ratio (>0.43) (62.5% vs 88.2%, p = 0.0067). Patients with both anemia and lymphopenia during concurrent chemoradiotherapy showed poor survival, independent of mid-squamous cell carcinoma antigen, and escalating high-dose-rate intracavitary brachytherapy ratio might improve survival.
Introduction
Quantitative measurements of tumor volume (TV)/regression and squamous cell carcinoma antigen (SCC Ag) measurements during and after conventional pelvic external beam radiotherapy (EBRT) are accepted prognosticators of radiotherapy (RT) treatment outcome in cervical squamous cell carcinoma (SCC).1–3 However, radioresistance frequently undermines treatment efficacy. Studies linking tumor repopulation mechanisms and anemia during RT attempt to explain radioresistance but were only partially successful.4,5
Tumor hypoxia is associated with poor treatment response and unfavorable prognosis due to angiogenesis, anaerobic glycolysis, and hypoxia-inducible factor 1 alpha (HIF-1α)-stimulated proliferation.6,7 In addition, hypoxic regions of heterogeneous tumors are less sensitive to RT than normoxic regions. Fractionated RT was developed to cause acutely hypoxic radioresistant tumor cells to become radiosensitive. 8 Nevertheless, RT-induced HIF-1α expression in residual chronic hypoxic or anoxic cancer cells could be boosted by microvascular reactive oxygen species (ROS) and inflammatory damage resulting from RT.6,9 Subsequent HIF-1α signaling could lead to immune suppression.9–11 Therefore, investigating intratumoral hypoxia and immunity during RT may guide therapy and reveal potential prognostic markers in cervical cancer.
A previous study by the authors identified hemoglobin (Hb), absolute neutrophil count (ANC), and absolute lymphocyte count (ALC) in the second week of concurrent chemoradiotherapy (CCRT) as prognostic factors in stage IIB cervical cancer. 12 However, this study could not correlate hematological parameters with CCRT resistance because these parameters were not compared with quantitative tumor measurements. Therefore, we investigated whether anemia, neutrophilia, and lymphopenia in the second week of CCRT were independently associated with poor survival. We further compared these parameters with SCC Ag levels before, during, and after CCRT in stage IB–III SCC.
Methods and materials
This investigation was approved by the International Review Board (xxxxx-xxx-xxx-xx-xxx). Patients diagnosed (by uterine cervical biopsy) with newly invasive SCC and treated with pelvic cisplatin-based CCRT and high-dose-rate intracavitary brachytherapy (HDR IBT; N = 152) at our hospital, from August 2001 to July 2014, were studied. Patients had stage IB–III disease, as classified by the International Federation of Gynecology and Obstetrics (FIGO) staging system. Three patients with autoimmune disease or infection at diagnosis were excluded. All patients underwent physical examination, comprehensive blood analysis with/without differential count, pelvic magnetic resonance imaging (MRI), chest radiography, and optional positron emission tomography–computed tomography (PET CT) scanning. Sigmoidoscopy and cystoscopy were performed in suspected rectal or bladder invasion cases, respectively. Concurrent with whole pelvis RT, cisplatin was administered for 4–6 weeks at 40–70 mg/m2 per week. Forty-one patients were treated with 45 (or 45.6) Gy in 27 fractions including partial hyperfractionation (18 or 15 Gy) in 12 (or 10) fractions twice a day in the third week, to reduce overall treatment time (OTT; time interval between RT start and RT end). 13 Parametrium and lymph node (LN) boost irradiations of 5.4–10 Gy in 3–5 fractions were performed sequentially. Midline block was applied after 36–53 Gy in 137 patients. Two-dimensional HDR IBT (iridium 192 (192Ir); Microselectron; Nucletron Elekta Instrument AB Stockholm, Sweden) was performed biweekly after patients underwent pelvic EBRT for 3–6 weeks. As per the International Commission on Radiation Units (report 38), all patients were treated with HDR IBT of 5–30 Gy to point A in 1–7 fractions. Point A was changed to 1 or 1.5 cm in patients with a small uterus. The equivalent dose in 2 Gy using an alpha/beta ratio (α/β) of 3 for the bladder and rectal points was delivered to <80.0 or <70.0 Gy in two-dimensional simulations, respectively. Total dose (TD) was defined as the sum of central pelvic EBRT equivalent dose in 2-Gy fractions using an α/β ratio of 10 (EQD2) and HDR IBT EQD2. The HDR IBT ratio was the ratio of the HDR IBT EQD2 to the TD. After treatment completion, patients were followed up every 3 months for 1 year, and every 6 months thereafter, (median follow-up: 58 months, range: 3–144 months). Progression was detected by physical examination, Pap smear, tumor markers, and imaging (CT, MRI, or PET CT). Patients with disease progression were managed with chemotherapy, RT, or conservative care. The endpoints of this study were disease-specific survival (DSS) and progression-free survival (PFS).
