Abstract
Various epidemiological and preclinical studies have already established the cancer chemopreventive potential of vanadium-based compounds. In addition to its preventive efficacy, studies have also indicated the abilities of vanadium-based compounds to induce cell death selectively toward malignant cells. Therefore, the objective of the present investigation is to improve the therapeutic efficacy and toxicity profile of an alkylating agent, cyclophosphamide, by the concurrent use of an organovanadium complex, vanadium(III)-
Keywords
Introduction
In this horizon of 21st century, cancer has become one of the alarming diseases to human civilization throughout the world. In spite of advancement of other treatment approaches, chemotherapy plays the most crucial role in the management of various forms of malignancy. However, failure to conventional chemotherapeutic regimen occurs due to the development of drug resistance and severe adverse effects. 1 Cyclophosphamide is one of the most commonly used chemotherapeutic drugs. But, its therapeutic application is limited due to severe organ toxicity and acquired drug resistance. 2 Regardless of that, cyclophosphamide remains a standard component of treatment regimens for different forms of leukemia, breast cancer, Hodgkin’s and non-Hodgkin’s lymphoma, multiple myeloma, ovarian cancer, neuroblastoma, and retinoblastoma. 2 Therefore, there is a real need to develop new sensitizing agents that can selectively enhance the cytotoxicity of cyclophosphamide toward cancer cells.
Vanadium is an important ultra-trace element which is widely distributed in nature as well as prevalent in the human body.3,4 In addition, it has received special attention in the pharmacological research, including the regulation of intracellular signaling, as a cofactor of enzymes essential in energy metabolism, as an alternative therapeutic agent for the treatment of diabetes mellitus, and as a potential cancer chemopreventive agent owing to its unique biological functions.3,5 From the toxicological point of view, it is found that no obvious toxic effects were reported following vanadium supplementation in humans. 4 Recently, vanadium complexes as anticancer agents have received increasing attention. This notion is based on the observations that vanadium-based compounds can specifically induce cell death in malignant cells at dose levels non-toxic to non-malignant cells.6,7 Vanadium-based compounds exert their antineoplastic effects through several mechanisms which include selective induction of apoptosis in cancer cells over normal cells, 6 halting the cell cycle progression of the proliferating cancer cells, 8 blocking of neovascularization, 9 and inhibition of metastasis. 10 Apart from its antitumor activity, few studies have also shown that vanadium-based compounds can provide adequate protection to the host from chemotherapeutic drug-induced toxicity.11,12
Previously, we have reported the synthesis, toxicity profile, and protective efficacy of an organovanadium compound, namely, vanadium(III)-

(a) Chemical structure of vanadium(III)-
Materials and methods
Reagents and antibodies
VC-III was synthesized following the literature procedure of Papaioannou et al.
14
Primary antibodies against p53, Bax, Bcl-2, cytochrome
Animals
Adult (6–8 weeks) female Swiss albino mice (25 ± 2 g), bred in the animal colony of Chittaranjan National Cancer Institute (Kolkata, India) were used for this study. The mice were maintained at controlled temperature (22 ± 2°C) and humidity (60 ± 5%) under alternating light and dark conditions (12 h/12 h). Standard food pellets (EPIC rat and mice pellet from Kalyani Feed Milling Plant, Kalyani, West Bengal, India) and drinking water were provided ad libitum. The experiments were carried out strictly following the guidelines of Institutional Animal Ethics Committee (Committee for the Purpose of Control and Supervision of Experiment on Animals (CPCSEA; Registration No. 1774/GO/RBi/S/14/CPCSEA), India).
Tumor cells
Murine breast adenocarcinoma cell line (Ehrlich ascites carcinoma (EAC)) was maintained in Swiss mice by weekly intraperitoneal transplantation of 1 × 106 viable tumor cells suspended in isotonic phosphate-buffered solution (PBS).
Experimental design
Animals were distributed into seven groups. Vehicle control (VC) group contained six animals and other groups (tumor inoculated) contained twelve animals each. Six animals from the later groups were kept to check the survivability, while six animals from each group were taken for studies at molecular and biochemical level. Animals of each group (except VC) were injected with tumor cells (1 × 106 cells/mouse) intraperitoneally. Generally the EAC cell line has the doubling time of 18–24 h. 15 So, no treatment was given on the day of tumor cell inoculation and it was counted as “day 0.” Mice were distributed in the following groups (Figure 1(b)):
VC group: Healthy mice were treated with the vehicle, that is, distilled water orally from day 1 to day 10.
EAC control group (E): EAC cell inoculated mice were treated with distilled water orally from day 1 to day 10.
Only VC-III concomitant treated group (ED): Tumor inoculated mice were treated only with VC-III (1 mg/kg body weight, per os) 24 h after tumor inoculation from day 1 to day 10.
Only VC-III pretreated group (PED): The mice were pretreated with VC-III (1 mg/kg body weight, per os) 7 days prior to tumor inoculation, and following tumor inoculation, the treatment was again continued from day 1 to day 10.
Only cyclophosphamide-treated group (EC): Tumor inoculated mice were treated with cyclophosphamide (25 mg/kg body weight, intraperitoneal) and distilled water orally from day 1 to day 10.
Concomitant treatment combination group (ECD): VC-III (1 mg/kg body weight, per os) along with cyclophosphamide (25 mg/kg body weight, intraperitoneal) was administered 24 h after tumor inoculation from day 1 to day 10.
