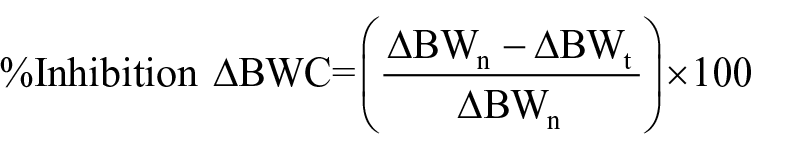Abstract
Peritoneal carcinomatosis is considered as a potentially lethal clinical condition, and the therapeutic options are limited. The antitumor effectiveness of the [Ru(
Introduction
Peritoneal carcinomatosis is a locoregional cancer widespread within the peritoneal cavity and may occur as result of metastatic primary lesion from the stomach, ovary, intestine, colorectal, and, less commonly, lung and breast. It is considered potentially lethal clinical condition.1–3 Usually, progression of the disease is associated with formation malignant ascites that causes abdominal pain and pressure, bloating, fatigue, nausea, and anorexia.4,5
The therapeutic options for peritoneal carcinomatosis are limited because complete surgical removal is not possible and systemic chemotherapy is not effective due to low concentration within the peritoneal cavity. 6 However, currently the treatment options have improved significantly. Intraperitoneal chemotherapy was introduced in treatments of peritoneal carcinomatosis to increase drug levels locally. 7
Thus, metallodrugs are being developed to help overcome the limitations of the efficacy and safety of current cancer therapies.8,9 In recent years, ruthenium complexes have attracted much attention as new transition-metal-based antitumor agents, as they have certain advantages over the platinum complexes that are currently used in cancer chemotherapy. 10 They show less toxicity, they are selective of tumor cells, and present a novel mechanism of action, the prospect of non-cross-resistance and a different spectrum of activity.11–19
In a previous study, we recently reported that complexes with the general formula [Ru(AA-H)(dppb)(bipy)]PF6 (AA-H = amino acid anion) inhibited the growth of Ehrlich carcinoma cells in vitro.
20
Among the Ru(II)/amino acid/diphosphine complexes evaluated, those with
Ehrlich carcinoma is a murine mammary adenocarcinoma, referred to as an undifferentiated carcinoma, has high transplantable capability, no-regression, rapid proliferation, shorter life span, and 100% malignancy.21,22 The ascitic form of the tumor is widely used as an experimental model for new chemotherapeutic studies. Ehrlich ascites carcinoma (EAC) is considered similar to the human tumors which are the most sensitive to chemotherapy due to its undifferentiated feature and rapid proliferation rate.22,23
Thus, the aim this study was to analyze the antitumor efficacy and side effects of the [Ru(
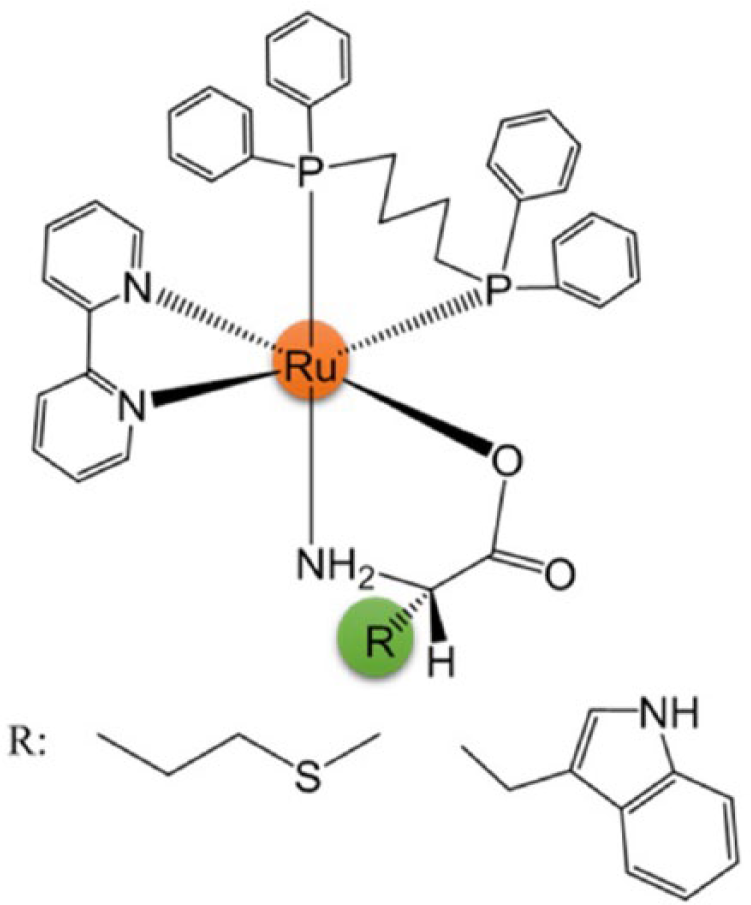
Structure of [Ru(AA-H)(dppb)(bipy)]PF6 complexes: AA = methionine
Materials and methods
Synthesis and characterization of compounds
The synthesis and characterization of [Ru(
In vivo studies
Animals
The 6–8 week-old Swiss albino mice with an average body weight of 25–35 g were used for the experiments. The animals were maintained under standard laboratory conditions (22–25°C with dark/light cycle, 12/12 h) and allowed free access to a standard dry pellet diet and water ad libitum. They were acclimatized to laboratory conditions for 5 days before the experiments.
Mouse tumor model
The Ehrlich tumor, murine mammary carcinoma, was introduced intraperitoneally (i.p.) as ascites tumors (peritoneal carcinomatosis) into the Swiss albino mice. After 7 days of cell inoculation, the peritoneal fluid of an animal with EAC was aspirated, the cells were washed in sterile phosphate-buffered saline (PBS), and an aliquot of the cell suspension was put into 1% (m/v) Trypan blue (Sigma-Aldrich, St. Louis, MO, USA) and counted in a Neubauer Chamber. Only cell dilutions with ≥90% viable cells were used for both the in vitro and in vivo studies. 24
Antitumor activity in vivo
The animals were divided into seven all-male groups of eight. They were inoculated i.p. with 6 × 106 viable EAC cells per mouse, in a volume of 0.2 mL, 24 h after the treatment was initiated. A negative control served as tumor control and received PBS, while the vehicle group was treated with PBS containing 10% dimethyl sulfoxide (DMSO; Sigma-Aldrich). A third group was treated with cisplatin, 2 mg kg−1 body weight. Solutions of complexes
where ΔACn and ΔACt are the changes in abdominal circumference from the negative and treated groups, respectively. ΔBWn and ΔBWt represent the body weight changes from the negative and treated groups, respectively.
