Abstract
Xanthones are phytochemical compounds found in a number of fruits and vegetables. Characteristically, they are noted to be made of diverse properties based on their biological, biochemical, and pharmacological actions. Accordingly, the apoptosis mechanisms induced by beta-mangostin, a xanthone compound isolated from Cratoxylum arborescens in the human promyelocytic leukemia cell line (HL60) in vitro, were examined in this study. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay was done to estimate the cytotoxicity effect of β-mangostin on the HL60 cell line. Acridine orange/propidium iodide and Hoechst 33342 dyes and Annexin V tests were conducted to detect the apoptosis features. Caspase-3 and caspase-9 activities; reactive oxygen species; real-time polymerase chain reaction for Bcl-2, Bax, caspase-3, and caspase-9 Hsp70 genes; and western blot for p53, cytochrome c, and pro- and cleavage-caspase-3 and caspase-9 were assessed to examine the apoptosis mechanism. Cell-cycle analysis conducted revealed that β-mangostin inhibited the growth of HL60 at 58 µM in 24 h. The administration of β-mangostin with HL60 caused cell morphological changes related to apoptosis which increased the number of early and late apoptotic cells. The β-mangostin-catalyzed apoptosis action through caspase-3, caspase-7, and caspase-9 activation overproduced reactive oxygen species which downregulated the expression of antiapoptotic genes Bcl-2 and HSP70. Conversely, the expression of the apoptotic genes Bax, caspase-3, and caspase-9 were upregulated. Meanwhile, at the protein level, β-mangostin activated the formation of cleaved caspase-3 and caspase-9 and also upregulated the p53. β-mangostin arrested the cell cycle at the G0/G1 phase. Overall, the results for β-mangostin showed an antiproliferative effect in HL60 via stopping the cell cycle at the G0/G1 phase and prompted the intrinsic apoptosis pathway.
Introduction
Leukemia is characterized by the formation and production of immature and non-functioning leukocytes by the bone marrow. The overproduced leukocytes surpass the formation of the red blood cells and platelets which eventually leads to the development of anemia, 1 infection, and excess bleeding. 2 Accordingly, leukemia is classified into four groups which comprises acute and chronic cases of both lymphocytic and myelogenous leukemia. 3
In Malaysia, records available at government hospitals indicate that leukemia was the third leading cause of death between 2003 and 2005. 4 The treatment strategies of hematological malignancy include chemotherapy and radiotherapy. 3 Basically, these strategies are meant to eliminate leukemic cells from the bloodstream through various mechanisms. The mechanisms are the promotion of normal cell communication, the enhancement of the immune response and the reduction of genetic instability, and the inhibition of abnormal gene expression, abnormal transduction signal, tumor angiogenesis, and apoptosis. Additionally, the strategies induce apoptosis via deactivation of antiapoptotic proteins concomitant with the stimulation of apoptotic proteins. 5
Apoptosis, also referred to as the program of cell death, is a process which occurs in a multi-cellular form, as well as in protozoa in response to stresses such as heat, radiation, nutrient depletion, lack of oxygen, and infection. 6 Basically, these factors induce apoptosis via two pathways. The first pathway is the extrinsic pathway which is activated by extracellular promoters that bind the death receptors on the cell membrane. This process leads to switch on cascade reactions, mediated by cysteine–aspartic proteases proteins (caspases) to change the expression profile of certain genes 7 to exacerbate the death of cells. Relatedly, the second pathway, which is also referred to as the intrinsic or mitochondria pathway, involves intracellular stimuli targeting the mitochondria, the endoplasmic reticulum (ER), and the nucleus. Interestingly, it is regulated by different apoptosis inducer caspases. 8
Characteristically, chemotherapy is the most common treatment for leukemia. Although it is capable of eradicating the flow of cancerous cells, it produces toxic side effects, and over time, the cancer cells develop resistance. Hence, the exploration of novel chemotherapeutic agents with less toxicity is advised. In the last two decades, researchers have focused on the anticancer properties of natural compounds isolated from plants and herbs in accordance with traditional medicine. 9 Among these compounds are xanthones. Xanthones are mostly naturally available in many vegetables and fruits. Accordingly, several studies have found that xanthones demonstrate anti-inflammatory, antioxidant, antitumor, antiallergic, antibacterial, antifungal, and antiviral activities. 10 A number of studies reported that xanthone molecules alpha and gamma mangostin, isolated from mangosteen fruit, exert anticancer properties. Particularly, alpha mangostin can reduce prostate malignant hyperplasia. 11 However, gamma mangostin can suppress colon 12 and hepatocellular cancers. 13
β-mangostin (BM), a xanthone compound isolated from Cratoxylum arborescens, is traditionally used to treat fever, cough, diarrhea, itches, ulcers, and abdominal disorders in Malaysia, South Burma, Sumatra, and Borneo. 14 A recent study reported that BM could inhibit the growth of MCF-7, a breast cancer cell line. 15 In addition, it showed a cytotoxic effect against the murine leukemia (WEHI-3) cells. 16 The in-depth investigation in this study was conducted to examine the apoptosis induction mechanisms via BM in HL60 cell line in vitro.
