Abstract
Considerable attention has been paid to the introduction of novel naturally occurring plant-derived radiosensitizer compounds in order to augment the radiation efficacy and improve the treatment outcome of different tumors. This study was therefore undertaken to evaluate the antitumor, antiangiogeneic, and synergistic radiosensitizing effects of apigenin, a dietary flavonoid, and/or cryptotanshinone, a terpenoid isolated from the roots of Salvia miltiorrhiza, against the growth of solid Ehrlich carcinoma in female mice. Apigenin (50 mg/kg body weight) and/or cryptotanshinone (40 mg/kg body weight) was intraperitoneally (i.p.) injected into non-irradiated or γ-irradiated (6.5 Gy whole-body γ-irradiation) solid Ehrlich carcinoma-bearing mice for 30 consecutive days. Investigations included molecular targets involved in proliferation, inflammation, angiogenesis, and tumor invasiveness. Treatment with apigenin and/or cryptotanshinone significantly suppressed the growth of solid Ehrlich carcinoma tumors and demonstrated a synergistic radiosensitizing efficacy together with γ-irradiation. These effects were achieved through downregulating the expression of angiogenic and lymphangiogenic regulators, including signal transducer and activator of transcription 3, vascular endothelial growth factor C, and tumor necrosis factor alpha, suppressing matrix metalloproteinase-2 and -9 activities, which play a key role in tumor invasion and metastasis, and enhancing apoptosis via inducing cleaved caspase-3 and granzyme B levels. Histological findings of solid Ehrlich carcinoma tumors verified the recorded data. In conclusion, a synergistic radiosensitizing efficacy for apigenin and cryptotanshinone was demonstrated against Ehrlich carcinoma in the current in vivo murine model, representing therefore a potential therapeutic strategy for increasing the radiation response of solid tumors.
Introduction
Cancer is the second leading cause of death worldwide behind cardiovascular diseases. 1 Modern surgery, chemotherapy, and radiotherapy are the most common types of cancer treatments available nowadays that significantly reduced the cancer mortality.2,3 A benefit of the radiotherapy is the possibility to apply the ionizing radiation locally, thereby preventing systemic toxicity. However, like chemotherapeutic agents, ionizing radiation does not affect all target cells, which can lead to severe side effects in the surrounding tissue. In addition, a large number of human malignant tumor cells responds poorly to ionizing radiation. 4 Accordingly, the effect of radiation on tumor tissues can be optimized by adding radiosensitizing agents, in order to achieve a greater degree of tumor damage than expected from radiation only, thus minimizing large doses of radiation and also sparing normal tissues. Most of the routinely used synthetic radiosensitizers are commonly toxic per se, non-selective, and are not tumor specific. Because of their negligible toxicity, herbal drugs have gained attention to replace the highly toxic radiosensitizers. 4 Apigenin (AP; 4,5,7-trihydroxyflavone), one of the most commonly occurring flavonoids, is found in numerous plants. 5 Like most flavones, AP possesses antioxidant, anti-inflammatory, and antidepressant properties,6–8 and several epidemiologic studies indicate that a diet rich in flavones, especially AP, is associated with a decreased risk of various human cancers. 9 In vitro studies demonstrated that AP suppresses cell growth in a number of human cancer cell lines.10–13 In addition, AP was reported to suppress prostate tumorigenesis in the transgenic adenocarcinoma of the mouse prostate (TRAMP) model through the phosphoinositide 3-kinase (PI3K)/Akt/forkhead box O (FoxO)-signaling pathway. 14 Cryptotanshinone (CPT), a major terpenoid tanshinone isolated from the roots of Salvia miltiorrhiza Bunge, has been used in traditional Chinese medicine for the treatment of a wide variety of clinical conditions, including acute ischemic stroke, coronary artery disease, hyperlipidemia, and Alzheimer’s disease.15,16 Several studies documented the ability of CPT to inhibit signal transducer and activator of transcription 3 (STAT3) phosphorylation, arrest the cell cycle, and induce apoptosis in different cancer cell lines.17–19 CPT has also demonstrated sensitizing effects to a broad range of anticancer agents, including cisplatin and 5-fluorouracil, highlighting its therapeutic potential in the treatment of human cancers. 20 This study was undertaken to evaluate the antitumor and radiosensitizing abilities of AP and/or CPT against solid Ehrlich carcinoma (EC) in female mice and to explore some of the molecular targets involved in these effects.
Materials and methods
Experimental animals
Female Swiss albino mice weighing 20–30 g were obtained from the National Cancer Institute (Cairo University, Giza, Egypt). Animals were housed in plastic cages and maintained under standard conditions of temperature, humidity, and 12 h light/dark cycles. They were provided with a commercial standard pelleted diet and water ad libitum. Mice were left for an acclimatization period of 1 week before the start of the experiment. All animal procedures were performed in accordance with the Guide for the Care and Use of Laboratory Animals 21 and approved institutional guidelines were followed.
