Abstract
Leaf extracts of Pseuderanthemum palatiferum (Nees) Radlk were investigated for their effects on human breast cancer MDA-MB-231 cell growth inhibition. Pseuderanthemum palatiferum (Nees) Radlk extracts were prepared using fresh or dried leaves and extracted by either water or 95% ethanol, respectively. Fresh leaf ethanolic extract was the most toxic to MDA-MB-231 cells measured by 3-(4,5-dimethyl)-2,5-diphenyl tetrazolium bromide assay. Fresh leaf ethanolic extract–treated MDA-MB-231 cell death was stained with propidium iodide and examined under fluorescence microscopy. Cell death was confirmed by annexin V-fluorescein isothiocyanate/propidium iodide and propidium iodide–stained cells employing flow cytometry. The mitochondrial transmembrane potential was disrupted in fresh leaf ethanolic extract–treated MDA-MB-231 cells and the percentage of cells with reduced mitochondrial transmembrane potential increased according to concentrations. Mitochondrial transmembrane potential–driven regulated cell deaths were in the form of both apoptosis and necrosis. Oxidative stress probe, 2′,7′-dichlorodihydrofluorescein diacetate, was used to indicate the redox status. Dichlorofluorescein level was significantly lower at high fresh leaf ethanolic extract concentrations. Total phenolic contents were found in all Pseuderanthemum palatiferum (Nees) Radlk extracts, whereas Ca2+ level in the cytosol increased, indicating Ca2+ overload and endoplasmic reticulum stress involvement with the activation of caspase-3, -8, and -9. In conclusion, fresh leaf ethanolic extract induced human breast cancer MDA-MB-231 programmed cell death via endoplasmic reticulum and oxidative stress by activating both extrinsic and intrinsic signaling pathways.
Keywords
Introduction
Cancer is a major global cause of death with incidence of the disease increasing annually. 1 Chemicals, toxins, radiation, ultraviolet light, viral, parasitic, and bacterial infections, and oxidative and mental stresses all cause cancer. 2 In women, breast and cervical cancer incidences are at the highest ranks. 3 Breast cancer can be caused by dioxins, oxidative stress, and oral contraceptives. Treatment and prevention of cancer prolong and enhance the quality of life, which is a very important issue in an elderly community. Chemotherapeutic drugs, surgery, immunotherapy, and radiotherapy are the conventional treatment methods for breast cancer depending on the stages of disease. However, cancer treatment remains limited due to its side effects and efficacy, which results in thermal and chemical burns of normal skin epithelium and hair cell death due to chemo- and radiotherapy. Another obstacle of chemotherapeutic drugs is the prohibitive high cost for low-income populations. 4
Many researchers have searched for new natural products to inhibit cancer cell growth and proliferation, which do not affect normal cells and hematopoietic cell production. Currently, the prevalent treatment for cancer or folk medicine for other diseases as health promotion involves chemoprevention. Herbal extracts such as natural products with antibiotic and anticancer activities have been shown to inhibit cancer cell growth and proliferation, allowing reduced dosages of chemotherapeutic drugs and ameliorating inherent severe side effects. 5 Mechanisms of these herbal extracts as anticancer agents might be via immune promotion and inhibition of the human telomerase enzyme. 6
The breast cancer cell death–inducing effect by natural extracts of Pseuderanthemum palatiferum (Nees) Radlk (PP) in the family Acanthaceae was never investigated before. Actually, PP extracts contain activities to treat diarrhea, hypertension, rheumatoid arthritis, and pharyngitis. 7 In Thailand, PP administered as a tea made from fresh leaves or even eating the leaves is used to treat several symptoms and diseases including stomachache, diabetes mellitus, and nephritis. PP leaves have been reported to consist of phytochemicals including flavonoids; beta-sitosterol, stigmasterol, kaempferol, apigenin, and compounds belonging to triterpenoid; saponins and phytol; and salicylic and malmitic acid groups.8,9 These active compounds inhibit cancer cell growth and proliferation but the mechanism remains unclear.
Cell death can be divided into three major classes as apoptosis, autophagic cell death, and necroptosis, 10 all of which are regulated or programmed cell death. The aims of this study were to investigate whether PP extracts inhibited human breast cancer MDA-MB-231 cell growth, mode of cell death, and mechanism of cell death using an in vitro model. Fresh or dry leaf extracts were prepared using different solvents (ethanol or water); the fresh leaf ethanolic extract (FEE) was used with the most potency for inhibiting MDA-MB-231 cell growth and inducing MDA-MB-231 cell death, that is, with the least concentration at IC50 to induce cancer cell death at various time intervals of incubation. The mode of cell death was regulated by programmed cell death with the phenotypes of both apoptosis and necrosis through the endoplasmic reticulum (ER) and oxidative stress via both death receptor and mitochondria-mediated signaling pathways.
Materials and methods
Reagents
Dulbecco’s modified Eagle medium (DMEM), Roswell Park Memorial Institute (RPMI) 1640 medium, Dulbecco’s modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F-12), fetal bovine serum (FBS), horse serum, streptomycin, and penicillin G sodium were obtained from Gibco BRL (Thermo Fisher Scientific Inc., Waltham, MA, USA). Hydrocortisone, insulin, epidermal growth factor (EGF), dimethyl sulfoxide (DMSO), 3-(4,5-dimethyl)-2,5-diphenyl tetrazolium bromide (MTT), propidium iodide (PI), 3,3′-dihexyloxacarbocyanine iodide (DiOC6), and 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) were obtained from Sigma-Aldrich (St Louis, MO, USA). Annexin V-FLUOS Kit was obtained from Roche (Indianapolis, IN, USA). Dihydroethidium (DHE), Rhod-2 AM, Fluo-3 AM, and the substrates of caspase-9 (LEHD-para-nitroaniline, LEHD-p-NA), caspase-8 (IETD-p-NA), and caspase-3 (DEVD-p-NA) were obtained from Invitrogen (Thermo Fisher Scientific Inc.). Folin–Ciocalteu (F-C) reagent was obtained from Fluka (Milwaukee, WI, USA). Cholera Toxin, Vibrio cholerae Inaba 569B was obtained from United States Biological, Salem, MA, USA.