Materials and methods
The human promyelocytic leukemia cell line (HL60) was obtained from the American Type Culture Collection (ATCC). HL60 was subcultured in a 75-cm tissue culture flask with RPMI 1640 media mixed with 10% bovine serum albumin and 1% penicillin–streptomycin (Nacalai Tesque, Inc., Japan). The subculture was then kept in an incubator at 5% CO2 saturation and 37°C temperature. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was obtained from Nacalai, USA. The antibodies used for western blot, caspase-3 and caspase-9, cytochrome c, β-actin, and secondary antibodies were obtained from Abcam (UK). p53 was purchased from Novus Biologicals (USA) and cleaved caspase-3 and caspase-9 from GeneTex, Inc. (USA). Caspase activity kits were obtained from R&D Systems (USA). The BD Pharmingen Annexin (USA) V-FITC Apoptosis Detection Kit (APO Alert Annexin V; Clontech Laboratories, Inc., USA) was used in the Annexin V experiment.
BM collection, purification, and identification
The pure beta-mangostin (BM) compound was isolated from the Cratoxylum arborescens plant and the purity was analyzed in a UFLC system equipped with a PDA-UV) detector and Ion-Trap time-of-flight (TOF) mass spectrometer (Shimadzu, Japan). Flow rate: 0.50 mL/min. Column temperature: 40°C. Gradient: 10%–100% MeCN over 7 min. Column: Water Xbridge 50 × 2.1 mm, 2.5 µM. Mobile phase: H2O (0.1% formic acid):MeCN (0.1% formic acid); more details have been reported in one of our group study. 17
Cell viability assay
The principle of cell viability assay is based on the reduction of yellow MTT into purple formazan derivatives by mitochondrial succinate dehydrogenase in the survival cells. The amount of formazan salts formed could be detected by a spectrophotometer after the salts were dissolved by dimethyl sulfoxide (DMSO). Briefly, 5 × 103 cells were seeded per well in 96-well plates; these cells were treated with BM (dissolved in 1% DMSO) at different concentrations (100, 50, 25, 12.5, 6.3, 3.5, and 1.5 µg/mL). In general, the negative control for all the assays was represented by the untreated medium containing DMSO (0.1%), which was then incubated at 37°C with 5% CO2 saturation for 24 h. After the incubation periods, 20 µL of MTT solution (5 mg/mL) was added to each well, followed by 4 h incubation. Also, 100 µL of DMSO was added to each well to dissolve the formazan crystals. The optical density was measured at a wavelength of 570 nm using a microplate reader (Tecan Group Ltd., Switzerland). The viability of the cell was thus determined. 18
Detection of morphological changes of apoptotic cells by acridine orange/propidium iodide double staining
Acridine orange (AO) is a cell membrane–permeable dye in viable and early apoptotic cells. The AO emits green fluorescence, whereas propidium iodide (PI) dye emits orange fluorescence when intercalating with DNA of late apoptotic and secondary necrotic cells. Accordingly, these double-staining dyes were used to detect the apoptotic cells of treated HL60. Briefly, HL60 cells were seeded at a concentration of 2 × 105 cells/mL in a 25-mL culture flask and treated with BM at 23, 35, 47, and 58 μM. The cells were then incubated with 5% CO2 at 37°C for 24 h and then collected and centrifuged at 1500 r/min. The supernatant was discarded, and the cell pellet was washed twice with cold phosphate-buffered saline (PBS). Up to 10 µL of a mixture of fluorescent dyes, AO (10 μg/mL) and PI (10 μg/mL) were added to the re-suspension pellet, and then, the stained cell was placed on a glass slide and covered with a cover slip. Before the fluorescent dye faded, the slides were examined within 30 min under a UV-fluorescence microscope (Leica attached with Q-Floro software) in accordance with standard procedure. Viable cells appeared with a green nucleus and an intact structure, whereas early apoptotic cells exhibited a bright-green nucleus showing condensation of the nuclear chromatin. Late apoptotic cells displayed dense orange areas of the chromatin condensation.19–21
Hoechst 33342 staining
Bisbenzimidazole (Hoechst 33342) stain was used to reveal the chromatin condensation, which is a hallmark of apoptosis. The treated HL60 cells with 23, 35, 47, and 58 μM of BM were collected and centrifuged at 1500 r/min, and the pellet was washed twice with cold PBS and then centrifuged again. Hoechst 33342 dye (10 µg/mL) was subsequently added. The stained cells were suspended and placed on a slide, covered with a cover slip, and examined under a UV-fluorescence microscope (Leica attached with Q-Floro Software).