Pretreatment Hb, ANC, ALC, and SCC Ag were acquired before CCRT and thereafter measured weekly for 4 weeks during treatment. The values of these parameters in the second week were designated as Hb2, ANC2, and ALC2. In the third week of CCRT and 2–3 months after treatment, SCC Ag levels were quantitated and designated as mid-SCC Ag and end-SCC Ag, respectively. Pretreatment TV was measured from contouring the gross tumor on T2-weighted MRI scans obtained using the Eclipse™ Treatment Planning System, version 10.0 (Varian Medical Systems, Palo Alto, CA, USA).
Statistical analyses were performed using the R software version 3.3.3 (R foundation for Statistical Computing, https://www.r-project.org/). Receiver operating characteristic (ROC) curves were generated for 5-year DSS with Hb2, ANC2, ALC2, HDR IBT ratio, pretreatment-SCC Ag, mid-SCC Ag, end-SCC Ag, and pretreatment-TV. Variables with area under the curve (AUC) ≤0.5 were excluded from analysis. Youden’s index (sensitivity + (specificity – 1)) was used to set cutoff values, and “high” and “low” groups were delineated for each parameter. The 5-year DSS rate (5DSSR) and 3-year PFS rate (3PFSR) were compared between groups using log-rank tests (low Hb2 vs high Hb2, low ALC2 vs high ALC2, low Hb2 and low ALC2 vs high Hb2 and/or high ALC2). Age, pelvic LN status, stage, OTT, TD, SCC Ag, HDR IBT ratio, and TV of low Hb2 and low ALC2 groups were compared with other groups using Fisher’s exact test and χ2 test. Those with a significant p value in univariate analyses of variables were included in multivariate analyses using Cox proportional hazards model. Three Kaplan–Meier plots were generated for “high/low” subgroups for variables: mid-SCC Ag and both Hb2 and ALC2, mid-SCC Ag and HDR IBT ratio, and Hb2 and ALC2 and HDR IBT ratio. Two-sided p values <0.05 were considered to be statistically significant.
Results
Patient characteristics
Table 1 summarizes patient characteristics and circulating and volumetric factors. Details of ROC analyses are described in Supplementary Table 1. Due to an AUC ≤0.5, ANC2 was excluded.
Patient characteristics.

SCC Ag: squamous cell carcinoma antigen; LN: lymph node; Hb: hemoglobin; CCRT: concurrent chemoradiotherapy; FIGO: International Federation of Gynecology and Obstetrics; ALC: absolute lymphocyte count; EQD2: equivalent does in 2 Gy fractions using an α/β ratio of 10; HDR IBT ratio: the ratio of high-dose-rate intracavitary brachytherapy dose to total dose.
Treatment outcome
Among 152 patients, 31 showed disease progression, leading to 23 mortalities. Local recurrence without distant metastasis (DM) was observed in four patients, while five patients showed cervical progression and DM to the para-aortic LN (three cases), liver (two), peritoneum (two), and bone (one). Overall, 22 patients exhibited DM: para-aortic LN (10 cases), lung (9), bone (5), peritoneum (3), liver (2), supraclavicular LN (2), muscle (2), brain (1), and adrenal gland (1). The 5DSSR and 3PFSR of all patients were 84.8% and 82.8%, respectively. The 5DSSR of patients with stage IB–IIA, IIB, and III was 88.8%, 87%, and 62.3%, respectively (p = 0.023). The 3PFSR of patients with stage IB–IIA, IIB, and III was 88.8%, 85.7%, and 52.9%, respectively (p < 0.001).