The antitumor activity was classified according to the percentage of tumor growth inhibition: absent (when the percentage inhibition of tumor equals 0), mild (when the percentage of tumor inhibition ≤25%), moderate (when the percentage of tumor inhibition ≥ 26%–50%), and high (when the percentage of tumor inhibition ≥51%). Throughout the period, the animals were observed, and all behavioral changes and signs of toxicity or deaths were recorded during the first 4 h and then daily. 27
Hematological and biochemical parameters
After 7 days of treatment, hematological and clinical chemistry examinations were performed on all surviving animals with saline solution, complexes
Histopathological examination
The liver and kidneys were removed, washed with 1× PBS and fixed in 10% (v/v) formalin. All tissues were embedded in paraffin blocks, sectioned into 5 µm in thickness and placed on glass slides. After hematoxylin–eosin staining, the slides were observed and photos were taken using an optical microscope (Carl Zeiss Axiovert w, Germany) containing AxioVision 4.8 software (Carl Zeiss).
Evaluation of the apoptosis using Annexin V–fluorescein isothiocyanate double staining by flow cytometry
After treatment, the ascitic fluid collected from the peritoneal cavity of the animals was used to determine the cell death of EAC cells by flow cytometer. The population of interest was selected, and the cells undergoing apoptosis were determined by quantification of phosphatidylserine exposure on the cell surface using staining with fluorescein isothiocyanate (FITC)-conjugated Annexin V and propidium iodide (PI; BD Biosciences, San Diego, CA, USA). A volume of 5 × 105 cells were stained with 2.5 µL of FITC-conjugated Annexin V plus 3 µL of PI (10 mg mL−1) for 15 min at room temperature in the dark. Cells were analyzed using a flow cytometer (FACSCalibur™; BD Biosciences) acquiring 10,000 events/sample using Cell Quest software. The criteria for positivity in cells at the early stages of apoptosis were Annexin V positive and PI negative, whereas the criteria for cells at the later stages of apoptosis were both Annexin V and PI positive. The criteria for cells in necrosis were Annexin V negative and PI positive. Animals which did not have ascites underwent peritoneal lavage using 9% (w/v) NaCl in order to capture the tumor cells found in the abdominal cavity for the analyses.
Assessment of apoptosis and necrosis
The ascitic fluid collected from the peritoneal cavity of the animals (n = 4 per group) was washed with PBS. A volume of 2 × 105 EAC cells were suspended in Hoechst (HO) 33342 (10 μg mL−1) and PI (2.5 μg mL−1) solution and then incubated at 37°C for 10 min in total darkness. The slides were observed using a fluorescence microscope (Leica, Germany). At least 300 target cells were counted for each animal (n = 4 per group). The percentages of apoptotic (early and late apoptosis) and necrotic cells were determined from the total number of cells: (1) viable—blue chromatin with organized structure, (2) early apoptosis—bright blue chromatin that is highly condensed or fragmented, (3) late apoptosis—bright pink chromatin that is highly condensed or fragmented, and (4) necrosis—pink chromatin with organized structure.28,29
Cell cycle analyses
In order to discriminate cell cycle phase distribution after treatment in vivo, 5 × 105 cells of the ascitic fluid were harvested by centrifugation, washed with PBS, fixed with 70% (v/v) cold aqueous ethanol, and stored overnight at −20°C. The fixed cells were then washed with PBS and incubated with PI (BD Biosciences) containing 0.05% RNase. Samples were incubated at 4°C in the dark and analyzed by flow cytometry. The percentage of cells in the sub-G1, G0/G1, S, and G2/M phases were analyzed using a flow cytometer (FACSCalibur™; BD Biosciences) acquiring 10,000 events/sample using ModFit software.
Mean survival time and increased life span
Survival was determined using the same experimental procedure for the treatment of antitumor activity. The death of the mice was noted daily up to 30 days after each treatment to determine mean survival time (MST) and percentage increase in life span (% ILS). The MST of each group was monitored by recording mortality daily for 30 days. The % ILS was calculated using the following equation 30
where MST = Σ survival time (days) of each mouse in a group/total number of mice. 30 An enhancement of % ILS ≥25% in the control was considered as an effective antitumor response.
Chick chorioallantoic membrane angiogenesis assay
The CAM model was used to assess complexes
In vitro studies
Cell culture
EAC cells were aspirated from the peritoneal cavity of the animals and washed with1× PBS solution, and then maintained in a humidified atmosphere (Thermo Fisher Scientific, Waltham, MA, USA) for 24 h at 37°C in 5% CO2. RPMI-1640 medium was supplemented with 10% fetal calf serum, 100 IU mL−1 penicillin G, and 100 μg mL−1 streptomycin (Sigma-Aldrich).
Western blot assay
To evaluate whether complexes
Glucose uptake assay
To verify whether glucose uptake can be modified by the presence of complexes
Evaluation of competitive inhibition
To evaluate responses to the system L competitive inhibitor, 2-aminobicyclo-(2,2,1)-heptane-2-carboxylic acid (BCH; Sigma-Aldrich), 1 × 105 EAC cells were treated with or without 5 µM of BCH in 1N NaOH. Complexes
Statistical analysis
Data were analyzed for statistically significant experimental differences using an analysis of variance (ANOVA) followed by the Tukey or Bonferroni test. Statistical significance was considered at p < 0.05. All statistical analyses were performed using the statistical software GraphPad Prism, version 5 for Windows (GraphPad Software Inc., La Jolla, CA, USA).
Results
Peritoneal carcinomatosis inhibition and clinical symptoms
The antitumor activities of complexes
Antitumor activity of Ru(II)/amino acid/diphosphine complexes against EAC-bearing Swiss mice according to abdominal circumference and body weight.