Annexin V assay
To determine the different stages of apoptosis, HL60 was stimulated with various concentrations (23, 35, 47, and 58 µM) of BM for 24 h, and then, the cells were collected and centrifuged at 1500 r/min. The pellet was resuspended in 1× binding buffer and incubated for 1 h. Afterward, Annexin V (5 μL) and PI (10 μL) were added. The cells were kept in the dark at room temperature for 15 min. Samples were then run and analyzed which led to fluorescein isothiocyanate (FITC) being detected in the 530/30 nm channel and PI at 585/42 nm channel on FACS Canto II cytometry (BD Biosciences, USA).
Determination of reactive oxygen species production
The capability of BM to produce reactive oxygen species (ROS) was evaluated using 2′,7′-dichlorofluorescin diacetate (DCFH-DA). HL60 cells (5 × 103 cells/mL) were seeded in each black 96 wells. Then, cells were treated with specific doses of BM. After incubation periods, DCFH-DA (100 µL) was subsequently added, and the suspensions were incubated for 30 min at 37°C. The fluorescence was measured at 485 nm via a fluorescence microplate reader (Tecan Infinite M 200 PRO, Switzerland).
Activities of caspase-9 and caspase-3/7
To assess the susceptibility of the BM to stimulate the enzymatic activity of caspase-9 and caspase-3/7 at different concentrations (23, 35, 47, and 58 µM) of BM compared to the control, cells were centrifuged at 1500 r/min for 5 min after incubation for 24 h. The cell pellet was washed in cold PBS, mixed with 25 µL of manufactured lysis buffer, and then incubated on ice for 10 min. At 10,000g, the mixture was centrifuged for 1 min, and the supernatant was collected and transferred to 96-well plates. Thereafter, 50 μL of reaction buffer and 5 μL of substrate for caspase-9 (LEHD-PNA) and caspase-3 (DEVD-PNA) were added. The plates were incubated at 37°C for 2 h. After incubation at 37°C for 2 h, the plates were read by a Tecan-Infinite® 200 Pro microplate reader (Tecan Trading AG, Switzerland) at excitation wavelength of 405 nm.
Real-time polymerase chain reaction
In order to assess the effects of BM on the apoptosis-controlling genes, such as Bax, caspase-3 and caspase-9, Bcl-2, and HSP70, real-time polymerase chain reaction (PCR) was done. After treating the HL60 with the different concentrations of BM, RNAs were extracted using the RNeasy Mini Kit (Qiagen, Germany), and then, the total RNAs were converted by the synthesization of complementary DNAs using a High-Capacity RNA-to-cDNA kit (Applied Biosystems, USA). This was done in accordance with the protocol proposed by the manufacturer. Additionally, the gene expression assay was run using TaqMan® Fast Advanced Master Mix, and genes β-actin, Bax, Bcl-2, Hsp70, caspase-3, and caspase-9 were obtained from TaqMan. The real-time PCR reaction program was set as reverse transcriptase for 2 min at 50°C, activation of polymerase at 95°C for 20 s, denaturation at 95°C, and finally, the annealing reaction for 1 and 20 s, respectively. Interestingly, this procedure was completed in 40 cycles in the StepOnePlus machine (Applied Biosystems).