Pretreatment combination group (PECD): The mice were treated with VC-III (1 mg/kg body weight, per os) for 7 days prior to tumor inoculation, and following tumor inoculation, the treatment was continued from day 1 to day 10 along with cyclophosphamide (25 mg/kg body weight, intraperitoneal).
The mice of all the groups were sacrificed on day 11 except the mice reserved for survivability study. All the experiments were repeated twice.
Tumor growth response and survivability
The antitumor efficacy of VC-III was assessed by measuring the inhibition in ascites tumor volume, packed cell volume, and viable tumor cell count by trypan blue dye exclusion method. Mean survival time (MST) of each group was monitored by recording the mortality daily and was expressed by Kaplan–Meier method. The end point of the experiment was determined by spontaneous death of animals and MST was calculated according to the equation: MST = (day of first death + day of last death)/2. The percentage of ILS (increase in life span) was calculated using the equation: ILS% = (T − C) / C × 100, where T represents MST of treated animals and C represents MST of tumor control group. T/C% (treated vs tumor control) was calculated as MST of treated animals/MST of tumor control group. TIR% (tumor-growth inhibition rate) = (A − B) / A × 100, where A represents mean tumor volume of tumor control group and B represents mean tumor volume of treated group. According to the criteria of National Cancer Institute (NCI), T/C > 125% and ILS > 25% indicate that the drug has a significant antitumor activity. 16
Detection of apoptosis by Annexin V positivity
Double staining for Annexin V–FITC and PI was performed using FITC Annexin V Apoptosis Detection Kit I (BD Pharmingen) according to the manufacturer’s protocol. Briefly, tumor cells were collected in PBS, centrifuged, and washed. Then, the cells were resuspended in Annexin V binding buffer and after that Annexin V–FITC and PI were added according to the manufacturer’s instructions. The cells were incubated for 15 min in the dark at room temperature. 17 The cells were acquired in a flow cytometer (BD FACSCalibur; BD Biosciences, Franklin Lakes, NJ, USA). The data were evaluated by FLOWJO, LLC, Data Analysis Software (Ashland, OR, USA).
Mitochondrial membrane potential (ψ) assay
Mitochondrial membrane potential (ψ) was measured by the uptake of JC-1 which is a dye that stains mitochondria in a membrane potential–dependent manner. 1 × 106 tumor cells were harvested in PBS containing JC-1 (5 µg/mL). The samples were incubated at 37°C for 15 min in dark, washed twice with PBS, and then analyzed by fluorescence measurement through spectrofluorimeter (Cary Eclipse; Varian, Mulgrave, Victoria, Australia) using 490 and 530 nm as excitation and emission wavelengths, respectively, for JC-1 monomers, and 525 and 590 nm as excitation and emission wavelengths, respectively, for JC-1 aggregates. The ratio of the reading at 590 nm to the reading at 530 nm (aggregate/monomer) was considered as the relative mitochondrial membrane potential (ψ) value. 18
Immunofluorescence
The immunocytochemical expression of key regulators associated with p53-mediated mitochondrial apoptotic pathway, such as, p53, Bax, Bcl-2, cyt
Immunoblotting
Tumor cells were lysed and whole cell protein was extracted using extraction buffer containing Tris–HCl, NaCl, ethylenediaminetetraacetic acid (EDTA), NaH2PO4, 1% Triton X-100, ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid (EGTA; pH 8.0), and protease inhibitor cocktail (1 mM phenylmethylsulfonyl fluoride, 1 mM sodium orthovanadate, 1 mM dithiothreitol, 50 mM sodium fluoride, 1 µg/mL aprotinin, and 1 µg/mL leupeptin). The lysate was then centrifuged at 14000
Angiogenesis study with blood vessels
To study the role of VC-III on tumor angiogenesis, the mice were sacrificed and the skins were removed carefully from peritoneal region without disturbing the angiogenic vessels. These blood vessels were counted macroscopically using convex lens, and the angiogenic index was calculated by number of blood vessels per mouse for each groups.
Immunoassay
The levels of VEGF-A and matrix metalloproteinase 9 (MMP-9) were measured in the ascites fluid of tumor-bearing mice using quantitative sandwich enzyme immunoassay technique (ELISA Kit) according to the manufacturer’s protocol.
Detection of DNA damage
Qualitative and quantitative measure of DNA damage was carried out by single-cell gel electrophoresis or comet assay. 22 Briefly, tumor cells and bone marrow cells were layered over a frosted microscopic slide previously coated with a layer of 1.0% normal melting agarose. The slides were then immersed in a lysis buffer of pH 10 and left overnight. Slides were transferred into a horizontal electrophoresis chamber containing alkaline solution (300 mM NaOH and 1 mM Na2EDTA; pH 13.0). A presoaking of 20 min was done in order to unwind the DNA. Electrophoresis was then carried out for 20 min (300 mA, 20 V). Slides were then washed thrice with neutralizing buffer (Tris buffer 0.4 M, pH 7.5) followed by staining with EtBr (final concentration 20 µg/mL). Finally, the slides were examined under a fluorescence microscope (DM4000 B; Leica) and subjected to image analysis using comet assay software program Komet 5.5 (Andor Technology, Concord, MA, USA). To avoid any selection bias, at least 100 cells from each sample were counted to determine different parameters such as damaged cell (%), comet tail length, and olive tail moment.