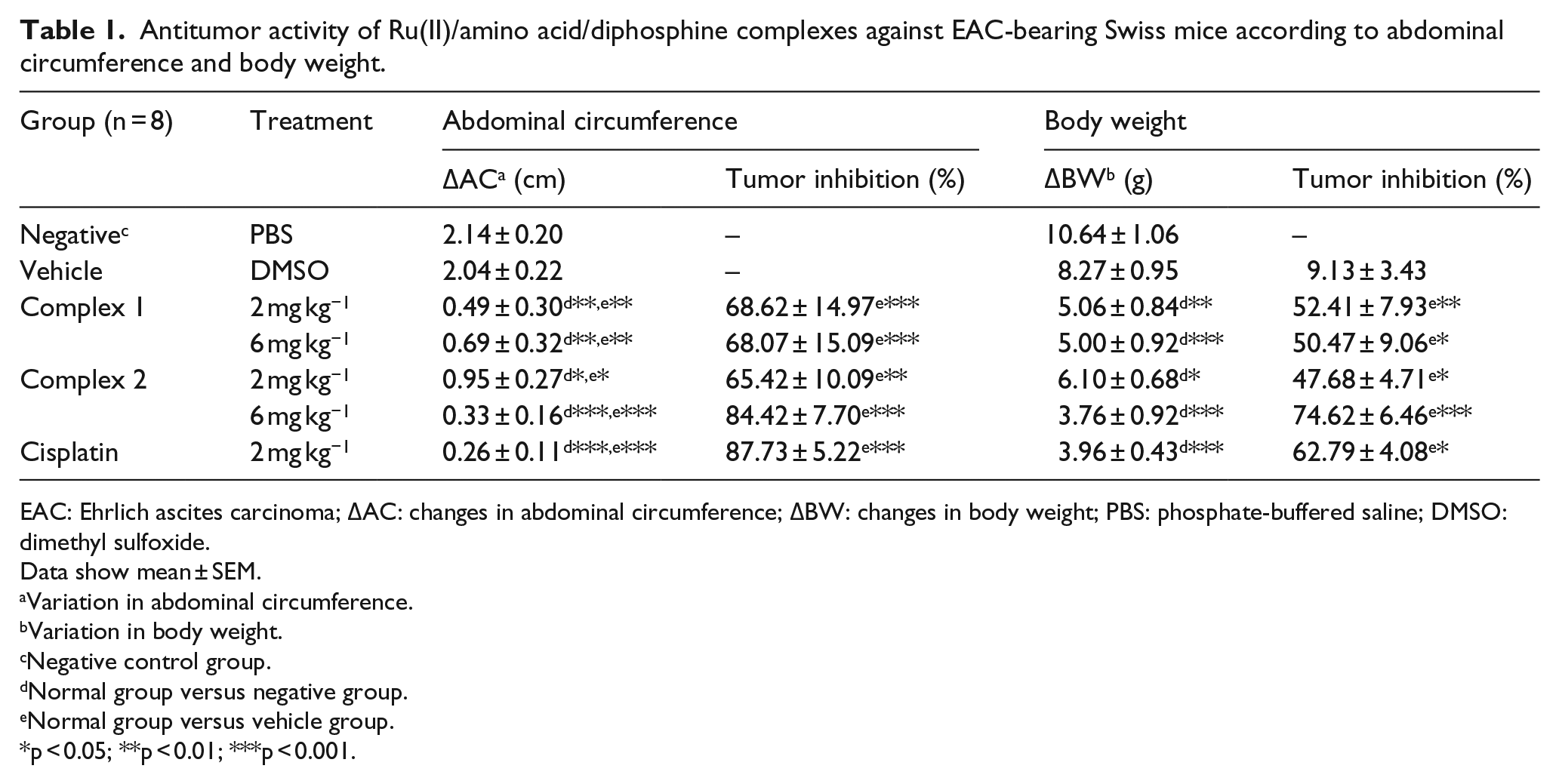
EAC: Ehrlich ascites carcinoma; ΔAC: changes in abdominal circumference; ΔBW: changes in body weight; PBS: phosphate-buffered saline; DMSO: dimethyl sulfoxide.
Data show mean ± SEM.
Variation in abdominal circumference.
Variation in body weight.
Negative control group.
Normal group versus negative group.
Normal group versus vehicle group.
p < 0.05; **p < 0.01; ***p < 0.001.
The animals treated for 7 days with complexes
Evaluation of hematological, biochemical, and histopathological parameters
Induction of the EAC changed the hematological and biochemical parameters of the mice (Tables 2 and 3). The values of erythrocytes, Hb, and HCT obtained from the negative control and vehicle groups were lower than the reference values for Swiss male tumor-free mice.
34
In addition, there were increased values of AST/SGOT and ALT/SGPT in the animals in the negative control and vehicle groups. Animals treated with complexes
Effect of Ru(II)/amino acid/diphosphine complexes on the hematological parameters in peripheral blood of EAC-bearing mice.
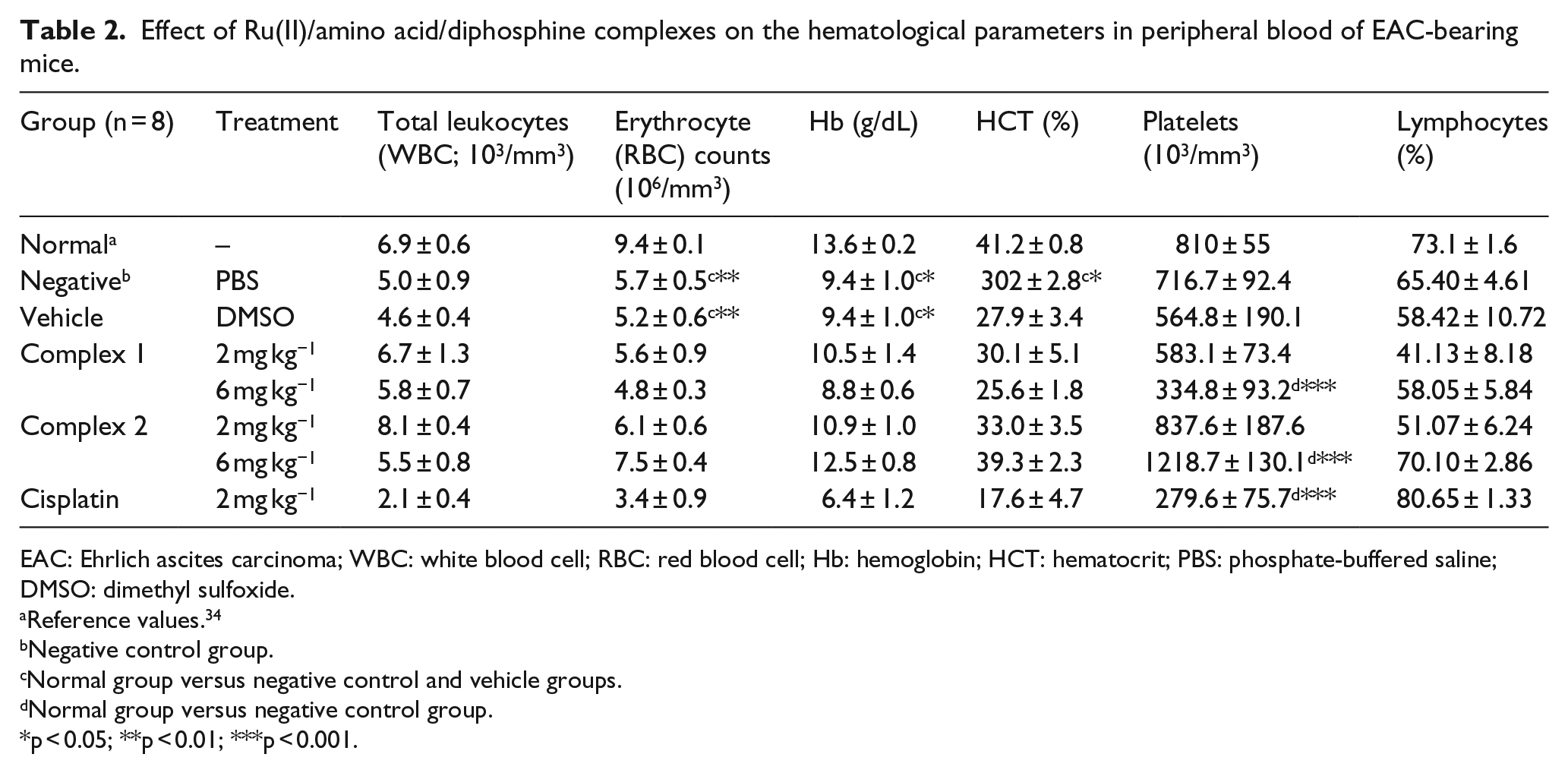
EAC: Ehrlich ascites carcinoma; WBC: white blood cell; RBC: red blood cell; Hb: hemoglobin; HCT: hematocrit; PBS: phosphate-buffered saline; DMSO: dimethyl sulfoxide.