Western blot
Western blot was run to evaluate the effect of BM on the apoptotic proteins, pro- and cleaved caspase-3 and caspase-9, p53, and cytochrome C. A 75-mL cell culture flask was used to seed the control and treat the cells with 23, 35, 47, and 58 µM of BM. Subsequently, these cells were incubated for 24 h. The PRO-PREPTM (iNtRON, Biotechnology, Korea) kit was used to extract the whole proteins, and the Bio-Rad kit was employed for protein quantification. Approximately 10%–14% of sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was used to run the samples to obtain the separate bands of targeting proteins for 2 h. Thereafter, the bands were transferred from the gel to a polyvinylidene fluoride (PVDF) membrane (Bio-Rad, USA). The membrane was washed with distilled water, and bovine serum albumin was used to block the membrane for 60 min. Each primary antibody for targeting proteins was diluted 1:1000 and incubated with the membrane for 1 h overnight and then washed with Tween 20 phosphate-buffered saline (TPBS). The membrane was incubated for 1 h with a suitable secondary antibody 1:2000. To visualize the protein bands on the membrane, the colorimetric horseradish peroxidase (HRP) substrate, 4-chloro-1-naphthol (4CN; Bio-Rad) kit, was used. To capture images of the membrane, a UV gel documentation system (Biospectrum 410, UVP) was used.
Cell-cycle analysis
To determine the phase at which BM can arrest the cell cycle of treated HL60 cells, 2 × 105 cells/mL of treated and untreated cells were harvested and centrifuged at 1800 r/min for 5 min. The pellet was washed twice with pre-warmed PBS (37°C) and then centrifuged. Subsequently, 700 µL of 90% cold ethanol was added, and the mixture was kept overnight at 4°C. After incubation, the cell suspension was centrifuged at 1800 r/min for 5 min, and the ethanol was discarded. The cell suspension was then washed with PBS, and 700 µL of pre-warmed PBS was added to the pellet. This was followed by the addition of 25 µL of RNase (10 mg/mL) and 50 µL of PI (1 mg/mL) prior to incubation for 1 h. The sample was then run and analyzed using FACS Canto II cytometry (BD Biosciences) at excitation wavelength of 488 nm and emission at 519 nm.
Statistical analysis
The SPSS was used to analyze the data and expressed as mean ± SD. Also, the one-way analysis of variance (ANOVA) was used to evaluate the differences between groups. Values of *p < 0.05 were considered statistically significant. GraphPad Prism 5.0 was used to present the data.
Results
BM inhibited growth of HL60 cells
The cytotoxic effects of BM on HL60 cells were assessed by MTT assay. As shown in Figure 1, BM inhibited the cell growth significantly at a concentration of 58 µM in 24 h.

Effects of BM on the HL60 cell viability.
BM induced apoptosis in HL60 cells
Morphological changes occurred in the cells due to exposure to BM as observed under fluorescence microscope. The status of the cells can be categorized as viable, early apoptosis, late apoptosis, and secondary necrosis cells.
The viable cells are structurally compact and has undamaged nucleus, which is presented by a homogenized green color. More so, the morphological changes appeared when the cell lines were exposed to BM. Furthermore, early apoptosis can be seen in bright-green cells, presented by cells showing membrane blabbing and DNA break-up. Meanwhile, cells stained with reddish-orange color due to AO binding to denatured DNA have represented the late stage of apoptosis. The apoptotic bodies representing the final stage of the apoptosis process have been observed and the necrotic cells manifested.
Quantitatively, the viable cells are most commonly observed in untreated cells, whereby its percentage has decreased to 70% in concentration of 35 µM in HL60. This decline continued until 35% at 58 µM. In contrast, the early apoptotic cell count elevated parallel to the increase of BM concentration to reach 18% at 58 µM.
The late apoptosis have been visibly exhibited with more than 30% of the total cells at the concentration of 58 µM. The necrotic cells were increased obviously at 47 and 58 µM to be more than 14% and 18%, respectively (Figure 2).

Morphological changes of HL60 cells due to apoptosis after treating with BM. This was followed by stained AO/PI dyes: (a) control (untreated) cells showed the normal structure with no remarkable apoptosis or necrosis feature. (b) At 23 µM of BM, AO interconnected with fragmented DNA (bright-green), and blabbing cells as an indicator of early apoptosis are noticed. (c) The orange color represents the late apoptosis at 35 µM. At (d) 47 µM and (e) 58 µM concentration, most of the cells in the late apoptosis secondary necrotic (bright red color) status are highlighted. (f) The percentages of viable, early, late apoptosis, and secondary necrotic cells after treating the HL60 with BM at different concentrations for 24 h. Apoptosis increased significantly (*p < 0.05) in a concentration-dependent manner.