Preparation of plant extracts
The fresh leaves were harvested in August 2016 from a farm operated by Prolac (Thailand) Company Ltd. The plant was identified by a botanist at the Queen Sirikit Botanical Garden with voucher specimen no. WP2615. Fresh leaves were extracted with water to obtain fresh water extract (FWE) or with 95% ethanol to obtain FEE. Dried leaves were extracted with water and 95% ethanol to obtain dried water extract (DWE) and dried ethanolic extract (DEE), respectively. All extracts were separately lyophilized and evaporated. Percentage yield of each extract is shown in Table 1.
Percent yield of each extract of Pseuderanthemum palatiferum (PP).

Each extract was obtained using the following procedure adapted from the protocol of Sittisart and Chitsomboon. 11 Briefly, fresh or air-dried leaves (300 g for each sample) were ground and then dried in a hot air oven at 50°C. The extraction was performed with 1 L deionized distilled (DI) water for 24 h at room temperature and filtered through Whatman filter paper No. 1. The water fractions were dried with a freeze-dryer (lyophilization) to obtain FWE and DWE, respectively. The ground leaves were macerated and extracted with water and subsequently with 95% ethanol (1 L) successively at room temperature and filtered through Whatman filter paper No. 1. The extraction process was repeated for 4 days until the percolated solvent was clear. The ethanolic fractions were evaporated under vacuum to obtain FEE and DEE, respectively; then the extracts were collected and stored at −20°C, before further experiments.
Cell culture
Human invasive triple-negative breast cancer MDA-MB-231 cells were obtained from Prof. Prachya Kongtawelert at Excellence Center of Tissue Engineering and Stem Cells, Department of Biochemistry, Faculty of Medicine, Chiang Mai University. MDA-MB-231 cells were cultured and used in the 3rd–6th passages in DMEM supplemented with 10% FBS, 25 mM sodium bicarbonate (NaHCO3), 20 mM HEPES buffer, 100 U/mL penicillin G, and 100 µg/mL streptomycin at 37°C and 5% CO2. The human normal mammary epithelial MCF10A cells were obtained from American Type Culture Collection (ATCC), Manassas, VA, USA. MCF10A cells were cultured and used in the 3rd–6th passages in DMEM/F-12 medium supplemented with 5% horse serum, 20 ng/mL EGF, 0.5 mg/mL hydrocortisone, 100 ng/mL cholera toxin, 10 µg/mL insulin, and 2.5 mL of penicillin–streptomycin (10,000 U/mL) at 37°C and 5% CO2.
Isolation of peripheral blood mononuclear cells
Peripheral blood mononuclear cell (PBMC) isolation was from buffy coats, which were isolated from whole blood at the Blood Bank of Maharaj Nakorn Chiang Mai Hospital, Chiang Mai. PBMCs were obtained from volunteers at Blood Bank Unit, with the informed consent signed, whereas the approval was available from the Ethic Committee of the Maharaj Nakorn Chiang Mai Hospital, Faculty of Medicine, Chiang Mai University.
PBMCs were isolated from heparinized blood by density gradient centrifugation using the standard protocol 12 with Ficoll-Hypaque reagent. The cells were cultured in 10% FBS in RPMI-1640 medium supplemented with penicillin G (100 U/mL) and streptomycin (100 μg/mL) at 37°C in a humidified atmosphere containing 5% CO2. 13
Cytotoxicity assay
Human breast cancer MDA-MB-231 cells were cultured in DMEM until 80% confluent and then plated in a 96-well plate at a concentration of 2 × 103 cells/well. Then the cells were treated with or without the extracts at various concentrations (100–500 µg/mL) for 24, 48, and 72 h. MTT dye at a stock concentration of 5 mg/mL was added and incubated for 4 h. The formazan crystal was developed and the media were removed gently before DMSO was added to dissolve the crystal. Next, absorbance was measured at a wavelength of 540 nm and compared to 630 nm by a spectrophotometric microplate reader (BioTek, Winooski, VT, USA). 14
Percentage cell viability was calculated from the equation and inhibitory growth curve and these ICs were used for further experiments of each extract. Cell viability was determined by calculation as follows. The cancer cells were treated with various doses (100–500 µg/mL) of extracts and determined for percentage of cell viability according to the equation given below. The absorbance of the extract at various inhibitory concentrations such as 10%, 20%, and 50% was calculated, converted using the equation, and then plotted as graphs; then the concentrations at each point were recalculated from three independent experiments performed in triplicate and the final stock concentrations of the extracts were recalculated and added to obtain various final concentrations to obtain at IC10 means 90% viability, IC20 means 80% viability, and IC50 means 50% viability; further experiments were then performed with the amount of cells as required.
Inhibitory concentration (IC) values at IC10, IC20, and IC50 were determined from the equation and inhibitory growth curve and these ICs were used for further experiments of each extract. Percentage cell viability was determined as follows

For the normal human cells, human mammary epithelial cell MCF10A and normal human PBMCs were cultured in DMEM/F12 and RPMI 1640 media, respectively. The cells were plated in a 96-well plate at a concentration of 5 × 103 and 1 × 105 cells/well, respectively, and then treated with or without the most effective (lowest IC50) extract at various concentrations for 24 h, and the cell viability was determined using MTT assay.
The cancer cells were treated with various doses (100–500 µg/mL) of extracts and determined for cell viability according to the given equation. The absorbance of the extract at various inhibitory concentrations such as 10%, 20%, and 50% was calculated using the plotted graphs by the equation, after which the concentrations at each point were recalculated from three independent experiments performed in triplicate, and the final stock concentration of the extracts was recalculated and added to obtain various final concentrations at IC10 means viable 90%, IC20 means 80% viability, and IC50 means 50% viability for further incubation experiments.