Hb and lymphocytes
Difference in 5DSSR and 3PFSR between high and low groups of both Hb2 and ALC2 was larger than when high and low groups of either Hb2 or those of ALC2 were compared (Table 2). The ROC curve considering both Hb2 and ALC2 had a larger AUC than that of Hb2 alone (Supplementary Figure 1). Sensitivity, specificity, and Youden’s index were 0.667, 0.719, and 0.386, respectively, as per the cutoff values for both Hb2 and ALC2 (≤11 g/dL and ≤639 cells/µL vs >11 g/dL and/or >639 cells/µL). Youden’s index for both Hb2 and ALC2 was higher than that of either Hb2 or ALC2 alone (0.378 vs 0.299 or 0.213). Most patients with both low Hb2 and low ALC2 were stage IIB/III, with higher pretreatment-SCC Ag, higher mid-SCC Ag, higher end-SCC Ag, and larger TV, than those with high Hb2 and/or high ALC2 (Table 3).
Comparison of survival rates according to cutoff values of hemoglobin, lymphocyte count, and both hemoglobin and lymphocyte count in the second week of concurrent chemoradiotherapy.

CCRT: concurrent chemoradiotherapy; 5-DSS: 5-year disease-specific survival; 3-PFS: 3-year progression-free survival; Hb: hemoglobin; ALC: absolute lymphocyte count.
Comparison of variables between patients with both anemia and lymphopenia during the second week of concurrent chemoradiotherapy and those without anemia and lymphopenia.
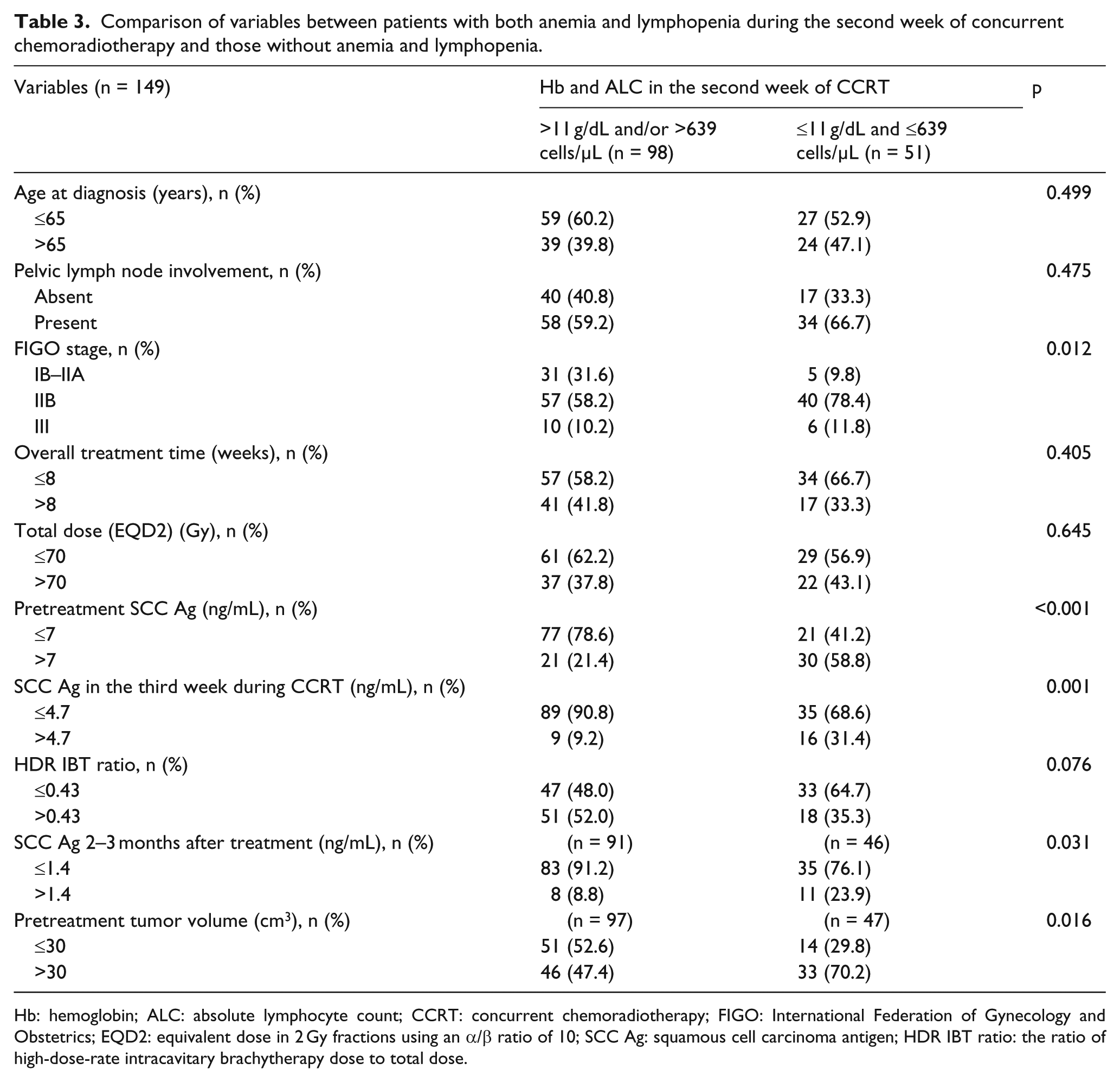
Hb: hemoglobin; ALC: absolute lymphocyte count; CCRT: concurrent chemoradiotherapy; FIGO: International Federation of Gynecology and Obstetrics; EQD2: equivalent dose in 2 Gy fractions using an α/β ratio of 10; SCC Ag: squamous cell carcinoma antigen; HDR IBT ratio: the ratio of high-dose-rate intracavitary brachytherapy dose to total dose.
Cox regression analyses
Univariate analyses revealed that stages, TV, pretreatment-SCC Ag, both Hb2 and ALC2, mid-SCC Ag, HDR IBT ratio, and end-SCC Ag correlated significantly with both DSS and PFS (Supplementary Table 2). Mid-SCC Ag and both Hb2 and ALC2 correlated significantly with DSS and PFS in multivariate analyses (Table 4). Significant association between HDR IBT ratio and PFS was also observed.