Determination of reactive oxygen species generation
Intracellular levels of reactive oxygen species (ROS) was measured in tumor cells and liver tissue homogenate using DCFH-DA probe.23,24 Briefly, tumor cells (1 × 107) and liver tissue homogenate (10% w/v) were incubated with 10 µM DCFH-DA for 30 min at 37°C. The levels of ROS were measured using spectrofluorimeter (Cary Eclipse; Varian; with an excitation set at 485 nm and emission at 529 nm) to estimate the conversion of non-fluorescent DCFH-DA to the highly fluorescent compound 2′,7′-dichlorofluorescein (DCF).
Measurement of nitric oxide production
The production of nitric oxide (NO) in tumor cells and liver tissue homogenate was estimated by estimating the level of stable NO metabolites, namely, nitrate (NO3−) and nitrite (NO2−) ions by reaction with Griess reagent using NaNO2 as standard.24,25 The absorbance was taken at 545 nm using the ultraviolet (UV)–visible spectrophotometer (Infinite® 200 PRO, TECAN, Männedorf, Switzerland) and expressed as micromolar nitrite per milligram of protein.
Estimation of lipid peroxidation
Spectrophotometric method was applied to estimate the level of lipid peroxidation (LPO) in liver microsomes by estimating the formation of thiobarbituric acid reactive substances (TBARS) using thiobarbituric acid (TBA; 0.8%) and expressed as nanomolar TBARS formed per milligram of protein using an extinction coefficient of 1.56 × 105/M/cm/ at 532 nm. 26
Antioxidant status
The level of reduced glutathione (GSH) was estimated in liver cytosol spectrophotometrically by the determination of dithiobis(2-nitro)-benzoic acid (DTNB) reduced by –SH groups by measuring the absorbance at 412 nm. The level of GSH was expressed as nanomolar per milligram of protein. 27
The activity of superoxide dismutase (SOD) was determined in liver cytosol by monitoring the auto-oxidation of pyrogallol (20 mM) at 420 nm and expressed as unit per milligram of protein. 28
The activity of catalase (CAT) in liver cytosol was quantified spectrophotometrically at 240 nm using H2O2 as the substrate and expressed as unit per milligram of protein. 29
The activity of glutathione peroxidase (GPx) in liver cytosol was estimated by nicotinamide adenine dinucleotide phosphate (NADPH) oxidation using a coupled reaction system consisting of GSH (10 mM), glutathione reductase (2.4 unit), and H2O2 (12 mM) and expressed as micromolar NADPH utilized per minute per milligram of protein using extinction coefficient of NADPH at 340 nm as 6200/M/cm/. 30
Glutathione-S-transferase (GST) activity in kidney cytosol was assessed from the increase in absorbance at 340 nm with 1-chloro-2,4-dinitrobenzene (CDNB; 30 mM) as the substrate, and specific activity of the enzyme was expressed as formation of CDNB–GSH conjugate per minute per milligram of protein. 31
Protein determination
Protein content in liver tissue homogenate was measured by Lowry et al.’s 20 method with BSA as the standard using spectrophotometer (Infinite® 200 PRO).
Histopathology
After 24 h of fixation, the tissue samples were dehydrated in ascending concentrations of ethanol, cleared in xylene and embedded in paraffin to prepare the blocks. Liver tissues were sectioned at 5 µm, mounted on slides, stained with hematoxylin and eosin (H&E), and observed under light microscope (DM 1000; Leica).
Hepatotoxicity markers
The serum activity of alanine transaminase (ALT), aspartate transaminase (AST), and alkaline phosphatase (ALP) were measured spectrophotometrically by standard methods using commercial kits (Span Diagnostics Ltd., Surat, India).
Chromosomal aberration from bone marrow cells
Cells arrested in metaphase were examined microscopically (DM 1000; Leica) for structural chromosomal aberrations. Mice were injected with 0.03% colchicine (1 mL/100 g body weight, intraperitoneal) and were kept for 90 min prior to sacrifice. Bone marrow preparations for the analysis of chromosome aberrations were carried out according to the technique of Ford and Hamerton 32 followed by Giemsa staining (5:24 dilution in Sorenson’s phosphate buffer). Only well-spread metaphase presenting chromosomes were analyzed. At least 100 metaphase plates per animal (over 600 for each group) were screened to obtain the total number of chromosomal aberrations.
Hematological studies
Hemoglobin (Hb) content of blood sample was measured following Sahli’s method. Red blood cell (RBC), white blood cell (WBC), and bone marrow cell counts were made following a literature procedure. Differential WBC count was carried out from Leishman stained blood smears. 24
Statistical analysis
All data were expressed as mean ± standard deviation (SD),
Results
VC-III reduced tumor burden and enhanced survivability of tumor-bearing mice
Treatment of tumor-bearing mice with VC-III (ED and PED) significantly (
Effect of VC-III during monotherapy or in combination with cyclophosphamide on the tumor burden and survivability of tumor-bearing mice.