The apoptotic properties possessed by BM have been confirmed by analyzing the DNA profile for treating HL60 via Hoechst 33342 staining. Figure 3 displays the differences seen in the nuclei of the treated and untreated cells after staining. HL60 cells have normal intact nuclei stained with dim blue dye. After BM application on the cells, the changes in the nucleus structure were detected at all concentrations.

Effect of BM on nuclear morphological changes of HL60 cells. (a) Control. And treated cells in (b) (23 µM), (c) (35 µM), (d) (47 µM), and (e) (58 µM) were stained with Hoechst 33342 after 24 h of treatment. Apoptotic cells have been represented brightly (bright blue arrows) due to DNA condensation in apoptotic cells, and the number increased through different concentrations of BM (magnification 100×).
Throughout apoptosis, the equilibrium distribution of phosphatidylserine (PS) is predominantly disturbed by an increased appearance of PS in the outer layer of the cell membrane. This asymmetric distribution is proportional to the different stages of apoptosis. Furthermore, PS displays a high affinity to bind selectively with Annexin V in the presence of Ca++. This property is used practically to distinguish viable cells from apoptotic and necrotic cells. Consequently, the detection is performed using flow cytometry. The results of Annexin V in Figure 4 is an indication that the HL60 cells were scattered across four quadrants. The viable cells Annexin V−/PI− accumulated in Q3, while early apoptotic cells Annexin V+/PI− displayed in Q4 due to Annexin V-FITC binding to the cell’s membrane. During late apoptosis, both Annexin V and PI are intercalated within the cells (Annexin V+/PI+; Q2), whereas the necrotic cells are (Annexin V−/PI+; Q1). The untreated cells of both cell lines (control) which initially gathered in Q3 moved to Q4, Q2, and Q1 in a concentration-dependent manner for HL60 cells upon BM treatment.

The influence of BM to induce apoptosis in HL60. After HL60 exposure to the different concentrations of BM for 24 h, flow cytometry was used to analyze early or late apoptosis. FITC-conjugated Annexin V and PI were added to each of the control and treated cells. The status of the cells was presented as healthy (Annexin V−/PI−) condensed in the quadrant (Q3-1), early apoptotic (Annexin V+/PI−; Q4-1), and late stage of apoptosis (Annexin V+/PI+; Q2-1), where the necrotic cells were (Annexin V−/PI+) in Q1-1. (a) Control, (b) 23 µM, (c) 35 µM, (d) 47 µM, and (e) 58 µM. (f) Representative bar chart illustrates the significant increase in the number of apoptotic and necrotic cells. Statistical significance is expressed as *p < 0.05.
Quantifying analysis of Annexin V-FITC has presented the percentage of viable cells, early apoptotic cells, late apoptotic cells, and necrotic cells of HL60 accordingly. Approximately 17% of treated cells at 23 µM entered the early apoptosis stage, which elevated to 22% at 35 µM. However, it peaked at 29% at the 58 µM concentration. Furthermore, the percentage of late apoptosis cells elevated to 13% at the 47 and 58 µM concentration. Moreover, the number of necrotic cells increased up to 10% at 58 µM concentration. This is expected since it is incubated with high concentration of BM, the cytotoxic effects of which manifested necrotic cells that are distinct. Thus, the number of viable cells were decreased automatically especially at concentrations of 35, 47, and 58 µM.
BM induced cell death with increased ROS formation
Overproduction of intracellular ROS disrupted mitochondrial membrane potential (MMP) and induced apoptosis. The result shows the elevation of fluorescence intensity unit of the treated cells in a concentration-dependent manner (Figure 5). Thus, excessive production of ROS in HL60 displayed significant differences compared to the untreated cells.

Effect of BM on ROS production. In treating HL60 cells at concentrations of 23, 35, 47, and 58 µM. For 24 h, the DCFH-DA was added to the cells, and the fluorescence was measured using a fluorescence microplate reader. Each treatment was triplicated. The statistical significance is *p < 0.05.
Determination of caspase-3/7 and caspase-9 activities
Caspases are intracellular protease enzymes involved in apoptosis. The findings have indicated that caspase-9 activities increased while treating HL60 cells to 0.40 in 58 µM from 0.25 in 23 µM of BM. Furthermore, β-mangostin has also been incorporated to stimulate caspase-3 activity at concentrations 23, 35, 47, and 58 µM as compared with the untreated cells (p < 0.05) (Figure 6).