Chemicals
AP was purchased from Sigma-Aldrich (Gillingham, UK) and CPT was obtained from Shaanxi Sciphar Hi-Tech Industry Co., Ltd. (Shaanxi, China). AP was dissolved in dimethyl sulfoxide (DMSO) diluted in sterile physiological saline (1:10 v/v), and each mouse received a daily i.p. injection of 50 mg/kg body weight for 30 consecutive days. 22 Similarly, CPT was dissolved in DMSO diluted in sterile physiological saline (1:10 v/v) and injected i.p. at a dose of 40 mg/kg body weight for 30 consecutive days.19,23
Tumor transplantation
The murine Ehrlich ascites carcinoma (EAC) cells were kindly provided by the National Cancer Institute (Cairo University, Giza, Egypt) and were maintained in our laboratory by the weekly i.p. injection of Ehrlich carcinoma (EC) cells into female Swiss albino mice. 24 Solid EC tumors were produced by the intramuscular inoculation of 2.5 × 106 viable EC cells into the right thigh muscle of the lower limb of a healthy female Swiss albino mouse. 25 The tumor volume was measured with a Vernier caliper, 26 and mice with a palpable solid tumor mass of approximately 1 cm3 (that developed 14 days after EC inoculation) were subsequently used in the study.
Radiation exposure
Whole-body γ-irradiation was performed to mice using a Gammacell 40 Cesium Irradiator (Atomic Energy of Canada Ltd, Chalk River, ON, Canada) installed in the National Center for Radiation Research and Technology (NCRRT) of the Egyptian Atomic Energy Authority (EAEA, Cairo, Egypt). Animals were first restrained, placed in a well-ventilated canister (31.2 cm diameter × 10.5 cm height), and then exposed to a total of 6 Gy γ radiation (cesium-137 located inside a cylindrical stainless steel double capsule) at a dose rate of 0.43 Gy/min. 27
Experimental design
A total number of 90 female Swiss albino mice were randomly allocated into 9 equally sized groups as follows:
Group 1 (Control, C): Mice received a daily i.p. injection of DMSO diluted in sterile physiological saline (1:10 v/v; 8 mL/kg body weight) for 30 consecutive days.
Group 2 (E): Mice bearing solid EC tumors were daily i.p. injected with DMSO diluted in sterile physiological saline (1:10 v/v; 8 mL/kg body weight) for 30 consecutive days.
Group 3 (E + AP): Mice bearing solid EC tumors were injected i.p. with 50 mg/kg body weight AP for 30 consecutive days starting from the 15th day following EC inoculation.
Group 4 (E + CPT): Mice bearing solid EC were injected i.p. with 40 mg/kg body weight CPT for 30 consecutive days starting from the 15th day following EC inoculation.
Group 5 (E + AP + CPT): Mice bearing solid EC were first injected i.p. with AP (50 mg/kg body weight) followed by an i.p. injection of CPT (40 mg/kg body weight) 2 h later for 30 consecutive days starting from the 15th day following EC inoculation.
Group 6 (E + R): Mice bearing solid EC were exposed to a single 6.5 Gy whole-body γ-irradiation at the 15th day following EC inoculation.
Group 7 (E + AP + R): At the 15th day following EC inoculation, mice were first injected i.p. with AP (50 mg/kg body weight), followed by a single 6.5 Gy whole-body γ-irradiation 30 min later. AP administration was then continued for 30 consecutive days.
Group 8 (E + CPT + R): At the 15th day following EC inoculation, mice were injected i.p. with CPT (40 mg/kg body weight), followed by a single 6.5 Gy whole-body γ-irradiation 30 min later. CPT administration was then continued for 30 consecutive days.
Group 9 (E + AP + CPT + R): At the 15th day following EC inoculation, mice were first injected i.p. with AP (50 mg/kg body weight), followed by an i.p. injection of CPT (40 mg/kg body weight) 2 h later and were finally exposed to a single 6.5 Gy whole-body γ-irradiation after 30 min. AP and CPT administrations were then continued for 30 consecutive days.
Blood collection and tissue sampling
At the end of the experiment, whole blood was withdrawn from animals by heart puncture and left to coagulate at 37ºC for 15 min. Serum was then separated and stored at −20ºC until further analysis. At autopsy, portions of the right thigh muscle (group 1) and solid EC tumors (groups 2–9) were dissected, rinsed in ice-cold isotonic saline, blotted dry with a filter paper, and preserved at −80ºC. Similarly, parts of the aforementioned specimens were kept in 10% formalin for histological analysis.
Preparation of tissue homogenate
A 10% whole-tissue homogenate was prepared by homogenizing a part of the right thigh muscle or solid EC tumors in 10 volumes of ice-cold isotonic saline in an electric homogenizer (Glas-Col LLC, Terre Haute, IN, USA). The homogenate was centrifuged at 1800g at 4ºC for 10 min and the supernatant was separated and stored at −20ºC.
Biochemical analysis
In the right thigh muscles and solid EC tumors, nitric oxide (NO) 28 and total protein 29 concentrations were assayed by colorimetric methods. Cleaved caspase-3 level was assayed using a Human/Mouse Cleaved Caspase (Asp175) DuoSet® IC ELISA kit (R&D Systems, Inc., Minneapolis, MN, USA) and granzyme B level was determined using a Mouse Granzyme B ELISA kit (RayBiotech, Norcross, GA, USA).