Reference values. 34
Negative control group.
Normal group versus negative control and vehicle groups.
Normal group versus negative control group.
p < 0.05; **p < 0.01; ***p < 0.001.
Effect of Ru(II)/amino acid/diphosphine complexes on the biochemical parameters in peripheral blood of EAC-bearing mice.

EAC: Ehrlich ascites carcinoma; AST: aspartate aminotransferase; ALT: alanine aminotransferase; PBS: phosphate-buffered saline; DMSO: dimethyl sulfoxide.
Reference values. 34
Negative control group.
Normal group versus negative control and vehicle groups.
Normal group versus negative control group.
p < 0.01; ***p < 0.001.
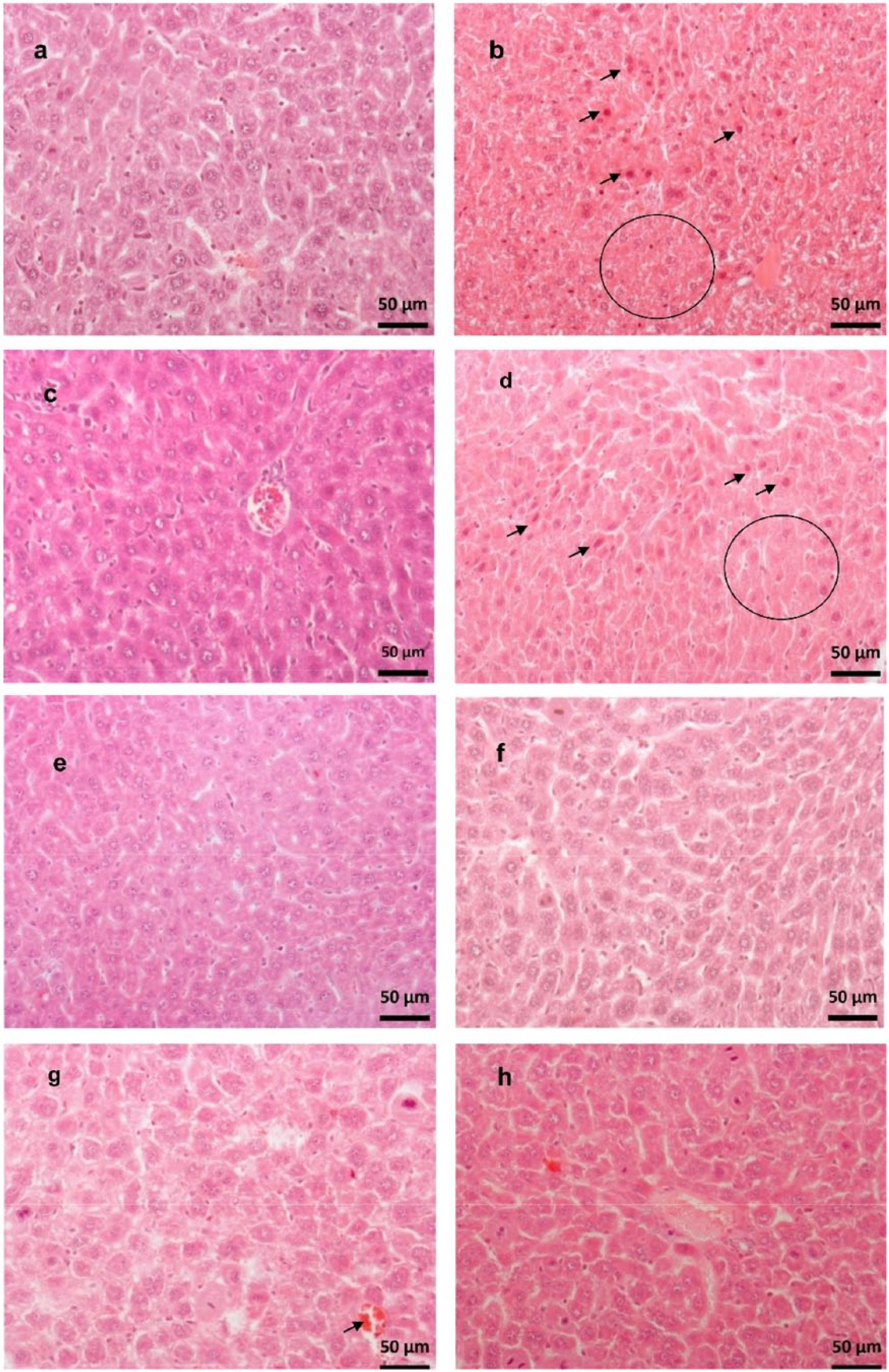
Effect of Ru(II)/amino acid/diphosphine complexes on the histopathological parameters of the livers of EAC-bearing mice. (a) Normal—hepatocytes with normal appearance and unchanged tissue. (b) Negative control—mild necrosis (circle) with eosinophilic hepatocytes with pyknotic nuclei (arrows) and cellular inflammation. (c) Vehicle—hepatocytes with normal appearance and mild cellular inflammation. (d) Cisplatin (2 mg kg−1)—mild necrosis (circle) with eosinophilic hepatocytes with pyknotic nuclei and cariorrexis (arrows), liver with mild cellular inflammation. (e) Complex
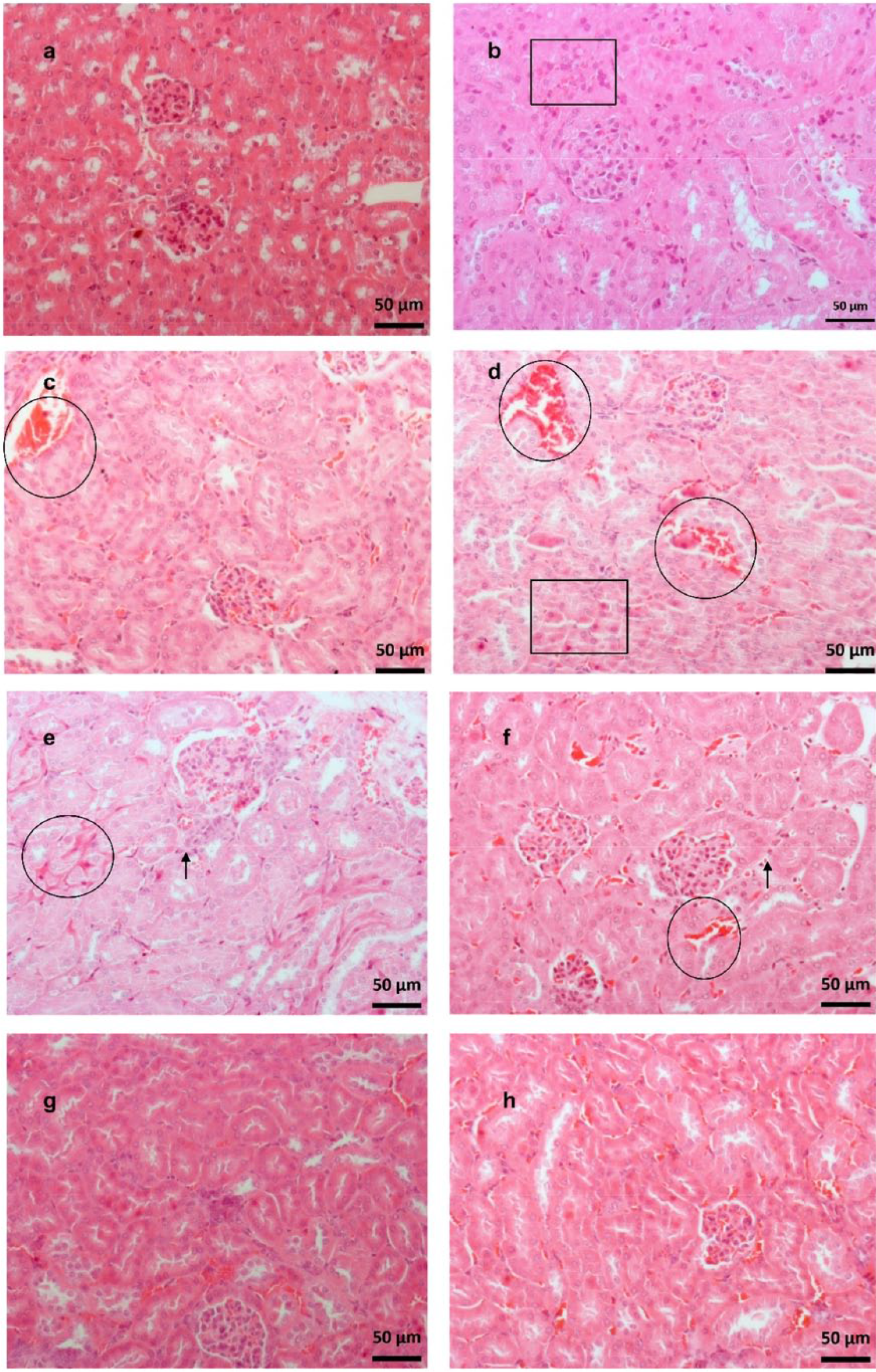
Effect of Ru(II)/amino acid/diphosphine complexes on the histopathological parameters of the kidneys of EAC-bearing mice. (a) Normal—renal cells with normal appearance and unchanged tissue. (b) Negative control—mild tubular necrosis (square) and tissue congestion. (c) Vehicle—mild tubular necrosis, hyperemia (circle), and tissue congestion. (d) Cisplatin (2 mg kg−1)—mild to moderate necrosis (square) with eosinophilic renal cells with pyknotic nuclei and cariorrexis and intensive hyperemia (circle). (e) Complex