The relative expression of caspase-9 and caspase-3 in HL60 cells after BM treatment at concentrations of 23, 35, 47, and 58 µM for 24 h, and results are represented as mean ± SD of three replicates. The statistical significance is *p < 0.05 from control.
Assessment of the effect of BM on the expression of selected apoptotic and antiapoptotic genes using the real-time PCR technique
The mRNAs of Bax in the treated HL60 significantly increased for the 23, 35, and 47 µM concentrations, while at 58 µM, extreme expression was observed (p < 0.05). Caspase-9 was also been significantly expressed (p < 0.05) at the concentrations of 35, 47, and 58 µM. Clearly, the caspase-3 expression was increased at the different concentrations (p < 0.05). The apoptosis repressor genes Bcl-2 and HSP70 significantly reduced mRNAs (p < 0.05) after treatment at the different concentrations of BM compared to the untreated cells (Figure 7).
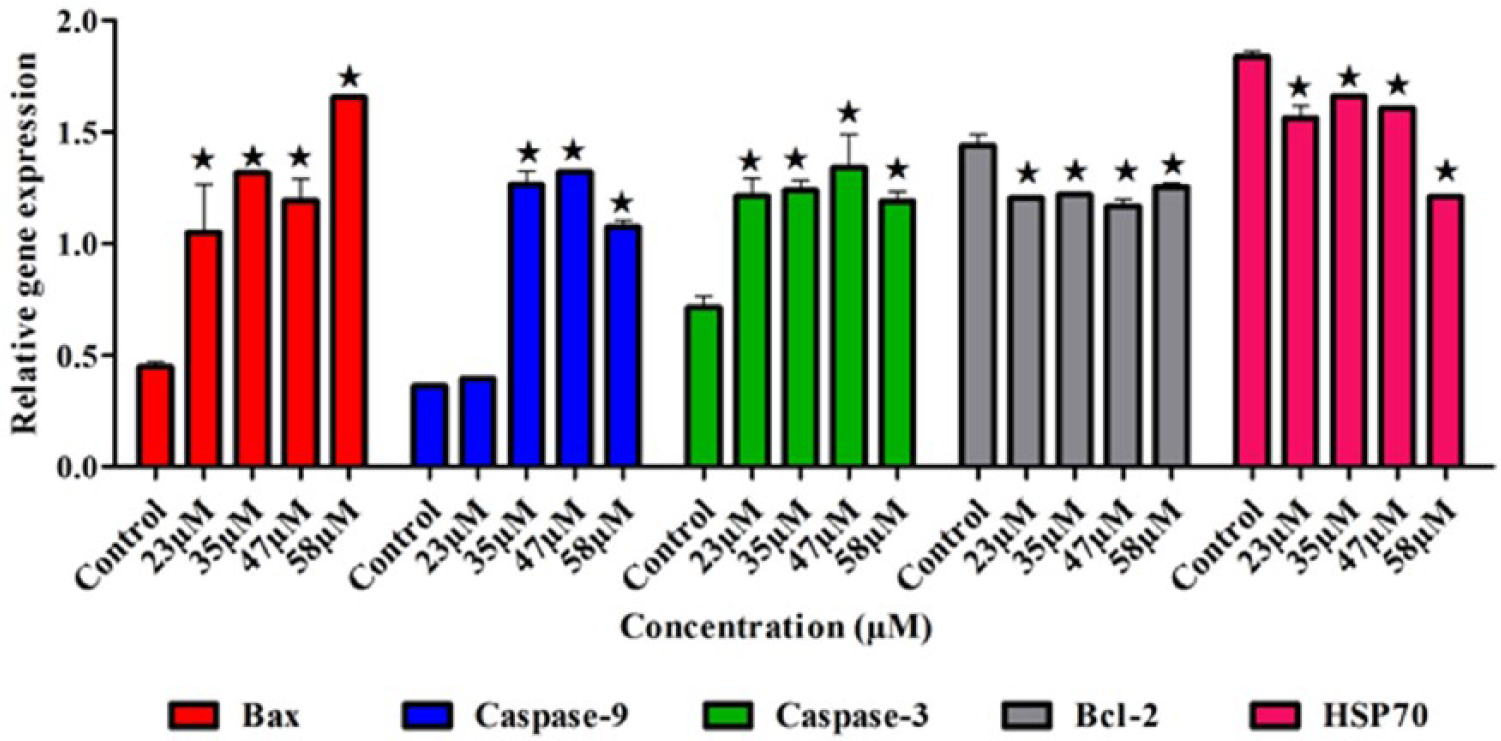
The influence of BM on the expression of the apoptotic and antiapoptotic genes in HL60. The expression of Bax, caspase-3, and caspase-9 genes were upregulated in treated cells. Contrariwise, the expression of antiapoptotic Bcl-2 and HSP70 genes was downregulated. The results were represented as mean ± SD of three replicates. Significant differences were compared to the untreated cells and expressed as *p < 0.05.