Multivariate analyses for disease-specific survival and progression-free survival.

HR: hazard ratio; CI: confidence interval; SCC Ag: squamous cell carcinoma antigen; Hb: hemoglobin; ALC: absolute lymphocyte count; CCRT: concurrent chemoradiotherapy; HDR IBT ratio: the ratio of high-dose-rate intracavitary brachytherapy dose to total dose.
Kaplan–Meier plots
High mid-SCC Ag conferred significantly lower 5DSSR and 3PFSR than low mid-SCC Ag (52% vs 92.2%, p < 0.001 and 48% vs 89.6%, p < 0.001). The 5DSSR and 3PFSR for the high HDR IBT ratio (>0.43) versus low HDR IBT ratio (≤0.43) groups were 91.1% versus 78.2% (p = 0.002) and 92.8% versus 74.2% (p < 0.001), respectively. Figure 1(a) shows risk stratification for the Hb2 and ALC2 subgroups, classified by mid-SCC Ag. In the low mid-SCC Ag group, for Hb2 and ALC2, the both low subgroup had significantly lower 5DSSR and 3PFSR than the either or both high subgroup (85.5% vs 94.6%, p = 0.0096 and 79.8% vs 93.1%, p = 0.0174). In the high mid-SCC Ag group, for Hb2 and ALC2, both the low subgroup tended to have lower 5DSSR and 3PFSR than the either or both high subgroup (43.8% vs 66.7%, p = 0.192 and 37.5% vs 66.7%, p = 0.268, respectively). In the mid-SCC Ag high as well as in the mid-SCC Ag low groups, there was differential risk between the high HDR IBT ratio and low HDR IBT ratio subgroups (Figure 1(b)). When both Hb2 and ALC2 were low, there was differential risk between low and high HDR IBT ratio subgroups, but this was not seen when either or both Hb2 and ALC2 were high (Figure 1(c)). In the low mid-SCC Ag group, 5DSSR and 3PFSR were significantly lower with a low compared to a high HDR IBT ratio (90.6% vs 94.5%, p = 0.033 and 82.8% vs 96.7%, p = 0.0017). When mid-SCC Ag was high, 5DSSR and 3PFSR tended to be lower in patients with a low compared to a high HDR IBT ratio (41.2% vs 75%, p = 0.149 and 41.2% vs 62.5%, p = 0.186).