Tumor cells adhered to the kidney capsule (arrow) and marked hyperemia, tissue congestion, and cellular inflammation.
Effect of complexes 1 and 2 on the reduction of tumor cell viability and cell death in vivo
The cell death type induced by complexes

Effect of Ru(II)/amino acid/diphosphine complexes on the mechanism of cell in Ehrlich ascites carcinoma cells after treatment of EAC-bearing mice. (a) Flow cytometric analysis using the Annexin V–FITC/PI double staining: Q1—viable cells, Q2—Annexin V+/PI−, Q3—Annexin V+/PI+, Q4—Annexin−/PI+. Data show the mean ± SEM of eight animals per treatment. (b) Western blot analysis of caspase-3 protein levels in EAC cells after incubation with complexes
To confirm the type of cell death induced by compounds, the cleavage of caspase-3 was investigated by western blotting and morphological analysis was analyzed using the HO/PI double staining. As shown in Figure 5(b) the protein levels of caspase-3 were significantly increased in response to both treatments. EAC cells with complex
The typical morphological features of apoptosis and necrosis were considered: apoptosis—nuclear condensation, cell shrinkage, membrane blebbing, and DNA fragmentation; and necrosis—swollen enlarged cells with disintegrated cell membrane (Figure 5(c)). Both compounds, when tested, caused cell death by apoptosis (Figure 5(d)). The treatment with complex 1 at doses of 2 and 6 mg kg−1 bw induced apoptosis in 29% ± 5% (p < 0.001) and 19% ± 4% (p < 0.01) of the EAC cells, respectively. While, 23% ± 7% (p < 0.001) and 23% ± 2% (p < 0.001) of EAC cells collected from animals treated with complex 2 (2 and 6 mg kg−1 bw, respectively) also undergo apoptosis.