Mode of cell death determination
Human breast cancer MDA-MB-231 cells were plated in a 24-well plate at a concentration of 1 × 104 cells/well, treated with the most effective (lowest IC50) extract at 24-h incubation, and then stained with PI. Cell morphology was examined under fluorescence microscopy (Olympus, Tokyo, Japan). 15 Positive condensed nuclei and fragmented bodies were demonstrated under a fluorescence microscope.
Determination of cell death mode using annexin V-fluorescein isothiocyanate/propidium iodide and flow cytometry technique
For an evidence-based rather than a subjective preference method to quantify regulated cell death, the most effective extract-treated cells were stained with annexin V-fluorescein isothiocyanate (FITC)/PI and analyzed by a flow cytometer (Becton Dickinson, Frankin Lakes, NJ, USA). 16 The conditions of varying concentrations (various ICs) and fixed time of incubation period at 24 h were employed.
Determination of mitochondrial transmembrane potential disruption
MDA-MB-231 cells were plated in a 24-well plate at a concentration of 1.5 × 105 cells/well and then incubated with the most effective extract at various concentrations (IC10, IC20, and IC50) for 24 h. Reduction of mitochondrial transmembrane potential (MTP) was measured by staining with DiOC6 fluorescent dye using a flow cytometer and analyzed by CellQuest software (Becton Dickinson). 17
Measurement of reactive oxygen species
MDA-MB-31 cells were plated and cultured overnight in a 96-well plate (final concentration 5 × 103 cells/well) and pretreated with DCFH-DA at 20 µM (final concentration) for 30 min and then with the most effective (lowest IC50) extract for various incubation times. After 2, 4, and 12 h of incubation with the most effective extract, the cells were washed with phosphate-buffered saline (PBS) twice. Reactive oxygen species (ROS) in the form of peroxide radicals were measured using a fluorescence plate reader at 485 and 530 nm for excitation and emission wavelengths, respectively (BioTek). 18
Superoxide anion radical generation was also investigated similar to that of DCFH-DA but using DHE fluorescent dye instead. Briefly, the cancer cells were incubated with DHE to the final concentration of 0.1 µM and incubated for 30 min. Then the cells were washed twice with PBS and the most effective extract was added at IC50 and incubated for 2, 4, and 12 h. The fluorescence was determined by a fluorescence plate reader at 535 nm excitation and 635 nm emission wavelengths (BioTek). 19
Determination of Ca2+ ions in mitochondrial or cytosolic compartment
MDA-MB-231 cells were plated in 24-well plates at a concentration of 1.5 × 105 cells/well and treated with the most effective extract; then Ca2+ levels were determined using Rhod-2 AM (40 nM) and Fluo-3 AM (4 µM) fluorescent dyes as specific markers of Ca2+ in mitochondria and cytoplasm, respectively, incubated for 15 min. Fluorescence intensity was determined by flow cytometry technique. 20
Determination of caspase activities
MDA-MB-231 cells were plated and cultured overnight in a six-well plate (final concentration 1 × 106 cells/well) and then treated with the most effective extract at IC10, IC20, and IC50. Activities of caspase-3, -8, and -9 were measured using the specific substrates DEVD-p-NA, IETD-p-NA, and LEHD-p-NA, respectively. Equal amounts of protein from the cells were incubated with the corresponding tetrapeptide substrate for 2 h and absorbance was measured using a spectrophotometric microplate reader at a wavelength of 405 nm (BioTek). 21
Measurement of total phenolic contents
Four extracts of PP were investigated for total phenolic contents using gallic acid as a standard control. Briefly, 75 µL of deionized distilled (DI) water was added to each of the 96 wells followed by 25 µL of either sample or standard and 25 µL of Folin–Ciocalteu reagent (diluted 1:1 (v/v) with deionized-distilled water (DDW)). The solutions were mixed and left for 6 min. Then, 100 µL of 75 mg/mL sodium carbonate (Na2CO3) was added to each well. The plate was incubated in the dark for 90 min after mixing. Absorbance was measured with a spectrophotometric microplate reader at 765 nm (BioTek). Gallic acid was used as a standard at concentrations in the range of 12.5–200 µg/mL. 22
Statistical analysis
All data were expressed as mean ± standard deviation (S.D.) of the mean from three independent experiments performed in triplicate with the same results. Biochemical data were assessed by one-way analysis of variance (ANOVA) and Dunnett’s post hoc test with a p value less than 0.05 considered significant. All analyses were conducted using commercially available SPSS software (version 22.0).
Results
Cytotoxic effects on human breast cancer MDA-MB-231 cells by PP leaf extracts
From the results of MTT assay, DWE at 100–500 µg/mL and FWE at 100–300 µg/mL have no cytotoxic effect on MDA-MB-231 cell growth or viability, except FWE at 72 h, where the cell viability was reduced to 70%–75% at 300 and 500 µg/mL (p < 0.05), respectively (Supplement Figure 1(a) and (b)). FWE caused the cells to proliferate less or increase cell death significantly more than that of control at 300 and 500 µg/mL when incubated for 3 days (p < 0.05).
For the term “the most effective extract” means that the extract which, contained the most toxic activity against MDA-MB-231 cancer cells viability and the one that provided the lowest IC50 with the percentage of dead cells occurs 50% or the least concentration that provided equal 50% cell dead population. The most effective extract was FEE, with lowest IC50 value (Figure 1(a)–(c) and Supplement Table 1).

Cytotoxicity of the ethanolic extracts from dried and fresh leaves of PP on human breast cancer MDA-MB-231 cells. Percent cell viability after MDA-MB-231 cells were incubated with dried leaf ethanolic extract at 24, 48, and 72 h (DEE; (a), (b), (c), blue line) and fresh ethanolic extract for 24, 48, and 72 h (FEE; (a), (b), (c), red lines) by MTT assay.
FWE and DWE were not toxic to MDA-MB-231 cells with the percent cell viability not reaching 50% at 24 h to the concentration up to 500 µg/mL (Supplement Figure 1(a) and (b)). Fresh leaf extract was more effective than dried leaf extracts toward MDA-MB-231 cancer cells with ethanolic extracts being more effective than the water ones (Figure 1(a) and (c)).