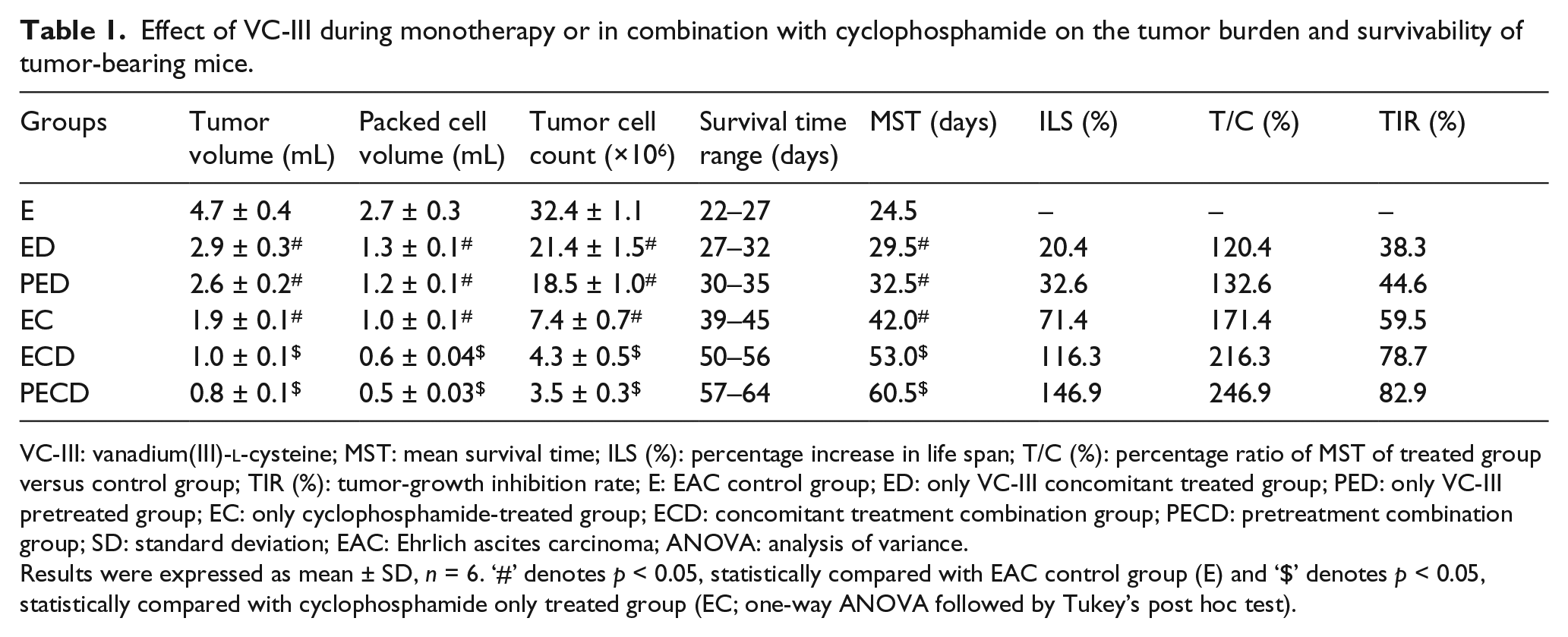
VC-III: vanadium(III)-
Results were expressed as mean ± SD,
VC-III intensified generation of ROS and NO by cyclophosphamide in tumor cells
Reactive oxygen and nitrogen free radicals act as important signaling and effector molecules, and can be cytotoxic.
33
Tumor cells have been reported to have inherently higher levels of ROS and NO in comparison to normal cells and could be expected to have greater sensitivity to oxidative and nitrosative assault.
34
Accordingly, we measured the levels of ROS and NO in tumor cells following different treatments. As shown in Figure 2(a), after treatment with VC-III (ED and PED), a schedule dependent increase (

Modulation of oxidant status and induction of mitochondrial membrane depolarization and DNA damage in tumor cells by VC-III. (a) Treatment of VC-III in combination with cyclophosphamide enhanced the levels of ROS and NO in tumor cells. (b) Photomicrographs showing comet in tumor cells from different groups of mice (EtBr staining, 400× magnification). Tumor control group (E) showed intact DNA with no tail. Concomitant and pretreatment with VC-III (ED and PED) was able to induce DNA damage in tumor cells as shown from the tail length and Olive tail moment. Cyclophosphamide treatment (EC) alone showed prominent tail length with less percentage of DNA in the head and greater in the tail. Combination treatment with VC-III and cyclophosphamide (ECD and PECD) showed more migrated DNA with severely damaged tail. (c) Effect of VC-III on mitochondrial membrane potential in tumor cells. Reduction of JC-1 fluorescent ratio (aggregate/monomer) by VC-III indicated induction of depolarization of mitochondrial membrane. Results were expressed as mean ± SD,
VC-III enhanced the cyclophosphamide-induced DNA damage in tumor cells
Single-cell gel electrophoresis or comet assay was performed to assess the DNA damage induced by free radicals as a result of oxidative stress following treatment with VC-III and cyclophosphamide (Figure 2(b)). The results showed that both pretreatment and concomitant treatment with VC-III could cause DNA strand breaks as indicated by higher frequency (
VC-III induced depolarization of mitochondrial membrane potential
The loss of mitochondrial membrane potential is a characteristic feature of apoptosis which can be measured using JC-1, as the ratio of J-aggregates/monomers, which serves as an effective indicator of the cellular mitochondrial transmembrane potential, and this allows apoptotic cells to be easily distinguished from their non-apoptotic counterparts.