Effect of complex 1 and 2 on cell cycle kinetics
Treatment of the negative control group with saline showed that most cells were viable, with a typical distribution in the sub-G1, G0/G1, S, and G2/M phases (Figure 6). However, after treatment of the mice with complexes
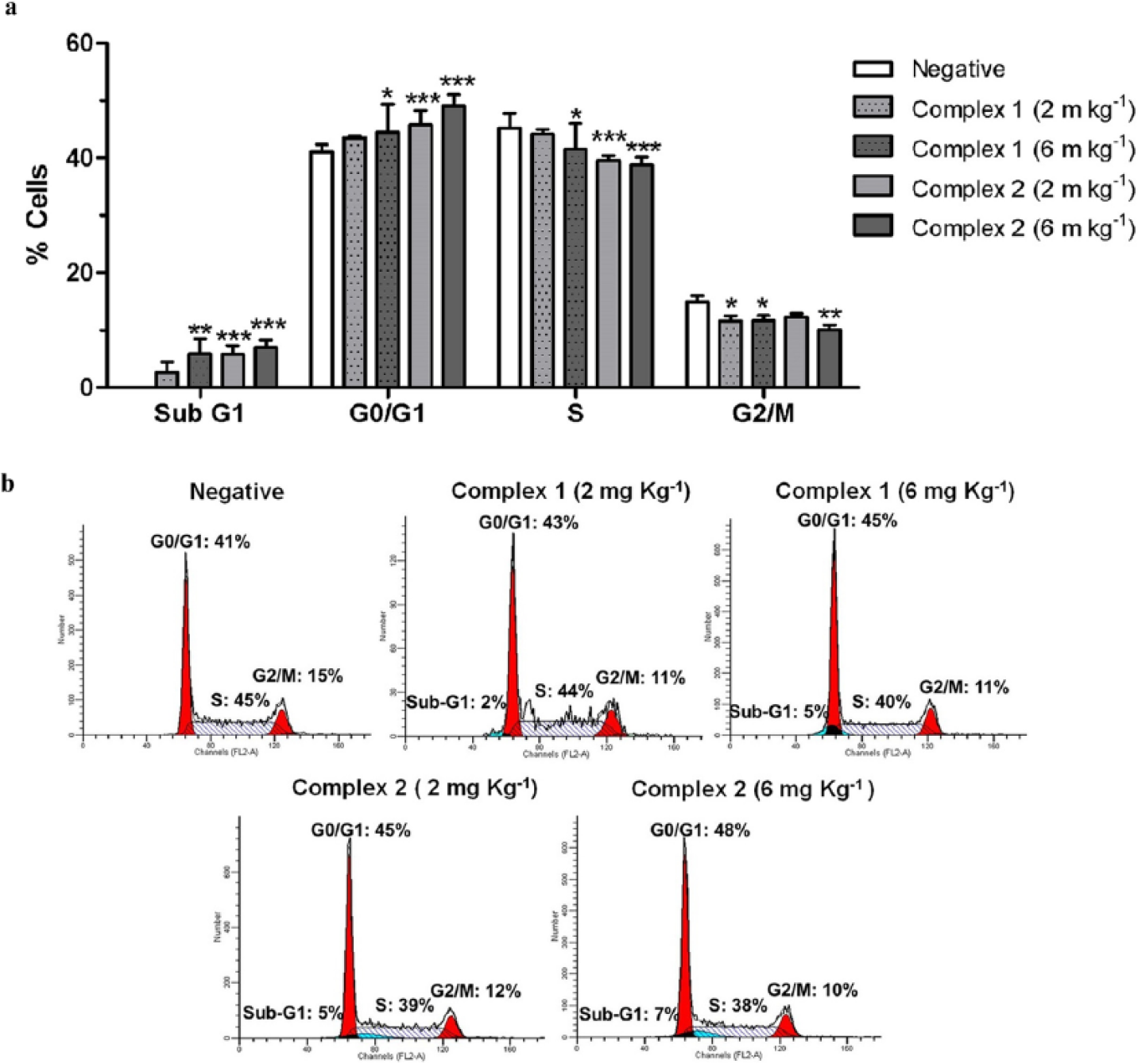
Effect of Ru(II)/amino acid/diphosphine complexes on cell cycle progression in Ehrlich ascites carcinoma cells after treatment of EAC-bearing mice was evaluated by flow cytometry. (a) Data show the mean ± SEM of eight animals per treatment (*p < 0.05, **p < 0.01, and ***p < 0.001 indicate statistically significant differences with the negative control). (b) A histogram for the cell cycle kinetics of the tumor cell in response to treatment with complexes
MST and ILS
The survival rate of EAC-bearing mice treated with complexes
Effect of Ru(II)/amino acid/diphosphine complexes on the MST and ILS of EAC-bearing mice.

EAC: Ehrlich ascites carcinoma; MST: mean survival time; ILS: increase in life span; PBS: phosphate-buffered saline; DMSO: dimethyl sulfoxide.
Animals (male) are monitored daily for 30 days.
Negative control group.
Normal group versus negative control group.
Normal group versus vehicle group.
p < 0.001.

Effect of Ru(II)/amino acid/diphosphine complexes on the Kaplan–Meier survival estimate of EAC-bearing mice. After EAC inoculation (6 × 106 cells/mouse i.p.), mice were treated with different doses of Ru(II)/amino acid/diphosphine complexes. Animals (male; n = 8) were monitored daily for 30 days.  negative group,
negative group,  vehicle group,
vehicle group,  complex
complex  complex
complex  complex
complex  complex
complex  cisplatin (2 mg kg−1).
cisplatin (2 mg kg−1).
Antiangiogenic activity in the CAM model and glucose uptake in vitro
Angiogenesis is critical to cancer progression because new blood vessels provide nutrition and oxygen, and elimination of accumulated metabolic wastes.35,36 CAM model is an attractive model to rapidly evaluate the efficacy of novel therapeutic drug and the in vivo inhibition of several tumor types.
37
As shown in Figure 8, the EAC cells induced angiogenesis in the CAM model with vascularization percentage of 34% ± 5% (p < 0.001 compared to control). The co-treatment of the EAC cells with complexes

Antiangiogenic potential of Ru(II)/amino acid/diphosphine complexes. (a) Representative photomicrographs of different CAMs after 72 h of treatment with complexes
To investigate whether antiangiogenic activity can change the glucose consumption, EAC cells were treated with complexes

Effect of Ru(II)/amino acid/diphosphine complexes on the glucose consumption of Ehrlich carcinoma cells after treatment for 48 h at IC50 values in vitro.
Effect of the inhibition of LAT1 on the viability of tumor cells in vitro
The neutral amino acid transporter, LAT1, can regulate cell proliferation and cancer progression, such as cell growth, invasion, and angiogenesis.