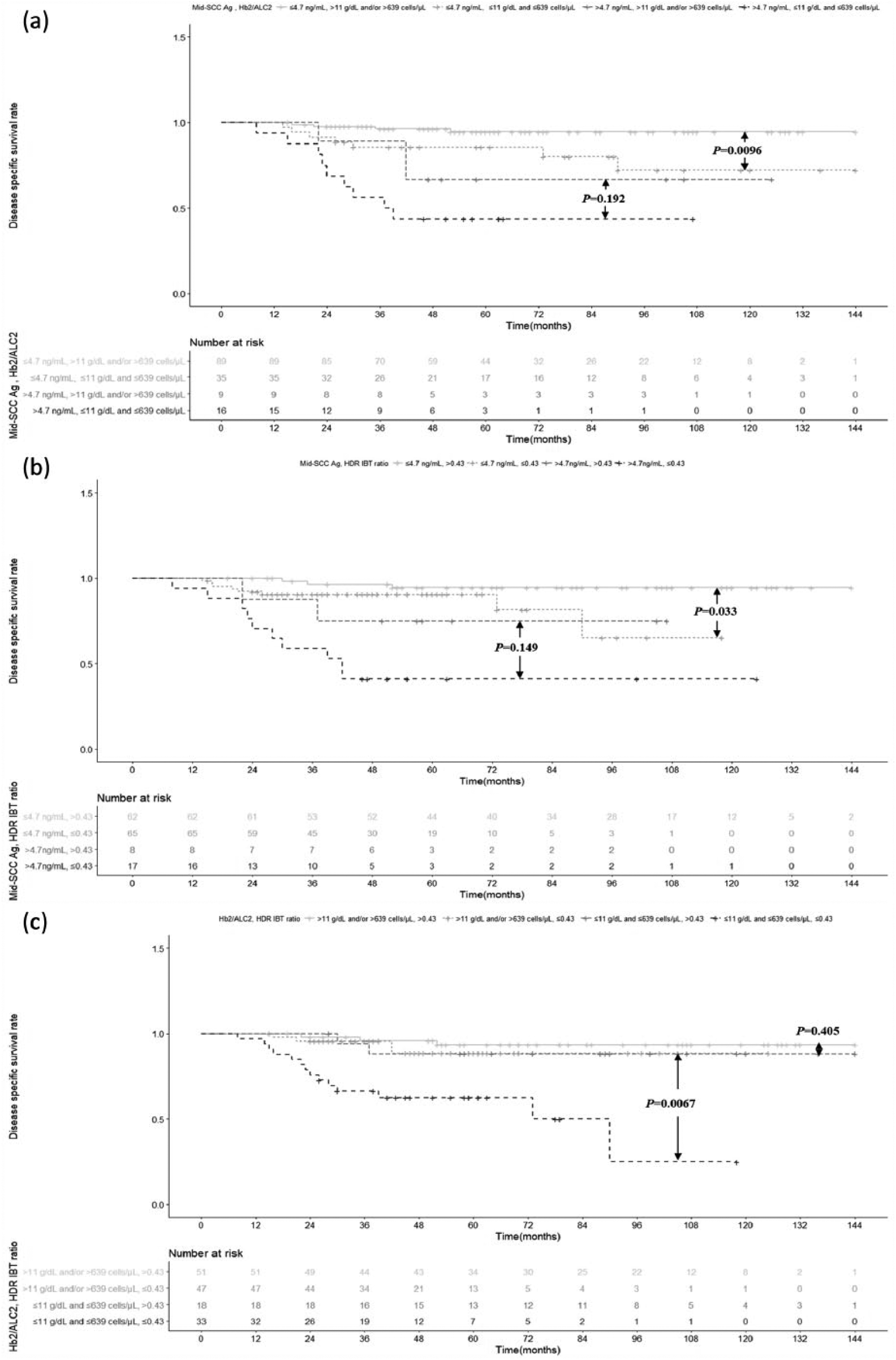
Kaplan–Meier plots of data from 152 cervical SCC patients treated with CCRT and high-dose-rate intracavitary brachytherapy (HDR IBT). Kaplan–Meier plots for disease-specific survival according to (a) subgroups of the “both Hb2 and ALC2’ group, classified by mid-SCC Ag, (b) subgroups of the “mid-SCC Ag” group, classified by HDR IBT ratio, (c) subgroups of “both Hb2 and ALC2” group, classified by HDR IBT ratio.