RNA extraction and real-time quantitative polymerase chain reaction
Total cellular RNA was extracted from frozen right thigh muscles and solid EC tumors using RNeasy® Mini kit according to manufacturer’s instructions (Qiagen, Hilden, Germany). The concentration and purity of total RNA were assessed by measuring absorbance at 260 and 280 nm, respectively, in a spectrophotometer (Nano Drop 2000; Thermo Fisher Scientific, USA). First-strand complementary DNA (cDNA) was synthesized using Thermo Scientific™ RevertAid™ First-Strand cDNA Synthesis Kit (Fermentus, Thermo Fisher Scientific Inc, Runcorn, UK). Real-time polymerase chain reaction (PCR) amplification and analysis were performed in an optical 96-well plate in ABI PRISM 7500 Fast Sequence Detection System Thermal Cycler (Applied Biosystems, Foster City, CA, USA) using Power SYBR® Green PCR Master Mix (Applied Biosystems). The amplification protocol consisted of 40 cycles (denaturation at 95ºC for 15 s, annealing at 55ºC for 20 s, and extension at 72ºC for 20 s). Primers used for STAT3, vascular endothelial growth factor C (VEGF-C), tumor necrosis factor alpha (TNF-α), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH, endogenous reference gene) are represented in Table 1. The relative expression of selected genes was determined by the ΔΔCT method. 30
Primers used for real-time polymerase chain reaction.

Gelatin zymography
A volume of 5 µL serum was mixed with 15 µL sample buffer and then separated onto a 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) containing 0.1% gelatin (type B from bovine; Sigma-Aldrich, St. Louis, MO, USA). After electrophoresis, the gel was soaked at room temperature in the washing buffer for 30 min and then incubated overnight at 37ºC in the reaction buffer in a horizontal shaker. Digestion bands were visualized by staining the gel with 0.1% Coomassie Brilliant Blue R-250 and then destained with methanol and acetic acid. Matrix metalloproteinase-2 (MMP-2; 72 kDa) and -9 (MMP-9; 92 kDa) proteins were defined on the slab gel by their gelatinolytic enzyme activity, which appeared as transparent bands on the blue background and were compared to a standard human purified collagenase IV (Sigma-Aldrich, USA) and a protein marker (Sigma-Aldrich, USA). The bands were quantified by densitometry using Gel-Pro Analyzer.
Histological examination
Fixed right thigh muscle and solid EC tumor specimens were processed and embedded in paraffin. Serial 4 μm sections were stained with hematoxylin and eosin and examined under a light microscope (Olympus, Tokyo, Japan).
Statistical analysis
The Shapiro–Wilk test for normality (p > 0.05) showed that all data were normally distributed. 31 Statistical analysis of the difference between means was carried out using one-way analysis of variance (ANOVA). In case of a significant F-ratio, post hoc Duncan’s test for multiple comparisons was used to evaluate the statistical significance between treatment groups at p < 0.05 level of significance. All the statistical analyses were done using Statistical Package for Social Science (SPSS) version 20.0 (SPSS Inc., Chicago, IL, USA).
Results
Tumor growth
At the end of the experiment (30 days starting after the tumor volume had reached 1 cm3), the size of solid EC tumor in untreated mice enlarged to 2125.88 mm3. A gradual significant decrease was demonstrated in the tumor volume following treatment of EC-bearing mice with AP or CPT, showing a synergistic suppressive effect with both compounds, compared to untreated EC-bearing mice. Similarly, exposure of EC-bearing mice to γ-irradiation produced a significant reduction in the tumor volume, and a radiosensitizing effect was noticeable by decreasing the tumor growth of γ-irradiated-EC-bearing mice co-administered AP, CPT, or both drugs, respectively, compared to untreated EC-bearing mice (Figure 1).