18
A decrease (
VC-III increased externalization of phosphatidylserine
To understand the nature of cell death, double labeling techniques using Annexin V–FITC/PI was performed (Figure 3). In tumor control group (E), there were very small amount of dead cells, in contrast to very large amount of double negative live cells. The population of Annexin V–bound cells were increased steadily (

Induction of apoptosis and cell cycle arrest by VC-III. (a) Externalization of phosphatidylserine by VC-III. Increase in population of cells in second and third quadrant indicated high incidence of apoptosis in tumor cells by VC-III. The figure is a representative profile of at least three experiments in duplicate. (b) Percentage of apoptosis observed in tumor cells following different treatments. The figure is a representative profile of at least three experiments in duplicate.
VC-III activated p53 and intensified intrinsic apoptotic pathway in tumor cells
As a tumor suppressor gene, p53 plays a pivotal role in DNA damage and apoptosis and acts as a key regulator of cellular death mechanism.
35
Therefore, we compared the expression of p53 after monotherapy and combination therapy against the untreated control group (Figures 4 and 5). VC-III treatment (ED and PED) moderately increased the expression of p53; however, combination treatment groups (ECD and PECD) showed more pronounced effect. Further analysis of mitochondrial pathway of apoptosis revealed that VC-III treatment decreased the expression of prosurvival proteins Bcl-2 and moderately increased the expression levels of proapoptotic Bcl-2 family protein, Bax.
36
Thus, VC-III induced release of cyt

Regulation of p53-mediated mitochondrial apoptotic pathway by VC-III. (a) Immunofluorescence staining of tumor cells showing p53, Bax, Bcl-2, cytochrome
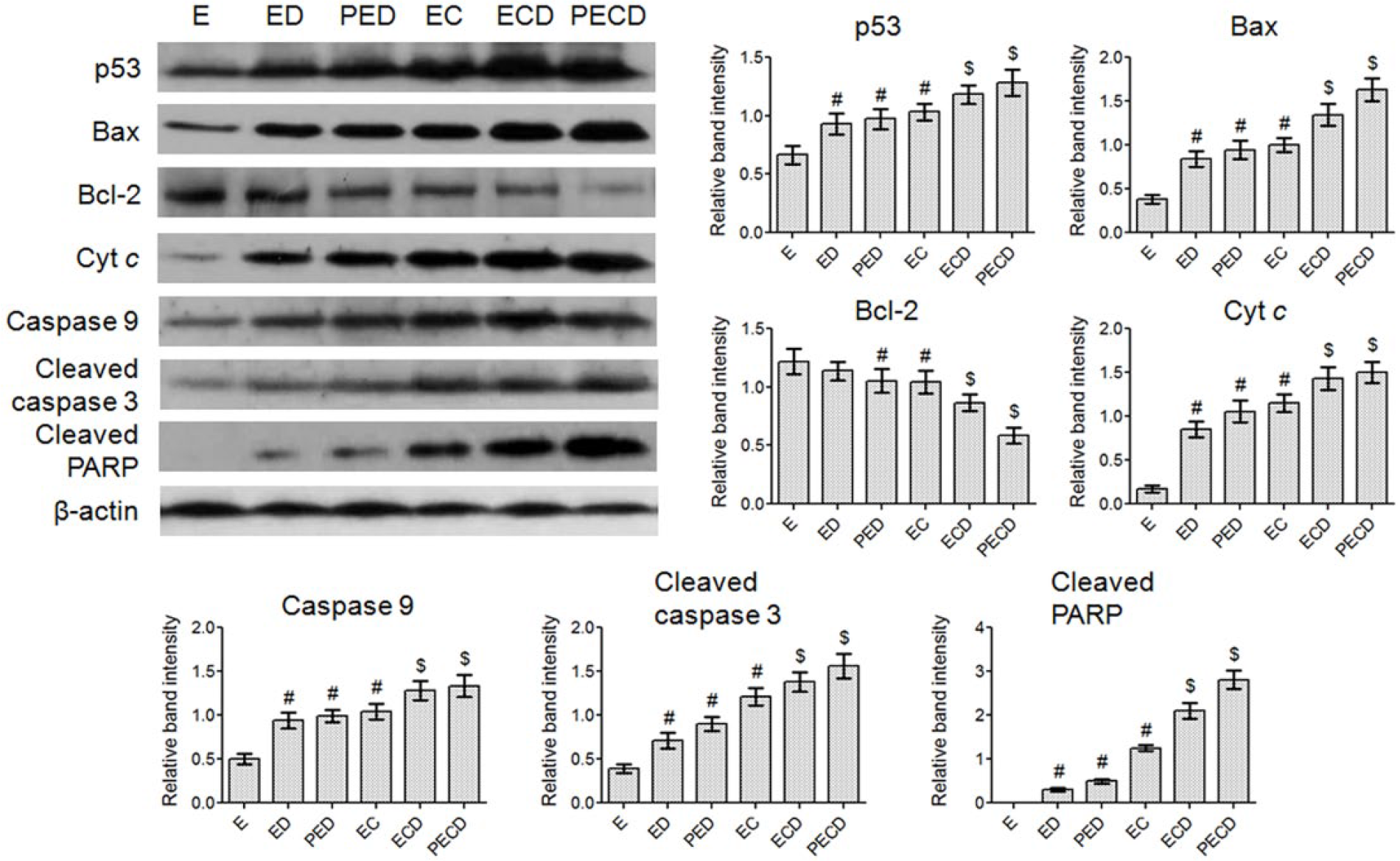
Western blot analysis was carried out to detect the expression profiles of p53, Bax, Bcl-2, cytosolic cytochrome c, cleaved caspase-3, and cleaved PARP in tumor cell lysates of treated and untreated mice. β-actin was used as internal reference. The bar diagrams show relative protein expression from western blot analysis. The data were normalized with β-actin expression. Values were expressed as mean ± SD,
VC-III could efficiently block angiogenesis and lowered the levels of proangiogenic mediators
The use of vascularity as a measure for angiogenesis is widely accepted. Here, we observed that the peritoneum of mice bearing EAC cells, when compared to that of normal mice, was rich in blood vessels (Figure 6(a) and (b)) due to angiogenesis. This enables the delivery of oxygen and nutrients essential for growth and metastasis of tumor. However, reduction in sprouting due to inhibition of the neovasculature in peritoneum was observed in mice that were treated with VC-III during concomitant and pretreatment schedule in both monotherapy (ED and PED) and combination therapy (ECD and PECD; Figure 6(a) and (b)). Moreover, the observed inhibition of angiogenesis was also attributed to VC-III-mediated inhibition of the levels of VEGF-A and MMP-9 in the ascitic fluid, which was more prominent in combination treatment groups (ECD and PECD; Figure 6(c)).