The factors determining the cell viability include the following: the metabolic change of dead cells leading to the inactive enzyme reaction in mitochondria and the mode and mechanism(s) of cell death, which were then investigated. FEE was more effective than DEE at >200 µg/mL toward MDA-MB-231 cancer cell death induction (Figure 1(a)–(c)). But FEE caused cell proliferation and growth at 100 µg/mL when incubated for 24 and 48 h (Figure 1(a) and (b)).
FWE and DWE were not toxic to MDA-MB-21 cells with 100% cell viability at 24 h, which might be due to the growth inducer or metabolic effector on mitochondrial succinate dehydrogenase or both to change the MTT dye to be formazan crystal and dissolve in DMSO to obtain violet color. The absorbance of the dye was measured by a spectrophotometric microplate reader. FWE and DWE were not toxic to MDA-MB-231 cells with the percent cell viability not reaching 50% at 24 h to the concentration up to 500 µg/mL (Supplement Figure 1(a) and (b)). Fresh leaf extract was more effective than dried leaf extracts toward MDA-MB-231 cancer cells with ethanolic extracts being more effective than the water ones at 300 and 500 µg/mL (Figure 1(a) and (b)).
Comparing cell viabilities, FEE was more effective than DEE on cytotoxic induction at the concentrations of 300 and 500 µg/mL. Hence, the most cytotoxic and effective extract regarding MDA-MB-231 cell growth inhibition was FEE at 24 h (Figure 1), considering the least value of IC50 concentration. At 24 h of incubation, FEE was the most toxic in a dose-dependent manner to MDA-MB-231 cells with IC10, IC20, and IC50 values at 199 ± 7.21, 216 ± 6.84, and 260 ± 7.04 µg/mL to reduce the cell viability, respectively, in a concentration-dependent manner, but not in a time-dependent manner (Supplement Table 1).
DEE was less toxic to MDA-MB-231 cells, with less ability to induce cell death at 100 and 200 µg/mL and less than 50% at 500 µg/mL; therefore, DEE caused the cells to be more viable than FEE at 100 and 200 µg/mL and at the same incubation time. At 300 and 500 µg/mL, FEE was more toxic than DEE and reached IC50, whereas at 100 and 200 µg/mL it was less toxic. Linear graphs of PP extracts (FEE) showed the percentage of viability at the same inhibition potential on MDA-MB-231 cancer cell growth or proliferation curves at all three incubation times, namely, 24, 48, and 72 h; hence, the inhibitory concentrations (IC10, IC20, and IC50) and the time of 24 h were selected for further experiments.
The IC50 at 24 h of FEE incubation was the objective or aim to be interpreted and compared as the most cytotoxicity condition of the extracts toward the breast cancer MDA-MB-231 cells, and the IC50 of FEE was lowest than the other three extracts, therefore FEE was selected as shown in Figure 1(a)–(c). The inhibitory concentrations of FEE at 199 (IC10), 216 (IC20), or 260 (IC50) µg/mL at 24 h were used to compare with those of others to study the concentration influence. The FEE toxic dose was chosen to be at 260 µg/mL (IC50) for further studying the mode and mechanism(s) of cell death or the concentrations at 199, 216, and 260 µg/mL at 24 h to obtain the concentration effects of FEE.
Even though the IC50 value was very high >150 µg/mL, which was hard to apply or translate for clinical use, according to the National Institute of Cancer (NIC) considering <20 µg/mL as the good potential. However, the result of cytotoxicity on normal human cells, human mammary epithelial cells (MCF10A; IC50 values at 306 ± 29.66 µg/mL), and normal human PBMCs (IC50 values at 319 ± 12.12 µg/mL) were less toxic (Supplement Figure 2(a) and (b)), respectively, when compared to that of the MDA-MB-231 cells, that is, 260 µg/mL. Since MDA-MB-231 cells were invasive and aggressive, the IC50 value was therefore very high, which also unavoidably affected the normal cell viability.
The toxic effect of FEE was less toward normal mammary epithelial MCF10A cells (306 µg/mL); however, for the conventional chemotherapeutic drugs, such as doxorubicin (DOX), it was toxic to MDA-MB-231 cells in a dose–response manner with an IC50 value of 9.15 µM (Supplement Figure 3). The combined effect of FEE plus conventional chemotherapeutic drug(s) may be useful to lessen the potential dosage of chemo drugs and attenuate the adverse side effects; however, further study on this issue in vivo is still required.
Since the IC50 values for normal human mammary epithelial MCF10A cells and PBMCs were higher than that of MDA-MB-231 cells, we used the dose at 260 (IC50) µg/mL for further studying the mode and mechanism(s) of cell death, which should allow a significant number of cell populations for the following experiments: flow cytometry, microplate reader, or fluorescence plate reader. However, this is an in vitro model and it should mimic human beings when using an in vivo model.
The quality control between batches of extracts could be performed by the polyphenolic contents and the biological activity of MTT assay, which showed the same patterns and values.
Morphology of MDA-MB-231 cells after death induction with FEE for 24 h
When MDA-MB-231 cells were treated with FEE at 199 (IC10), 216 (IC20), and 260 (IC50) µg/mL for 24 h and stained with PI, the morphologies of apoptotic cells appeared as condensed nuclei with fragmented or apoptotic bodies 15 (indicated by arrows in Figure 2).

Cell morphology of FEE-treated MDA-MB-231 apoptotic cells after 24-h treatment. The typical morphology of MDA-MB-231 cells after treatment with FEE for 24 h exhibits condensed nuclei and apoptotic bodies (arrow), a representative result selected from three independent experiments.
Apoptotic or regulated cell death induction on MDA-MB-231 cells by FEE
The biochemical character of apoptotic cells showed externalization of phosphatidylserine (PS) to the outer layer from the inner layer. This was determined using annexin V (a lipoprotein that specifically binds to PS) tagged with FITC. 23 Therefore, FEE-treated MDA-MB-231 cancer cells for 24 h were stained with annexin V-FITC/PI employing the flow cytometry technique.
