Abstract
CIP2A is emerging as an oncoprotein overexpressed commonly across many tumours and generally correlated with higher tumour grade and therapeutic resistance. CIP2A drives an oncogenic potential through inhibiting protein phosphatase 2A, stabilizing MYC, and promoting epithelial-to-mesenchymal transition, although further biological mechanisms for CIP2A are yet to be defined. CIP2A protein expression was studied by immunohistochemistry in oestrogen receptor–positive primary breast cancers (n = 250) obtained from the Leeds Tissue Bank. In total, 51 cases presented with a relapse or metastasis during adjuvant treatment with tamoxifen and were regarded as tamoxifen resistant. CIP2A expression was scored separately for cytoplasmic, nuclear, or membranous staining, and scores were tested for statistically significant relationships with clinicopathological features. Membranous CIP2A was preferentially expressed in cases who experienced a recurrence during tamoxifen treatment thus predicting a worse overall survival (log rank = 8.357, p = 0.004) and disease-free survival (log rank = 21.766, p < 0.001). Cox multivariate analysis indicates that it is an independent prognostic indicator for overall survival (hazard ratio = 4.310, p = 0.013) and disease-free survival (hazard ratio = 5.449, p = 0.002). In this study, we propose the assessment of membranous CIP2A expression as a potential novel prognostic and predictive indicator for tamoxifen resistance and recurrence within oestrogen receptor–positive breast cancer.
Introduction
A large majority (~75%) of breast cancers are oestrogen receptor positive (ER+), 1 and therefore successful treatment of this group remains a key priority for improving breast cancer outcomes. Tamoxifen is established as the treatment of choice for many ER+ breast cancers (BCER+) in both pre- and post-menopausal patients. 2 More recently, aromatase inhibitors (AIs) have become a preferred choice of treatment in cases with a worse prognosis, as defined by high lymph node involvement (four or more axillary lymph nodes involved) and high tumour grade (grade 3). In post-menopausal cases, high Ki67 or human epidermal growth factor receptor 2 (HER2) positivity are also factors favouring the use of AIs over tamoxifen.3,4 Nevertheless, tamoxifen remains an extremely common treatment. However, despite the well-established efficacy of tamoxifen in this setting, 2 14.5% of patients on 5 years or 13.1% of patients on 10 years of adjuvant tamoxifen experience recurrences within 10 years of diagnosis. Cumulative recurrence rates increase to 21.4% at 15 years from diagnosis if receiving tamoxifen for 10 years, accompanied by a death rate of 12.2%. 5 Recurrences during treatment indicate resistance to therapy, which can be intrinsic or induced through various mechanisms namely, loss of ER, increased growth factor signalling, or inhibition of autophagy and apoptosis. 6
CIP2A is an emerging oncoprotein known to drive the malignant phenotype through specific pathways. CIP2A has been described to be overexpressed in 39% of breast cancer cases 7 and approximately 70% of de novo acute myeloid leukaemia (AML) patients 8 compared to normal tissue. Amplification/gain of CIP2A as well as overexpression of CIP2A is fairly common across various tumour types.9–11 Various studies have associated higher CIP2A expression with grade, highly proliferative tumours, and stage in distinct tumour types such as prostate, gastric, colon, breast, head and neck squamous cell carcinoma, and ovarian cancer.7,9,12–17 Our interest in CIP2A stems from its inhibitory activity on protein phosphatase 2A (PP2A), which results in protection of MYC from dephosphorylation thereby stabilising the oncogenic stimulation of MYC on cell proliferation.7,18,19
The mechanism of CIP2A-dependent inhibition of PP2A is not well understood, although some suggest that CIP2A binds to the PP2A catalytic subunit allosterically at the interface where it binds the regulatory B subunit, thus altering substrate specificity and/or limiting activity.8,19–22 In a chronic myeloid leukaemia (CML) model, knockdown of CIP2A results in decreased BCR-ABL1 tyrosine kinase activity. 16 Therapy-induced reduction of BCR-ABL1 activity (imatinib) in CML reduces the levels of SET, another PP2A inhibitor. Patients who still progress to blast crisis show elevated CIP2A levels showing that CIP2A maintains PP2A inhibition and leads to cancer progression. 16
A CIP2A transcriptional signature suggests that CIP2A promotes MYC signalling pathway but also other pathways as the c-jun N-terminal kinase 2 (JNK2).7,18 CIP2A is involved in the regulation of a major group of genes that mediate cellular migration. High CIP2A has been correlated with the mesenchymal phenotype in prostate cancer defined by low expression of E-cadherin and higher expression of N-cadherin and vimentin. 23 However, depletion of this endogenous PP2A inhibitor caused a significant inhibition of migration which was rescued by okadaic acid (an inhibitor of PP2A), hence indicating that this process is mediated through the suppressive interaction of CIP2A with PP2A. This may explain the correlation between CIP2A overexpression and more aggressive tumour phenotypes and lymph node positivity.18,24
In light of the involvement of CIP2A in cellular migration and therapeutic resistance, we aimed to investigate CIP2A expression within BCER+ patients. In particular, we were interested to assess whether CIP2A had a role in tamoxifen resistance and therefore whether its expression levels could be used to improve prognostication or therapy prediction in breast cancer.
