Abstract
Angiogenesis plays an important role in tumor growth, invasiveness, and metastasis. It is well established that prostate cancer is exposed to fluctuating oxygen tensions and both acute and chronic hypoxia exist, and these conditions can upregulate angiogenesis-associated proteins such as hypoxia-inducible factor 1 alpha and vascular endothelial growth factor A. Low-frequency low-intensity ultrasound with microbubbles can induce obvious microvessel damage in tumors, cause cell necrosis or apoptosis. However, there is no information about whether the blocking blood effect of low-frequency low-intensity ultrasound with microbubbles has an influence on hypoxia environment of prostate cancer. Therefore, we investigated the impact of different low-frequency low-intensity ultrasound with microbubbles radiation times on prostate tumors, observed the change in the hypoxia-inducible factor 1 alpha and vascular endothelial growth factor A protein levels, as well as cell proliferation, apoptosis, and tumor volume. The results indicated that as the radiation was repeated four times on each treatment day, the effects of interruption were durable, the cell proliferation was inhibited, and apoptosis was promoted, and the hypoxia-inducible factor 1 alpha and vascular endothelial growth factor A expression levels were lower in the treatment group than in the control group. When the radiation was carried out once per treatment day, the hypoxia response was stimulated, the hypoxia-inducible factor 1 alpha and vascular endothelial growth factor A expression levels were higher compared with the control group, and cell proliferation was promoted. In addition, the tumor volume increased obviously in the hypoxia-stimulated group, whereas tumors grew slowly in the hypoxia-suppressed group. The results of this work demonstrated that under the same conditions, different radiation times of low-frequency low-intensity ultrasound with microbubbles affect the hypoxia response differently, and the effect at least partly stimulates or inhibits tumor growth.
Keywords
Introduction
Prostate cancer (PCa) remains to be one of the most frequently occurring malignancies for men in the United States and was responsible for more than 2.75 × 104 deaths in 2015. 1 Temporal trend analysis of Asian men also shows a rapidly increasing incidence rate of PCa. 2 At first, PCa is sensitive to androgen deprivation therapy; unfortunately, a large portion of patients continue to present a metastatic castration-resistant progress after the initiation of androgen deprivation therapy, which affects the quality of life of patients with serious osteoblastic bone metastases.1,3
The pathogenesis of PCa and its molecular biological progression have not yet been clarified. Neoangiogenesis is an important character of progressing solid tumors, especially in PCa, which is an angiogenesis-dependent tumor. The hypoxic microenvironment of the tumor has been confirmed to be correlated with tumor metastasis and resistance to chemotherapy and radiotherapy.4,5 This is particularly true for PCa: Several clinical studies have proved that hypoxia is an independent prognostic indicator of poor outcome, which is detrimental to the efficacy of treatment.6 –8
Hypoxia within the tumors can be divided into two categories: chronic (diffusion-limited) hypoxia, in which cells far from the blood vessels are exposed to slowly decreasing concentrations of oxygen and increasing intracellular hypoxia; and acute (perfusion-limited) hypoxia, in which a sudden, intermittent, or complete reduction of blood flow lead to acute lack of oxygen and nutrients, leading to intracellular hypoxia.9,10 Hypoxia-inducible factor 1 (HIF-1) is a heterodimer that is composed of HIF-1α and HIF-1β subunits and plays a key role in O2 homeostasis.11,12 HIF-1α is a unique O2-regulated subunit that determines HIF-1 activity, and its levels are generally increased in hypoxic conditions and in aggressive tumors.11,13,14 HIF-1 transactivates many genes whose proteins function to increase the O2 availability or allow cell adaptation to O2 deprivation, for example, vascular endothelial growth factor (VEGF), inducible nitric oxidesynthase (iNOS), and heme oxygenase 1.11,14 Studies have revealed that HIF-1α and VEGF are highly expressed in PCa compared to benign prostatic hypertrophy, which are connected to biochemical failure in PCa patients.11,15,16
Currently, low-frequency low-intensity ultrasound with microbubbles (LFUM) has been considered as a novel anti-vascular method to PCa. 17 Several studies indicate that acoustic cavitation, which is a major effect of LFUM, can lead to mechanical damage of small blood vessels for the magnitude of expansion and collapse as the microbubbles oscillate.18,19 Feng et al. 20 and Liu et al. 21 demonstrated that ultrasound (US) enhanced with microbubbles could slow down or block blood perfusion in rabbit spleens for up to 1 h and could supply an obvious hemostatic effect for organ trauma such as liver injury. Our previous studies also prove that LFUM has the ability to block blood perfusion in PCa xenografts for 2 h. In addition, LFUM might promote tumor cell apoptosis and inhibit proliferation, thereby enhancing the effects of radiotherapy or chemotherapy to PCa.22,23 However, some studies revealed that US stimulation can induce angiogenesis both in murine subcutaneous tumors and in mice ischemic hind limbs.24,25 Reher et al. 26 certified that therapeutic US influences local hypoxia to enhance tissue repair. Both low-intensity and low-frequency US radiation have effects on healing tissue.27,28 Furthermore, US conditions in combination with microbubbles along with the treatment times and intervals in studies vary widely. More importantly, no study has demonstrated whether the effect of sudden reduction of blood flow due to LFUM could reverse or prevent hypoxia in PCa.