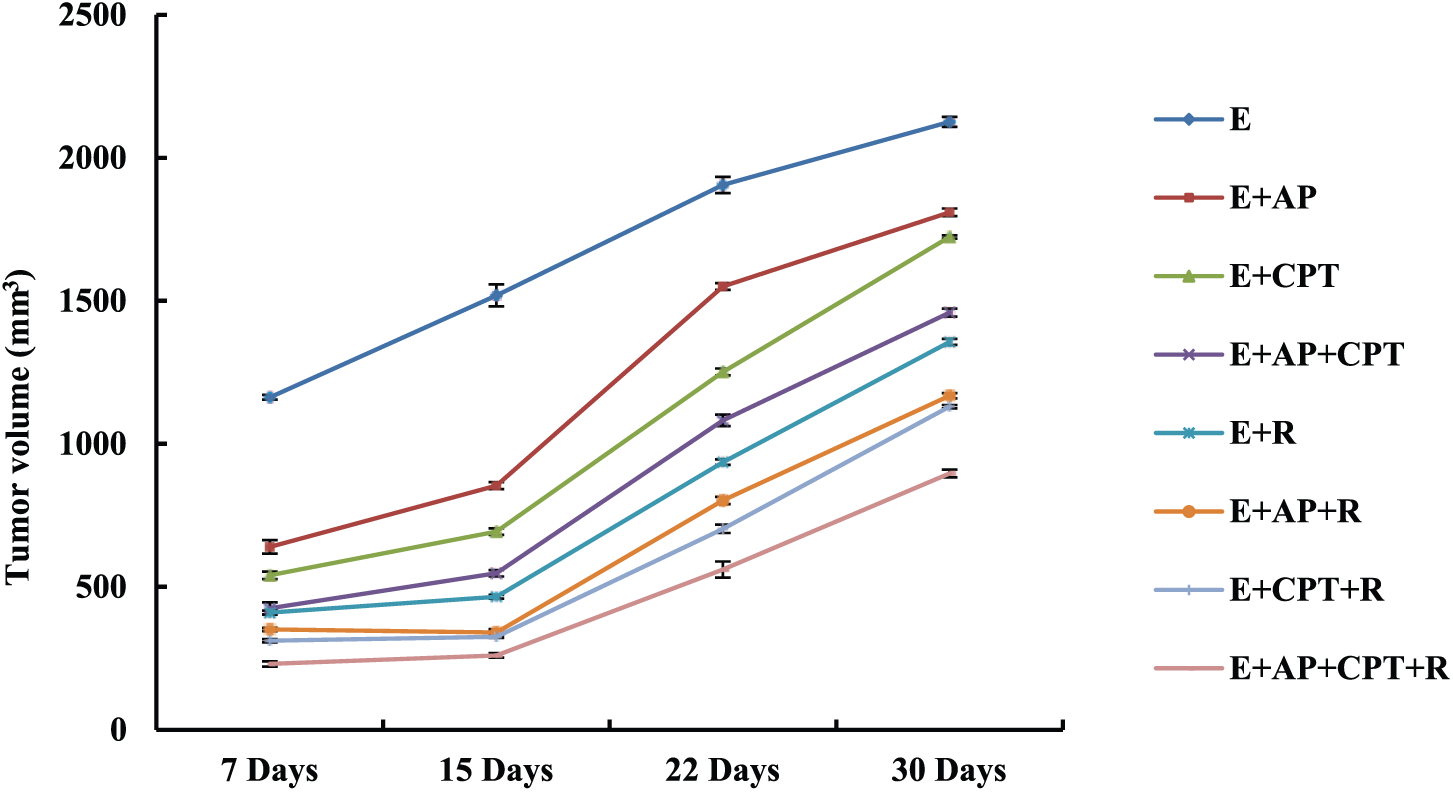
Change in the solid EC tumor growth in different groups throughout the experiment. Female mice were inoculated with EC tumor cells in the right thigh muscle of the lower limb. Mice with a palpable solid tumor mass of 1000 mm3 that developed 14 days after EC inoculation were subsequently assigned to the different treatment modalities, and the tumor volume was measured at four time intervals (7, 15, 22, and 30 days; n = 8).
Tumor nitric oxide, cleaved caspase-3, and granzyme B levels
Mice bearing solid EC tumors manifested a significant increase in the tumor nitric oxide level (386.80%), and in contrast, cleaved caspase-3 and granzyme B levels were significantly decreased (93.30% and 77.08%, respectively), compared to non EC-bearing mice. Treatment of solid EC-bearing mice with AP, CPT, or both compounds resulted in a significant reduction in nitric oxide level (39.87%, 51.52%, and 63%, respectively) and enhanced apoptosis as revealed by the significant elevation in cleaved caspase-3 and granzyme B levels (220%, 442.86%, and 622.86% for caspase-3; 72.59%, 134.07%, and 191.11% for granzyme B, respectively), compared to untreated EC-bearing mice. However, exposure of EC-bearing mice to γ-irradiation either alone or in combination with AP or CPT produced a significant suppression in tumor nitric oxide level (38.12%, 61.08%, and 62.68%, respectively), which is associated with a sharp increase in caspase-3 and granzyme B levels (385.71%, 460%, and 588.57% for caspase-3; 128.15%, 177.04%, and 215.56% for granzyme B, respectively), where the three treatment modalities showed the most pronounced significant decrease in the level of tumor nitric oxide (71.61%), as well as a significant increase in caspase-3 and granzyme B levels (740% and 267.59%, respectively), compared to untreated EC-bearing mice (Table 2).
Nitric oxide (NO), cleaved caspase-3, and granzyme B levels in the right thigh muscle (control) or solid EC tumor tissues in different groups.

Values are mean ± standard error (n = 8, p < 0.001 is highly significant). Values with dissimilar superscripts are considered significantly different.
%Change versus control group (C).
%Change versus untreated solid EC-bearing mice (E).
Relative expression of selected genes
A significant upregulation was demonstrated in the expression of STAT3, VEGF-C, and TNF-α genes (919.20%, 751.61%, and 650%, respectively) in solid EC tumors, compared to non EC-bearing mice. In contrast, treatment of EC-bearing mice with AP, CPT, or both agents resulted in a significant reduction in the expression of the aforementioned genes (41.52%, 33.96%, and 31.32% for AP; 48.12%, 47.22%, and 45.12% for CPT; and 57.69%, 60.98%, and 56.28% for both compounds, respectively), compared to untreated EC-bearing mice. However, exposure of EC-bearing mice to γ-irradiation either alone or in combination with AP or CPT produced a significant downregulation in the expression of tumor STAT3, VEGF-C, and TNF-α genes (36.73%, 39.39%, and 53.02%; 56.91%, 53.79%, and 66.82%; and 64.29%, 62.25%, and 73.49%, respectively), where the three treatment modalities caused the most pronounced significant downregulation in the expression of the selected genes (74.25%, 71.84%, and 78.14%, respectively), compared to untreated EC-bearing mice (Figure 2(a)–(c)).