Dot plot analysis of FEE-treated MDA-MB-231 cells showed regulated cell death with the cells stained with both annexin V-FITC/PI and with PI alone; it was found to be in the late apoptosis (upper right quadrant) and necrosis (upper left quadrant) (Figure 3(a)). The mean percentage of late apoptosis plus necrosis was higher in a dose-dependent manner when compared to those of control and IC10 shown as bar graphs indicating regulated cell death in the form of secondary necrosis (resulting from late apoptosis or secondary necrotic cell death) and necrotic cell death, which was regulated (or programmed) cell death (Figure 3(b)). The MTP reduction or disruption was able to cause regulated cell necrosis, 24 and hence MTP was further investigated to support this hypothesis.
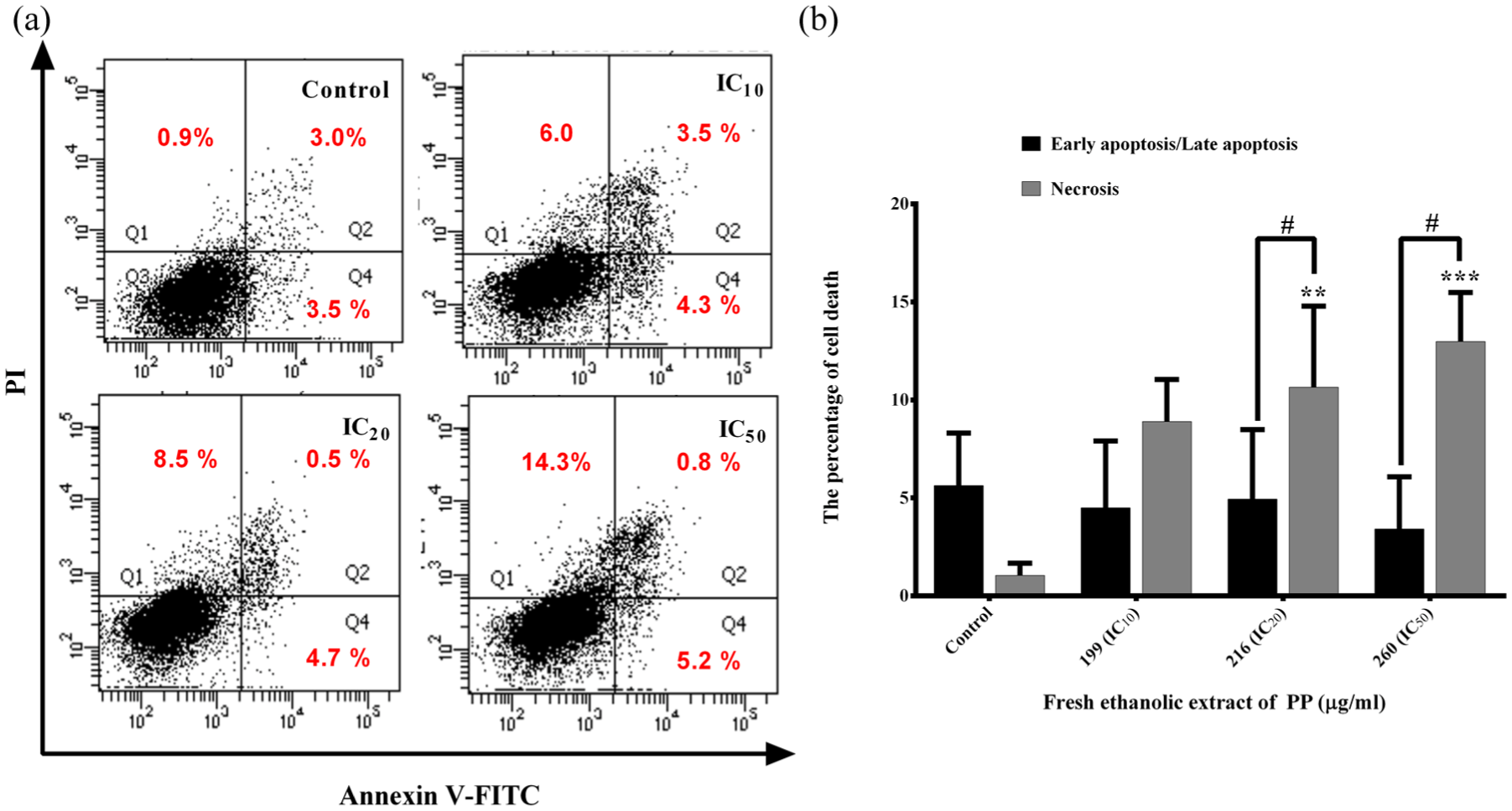
Regulated cell death induction in FEE-treated MDA-MB-231 cells determined by annexin V-FLUOS Kit employing flow cytometry. From dot plot analysis as a representative of three independent experiments, it is demonstrated that the cells underwent necrosis (left upper quadrant) (a) and early/late apoptosis (right lower and upper quadrants). Bar graphs represented early and late apoptotic cell population stained with annexin V-FITC and annexin V-FITC/PI (black bars), and necrosis (PI, gray bars) (both are representative of regulated or programmed cell death) (b) represented as mean ± S.D.
Disruption of MTP
The outer membrane permeability transition allows the mitochondrial signaling pathway for both the voltage-dependent anion channel (VDAC) and Bax/Bax, Bax/Bak, or Bak/Bak homodimer or heterodimer forming channels to open at both outer and inner mitochondrial membranes. 24 Mitochondrial membranous proteins such as cytochrome c, HtrA2, and Endo G are released from the intermembranous mitochondrial space 25 and mitochondrial membrane permeability transition (MMPT) is disrupted in FEE-treated MDA-MB-231 cells.
The percentages of cells with loss of MTP increased in a dose–response manner as shown in Figure 4. MTP-driven necrosis or apoptosis is a form of regulated cell death initiated by specific perturbation of intracellular milieu such as oxidative stress and cytosolic Ca2+ overload. 26 Therefore, ROS production and Ca2+ levels in the treated cells were further determined.