In the both Hb2 and ALC2 low group, patients with low HDR IBT ratio had significantly lower 5DSSR and 3PFSR than those with high HDR IBT ratio (62.5% vs 88.2%, p = 0.0067 and 54.6% vs 88.5%, p = 0.0061, respectively). When either or both Hb2 and ALC2 were high, 5DSSR and 3PFSR were not significantly different between low and high HDR IBT ratio groups (88.6% vs 93.5%, p = 0.405 and 86.9% vs 94.1%, p = 0.089, respectively).
Discussion
Simultaneous anemia and lymphopenia in the second week of CCRT and mid-SCC Ag correlated significantly with DSS and PFS. Risk stratification based on both Hb2 and ALC2 was independent of mid-SCC Ag, though having both low Hb2 and low ALC2 correlated with increased SCC Ag throughout the study.
Acute tumor response to fractionated EBRT depends on increased radiosensitivity through reoxygenation (min), reduced respiration of dying cells (h), and increased oxygenation due to lowered vessel-to-tumor distance (days).14,15 However, residual tumor tissue is irreversibly hypoxic rather than reversibly hypoxic or normoxic. During reoxygenation, RT may damage intratumoral microvessels by triggering ROS and inflammation, 9 although vasculopathy within the residual tumor might lower oxygen pressure to sub-pre-RT levels. Induction of HIF-1α could lead to T-cell apoptosis (resulting in possible lymphopenia), tumor revascularization, and proliferation.10,11 Thus, fractionated RT might not be an effective method to control hypoxic tumors with rapid repopulation capability. Since HDR IBT characteristically delivers high doses without direct damage to circulating lymphocytes (counteracting HIF-1α-induced proliferation and lymphopenia), it could be a better option to control primary tumors.16–18
Thus, evaluating tumor hypoxia, inflammation, and immunity in the second week of CCRT (putative repopulation period for cervical cancer) might aid in HDR IBT dose selection and evaluating disease prognosis. 4 Benefits of adding hypoxic sensitizers, hyperbaric oxygen, hyperthermia, or immunotherapy may also be evaluated using this data.19,20 However, measuring intratumoral oxygen pressure and HIF-1α during CCRT is impractical, requiring tumor biopsies, which nevertheless may not be representative of the entire tumor. 19 Due to this limitation, anemia as a cause of hypoxia has been investigated as a prognostic factor for cervical cancer in many studies.5,19,21–23
Since it was not practical for us to monitor inflammatory markers (interleukins, tumor necrosis factor, and nuclear factor kappa B (NF-κB)) during RT, 9 we measured ANC. For similar reasons, we used ALC to evaluate immunity instead of immune checkpoint markers or T-cell counts. Though Hb, ANC, and ALC are indirect markers of hypoxia, inflammation, and immunity, they are linked through HIF-1α signaling during RT. Hence, we believe that evaluating all three may provide a reliable picture of target parameters. Literature support for our assumption comes from a previous study evaluating Hb2, ANC2, ALC2, and HDR IBT ratio in stage IIB cervical cancer. 12
Our results reveal a correlation between hematological parameters (tumor quality) and SCC Ag (tumor quantity) and treatment outcome in cervical SCC patients of various stages. We excluded ANC2 from our analysis due to an AUC ≤0.5 in ROC analysis, contrary to our expectation. This inconsistency compared to our previous result might be because of the relatively small sample size in our previous study. 12 In addition, neutrophil counts may not truly represent inflammatory conditions, and there is controversial evidence linking neutrophils to both tumor suppression and tumor progression. 24 Moreover, inflammation during RT may influence cancer progression, fibrosis, and intratumoral microvascular damage, and ANC2 levels may not reflect these changes accurately. 9 Notably, there are no reports linking neutrophil counts during RT and survival, while the association between either anemia or lymphopenia during RT and survival in cervical cancer patients has been previously reported.5,22,25