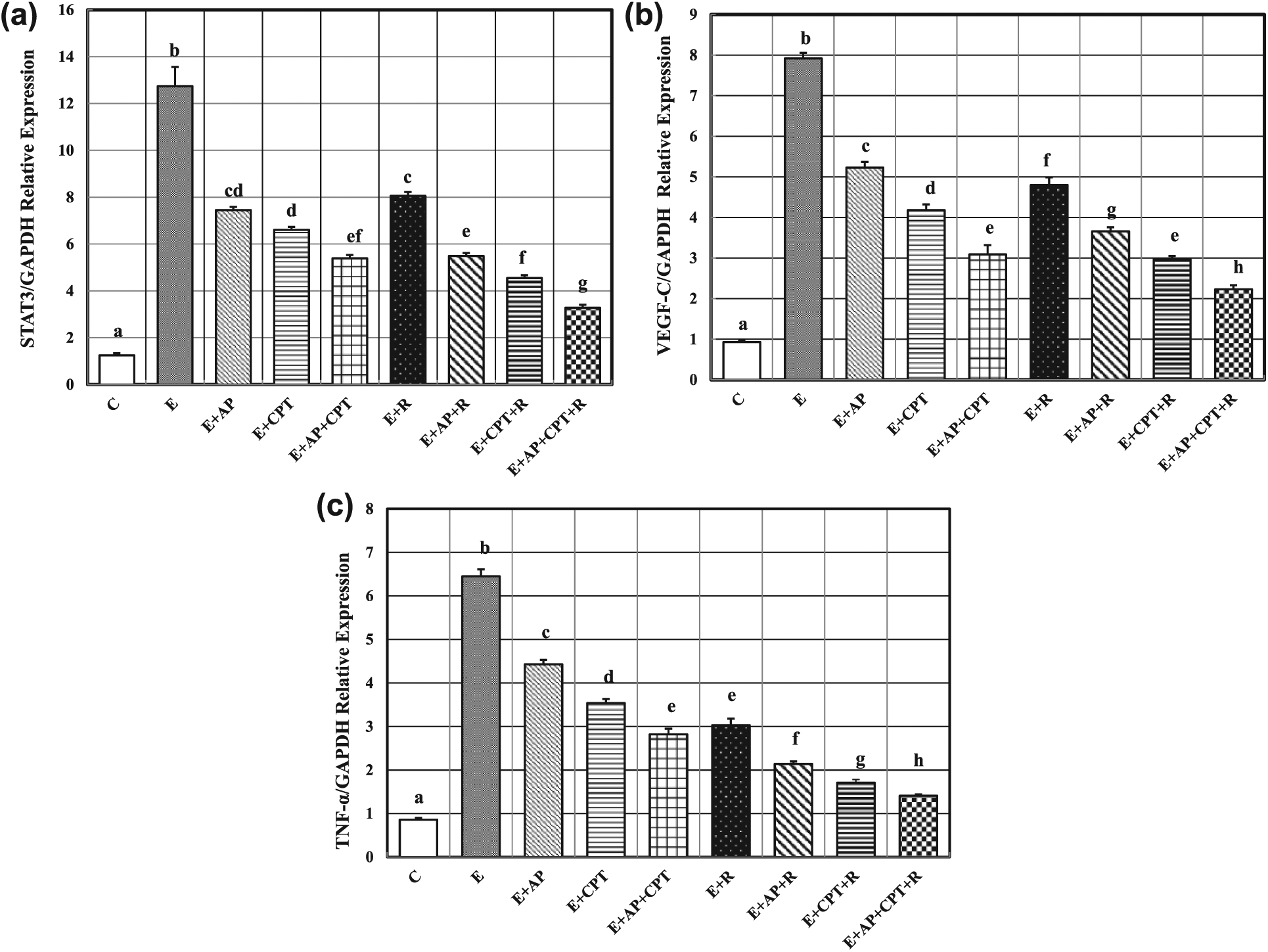
Relative mRNA expression of (a) STAT3, (b) VEGF-C, and (c) TNF-α in the right thigh muscle (control) or solid EC tumor tissues in different groups. Values are mean ± standard error (n = 8). Values with dissimilar superscripts are considered significantly different.
Activity of serum MMP-2 and -9
Mice bearing solid EC tumors demonstrated a significant sharp increase in the activity of serum MMP-2 and -9 (765.11% and 278.91%, respectively), compared to non EC-bearing mice. Treatment of solid EC-bearing mice with either AP, CPT, or both compounds resulted in a significant reduction in MMP-2 and -9 activities (43.35% and 34.62% for AP; 48.27% and 45.17% for CPT; and 61% and 62.22% for both drugs, respectively), compared to untreated solid EC-bearing mice. Similarly, whole-body γ-irradiation of solid EC-bearing mice either alone or in combination with AP or CPT produced a significant decrease in the activity of serum MMP-2 and -9 (43.06% and 57.49% for γ-irradiation; 59.77% and 64.02% for AP, and 58.53% and 64.77% for CPT, respectively), where the three treatment modalities showed the most pronounced significant reduction in MMP-2 and -9 activities (73.26% and 66.26%, respectively), compared to untreated EC-bearing mice (Figure 3(a)–(c)).