In this work, we treated PCa xenografts using the same LFUM treatment condition with different radiation times. According to our previous study, this condition, which causes microvessel disruption in PCa xenografts, was decided by orthogonal experimental design, and the blocking effect was of 2 h and the tumor growth was inhibited. This study aimed to further investigate whether this LFUM treatment condition could affect hypoxia within the tumor by influencing the HIF-1α-/VEGFA-dependent mechanism. We also aimed to determine the results of altered radiation time in PCa.
Materials and methods
Cell lines and subcutaneously transplanted tumor models
A human bone metastatic hormone refractory prostate cancer cell line, PC3, was used in this work. Cells were cultured in Dulbecco’s modified Eagle’s medium (Gibco, USA) with 10% fetal bovine serum (Gibco) in a humidified incubator at 37°C with 5% CO2. Briefly, 100 μL of phosphate-buffered saline (PBS) containing 1 × 107 PC3 cells was subcutaneously injected into the right flank of homogeneous nude mice (5–6 weeks old; weight: 24 ± 2 g) to build the xenograft tumor model. Experiments were initiated 10 days later, when the tumors grew to maximum and minimum diameters of approximately 4 and 3 mm, respectively. The experiment was approved by the committee on Ethics of Laboratory Animal Welfare of Shanghai Jiao Tong University.
Instruments and antibodies
Low-frequency ultrasound equipment was provided by Shanghai Institute of Ultrasound in Medicine (China). The ultrasonography experiment was carried out on MyLabTwice (Esaote; Italy) with a LA523/LA522 probe. The parameters were set as 4–13 MHz with the RES-L mode for acoustic frequency and 100% power with a mechanical index of 0.8. Microbubbles (11.8 mg/mL) were purchased from Sono Vue® (Bracco; Italy). The commercially available kit for deoxynucleotidyl transferase–mediated deoxyuridine triphosphate nick-end labeling (TUNEL) was bought from Roche (Germany). Rabbit anti-human Ki-67, mouse anti-HIF-1α, and mouse anti-VEGFA monoclonal antibodies, as well as anti-
Treatment procedures
The therapeutic parameters of LFUM were chosen according to our previous study: 22 microbubbles (0.2 mL), acoustic intensity (1 W/cm2), frequency (20 kHz), duty cycle (40%), and radiation duration (3 min). A total of 54 nude mice were recruited in the experiment, and the mice were randomly divided into the following three groups: the experimental groups were treated with LFUM every 2 days a total of three times. At each treatment day, the operation was carried out once in experimental group 1 (3 min × 1) but repeated four times with a 3-h interval in experimental group 2 (3 min × 4), indicating that tumors in this group received a total of 12 min radiation. Finally, the control group was treated with the same process but without US.
Contrast-enhanced ultrasound
Contrast-enhanced ultrasound (CEUS) of the tumors was carried out three times: before treatment, immediately, and 3 h after the first treatment immediately. The ultrasonography experiments are described in section “Instrument and antibodies,” and 0.1 mL of microbubbles were used each time. The CEUS images were analyzed by the Qontra Xt Software (Esaote) to determine the average peak intensity (API), which represents the average echogenicity at the peak contrast enhancement within a region of interest.
Tissue preparation and tumor volume
The mice were anesthetized with pentobarbital. Tumors were harvested on days 4 and 14 after the last treatment (three mice per group at each time). For immunohistochemistry, the samples were fixed in 4% formalin for paraffin-embedded sections. For protein preparation, samples were snap-frozen in liquid nitrogen. In addition, the tumor volumes were evaluated 14 days after treatment according to the following formula: Tumor volume (mm3) = (a × b2)/2.