Reduction of mitochondrial transmembrane potential (MTP). After treatment with the fresh ethanolic extract (FEE), the mitochondrial membrane potential was disrupted, which was measured using fluorescence dye DiOC6 staining and flow cytometer. Bar graphs represent the percentage of breast cancer MDA-MB-231 cells with reduced MTP. Data in each bar graph are represented as mean ± S.D.
Reduction of ROS in peroxide radicals
ROS cause oxidative stress when their levels are higher than those of antioxidants or an imbalance between these two factors occurs. Levels of ROS have been influenced by many factors, such as the glutathione (GSH) levels in the cells, redox state in ER, the amount of aggregated or unfolded proteins, mitochondrial membrane integrity, permeability, and potential, antioxidant levels, and types/quantity of phenotype contained in each type of extract (flavonoids, catechins, terpenoids, and so on). ROS are generated in normal metabolic pathways, such as electron transport chains, adenosine triphosphate (ATP) production, and mitochondrial respiration in mitochondria. 27 ROS induce both apoptosis and necrosis in cancer cells.28,29 When FEE-treated cells were incubated with DCFH-DA or DHE to measure the amount of peroxide or superoxide anion radicals, respectively, peroxide radicals decreased significantly (Figure 5), whereas the level of superoxide anion radicals did not alter (Supplement Figure 4).

Reactive oxygen species production in FEE-treated MDA-MB-231 cells. Fluorescence redox probe, DCFH-DA, was used to stain MDA-MB-231 cells, and FEE treatment for 2, 4, and 12 h was performed in oxidized condition after the probe were changed to be DCF with fluorescence emission from the cells indicating ROS generation. From the probes, it emitted fluorescence from DCF (peroxide radicals). Data in each bar graph are represented as mean ± S.D.
Increased Ca2+ level in cytosol of FEE-treated MDA-MB-231 cells
Ca2+ acts as a second messenger and plays a significant role in both physiological (such as muscle contraction) and pathophysiological (such as apoptotic and necroptotic cell death induction) pathways.30,31 For Ca2+ level in FEE-treated breast cancer MDA-MB-231 cells, Rhod-2 AM fluorescence intensity did not change, indicating that mitochondrial Ca2+ level did not significantly alter (Figure 6). But the Fluo-3 AM fluorescence intensity was greatly enhanced compared to control (as shown in Figure 7(a) and (b)), indicating a significant increase of Ca2+ influx in the cytoplasm, which is presumably mediated by PS exposure and followed by mixed lineage kinase domain-like pseudokinase (MLKL)-mediated channel formation at the cell membrane allowing loss of cell membrane integrity 32 or other organelles such as mitochondria and ER. This indicates the overload of cytoplasmic Ca2+ and ER stress–mediated regulated cell death at the same incubation time. Necrosis and apoptosis driven by MTP occurred as evidenced in the annexin V-FITC/PI-positive cells and PI-positive cells, which were FEE-treated MDA-MB-231 cells for 24 h.

No alteration of mitochondrial Ca2+ levels was observed in MDA-MB-231 cells after incubation with FEE for 24 h. Ca2+ levels were measured by the fluorescence dye Rhod-2 AM. Histograms of Rhod-2 AM intensity of the FEE-treated MDA-MB-231 cells after being treated with FEE at 199 (IC10, (a)), 216 (IC20, (b)), and 260 (IC50, (c)) µg/mL. Rhod-2 fluorescence intensities in FEE-treated MDA-MB-231 cells were compared with that of untreated cells shown in the bar graphs (d). The fluoresced cells were determined using a flow cytometer under PE settings. Data in each bar are represented as mean ± S.D.

Increased Ca2+ levels in the cytoplasmic compartment of FEE-treated MDA-MB-231 cells. Increased levels of Ca2+ in cytosol were measured by the fluorescence dye Fluo-3 AM. Histograms of Ca2+ levels in the cytoplasm after being treated with FEE at 199 (IC10, (a)), 216 (IC20, (b)), and 260 (IC50, (c)) µg/mL by employing the Fluo-3 AM fluorescence emission, respectively. Fluo-3 AM fluorescence intensities in FEE-treated MDA-MB-231 cells were compared with that of untreated cells shown in the bar graphs (d). The fluoresced cells were determined using a flow cytometer under FITC settings. Data in each bar are represented as mean ± S.D.
Cytosolic Ca2+ levels are demonstrated as a specific parameter for cytosolic Ca2+ overload and also indicate MTP-driven regulated cell death (necrosis and late apoptosis) and also via ER-stressed mechanistic pathway.
Activation of caspase-3, -8, and -9 activities
In cells undergoing regulated apoptotic cell death, the cysteinyl aspartate–specific proteases (caspases) are stimulated and reactions are activated. Caspase-3 functions to proteolyze structural cytoskeletal proteins such as actin, fodrin, and laminin leading the cells to apoptosis with typical morphology,33,34 whereas caspase-8 and -9 play vital roles in the initiating steps of extrinsic (death receptor) and intrinsic (mitochondrial) pathways, respectively. 35 The FEE-treated MDA-MB-231 cells demonstrated significant enhancements in all three caspases, which were caspase-3, -8, and -9, especially at IC50 as shown in Figure 8(a)–(c), respectively. Hence, this led to the hypothesis that the FEE induced MDA-MB-231 cells to undergo apoptosis and finally secondary necrosis, both of which are regulated cell death.

Activation of caspase-3, -8, and -9 in FEE-treated MDA-MB-231 cells. The cancer cells were treated with FEE for 24 h and then incubated with specific substrates of each caspase: (a) DEVD-p-NA, (b) IETD-p-NA, and (c) LEHD-p-NA, respectively. The increased caspase-3, -8, and -9 activities contributed to the effector caspase-3, death receptor–mediated, and mitochondria-mediated apoptotic death pathways as the data in each bar are represented as mean ± S.D.