The effects of BM on the specific proteins involved in apoptosis
Apoptosis is a biochemical event leading to characteristic cell changes (morphology) and death. This event is controlled by several apoptotic proteins, such as pro- and cleaved caspase-3 and caspase-9, p53, and cytochrome c. The results demonstrate that the pro- and cleaved caspase-3 and caspase-9 and p53 were upregulated at concentrations of 23, 35, 47, and 58 µM of BM. The release of cytochrome c from the mitochondria to cytosol to bind with apoptotic protease activating factor 1 (APAF1) was consolidated. Hence, the abundance of mitochondrial cytochrome c was decreased by the concentrations. However, β-actin protein was used to normalize the results (Figure 8).

(a) Effect of BM at concentrations of 23, 35, 47, and 58 µM on the expression of apoptosis induction proteins; pro- and cleaved caspase-9 and caspase-3, cytochrome c, and p53. (b) Quantitative analysis of the proteins. β-actin was used as a housekeeping protein, and the data were expressed as mean ± SD of three replicates.
BM arrests the HL60 cell cycle at the G0/G1 phase
The effect of BM on the DNA content of HL60 cells was determined through cell-cycle analysis. As shown in Figure 9, the cell cycle was arrested at the G0/G1 phase with a cell accumulation of (56.1% ± 0.77%) at 58 µM, (62.6% ± 0.57%) at 47 µM, (61.1% ± 0.72%) at 35 µM, and (62.0% ± 3.27%) at 23 µM. Moreover, most of the control cells were accumulated at G0/G1 phase (54.45% ± 1.35%).

Flow cytometry analysis shows that BM arrested the cell cycle of HL60 at G0/G1 phase (*p < 0.05). HL60 was treated for 24 h at (b) 23, (c) 35, (d) 47, and (e) 58 µM. This is presented in the histograms where (a) the control cell was also present. (f) Quantification of the different cell phases as G (gap), S (synthesis), and M (mitosis).
Discussion
In recent times, increased scientific attention has been paid to examining how the biological properties of xanthones, including mangostin, can emerge as chemotherapeutic agents. 22 Accordingly, this study has demonstrated that BM has cytotoxicity effect on the HL60. An examination aimed at examining the anticancer mechanism of BM in HL60 in vitro has thus been done.
Empirical underpinning reinforced that apoptosis and the genes that control it have a potential role in the malignant progress. As a result of the accumulation processes, disruptive mutations in apoptosis inducing genes and stimulative mutations in apoptosis inhibitor genes. The above processes can lead to the cancer cell overriding the program of cell death at different stages of carcinogenesis.
Over the last decade, a number of cytotoxic anticancer agents were explored with keen consideration of apoptosis induction properties. 23 That being noted, the cell undergoing apoptosis is characterized by special features. The special features are cell membrane blebbing, chromatin condensation, cell volume reduction, nuclear fragmentation, and apoptotic bodies’ formation. 24 Interestingly, there were the exact features that were observed under fluorescent microscope of treated HL60 after being stained with AO/PI and Hoechst 33342. Howbeit, the images of these dyes facilitated the differentiation between early apoptotic cells, late apoptotic, and necrotic cells.
In quantification study, Annexin V was applied being a well-established technique for apoptosis detection. Also, the Annexin V is a cellular calcium binding that has a high affinity to bind with phosphatidylserine (PS). Accordingly, in this study, the Annexin V was used to differentiate all apoptosis stages after being conjugated with FITC dye.25,26 The results indicated that BM can cause membrane disintegration via translocation of the PS from inner to an outer surface of the cell membrane. 27 Consequently, the populations of early and late apoptotic and even the necrotic cells were increased with BM concentrations. This finding is in agreement with previous studies.