Immunohistochemistry
The tumor specimens were fixed in 10% neutral formalin for 24 h. Then, several 5-μm-thick sections were obtained, dewaxed, and hydrated after embedding the tissues in paraffin. Endogenous peroxidase was blocked with 3% H2O2 for 20 min, and non-specific binding was blocked by incubating the samples with 10% goat serum in PBS for 20 min. The slides were incubated overnight with anti-HIF-1α (1:200) and anti-VEGFA (1:100) at 4°C. After washing, the slides were incubated with horseradish peroxidase–labeled goat anti-mouse IgG for 30 min. Finally, they were incubated in PBS containing 3,3′-diaminobenzidine (DAB) for 5 min. HIF-1α and VEGFA proteins were found to be predominantly localized in the cell cytoplasm. The HIF-1α and VEGFA protein expression levels were evaluated according to the percentage of positive cells, which were detected by brown-yellow or dark-brown staining of the cytoplasm. Five random and non-overlapping fields were viewed per group. Apoptosis of tumor cells was determined by TUNEL. Tumor cell proliferation was determined by anti-human Ki-67 rabbit monoclonal antibodies. The apoptosis index (AI) and proliferation index (PI), which were expressed as percentages, were defined as the ratio of TUNEL-positive tumor cell nuclei and the ratio of Ki-67-positive tumor cell nuclei to all tumor cell nuclei, respectively.
Western blot analysis
A protein extraction kit was used to obtain protein extracts. Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) was carried out, and the proteins were then transferred to polyvinylidene fluoride (PVDF) membranes. After being blocked with 5% fat-free milk, the membranes were incubated with specific anti-
Statistical analysis
Data were analyzed using SPSS 19.0 software and expressed as the mean ± standard deviation. One-way analysis of variance was carried out to compare the differences in API, tumor volume, AI, PI, HIF-1α, and VEGFA between the three groups. Multiple comparisons were conducted by the least significant difference test. p < 0.05 was considered to be statistically significant.
Results
Perfusion parameters within tumors using CEUS
As showed in Figure 1A(a)–(c), before LFUM treatment, all the tumors in the three groups showed abundant perfusion enhancement. Immediately after treatment, tumor perfusions of group 1 (3 min × 1) and group 2 (3 min × 4) were severely blocked. At 3 h after treatment, group 2 (3 min × 4) tumors demonstrated a little perfusion, while perfusion obviously increased in the tumors of group 1 (3 min × 1). The perfusion in tumors of the control group did not change.
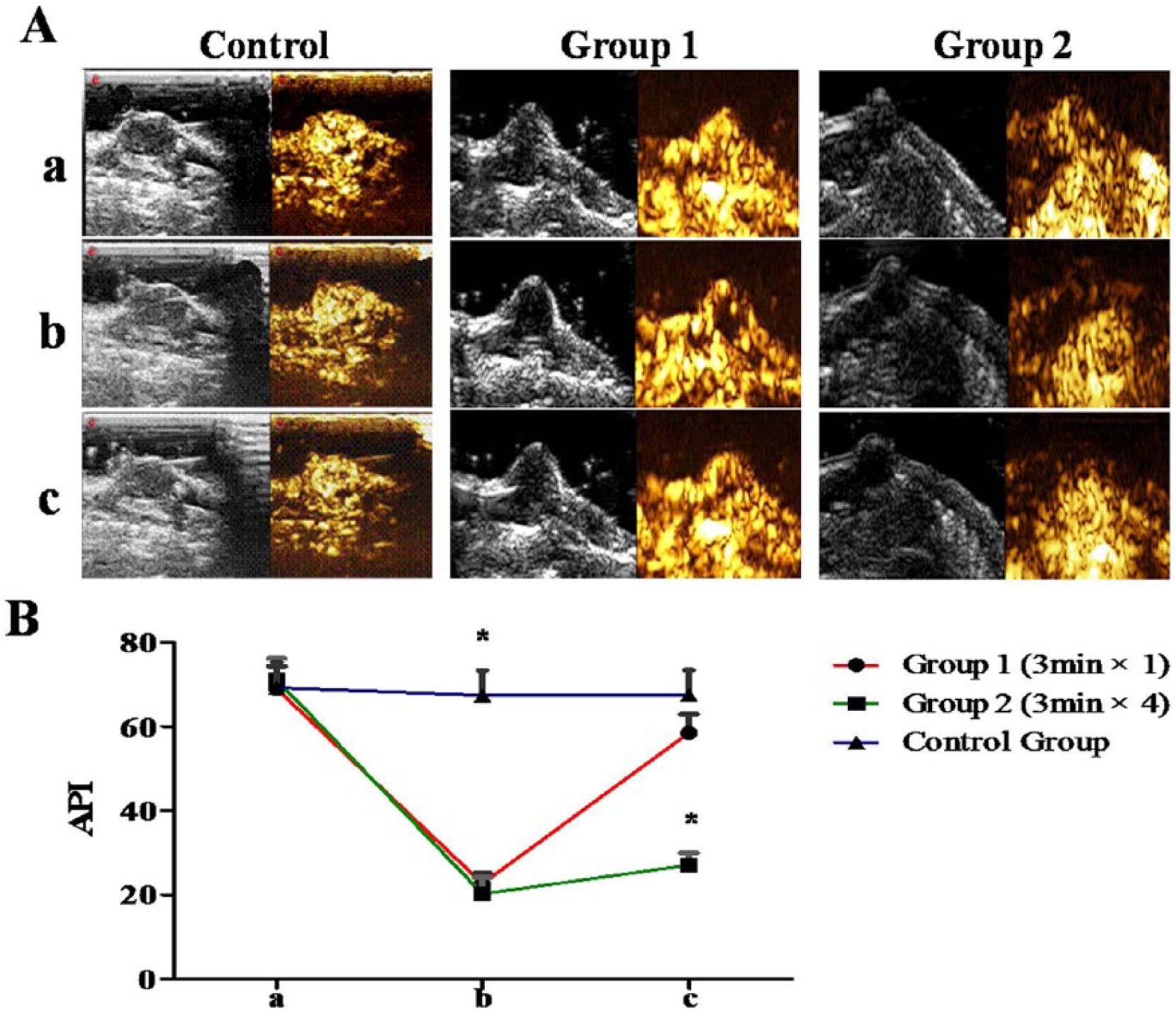
(A) Two-dimensional ultrasound and CEUS images and (B) average peak intensity (API) of prostate cancer xenografts in control group, group 1, and group 2. (a) Before the LFUM treatment, (b) immediately after the last treatment, and (c) 3 h after the last treatment (*p < 0.05 vs the other groups at the same time).