Inhibition of peritoneal neovascularization and angiogenic markers. (a) Peritoneal lining of tumor control group (E) shows sprouting of new capillaries and leaky blood vessels of the peritoneal membrane. Treatment with VC-III and cyclophosphamide showed reduction in the peritoneal vasculature and normalization of blood vessels. (b) Angiogenic index (number of blood vessels/mouse) in different treatment groups. (c) Effect of VC-III on the levels of VEGF-A and MMP-9 in ascites fluid of tumor-bearing mice. Results were expressed as mean ± SD,
VC-III ameliorated hematological parameters in tumor-bearing mice
Hematological parameters of tumor-bearing mice were altered significantly (
Hematological parameters, bone marrow cellularity, and hepatotoxicity markers in tumor-bearing mice following treatment with cyclophosphamide and VC-III as monotherapy and combination therapy.

VC-III: vanadium(III)-
Results were expressed as mean ± SD,
VC-III protected tumor-bearing mice from cyclophosphamide-induced hepatotoxicity
The hepatoprotective efficacy of VC-III was confirmed through estimation of hepatic function markers and liver histopathological examinations. Administration of cyclophosphamide to tumor-bearing mice elevated (
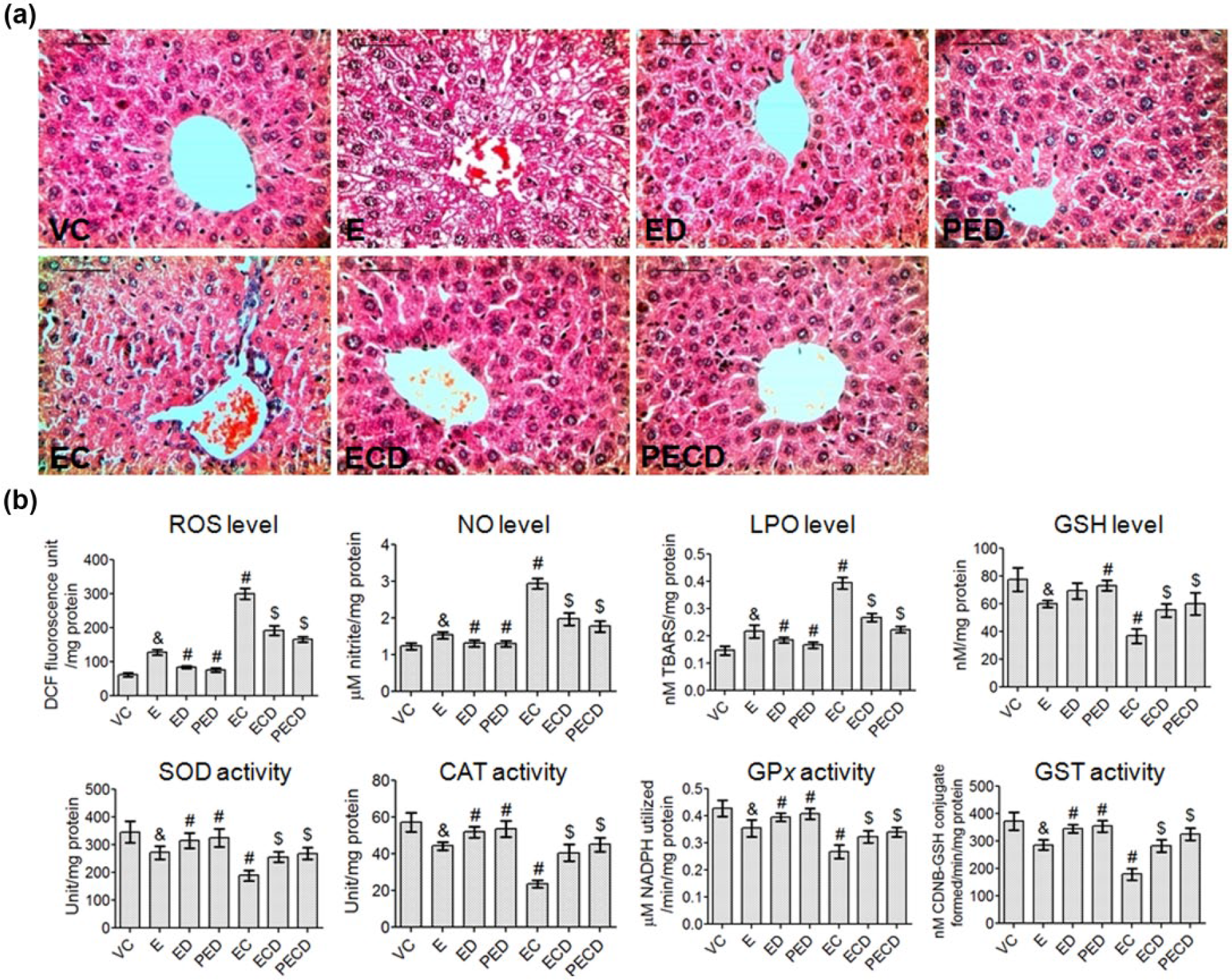
VC-III reduced cyclophosphamide-induced hepatotoxicity through attenuation of oxidative stress and enhancement of the activity of antioxidant enzymes. (a) Histopathological analysis of liver sections from different groups of mice (H&E staining, 400× magnification). Liver of mouse receiving vehicle (VC) showing normal hepatocellular architecture depicts hepatocytes radiating from the central vein. Tumor control mice (E) show microvacuolization, vacuolization of cytoplasm, pyknotic nuclei, and congestion. Concomitant treatment (ED) and pretreatment (PED) with VC-III reveals nearly the similar histological profile as in vehicle control (VC) group with no marked hepatotoxicity. Treatment with cyclophosphamide (EC) shows central vein dilatation, sinusoidal dilatation, leukocyte infiltration, congestion, and microvacuolization. Treatment with VC-III together with cyclophosphamide (ECD and PECD) markedly reduced the severity of the cyclophosphamide and tumor-induced hepatic damage. (b) Effect of VC-III on the cyclophosphamide-induced changes in the levels of prooxidant (ROS, NO, and LPO) and antioxidant (GSH) and in the activity of antioxidant enzymes (SOD, CAT, GP
VC-III significantly mitigated cyclophosphamide-induced alterations of hepatic redox status
Figure 7(b) represents the effect of VC-III on the cyclophosphamide-induced changes in the hepatic prooxidant levels and the activity of antioxidant enzymes in the liver tissues of all experimental mice. Cyclophosphamide treatment (EC) increased (
VC-III prevented cyclophosphamide-induced genotoxicity in bone marrow cells
Mice treated with cyclophosphamide (EC) showed appreciably significant (