38
In order to verify whether the in vitro18,20 and in vivo effects of the Ru(II)/amino acid/diphosphine complexes observed for EAC cells could be related to the LAT1, the cells were treated with complexes
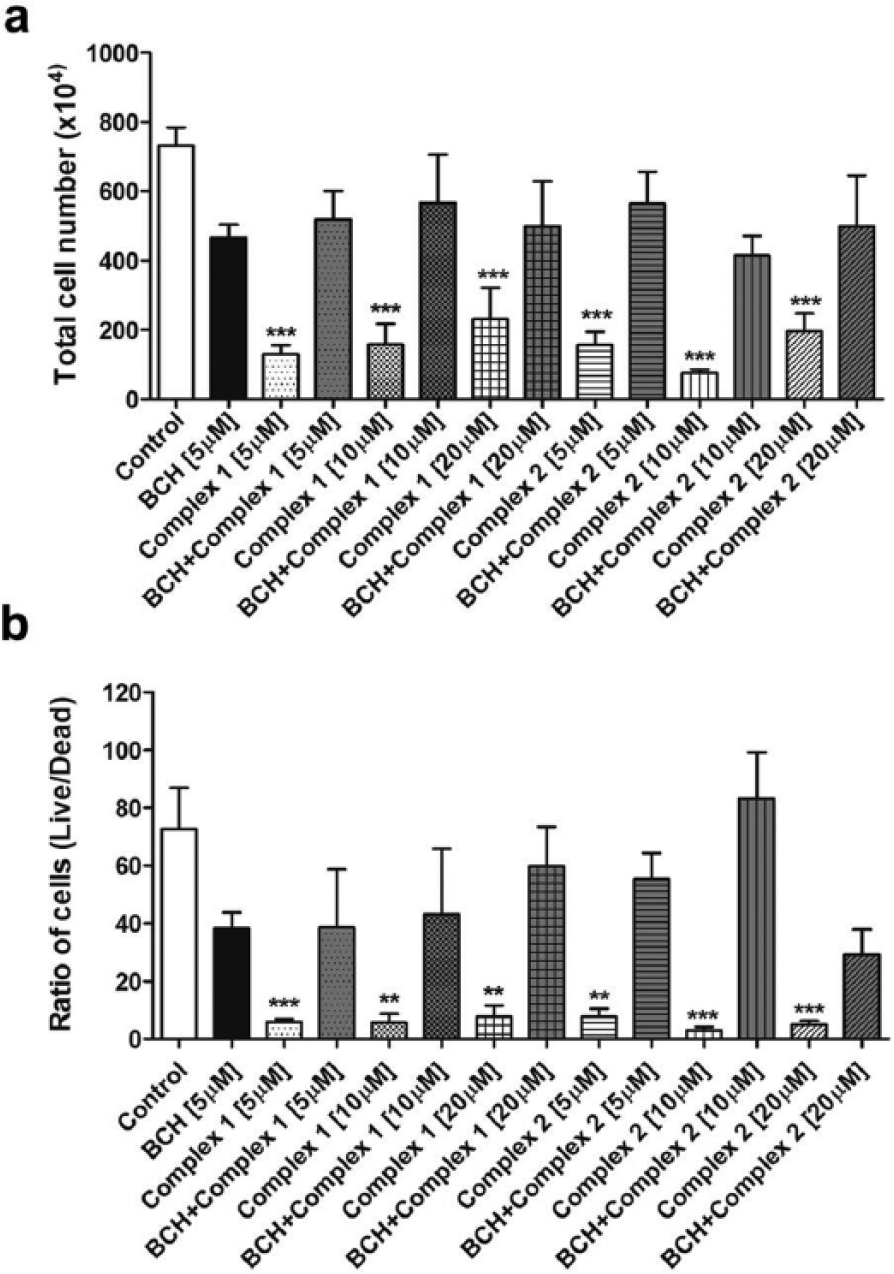
Effect of Ru(II)/amino acid/diphosphine complexes on the viability of Ehrlich carcinoma cells after inhibition of LAT1 in vitro. (a) Total Ehrlich tumor cell number after exposure to BCH and Ru(II)/amino acid/diphosphine complexes. (b) Ratio of cells (live/dead) after exposure to BCH and Ru(II)/amino acid/diphosphine complexes. All experiments were repeated in triplicate. Data show the mean ± SD (*p < 0.05, **p < 0.01, and ***p < 0.001 indicate statistically significant differences with the untreated control).
Discussion
In this study, the antitumor effects of complexes
Complexes
As demonstrated by other similar signs of toxicity studies with cisplatin,40,41 the results obtained for the antitumor evaluation with 2 mg kg−1 of cisplatin demonstrated high therapeutic potential (Table 1). However, the treatment triggered various side effects in the EAC-bearing mice, such as weakness. Treatment of the mice led to an MST of 21 days (p > 0.05) and an ILS of 17% (Table 4), which were not effective for prolonging the life span.
Progression of Ehrlich carcinoma has been reported in the literature by means of hematological changes, such as a decrease in Hb and the erythrocyte count,15,25,39 and the same were observed in our data (Table 2). The values of erythrocytes, Hb, and HCT obtained from the negative control and vehicle groups were reduced, when compared to values for Swiss male tumor-free mice (Table 2). The reduced values of erythrocytes, Hb, and HCT suggest anemia in EAC-bearing mice. Anemia seen in EAC-bearing mice is reported mainly through a reduction in erythrocytes or Hb.
42
Although the treatments with complexes
Disorders in bone marrow cells have been reported after using metallodrugs, such as cisplatin and carboplatin, causing myelotoxicity,
43
while after treatment with ruthenium complexes, hematologic toxicity due to erythropenia and leukopenia were reported.24,44 The leucocyte count for animals treated with complexes
Although modifications in kidney tissue were observed in the treatment with complex
According to the data obtained here, complex
Studies with ruthenium complexes found that the most lipophilic complexes exhibited the best cytotoxicity and highest cellular uptake.45–47 The partition coefficient (log p) between the water or buffer and n-octanol is the most widely used measure of chemical compound lipophilicity
48
as it is a major structural factor governing both the pharmacokinetics and pharmacodynamics of drugs, because up to a certain limit, compounds with higher lipophilicity have higher permeation across biological membranes (but lower aqueous solubility). Generally, for an effective anticancer drug to initiate cell death, it must reach viable cells in a tumor and be retained at a sufficient concentration on a relevant time scale. The partition coefficient values for complexes
Studies in vitro have reported the effects of Ru(II)/amino acid/diphosphine complexes on the mechanism of growth inhibition of tumor cells.17,18 It was found that the Ru(II)/glycine/diphosphine complexes induce apoptosis in 180 tumor sarcoma cells, as well as increases the number of Annexin V positive cells and causes G0/G1 phase cell cycle arrest, loss of mitochondrial membrane potential, activates caspase-3, caspase-8, and caspase-9, and causes a change in the messenger RNA (mRNA) expression levels of caspase-3, caspase-9 and Bax. It also increases the levels of the pro-apoptotic Bcl-2 family protein Bak.