(a) Gelatin zymography, (b) activity of serum MMP-2, and (c) MMP-9 in different groups (n = 5). Lane M: protein marker; lane 1: standard collagenase (350 U/mg protein); and lanes 2–9: control, E, E + AP, E + CPT, E + AP + CPT, E + R, E + AP + R, E + CPT + R, and E + AP + CPT + R, respectively.
Histological findings
Histological examination of the thigh skeletal muscle from a control mouse showed normal histological structure of striated bundles representing normal muscular fibers (Figure 4(a)). Solid EC tumor inoculated in the thigh muscle of an untreated mouse showed various bundles of muscular fibers intermingled with tumor patches, consisting of groups of large, round, and polygonal cells with pleomorphic shapes and hyperchromatic nuclei (Figure 4(b)). Solid EC tumors of AP-treated mice showed various degrees of muscle fiber lysis and ragged margins, along with apoptotic tumor cells (Figure 4(c)). Similarly, solid tumors of CPT-treated EC-bearing mice showed degenerated areas of tumor with highly pyknotic nuclei (Figure 4(d)). Furthermore, solid EC tumors from AP- and CPT-treated EC-bearing mice showed wide areas of muscular necrosis, regression of tumor cell invasion, and apoptotic tumor cells (Figure 4(e)). Tumor cell remnants and areas of muscular necrosis were noticed in solid EC tumor from γ-irradiated EC-bearing mice (Figure 4(f)). A more pronounced effect in tumor regression was noticed following treatment of γ-irradiated EC-bearing mice with either AP (Figure 4(g)), CPT (Figure 4(h)), or both compounds (Figure 4(i)) as revealed by the existence of degenerated areas of apoptotic EC tumor cells with pyknotic nuclei, inflammation, and lysis of muscular fibers.

Photomicrographs of sections from the skeletal thigh muscle from a (a) control mouse with normal histological structure of striated muscular fibers, a solid tumor in an (b) untreated EC-bearing mouse showing tumor cells within muscular tissues (*), (c) apigenin-treated EC-bearing mouse showing wide areas of muscle fiber lysis (arrow head) and apoptotic tumor cells (arrow), (d) cryptotanshinone-treated EC-bearing mouse demonstrating degenerated areas of tumor with pyknotic nuclei (arrow), (e) apigenin- and cryptotanshinone-treated EC-bearing mouse showing wide areas of muscular necrosis (arrow head) and apoptotic tumor cells (arrow), (f) γ-irradiated EC-bearing mouse showing tumor cell remnants (arrow) and areas of muscular necrosis (arrow head), (g) apigenin-treated γ-irradiated EC-bearing mouse, (h) cryptotanshinone-treated γ-irradiated EC-bearing mouse, and (i) apigenin- and cryptotanshinone-treated γ-irradiated EC-bearing mouse showing tumor cell remnants (arrow) and necrotic areas (arrow head; (hematoxylin and eosin stain, 40×).
Discussion
Although radiotherapy is one of the most effective strategies in the treatment of cancers, radiation resistance is considered as one of the major causes of radiotherapy failure and subsequent tumor relapse. Enhancing the radioresponsiveness of tumors using radiosensitizers, agents that sensitize tumor cells to radiation, is a promising approach to increase the efficacy of radiation therapy. 32
In an attempt to improve cancer therapeutic protocols, this study was undertaken to evaluate the synergistic antitumor effect of AP and CPT together with γ-irradiation against solid EC tumors in female mice. Different molecular targets were analyzed in order to explore the underlying mechanisms by which both compounds suppressed the growth of solid tumor and/or enhanced the radiosensitivity of cancer cells to γ-irradiation.
In this study, a significant upregulation was recorded in STAT3, VEGF-C, and TNF-α messenger RNA (mRNA) expressions in solid EC tumors, compared to non EC-bearing mice. STAT3 plays a fundamental role in tumor progression and angiogenesis, and its overexpression in different tumors had been previously reported.33–38 In addition, STAT3 directly regulates the transcription of the VEGF gene (the most potent proangiogenic stimulus that plays a critical role in tumorigenesis by stimulating tumor lymphangiogenesis and metastasis) and induces its overexpression in different tumors.39–41 STAT3 promotes tumor cell proliferation by upregulating the expression of the cyclin B1/cdc-2/c-myc/cyclin D1/c-jun/c-fos complex and downregulating the expression of the cyclin B1 kinase inhibitor cyclin-dependent kinase 1 (CDK1) in p21/p27 expressing cells, which drives rapid progression through the G1/S checkpoint. 42
It has been estimated that approximately 25% of the malignancies worldwide are induced by chronic inflammation, where altered levels of proinflammatory and proangiogenic factors are observed in different cancers. 43 Activation of key inducers of proinflammatory cascades, including the transcription factors STAT3 and nuclear factor kappa beta (NF-κβ), leads to enhancement of cell proliferation, apoptotic evasion, invasion, and metastasis, as well as angiogenesis, all being well-established hallmarks of cancer. 44 TNF-α is a key player in cancer-related inflammation and promotes lymphangiogenesis and lymphatic metastasis in clinically relevant human and mouse epithelial carcinoma tumor models through several mechanisms, including tumor inflammation, tumor angiogenesis, and inducing epithelial–mesenchymal transition (EMT).45,46 In addition, TNF-α is a major inflammatory mediator that induces multiple changes in endothelial cell gene expression, including induction of adhesion molecules, integrins, and MMPs, therefore acting as an autocrine growth factor for tumor angiogenesis. 47