Effect of VC-III on cyclophosphamide-induced genotoxic damage in murine bone marrow cells (a) Representative photomicrograph of metaphase chromosomes of bone marrow cells showing various types of structural aberrations (Giemsa staining, 1000× magnification). Arrows indicate stretching, break, gap, constriction, and terminal deletion. To the right, bar diagram shows percentage of chromosomal aberration of bone marrow cells (600 metaphase plates for each group was counted) in different groups. (b) Photographs showing comet of bone marrow cells in each group (EtBr staining, 400× magnification). Quantified representations of comet assay parameters revealed that treatment with VC-III mitigated damage cell (%), comet tail length (µm), and Olive tail moment. Results were expressed as mean ± SD,
Discussion
The success of cancer therapy with chemotherapeutic drugs used alone or in combination is limited due to lack of therapeutic selectivity, heterogeneity of tumor bed, resistance acquired by the cancer cells toward the therapy, reduction in the efficacy of the drug delivery, and drug-induced toxicity. 38 In this regard, this study investigated the potential role of an organovanadium compound, VC-III, in sensitizing tumor cells to chemotherapy-induced cytotoxicity. In this study, VC-III itself showed promising chemopotentiating efficacy in tumor-bearing mice as evident from suppression of tumor burden and prolongation of host survivability, which is one of the important criteria to evaluate the potency of anticancer drugs. 16 VC-III in combination with cyclophosphamide substantially enhanced the life span of tumor-bearing mice compared to the cyclophosphamide monotherapy, which reflects the potential advantage of this combination in chemotherapy. Although the precise molecular mechanisms by which VC-III modulates cyclophosphamide therapy are not fully understood at this moment, a number of factors which could have an impact are discussed herein.
Evasion of apoptosis is one of the hallmarks of cancer. It is responsible for tumor promotion and progression as well as for treatment resistance. 39 An apoptotic stimulus causes externalization of phosphatidylserine, detectable by increased binding of Annexin V. 17 VC-III alone and in combination with cyclophosphamide affected externalization of phosphatidylserine, corroborating that VC-III exerts its cytotoxic activity primarily via apoptosis. Thus, the induction of apoptosis by VC-III is probably playing a major role in sensitizing tumor cells to cyclophosphamide-induced cytotoxicity which in turn improved the efficacy of the combination therapy.
There is mounting evidence to suggest that enhanced generation of ROS plays an important role in cancer biology. In cancer cells, the basal levels of ROS are higher, and if an oxidative assault beyond a critical threshold is mounted, it actually leads to an imbalance in the redox homeostasis and can translate into apoptosis.33,34 In this study, concurrent administration of VC-III with cyclophosphamide intensified generation of ROS and NO in tumor cells. Thus, this oxidative assault contributed toward the redox imbalance in tumor cells necessary to induce apoptosis. Furthermore, we investigated whether DNA damage was associated with the observed apoptosis of tumor cells. In this study, we found that VC-III along with cyclophosphamide caused a significant elevation in the magnitude of damaged DNA of the tumor cells. The observed enhancement in DNA damage of tumor cells was associated with the cytotoxic efficacy of the combined treatment and thus sensitized tumor cells toward apoptosis.