17
For EAC cells, an in vitro study reported that Ru(II)/tryptophan/diphosphine complex (complex
In this study, a test using fluorescent Annexin V and HO/PI staining was carried out to determine whether the antitumor potential in vivo of complexes
The apoptotic pathway is initiated by cleavage of caspase-3 and causes typical morphological changes including cell shrinkage, chromatin condensation, membrane blebbing, the appearance of membrane-associated apoptotic bodies, and DNA fragmentation.49,51 Likewise, the morphological characteristics induced by complexes
As shown in the cell cycle analysis (Figure 6), complexes
CAM model has been widely used for monitoring tumor angiogenesis by inducing angiogenesis in several different types of tumor cells.52–55 However, the induction of angiogenesis by EAC cells using CAM model was reported for the first time in this study. Complexes
We demonstrated that the EAC cells treated with complexes
Likewise, cancer cells show reprogrammed energy metabolism, which results in a selective growth advantage and resistance to apoptotic death, since not only glucose but also other biomolecules, such as amino acids, are required for their survival and growth.36,59,60
The LAT1 are members of the sodium-independent amino acid transport system L, originally defined in the EAC cells, and are upregulated in human primary cancers and tumor cell lines, demonstrating an essential role for growth and survival. 61 Studies determining the function of LAT1 in cancer have used a well characterized LAT family inhibitor, BCH. 62 BCH is a model compound for the study of amino acid transporters, it is characterized as a system L competitive and selective inhibitor, particularly LAT1, capable of blocking the transport of amino acids into living cells and causing cell damage. 63 The inhibition of LAT1 activity has been proposed as oncology therapeutic implications, causing inhibition of cell growth and proliferation by deprivation of essential amino acids necessary for protein synthesis, cell growth, and proliferation.64–67
When LAT1 is blocked by BCH, it has been reported that the cell proliferation and DNA synthesis are suppressed,33,62 apoptosis is induced in several types cancer cells,67,68 and cleaved caspase-3 levels are increased.63,68 As shown in Figure 10, EAC cells were sensitive to complexes
The inhibition of EAC cell angiogenesis probably reduces the supply of essential nutrients required, like glucose, for cell growth and survival leading to cell death by apoptosis. Likewise, our results show that these metal complexes could be cytotoxic substrates that are delivered into the cell via LAT1 or can block transport by LAT1 and thereby promote tumor cell death. Our hypothesis for the inhibition of mechanisms of growth in EAC cells by complexes
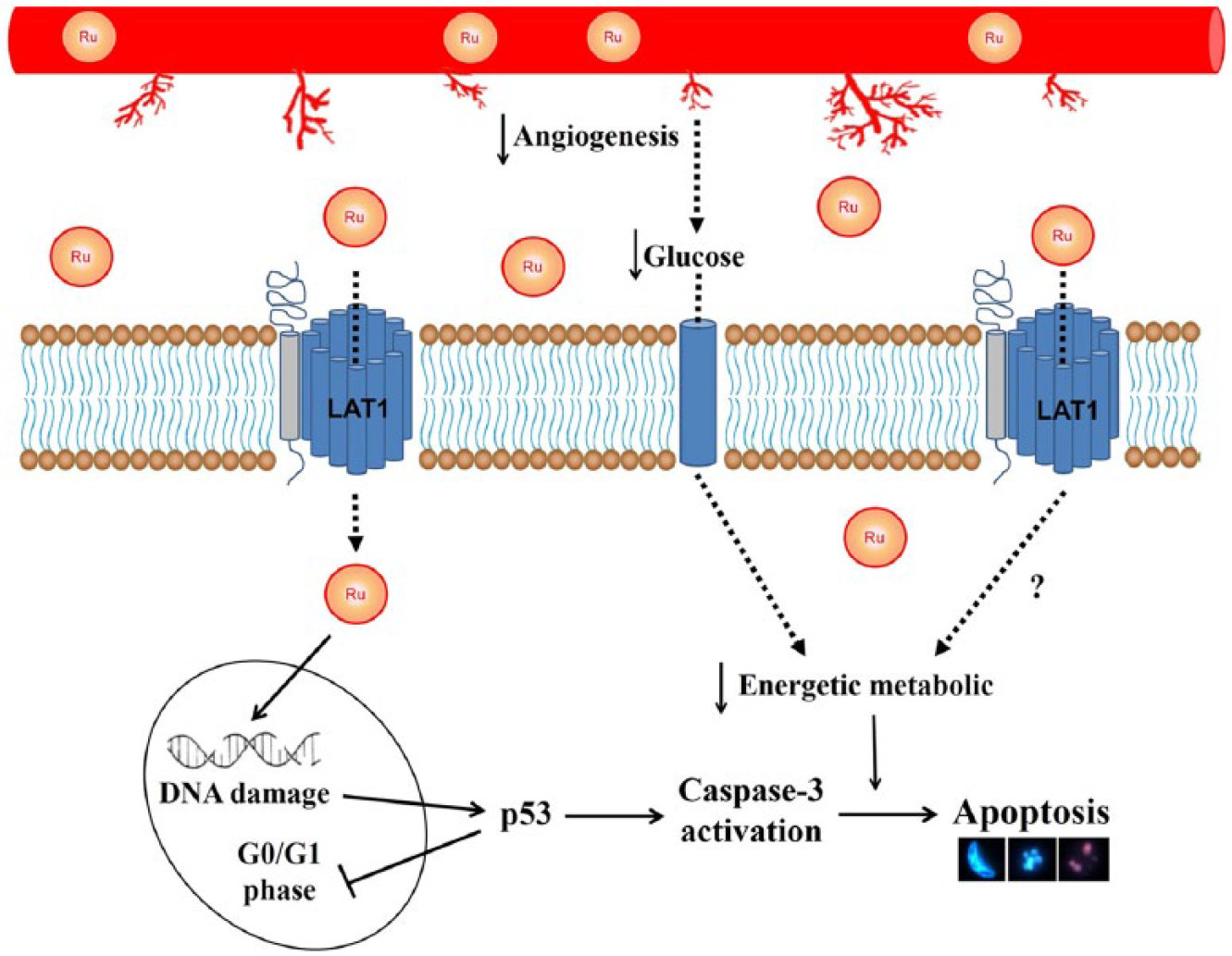
Hypothesis of effect of Ru(II)/amino acid/diphosphine complexes on tumor cells, suggesting that inhibition of angiogenesis reduces the glucose consumption in tumor cells and that LAT1 can be targeted by Ru(II)/amino acid/diphosphine complexes delivered into tumor cells, depriving supplementation of several growth factors and the nutrients needed for cell survival and proliferation, leading to apoptosis. Moreover, complexes
Conclusion
In summary, the [Ru(
Footnotes
Acknowledgements
The authors thank Julia Maria de Barros, Wildes Arantes de Moraes, and Vânia Beatriz Lopes Moura, PhD for technical assistance. The authors are grateful to Hugo Delleon da Silva, PhD for critically reading the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in the study involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted. The procedures described were reviewed and approved by the Research Ethics Committee (CEUA) at the Federal University of Goiás (No. 039/2012).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Foundation for Research Support of the State of Goiás (FAPEG 2012/12), São Paulo Research Foundation (FAPESP 2014/10516-7), and the Coordination for the Improvement of Higher Education Personnel (CAPES 2014/1267732).