Tumor cells acquire specific functions necessary for tumor growth and dissemination, where MMPs (a family of enzymes involved in matrix remodeling) constitute key players in these processes, allowing tumor cells to modify the extracellular matrix (ECM) and to release cytokines, growth factors, and other cell-surface molecules, ultimately facilitating protease-dependent tumor progression. 48 Several studies had shown that cancer cells that were attributable to metastasis to distant organs express high levels of MMP-9. Therefore, MMP-9 is not only important in identifying invasion symptoms and diagnosis, but also a promising therapeutic target to prevent cancer invasion and metastasis. 49 A mutual relationship between STAT signaling and MMP-2 was previously demonstrated, where overexpression of STAT3 enhances the invasiveness in less-invasive melanoma cells through increasing MMP-2 expression and activity, while inactivation of STAT3 remarkably impairs the invasive ability of invasive melanoma cells through decreasing MMP-2 expression and activity. 50 In addition, TNF-α has already been shown to be involved in cell–cell communication, and in a coculture of breast cancer cells and tumor-associated macrophages, macrophage-derived TNF-α increased the levels of MMP-2 and MMP-9 in tumor cells. 51
A significant effect on tumor lymphangiogenesis and lymphatic spread in addition to tumor angiogenesis is exerted by nitric oxide (NO). 52 The inducible form of nitric oxide synthase (iNOS) is expressed mainly through TNF-α activated pathway. 53 Nitric oxide induces the proliferation of lymphatic endothelial cells and prolongs their survival by increasing lymphangiogenic growth factors (such as VEGF) and VEGF receptors expression in tumor-associated lymphatic vessels. Furthermore, increased vascular permeability and vasodilatation by NO facilitates the spread of tumor cells via lymphovascular invasion. 52
In agreement with the current findings, induction of serum MMP-2 and -9 activities in solid EC-bearing mice had been previously reported. 54 The increase in the angiogenic regulators (STAT3, VEGF-C, and TNF-α) along with the induction of MMP-2 and -9 activities and increasing tumor NO level induced neovascularization and enhanced the ability of solid EC tumor to grow, as manifested by the increase in tumor volume of EC-bearing mice.
Our results demonstrated apoptosis suppression in solid EC tumors as evidenced by the significant reduction in the level of apoptotic molecules (caspase-3 and granzyme B), compared to non EC-bearing mice. Apoptosis is a programmed cell death that maintains the stability of the internal environment through removing genetic mutations and unstable cells. However, this process is inhibited in cancer, which leads to the accumulation of various genetically unstable cells. Constitutive STAT3 activation is closely associated with hyperproliferation and antiapoptosis in tumor cells. STAT3 exerts its antiapoptotic effect via upregulating the expression of antiapoptotic proteins, inducing the production of p100/p52, and suppressing p53-mediated apoptosis by binding to p53 promoter inhibiting its subsequent transcription. 42
The disturbance in the angiogenic and apoptotic regulators leads to tumor proliferation and growth. The current data revealed that the upregulation of STAT3, VEGF-C, and TNF-α genes and induction of MMPs’ activity along with apoptosis suppression in solid EC-bearing mice collectively enhance tumor cell proliferation and neovascularization, which ultimately resulted in the acceleration of tumor growth, invasiveness, and metastatic ability of tumor cells.
Many cancer cells, including lung, prostate, immortalized keratinocytes, and colon cancer cells, are committed to apoptotic cell death when exposed to radiation.55,56 In this study, exposure of EC-bearing mice to γ-irradiation produced a significant delay in tumor volume, which was achieved through the downregulation of angiogenic regulators expression, suppression of MMP activities, and induction of apoptosis. Radiation-induced DNA damage activates a variety of signaling pathways; the intrinsic apoptotic pathway has been considered the primary mechanism mediating ionizing radiation–induced apoptosis, making tumor cells unable to divide and grow.57,58
Current dietary guidelines to combat chronic diseases, including cancers, recommend increasing intake of plant foods that are rich in antioxidants. 59 The result of this study revealed that treatment of EC-bearing mice with AP and/or CPT per se significantly reduced the growth of solid tumors, and a synergistic effect was demonstrated following combination of both treatments with γ-irradiation. As a key player in tumor proliferation, angiogenesis, and inflammatory cascade induction, the downregulation of STAT3 expression following treatment of irradiated and non-irradiated EC-bearing mice with AP and/or CPT reduced tumor growth via multiple mechanisms, including suppression of tumor cell proliferation (reduction in tumor volume), angiogenesis (downregulation of tumor VEFG-C expression), production of proinflammatory cytokines (downregulation of tumor TNF-α expression), invasion (reduction of serum MMP-2 and -9 activities), and enhancing apoptosis (induction of tumor caspase-3 and granzyme B levels).