Our results suggest that a combination of Hb2 and ALC2 is superior to either Hb2 or ALC2 alone in predicting survival and supports our assumption about the link between tumor repopulation, immune suppression, and hypoxia during CCRT. In this study, patients with low Hb2 and ALC2 showed high SCC Ag and high TV, implying that qualitative and quantitative aspects of a heterogeneous tumor are inter-connected. Although mid-SCC Ag was significantly predictive of survival, Hb2 and ALC2 together may allow for independent risk stratification. This may also imply that Hb2 and ALC2, reflecting tumor hypoxia and immunity, may be clinically different from mid-SCC Ag, which reflects tumor burden. Moreover, Kaplan–Meier analysis revealed that escalating relative HDR IBT dose was more effective in improving survival in patients with both low Hb2 and ALC2 and was effective irrespective of mid-SCC Ag level. Hence, treating primary tumors with HDR IBT instead of EBRT might improve treatment outcome in patients with both low Hb2 and ALC2, probably by controlling hypoxic tumors without associated circulating lymphocyte damage. These results could also partially explain the improved treatment outcomes with high HDR IBT ratio in our previous study. 18
Past studies have illustrated that Hb during RT was associated with survival.5,22 However, the association between weekly Hb and survival was not analyzed, while in this study, a thorough analysis of these parameters was performed. Moreover, the modest association between anemia and low tumor oxygenation bring into question the proposed causative link between anemia and HIF-1α pathway activation. 26 Our attempt to evaluate Hb and ALC simultaneously may reinforce the clinical relevance of anemia during CCRT, without the need for measuring tumor oxygenation. The hypothesis that co-existence of anemia and lymphopenia during RT could influence treatment outcome might partially explain failure of clinical trials, which aimed to overcome anemia using transfusion or erythropoietin, to ultimately improve survival.26,27 In addition, past studies reported that adding hyperthermia or bevacizumab continuously during pelvic CCRT could expect improvement of treatment outcome in locally advanced cervical cancer.28,29 Our results suggest that the second week of CCRT is the optimal time to employ additional intervention modalities like blood transfusion, erythropoietin administration, hyperthermia, and bevacizumab for patients with simultaneous anemia and lymphopenia during pelvic CCRT. This would ensure maximum patient benefit with reference to time, cost, and toxicity. In a past study, the survival benefit of HDR IBT in cervical cancer has been explained by the ability to deliver high doses efficiently to the cervical tumor while sparing adjacent organs. 30 However, this may not explain the benefit of increasing the relative HDR IBT dose. 18 This study presents evidence to elucidate the survival benefit conferred by HDR IBT by comparing relative efficacies of HDR IBT doses in groups classified according to mid-SCC Ag and both Hb2 and ALC2.
This study had several limitations. First, Hb2 and ALC2 are indirect indices of intratumoral hypoxia and lymphopenia and may not accurately represent HIF-1α relevant immune modulation. Further experimental investigation should monitor intratumoral oxygen pressure, HIF-1α signaling, immune checkpoints, and T-cell subgroups, to confirm relevant correlations. Second, this was a retrospective study of a small population. Therefore, larger observational studies or effective prospective studies are warranted. Despite these limitations, as per our knowledge, this is the first study to suggest an association between simultaneous anemia and lymphopenia during CCRT, HDR IBT ratio, and survival, analyzed against SCC Ag in cervical SCC patients.
In conclusion, Hb2 and ALC2, independent of mid-SCC Ag, could predict treatment outcome in stage IB–III cervical SCC patients. Escalating relative HDR IBT dose might improve survival of patients with both anemia and lymphopenia in the second week of CCRT. Therefore, Hb2 and ALC2 may reflect intratumoral environment and quality, while mid-SCC Ag may be a quantitative measure of tumor size. Taken together, these parameters might be effective prognosticators of treatment response and survival in cervical cancer.
Footnotes
Author contributions
The article was conceptualized by Oyeon Cho. Methodology was by Oyeon Cho and Mison Chun, and formally analyzed by Oyeon Cho and O Kyu Noh. Investigation was carried out by all the authors. The original draft was written by Oyeon Cho, reviewed and edited by Oyeon Cho and Mison Chun. The resources are provided by Suk-Joon Chang, Hee-Sug Ryu, and Eun Ju Lee, and supervised by Mison Chun, Young-Taek Oh, and O Kyu Noh.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval and patient consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors. The need for informed consent was waived because of the retrospective nature of the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