Total phenolic contents
Natural products usually consist of phenolic compounds, flavonoids, alkaloids, terpenoids, and catechins, which contain anticancer, anti-inflammatory, and antioxidant activities. 36 Compared with the standard curve using gallic acid as control (Supplement Figure 5), PP extracts consisted of total phenolic contents as follows: DWE > FWE > DEE > FEE from equal amount of each extract (Figure 9). The polyphenolic contents were used as control for the batch to batch of each extract, whereas MTT assay was obtained for cytotoxicity as the biological activity control. FEE exhibited the most effective cytotoxicity and regulated cell death induction on MDA-MB-231 cells, in contrast to the least quantity of phenolic compounds, which require further identification and characterization of new responsible active compound(s).
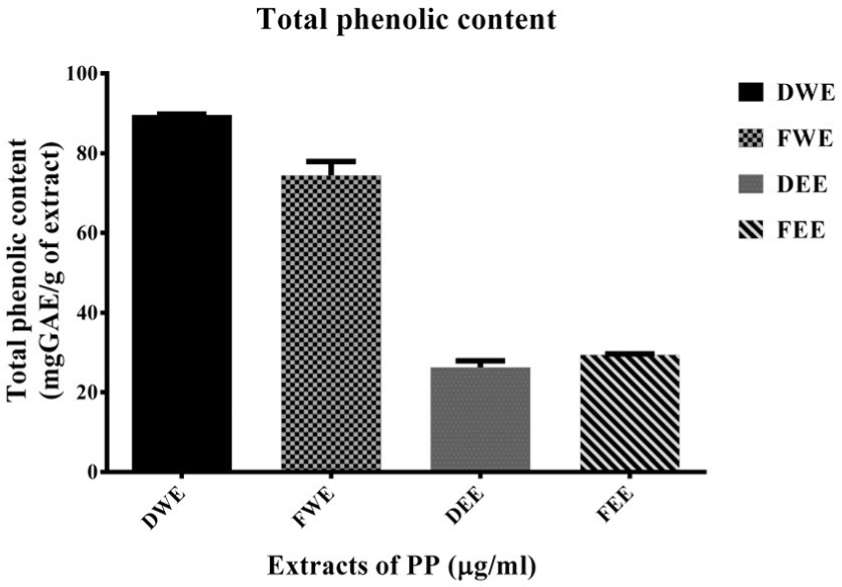
Total phenolic contents in various extracts of PP. The levels of phenolic compounds found in the dried water extract (DWE), fresh water extract (FWE), dried ethanolic extract (DEE), and fresh ethanolic extract (FEE), represented as bar graphs of mean ± S.D.
Discussion
FEE inhibited human breast cancer MDA-MB-231 cell growth and proliferation via regulated cell death or apoptosis/necrosis. Results proved that FEE of PP was the most effective compared to aqueous extracts (DWE and FWE). Fresh leaf extracts showed higher inhibition of cell proliferation/viability than the dried leaf ones. The polarity and types of solvents used for herbal extraction are significant to obtain the optimal quantity and quality of active ingredients for anticancer activity, which is called structure–activity relationship (SAR). Human breast cancer MDA-MB-231 cell line was selected as a model to investigate cell growth inhibition and cell death mechanism. Since human breast cancer is the highest incidence of cancer in woman, FEE of PP offers a high potential for breast cancer treatment as evidenced in an in vitro model.
Other biological effects of PP extracts include the following: for example, in 2016, inhibition of inflammatory cytokine production in the murine-derived myeloid cell line RAW264.7 induced by lipopolysaccharide (LPS). 37 Sittisart and Chitsomboon 11 reported that the water extract (WE) of PP has higher phenolic and flavonoid levels with increased the 2,2-diphenyl-1-picryl-hydrazyl hydrate (DPPH) radical scavenging activity. The EE shows higher reducing power and enhances the decrease of oxidative stress. Both aqueous and ethanolic extracts from PP suppress inflammation. 11
Khonsung et al. 38 reported that the water extract of PP reduces both mean arterial blood pressure and pulse rate of rodents. The extract possesses vasodilatation effects via suppression of nitric oxide synthase (NOS) expression, leading to decreased nitric oxide (NO), an oxidant/vasodilator. Mai et al. 39 purified and characterized two new lignans in the root of PP as palatiferin A and palatiferin B, five known triterpenes, such as epifriedelanol, lupeol, lupenone, betulin, and pomolic acid, and a dipeptide asperglaucide.
Several factors influence ROS generation including mitochondrial membrane potential and intracellular antioxidant levels such as GSH. These include multiple factors since all four extracts have phenolic components that are antioxidants. The parameters influence ROS generation or quantity, which are GSH levels, redox state in ER, the amount of unfolded/aggregated proteins/nitroso proteins, mitochondrial membrane integrity, permeability and membrane potential, and antioxidant levels (types, quantity of polyphenol) contained in each extract and active compounds such as catechins, flavonoids, terpenoids, and alkaloids found in the extract. Water extracts of dried and fresh leaves were less toxic to MDA-MB-231 cells up to 500 µg/mL, whereas the percentage of viable cells at 100 µg/mL at 24 h was more than 100%. This might be due to the redox state contributed by such factors inside the cells or its milieu.
At low concentrations of the total phenolic compound, it might act as an antioxidant but at high concentrations, it might be pro-oxidant, such as curcumin. The cell viability more than 100% at 24 h by 100 µg/mL FEE could be interpreted as FEE as a proliferator or growth factor and/or enzymatic regulator, which can change the MTT dye solution to be formazan crystal, to stimulate MDA-MB-231 cell growth, proliferation, and cell metabolic activity. At this dose and time, FEE acts as an antioxidant but at levels higher than this FEE acts as a pro-oxidant and cell death inducer.
However, the phenolic contents might not be considered to be good factors to represent oxidative stress or antioxidant status. But the measurement of the redox activity instead by using 2,2-diphenyl-1-picryl-hydrazyl-hydrate (DPPH) free radical scavenging method and antioxidant assay (2,2′-azino-bis-(3-ethylbenzthiazoline-6-sulfonic acid)) to generate a radical cation; 2,2′-azinobis-(3-ethylbenzthiazoline-6-sulphonate) (ABTS+) would be more precise to represent for the redox status in the cellular cytosol. Hence, the extract from PP might influence and lead the cells to undergo regulated cell death: apoptosis and necrosis.