AP is an important dietary flavonoid with strong antioxidant, chemopreventive, and anti-inflammatory activities. 60 It enhances the endogenous defense against oxidative stress, which is attributed to its ability not only to donate hydrogen to the harmful free radicals to prevent the oxidative damage but also to directly react with reactive oxygen species (ROS). 9 AP also induced caspase-dependent extrinsic apoptosis in BT-474 breast cancer cells by upregulating the levels of cleaved caspase-8 and cleaved caspase-3 and inducing the cleavage of poly (ADP-ribose) polymerase (PARP) through inhibition of STAT3 signaling and decreasing VEGF and MMP-9 production. 61 Furthermore, AP inhibited the expression of hypoxia-inducing factor (HIF-1) and VEGF in human prostate (PC-3, DU145, and LNCaP), ovarian (OVCAR-3), and colon (HCT-8) cancer cells and reduced tumor angiogenesis in vivo, which was associated with a decrease in HIF-1 and VEGF expression in tumor tissues. 62 Similarly, AP also induced apoptosis and inhibited melanoma cells’ invasiveness by decreasing MMP-2 activity and VEGF expression through suppressing the constitutively active STAT3. 50 AP was also reported to sensitize cancer cells to paclitaxel-induced apoptosis in different human cancer cell lines. 63 According to the structure-activity relationship of flavonoids and their anti-inflammatory effects, the C2–C3 double bond and hydroxylations at positions 5, 7, and 4′ along with a 4-oxo functional group of the C ring are essential to the higher anti-inflammatory effect of AP. 60
Compounds isolated from Danshen, including tanshinone I, tanshinone IIA, dihydrotanshinone, and CPT, have been reported to possess potent anti-inflammatory effects through suppression of NF-κB and iNOS signaling pathways and reducing proinflammatory mediators production.64,65 Lu et al. 18 reported that CPT significantly suppressed malignant glioma cell proliferation through inhibition of STAT3 phosphorylation and attenuation of STAT3 nuclear translocation, with subsequent downregulation of STAT3-regulated proteins (cyclin D1 and survivin) that ultimately arrest cells at the G1/G0 phase. In addition, CPT binds directly to the STAT3 monomer preventing its downstream dimerization in DU145 prostate cancer cells and inhibits STAT3 phosphorylation in HCT116 and SW480 colorectal cancer that reduces the expression level of antiapoptosis proteins (Bcl-2, CyclinD1, and survivin).66,67
Several studies demonstrated the antiangiogenic ability of CPT, which inhibited tube formation in murine lymphatic endothelial cells in an in vitro model of lymphangiogenesis. 68 It also inhibited the proliferation, migration, angiogenic sprouting, and tube formation of human umbilical vein endothelial cells (HUVECs). 69 Deng et al. 70 reported that CPT inhibited the proliferation of tumor cells in a human gastric cancer cell line (SGC-7901) and suppressed angiogenesis by downregulating the expression of VEGF mRNA expression. In addition, CPT reduced VEGF-induced neovascularization and sprouting in mouse B16F10 tumor cells xenografted into the right hind footpad of C57BL/6 mouse. 71 Furthermore, tanshinone IIA was reported to induce cell death and chemosensitizes colon cancer cells (HCT116 and COLO205) to fluorouracil therapy by suppressing NF-κB-regulated genes including VEGF and Bcl-2. 72
CPT, the most potent anticancer agent among the extracts of Danshen, inhibits the proliferation of cancer cells and induces cell apoptosis. Also, it enhances ROS production, which activates p38/JNK and inhibits extracellular signal–regulated protein kinases 1 and 2 (ERK1/2), leading to caspase-independent cell death in different human cancer cell lines (rhabdomyosarcoma cells (Rh30), hepatocellular carcinoma cells (HepG2), prostate cancer cells (DU145), and breast cancer cells (MCF-7)). The mechanisms involved in the inhibition of cellular proliferation and induction of apoptosis by CPT include arresting the cell cycle in G0/G1 phase, downregulation of cyclin D1 expression, hypophosphorylation of retinoblastoma (Rb), and inhibition of STAT3.20,73 It also induced cell apoptosis in Bel-7404 gastric cancer cell line and upregulated the activation of apoptosis-related cleaved caspase-3 and -9 and cleaved poly (ADP-ribose) polymerase. In addition, i.p. treatment with CPT (100 mg/kg) of Balb/C nude mice xenografted subcutaneously with Bel-7404 decreased the antiapoptotic protein Bcl-2 and increased the level of the proapoptotic Bax protein. 19
Concomitant with the improvement of biochemical markers, the histological examination of solid EC tumors following treatment with AP and/or CPT of irradiated and non-irradiated EC-bearing mice revealed noticeable amelioration in the cellular architecture of muscle bundles, in addition to the reduction of tumor cell infiltration and enhancing tumor cell apoptosis.
In conclusion, AP and CPT, as natural agents, demonstrated antitumor abilities in the current solid tumor–bearing murine model through the downregulation of fundamental molecular targets involved in proliferation, inflammation, angiogenesis, and tumor invasiveness, with ultimate induction of apoptosis. In addition, both compounds demonstrated a synergistic potential to enhance the sensitivity of tumor cells to ionizing radiation, and their combined administration may represent a potential therapeutic strategy for enhancing the response of solid tumors to radiations. Furthermore, the combined use of AP and/or CPT together with γ-irradiation may represent an effective way to decrease the dose of radiation taken.
Footnotes
Acknowledgements
The authors would like to acknowledge the histological evaluation provided by Dr Adel M. Bakeer, Professor of Pathology, Faculty of Veterinary Medicine, Cairo University, Egypt.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