Materials and methods
Patients
Ethical approval was obtained from Leeds, UK (East; REC: 06/Q1206/180). Formalin-fixed paraffin-embedded breast cancer tissue was obtained from the Leeds Tissue Bank. A cohort of 250 BCER+ patients was enrolled. ER staining was scored using the Allred scoring method, as previously described. 25 All patients were treated with tamoxifen, and 172/199 of the control group had a follow-up longer than 5 years. In total, 51 patients experienced a recurrence or metastasis while on tamoxifen treatment; therefore, these tumours are considered to be tamoxifen resistant (TAMR). 26 Tumour tissue samples were constructed into tissue microarrays (TMAs) with 0.6 mm triplicate cores for each case wherever available. Cases were annotated with histopathological and clinical information including survival, disease-free survival (DFS), and treatment accompanied by metastasis or relapse information (Supplementary Table 1).
CIP2A immunohistochemistry
TMA sections were dewaxed prior to heat-induced antigen retrieval in citrate buffer (pH 6.0–6.2) using the PT-link (Dako, Santa Barbara, CA) set at 102°C for 20 min. Endogenous peroxidase was then blocked for 20 min by 0.3% H2O2, followed by blocking with normal swine serum. TMAs were incubated with 1:200 anti-KIAA1524 (CIP2A; HPA039570; Atlas Antibodies, Sweden) at 4°C overnight in a humid chamber. Following a 1× Tris-buffered saline (TBS) wash, the sections were incubated for 1 h with biotinylated goat anti-rabbit antibody (Dako), followed by detection using the Avidin–Biotin Complex kit (VECTASTAIN; Vector laboratories, Burlingame, CA). The antibody was validated by western blotting. Whole sections were included as negative (primary antibody omitted) and positive controls (previously determined CIP2A positive breast tissue sections) to assess batch staining. Strong positive vascular endothelium within sections and cores was used as a staining internal control. CIP2A staining could not be assessed for three TAMR cases due to no tumour present within cores or cores were lost during processing. Cores were scored for positivity by two individuals (S.B. and L.M.W.). L.M.W. scored 25% of cases, and correlations between scorers were assessed to confirm scoring reproducibility (Supplementary Table 2). The