To further investigate the underlying molecular mechanism of apoptosis, proteins involved in apoptotic pathways were analyzed by immunofluorescence and western blot techniques. The tumor suppressor protein p53 plays an integral role in apoptosis.
35
We observed that the apoptosis induced by VC-III was initiated through the upregulation of p53. Upon activation, p53 can abrogate survival signals and induce apoptotic/antiproliferative effect via activation of the mitochondrial pathway. This mitochondrial pathway included inactivation of antiapoptotic protein Bcl-2, translocation of apoptotic protein Bax to mitochondria, and release of cyt
Inhibition of angiogenesis by VC-III is another major effector mechanism in potentiating the therapeutic efficacy of cyclophosphamide. Herein, the angiostatic property of VC-III was established by the considerable minimization of the neovascularization in the peritoneum lining of the tumor-bearing mice. Angiogenesis also plays a critical role in tumor growth and metastasis. 39 VEGF-A and MMP-9 play most potent and predominant role in angiogenesis through stimulation of vascular endothelial cell growth, survival, and proliferation. 40 The results showed that administration of VC-III caused significant reduction in the levels of VEGF-A and MMP-9 in the ascites fluid of EAC cell–bearing mice, which actually represents the microenvironment of the tumor cells. Various reports demonstrated that antiangiogenic compounds normalize the blood flow in dilated, chaotic, hemorrhagic, and leaky tumor vasculatures, ensuring proper homogeneous delivery of chemotherapeutic drugs to the tumor bed.38,39 Thus VC-III during combination therapy might be able to normalize the tumor vasculature and at the same time allowing cyclophosphamide to extravagate and traverse to different parts of the tumor.
Apart from the influence of VC-III on chemotherapy of tumor cells, we have also checked the protective effect of the compound on chemotherapy-associated toxicity to host organs and tissues. Malignancy and its corresponding chemotherapy inevitably lead to the reduction in bone marrow function which is reflected by a substantial decrease in the mature blood elements. 41 Chemotherapy with cyclophosphamide caused further decrease in the number of WBC, RBC, bone marrow cell counts, and hemoglobin content. VC-III effectively prevented tumorigenesis-induced and chemotherapy-induced alterations of these parameters, when applied either alone or in combination with cyclophosphamide in tumor-bearing animals. We also found that VC-III quite effectively mitigated cyclophosphamide-induced chromosomal aberration and DNA damage in murine bone marrow cells, thus inhibiting the probability of chemotherapy-induced secondary carcinogenesis.
Hepatic dysfunction is the most common regimen-related toxicity reported in patients treated with cyclophosphamide. 13 Hepatic activation of cyclophosphamide leads to the formation of toxic metabolites causing damage to the liver tissues. Due to such damage, the enzymes such as ALT, AST, and ALP leak from the damaged liver to the serum, 41 as found in this study in cyclophosphamide-treated tumor-bearing mice, suggesting hepatic injury. Treatment with VC-III restored these increased activity of serum ALT, AST, and ALP, which indicates the protective activity of the compound toward liver. Relationships between tissue injury, genotoxicity, chromosomal instability, and oxidative stress have been well demonstrated in many experimental animal models. 41 Treatment with cyclophosphamide leads to marked disruption in cellular redox homeostasis. However, the significant decrease in the levels of prooxidants coupled with the enhancement of GSH and antioxidant enzymes by the treatment with VC-III advocated its role in reducing cyclophosphamide-induced damages. The protection by VC-III was also confirmed by the histopathological evaluation of the liver. All these results together confirmed that VC-III successfully attenuated cyclophosphamide-induced systemic toxicity in the tumor-bearing host, thereby rejuvenating the intrinsic defense system of the host. As a cumulative effect of all these consequences, the tumor-bearing mice were extended with better survival advantage at this dose combination.
Thus, these results proposed that as a promising adjuvant, the organovanadium compound VC-III sensitizes cancer cells toward cyclophosphamide therapy through generation of oxidative stress in tumor cells, upregulation of p53 followed by promotion of mitochondrial caspase cascade promotion, and induction of apoptosis in tumor cells along with simultaneous protection to the host from chemotherapy-associated toxicity. Taken together, it can be said that combined treatment of the organovanadium compound and cyclophosphamide may serve as a potential strategy for sensitizing chemotherapy, and an adjuvant regimen of these two needs to be evaluated primarily in tumor xenograft models and clinical trials in future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving animals were carried out strictly following the guidelines of Institutional Animal Ethics Committee (Committee for the Purpose of Control and Supervision of Experiment on Animals (CPCSEA Registration No. 1774/GO/RBi/S/14/CPCSEA), India).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ab.B. gratefully acknowledges Indian Council of Medical Research (ICMR), New Delhi, India, for Senior Research Fellowship (No. 3/2/2/58/2011/NCD-III) and Council of Scientific and Industrial Research (CSIR), New Delhi, India, for Research Associateship (No. 09/030(0075)/2015 EMR-I). Ar.B. also gratefully acknowledges ICMR, New Delhi, India, for Senior Research Fellowship (No. 45/36/2008/PHA-BMS).
Informed consent
Informed consent was obtained from all individual participants included in the study.