The level of ROS in the form of peroxide radical production after FEE treatment for 4 and 12 h was altered significantly, and the cells were incubated with the probes (DCFH-DA) first and then FEE was added for 30 min. This might be due to the changes of ROS type (superoxide anion radicals to be peroxide radicals in the mitochondria) since the cells were incubated with FEE and mitochondrial dysfunction might occur during the process of apoptotic cell death via intrinsic (mitochondrial) pathway and electron transport chain disruption. Hence, the cellular treatment with the FEE for 4 and 12 h has an impact on redox status (peroxide rather than superoxide anion radical levels) significantly.
The Ca2+ influx or efflux from other organelles might also play roles. Furthermore, no previous findings were reported on the inhibitory effects of PP on human breast cancer cell growth and cell death mechanism(s) including the biological activity of this plant leaf ethanolic extract. The examination of FEE from PP was the first to discover its anticancer property and ability to activate regulated cell death via either apoptotic or/and necrotic cell death signaling pathways, which are regulated or programmed. The role of cytosolic Ca2+ level in regulated cell death induction in human breast cancer MDA-MB-231 cell line by FEE might be due to several factors that influence the increase of Ca2+ level not in a dose-dependent manner. This might be caused by various parameters influencing the cytoplasmic Ca2+ level, such as the status of cell membrane integrity allowing Ca2+ level changing due to influx or efflux, or Ca2+ level in ER and/or mitochondria due to changes in membrane potential or pumps/transporters in such membranes. The cellular milieu might also play various pivotal roles, such as the stimulators of cell death, intensity of triggers, and severity or concentrations of the inducers of regulated cell death.
Even though the FEE IC50 value was still too high to be used clinically, it might be the first step in developing anticancer drugs by preparing the effective extract and using fractionation with bioassay guide for more effective compound characterization to obtain a low dose of IC50 together with using vehicle such as the green nanoparticle technique, in the case if it is more probable in vitro. 40 The study is worthwhile to further investigate and identify the novel purified compound with anticancer activity to develop a single active anticancer agent or analogs/derivatives by chemical synthesis or modification and search for its biological death induction/activities at molecular mechanism(s) to be applied instead of the formula/extract as low oral dosage taking.
Moreover, in response to chemotherapeutic drugs, such as DOX, in human breast cancer MCF-7 and MDA-MB-231 cells when cultured in three-dimensional (3D) versus two-dimensional (2D) extracellular model, the cells exhibited significant reduction in cell growth and proliferation. The mechanism is signaling via integrin type switching/alteration and disruption. 41 DOX also induces apoptosis and oxidative stress in breast cancer cell lines. The comparison of mechanisms of apoptosis in DOX-induced breast cancer cell death between MCF-7 (estrogen, progesterone receptors: positive; HER2: negative) and MDA-MB-231 (triple negative: estrogen, progesterone receptors; HER2: negative) cells are found to be via pro-apoptotic protein Bcl-2 upregulation and anti-apoptotic down-regulations. ROS level decreases due to superoxide dismutase expression of both protein and gene levels. The significant adverse side effects of DOX are ROS generation and cardiovascular alteration including arrhythmia. The signaling is via a transcription factor, NF-kappaB, signaling pathway. 42 The study of molecular signaling in cancer cell death helps to focus and target the molecules mediated or signaling pathway with least effect on normal cells.
FEE of PP was determined as the most cytotoxic fraction against MDA-MB-231 cells and the mode of cell death was regulated cell death with the amphipathic phenotypes of apoptosis (MTP and caspase activities) and necrosis (annexin V-FITC/PI and PI staining of cells, MTP-driven necrosis via MLKL and Ca2+ overload) through the ER (high Ca2+) and oxidative stresses and through mitochondrial (caspase-9) and death receptor (caspase-8) signaling pathways, evidenced by both caspase activation of caspase-3 activity, allowing as a cascade of cell signaling.
The evidence exhibited the abrupt loss of permeability of the outer and inner mitochondrial membranes to small solutes, resulting in a rapid decrease of MTP, dissipation, and osmotic breakdown of both mitochondrial membranes, leading to regulated or programmed cell death.43,44 These research data support the present findings of regulated cell death ending in both apoptosis and secondary necrosis in human breast cancer MDA-MB-231 cell line induced by FEE of PP.
Conclusion
The FEE of PP was the most effective extract to induce regulated cell death with the phenotype of apoptosis and necrosis in MDA-MB-231 cells through intrinsic (caspase-9) and extrinsic (caspase-8) pathways, with the evidence of disruption of MPT-driven necrosis, overload of cytosolic Ca2+ level, and oxidative and ER stresses.
Supplemental Material
Tum_Bio_PPSupplement_data_edit_160718 – Supplemental material for Effect of Pseuderanthemum palatiferum (Nees) Radlk fresh leaf ethanolic extract on human breast cancer MDA-MB-231 regulated cell death
Supplemental material, Tum_Bio_PPSupplement_data_edit_160718 for Effect of Pseuderanthemum palatiferum (Nees) Radlk fresh leaf ethanolic extract on human breast cancer MDA-MB-231 regulated cell death by Phimphan Komonrit and Ratana Banjerdpongchai in Tumor Biology
Footnotes
Acknowledgements
This study was conducted at Department of Biochemistry and the Center of Medical Research and Instruments, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand. Phimphan Komonrit contributed to the conduct of the study and acquisition and analysis of data. Ratana Banjerdpongchai contributed to the experimental design, hypothesis, idea, supervision, and manuscript preparation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Research and Researcher for Industries (RRI) for Master Degree (MSD5810017). The RRI-MAG grant was partly shared by Prolac (Thailand) Corp., Ltd, for P.K. and the financial support from Research Endowment Fund of Faculty of Medicine, Chiang Mai University, Chiang Mai for R.B. (Grant No. 007/2017).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