H-score method was used to score the level of immunohistochemical staining in the nuclear, cytoplasmic, and membranous compartments separately. The H-score was computed as the combinatorial result of the degree of intensity multiplied by the percentage of cells at each intensity. The intensity is scored from 0, when no staining at all is seen through, to 3, when staining is very strong. Thus, if all cells (100%) staining strongly with intensity rated 3, the score would be 3 × 100, that is, 300.14,27,28
Statistical analysis
CIP2A staining in the cytoplasmic compartment was categorised as negative (H-score ≤ 30), weakly positive (H-score > 30 and ≤ 120), and strongly positive (H-score > 120). Membranous and nuclear compartments were categorised as negative when H-score was 10 or less and positive when above 10. These thresholds have shown the best Kappa agreement measures between scorers and implied the best statistical significance towards prediction of survival. The Kappa score was used to measure agreement between CIP2A categorised scores. When cytoplasmic CIP2A scores were categorised into negative, weak positive, and strong positive, a Kappa score of 0.697 (Supplementary Table 2) was obtained which is considered as substantial.29–31 The continuous H-scores of the two scorers showed a significant correlation for CIP2A expression (Pearson’s and Spearman correlations – Supplementary Table 2). This was confirmed by the two-way random effects model of intraclass correlation coefficient (ICC) which is used to estimate the agreement on continuous scores between observers where perfect agreement is denoted by 1 and 0 establishes no significant agreement under the null hypothesis (Supplementary Table 2).32–35 The ICC values obtained are above the 0.7 threshold for acceptable reliability as defined for ICC for group comparisons by the ISPOR (International Society for Pharmacoeconomics and Outcomes Research) task force. 36
Statistics were computed using the SPSS (IBM SPSS, Chicago, IL). Where two or more cores represented one case, the mean score was derived. The overall survival (OS) endpoint was taken to be deaths attributed to the breast cancer. Death from other causes, where known, was censored. Likewise, the endpoint for DFS was any relapse, metastasis, or death by breast cancer and any death by unknown or other causes was censored. Cox multivariate analysis was performed using the Enter method. Tests performed were two-tailed and considered significant when p < 0.05.