Apoptosis is a complex mechanism with various points of regulation, some of cancer therapies induce their action by manipulating on these adaptor points. Basically, apoptosis is processed via two different tracks, namely, extracellular and intracellular tracks or pathways. Characteristically, the intrinsic pathway is active in response to intracellular DNA damage or exposed to cytotoxic agents. The BCL-2 is a family of proteins that takes a potential role in intrinsic apoptosis regulation. The BCL-2 family is basically made of antiapoptotic proteins such as BCL-2 and BCL-xL as well as apoptotic proteins such as Bax and Bak. In a cell undergoing apoptosis, the apoptotic proteins Bax and Bak become active and Bax translocates from cytosol to mitochondria and conjugates with Bak to form oligomers and facilitate the release of the cytochrome c into cytosol. Bcl-2 can interfere to inhibit this step by binding with Bak and prevent the oligomer formation.
Released cytochrome c binds with Apaf-1 (apoptosis protease activating factor) protein to form the apoptosome, which converts procaspase-9 to the active form, which in turn activates the procaspase-3, and then, a stream of reactions were stimulated to induce apoptosis. 28
The data demonstrated that BM increases the cell membrane permeability of HL60 and cytochrome c release. Subsequently, it activates caspase-9 that cleaved procaspase-3 and activate caspase-3. Hence, BM induces apoptosis via the intrinsic pathway by activating caspase-9, downregulating Bcl-2, and upregulating Bax. These results were similar with the results of BM action on MCF-7 cells obtained by Suvitha et al. 15
The production of ROS is one of the mechanisms by which most chemotherapeutic drugs or radiotherapy kills malignant cells. It influences the mitochondrial membrane permeability that leads to cytochrome c release and subsequently activates the intrinsic apoptotic pathway. 29
The results of this study indicate that BM significantly stimulated the ROS accumulation. However, it supported the overproduction of ROS by downregulating the expression of HSP70 gene that protects the normal and cancerous cells from the oxidative stress. 30 Moreover, ROS declined the expression of HSP70 and subsequently prevented the interaction between HSP70 and CARD domain of Apaf-1. Consequently, Apaf-1 bound with procaspase-9 to form the apoptosome. 31
The finding of this study is consistent with Chang and Yang 12 who reported that γ-mangostin stimulated the ROS production in colorectal adenocarcinoma HT29 cells and consequently prompted the programmed cell death and prevented colon cancer formation. Many anticancer drugs work on the mechanism of cell-cycle arresting and/or a combination of both cell-cycle arrest and cell death mechanism 32 which is considered a potentially powerful strategy for tumor growth control. Our results showed that BM restrained the cell division cycle at the G0/G1 phase and could stop the cell cycle through upregulating the expression of the tumor suppressor gene p53. Shibata et al. 33 reported that α-mangostin has the same influence on the cell cycle of BJMC3879luc2 cells.
Conclusion
The current results provided evidence that BM is an apoptosis inducer agent of the promyelocytic leukemia HL60 cell line as it is found to stimulate the intrinsic apoptosis pathway. Interestingly, the BM upregulates p53 and Bax and suppresses Bcl-2 and HSP70 genes in addition to arresting the cell cycle in vitro. Overall, these findings demonstrated that BM is a promising treatment for leukemia. However, further in vivo investigations are needed to delineate the mechanisms of apoptosis in different animal models. More so, additional studies can be done to examine the activity of BM on another human malignant tumors with specific empirical attention paid to other unknown xanthones with a view to detecting their anticancer activities.
Footnotes
Acknowledgements
The authors thank the University of Malaya, especially Pharmacy Department and Faculty of Medicine, for providing all the necessary facilities to conduct this work. Fatima Abdelmutaal Ahmed Omer, Najihah Binti Mohd Hashim and Syam Mohan conceived and designed the experiments. Fatima Abdelmutaal Ahmed Omer and Landa Zeenelabdin Ali Salim performed the experiments. Fatima Abdelmutaal Ahmed Omer, Firouzeh Dehghan, and Hamed Karimian analyzed the data. Najihah Binti Mohd Hashim, Syam Mohan, and Maizatulakmal Yahayu contributed reagents/materials/analysis tools. Fatima Abdelmutaal Ahmed Omer, Mohamed Yousif Ibrahim, and Najihah Binti Mohd Hashim wrote the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was financially supported by the postgraduate research Institut Pengurusan dan Pemantauan Penyelidikan (IPPP; PG034-2012B) and UMRG (FL001E-13BIO) grants from the University of Malaya and Ministry of Higher Education for the High Impact Research grant (UM-MOHE MC/625/1/HIR/MOHE//SC/09).