Results
Membranous CIP2A expression predicts a worse survival in BCER+
CIP2A was weakly (H-score = 30–120) expressed in the cytoplasm in 40.0% of cases and strongly (H-score > 120) in 2.4%. Any positive expression in the nuclear and membranous compartments was less common, with positivity (H-score > 10) in only 2.8% and 6.1%, respectively (Figure 1(a)–(d)). Typical weak staining in normal tissue is illustrated in Figure 1(e). Nuclear and membranous staining were both associated with cytoplasmic staining (χ2 = 12.05, p = 0.002; χ2 = 12.04, p = 0.002, respectively). Conversely, there was no significant association between nuclear and membranous staining (χ2 = 0.85, p = 0.359).
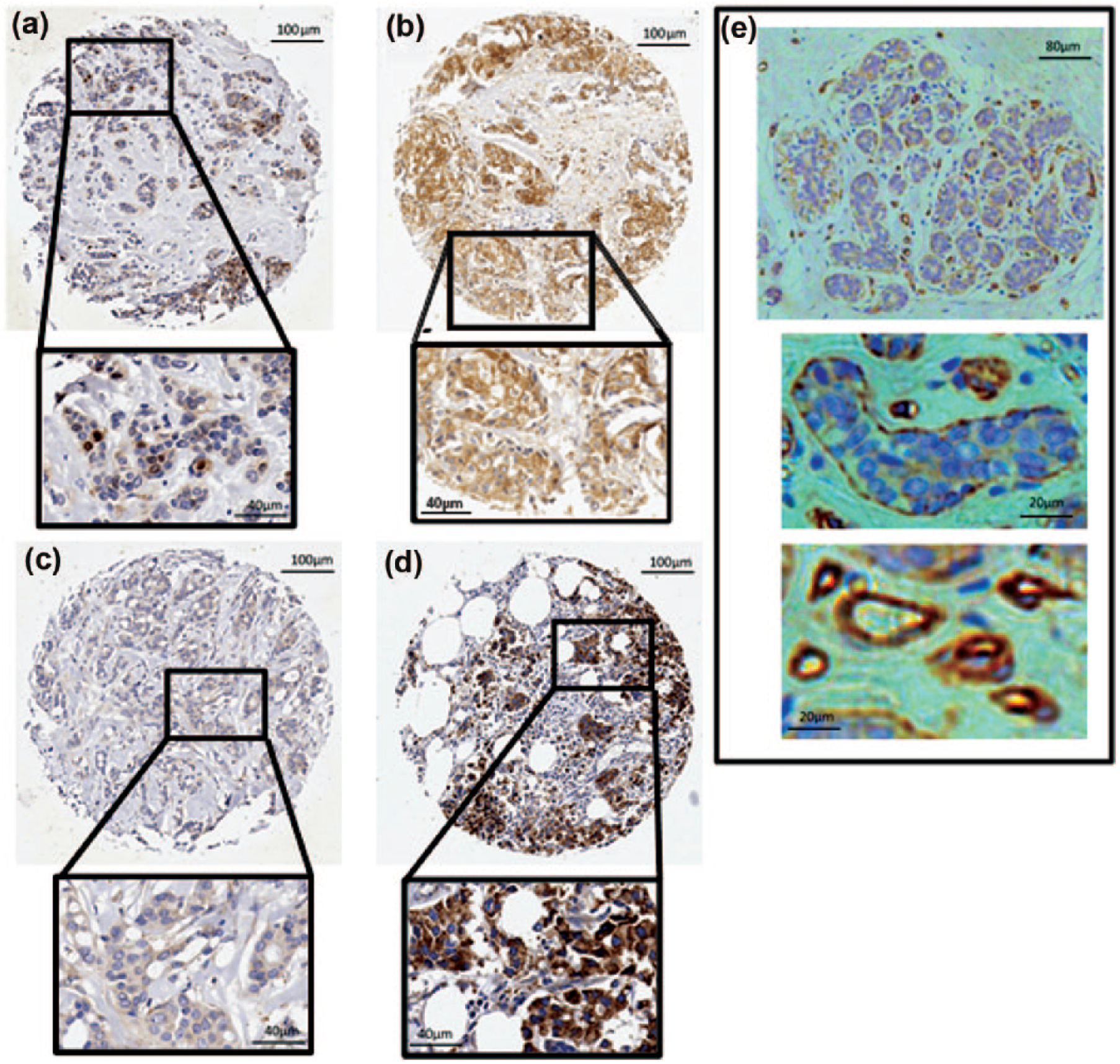
Immunohistochemistry staining patterns of CIP2A in (a-d) breast cancer and (e) normal breast ducts. (a) Nuclear staining, (b) membrane staining, (c) weak cytoplasmic staining, and (d) intense and prevalent cytoplasmic staining are presented. (e) Normal breast ducts show negative to weak cytoplasmic staining.
There was no association of cytoplasmic or nuclear CIP2A with histopathological features or survival (data not shown). Conversely, membranous CIP2A was found to be significantly associated with recurrence or metastasis during tamoxifen treatment (Table 1). CIP2A expression in the tumour cell membrane was detected in 20.8% of breast cancer that recurred while on tamoxifen treatment (TAMR) as opposed to 2.5% occurrence in the control group. In fact, membranous CIP2A was found to predict strongly a worse OS and worse DFS regardless of continued tamoxifen use (Figure 2). Breast tumours with no evidence of membranous CIP2A expression have an 85% and 77% 5-year and 10-year cumulative survival rate, respectively, while cases with membrane expression of CIP2A showed substantially lower survival rates of 60% and 39% at 5-years and 10-years, respectively.
Membranous CIP2A expression in breast cancer with recurrence during tamoxifen treatment (TAMR group) as opposed to breast cancer with no recurrence during treatment (Control group).

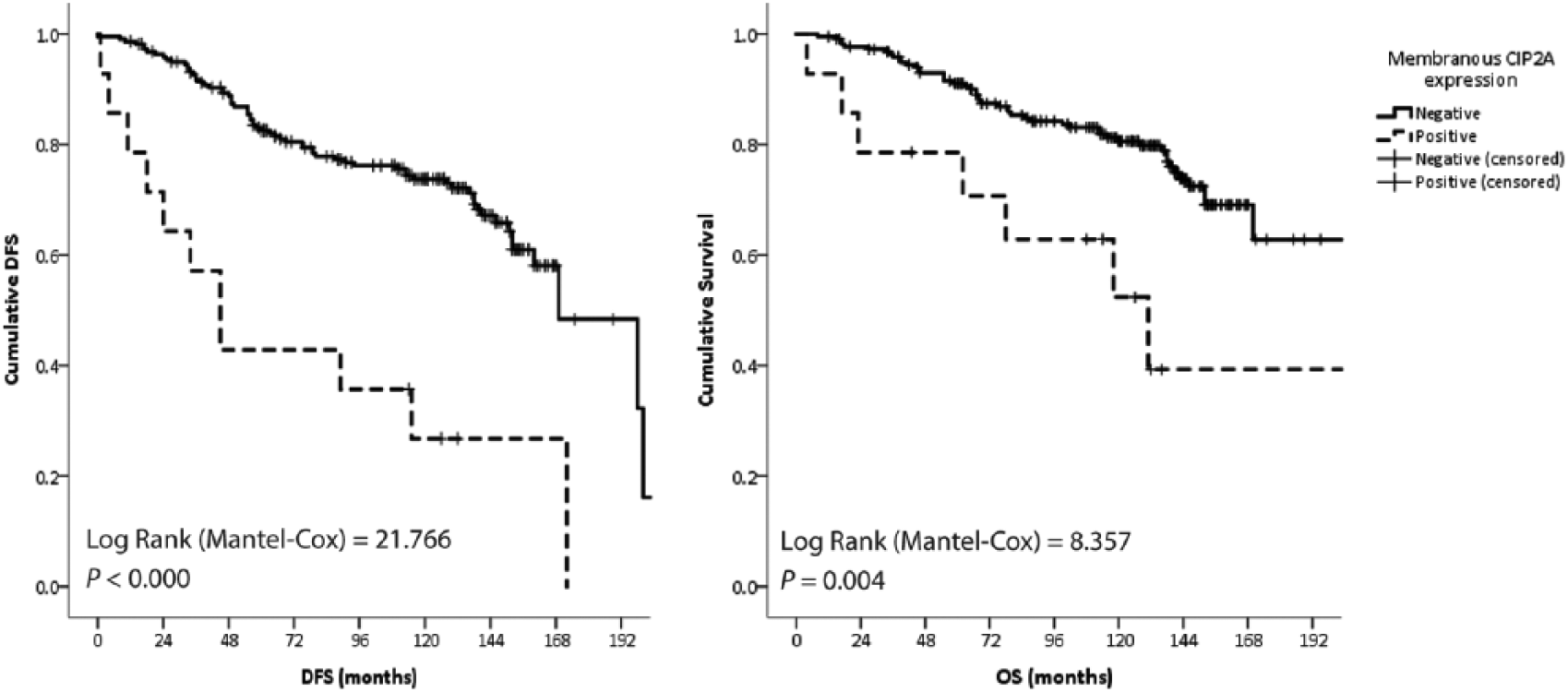
Kaplan–Meier survival curves for membrane CIP2A expression in breast cancer.
Membranous CIP2A is an independent predictor of recurrence
Cox multivariate analysis of available factors indicates that membranous CIP2A along with other histopathological factors is significant independent predictor of DFS (Table 2). Interestingly, there was no association between membrane CIP2A staining and Nottingham prognostic index (NPI), lymph node involvement, tumour size, grade, and histological type (χ2 test, respective p values: 0.353, 0.151, 0.614, 0.265, and 0.760). Membranous CIP2A shows the highest risk across all categorical variables.
Cox multivariate analysis of variables associated with breast cancer recurrence (DFS).

DFS: disease-free survival; NPI: Nottingham prognostic index.
Grade, positive lymph nodes, NPI, CIP2A staining (membranous, nuclear, and cytoplasmic), and histological type were considered as categorical indicators. Control group n = 163; Study group n = 24.
Discussion
Various studies have shown a preferential overexpression of CIP2A RNA in basal and HER2-enriched breast cancer when compared to BCER+.9,11,18 Of note, CIP2A is weakly expressed in normal breast tissue as protein and transcript.7,15 Similar to the study of Tseng et al. (2012) 37 and Choi et al., 38 in this study the H-score was used to obtain more information on the distribution of CIP2A scores and intensities. Although the H-score provides more information, there is a lack of consensus about the method used to score CIP2A immunohistochemical staining. Most studies score only the cytoplasmic compartment and/or only record intensity scores.12,39–41 Nuclear staining was scored independently from the cytoplasmic compartment in the study of Böckelman et al. 42 as positive or negative. Another study considered only the percentage of cells that were stained without considering localisation and intensity. 15 Harmonisation is required to allow multivariate analysis taking studies together to increase the power to characterise prognostic and therapeutic outcomes.
Overall, CIP2A expression has been correlated with aggressive breast cancer and also predicts a worse prognosis at 5-year follow-up.7,15 Oestradiol has been shown to promote CIP2A expression while in the absence of CIP2A, oestradiol-enhanced proliferation was compromised. 38 A study by Liu and colleagues,43,44 showed that tamoxifen-induced apoptosis was associated with downregulation of CIP2A and p-AKT in tamoxifen-sensitive breast cancer cells including the ER+ MCF7. Interestingly, we found that membranous expression of CIP2A was significantly higher in patients with tamoxifen resistance.
The biological mechanism of nuclear and membranous CIP2A expression still needs to be elucidated. Both staining patterns have been previously observed.42,45 Despite a correlation between nuclear and cytoplasmic staining, we expect CIP2A to have distinct roles in the different cellular compartments. Cytoplasmic CIP2A is available for PP2A regulation and therefore subsequent MYC activation, while nuclear CIP2A might be involved in transcription regulation or is transported into the nucleus while in complex with other protein subunits. Nuclear CIP2A staining significantly predicts a better survival in ovarian cancer, 13 although in this study the incidence of nuclear staining was too low to allow robust analysis of correlations with histopathological factors or prognosis.
Localisation of CIP2A to the cellular membrane might be caused by interaction with membrane receptors. CIP2A has been described to interact with cell surface receptors like UNC5H. Interaction of CIP2A with UNC5H in the presence of netrin-1 inhibits PP2A-mediated dephosphorylation of death-associated protein kinase (DAPk) thus evading apoptosis. 46 Transforming growth factor beta (TGFB) has also been known to interact with PP2A, which might contribute to the contradictory regulation of p-AKT and MYC.47–49 In metastatic breast cancer (MDA-MB-231), the transition of tumour-suppressive role to tumour-promoting role of TGFB has been attributed to the nuclear factor of activated T-cells (NFAT), which is shown to drive epithelial-to-mesenchymal transition (EMT) and c-MYC expression. 50 Here, we suggest another potential mechanism for this transition. Binding of PP2A to TGFB will promote tumour-suppressive effects which can be reversed if CIP2A also complexes with PP2A leading to c-MYC protection as well as upregulation of p-AKT. Interestingly, downregulation of AKT through expression of the TGFB growth factor was described as an off-target effect of tamoxifen.49,51
Of importance, Liu et al. 44 showed an induced sensitivity to tamoxifen in the triple-negative breast cancer cell line HCC-1937, when re-activating PP2A using forskolin. This suggests the potential for use of combinatory therapy for BCER+ with membranous CIP2A staining.
Concluding remarks and further recommendations
Membranous CIP2A is an indicator for resistance to tamoxifen within the hormone receptor–positive breast cancer setting. Membranous CIP2A might prove to be useful in selecting ER+ patients with risk of developing tamoxifen resistance for closer monitoring or even for assisting treatment decisions. Further investigation of the function of CIP2A in the cellular membrane might identify malignant mechanisms that are independent of ER signalling. In addition, CIP2A protein expression and cellular distribution need further investigation to assess clinical significance in tamoxifen resistance.
Footnotes
Acknowledgements
The authors thank the Breast Cancer Now Tissue Bank for helping with sample and data collection.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Breast Cancer Project Scholarship (2014-2016) funded by the Action for Breast Cancer Foundation and ALIVE 2013 through the Research, Innovation & Development Trust (RIDT) of the University of Malta, and by the Faculty of Medicine & Surgery, University of Malta.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