Further confirmation about the changing of perfusion within tumors was carried out using API analysis (Figure 1B). Before the LFUM treatment, the API in the control, group 1, and group 2 was calculated as 69.21 ± 6.01, 68.98 ± 5.37, and 71.1 ± 5.04, respectively. There were no significant differences among the three groups (both p > 0.05). Immediately after the first treatment, the value changed into 67.43 ± 5.96, 22.68 ± 2.51, and 20.26 ± 3.84, respectively. Compared with the control group, there was a significant decrease in the API in groups 1 and 2 (both p < 0.001), but this difference was not significant (p > 0.05). At 3 h after treatment, the perfusion value of three groups fluctuated again: 67.59 ± 5.87, 58.53 ± 4.39, and 27.07 ± 2.91, respectively. Tumor perfusion value had increased in the experimental groups, but the API value was significantly lower in group 2 than in group 1 (p < 0.001). The API value did not significantly differ between group 1 and the control group (p > 0.05).
Immunohistochemical detection of tumor apoptosis, cell proliferation, HIF-1α, and VEGFA
We used immunohistochemical methods to confirm the biological effect of LFUM used for different therapeutic times (Figure 2(a)). The degree of cell apoptosis was evaluated first (Figure 2(b)). Four days after the last treatment, the AI value of the control group, group 1, and group 2 were calculated to be 10.48% ± 1.23%, 9.25% ± 1.15%, and 20.37% ± 1.14%, respectively. Group 2 had the highest AI compared with other groups (both p < 0.001). After 14 days, the values changed to 11.12% ± 1.09%, 3.48% ± 1.17%, and 13.06% ± 2.06%, respectively. Group 2 still had a higher AI than group 1 (p < 0.001). The cell proliferation was also evaluated (Figure 2(c)). The PI values of control, group 1, and group 2 at 4 days after the last treatment were 24.94% ± 5.3%, 29.22% ± 3.68%, and 9.4% ± 2.01%, respectively. The PI values of the three groups 14 days after the last treatment were 28.97% ± 5.57%, 47.38% ± 6.34%, and 16.36% ± 3.17%, respectively. Group 1 had the highest PI compared to the other groups (both p < 0.001).

(a) Immunohistochemical detection of cell apoptosis and proliferation of prostate cancer xenografts in the control group, group 1, and group 2 at 4 and 14 days after the last LFUM treatment. TUNEL-positive nuclei and Ki-67-positive nuclei were stained brown or tan (magnification: 20×; scale bar: 20 μm). (b) AI and (c) PI of the control group, group 1, and group 2 at different times (*p < 0.05).
Further investigations on the positive expression of HIF-1α and VEGFA protein were conducted. As showed in Figure 3, at 4 days after the last treatment, HIF-1α expression levels in the control group, group 1, and group 2 were 20.16% ± 3.05%, 40.43% ± 5.18%, and 9.25% ± 1.42%, respectively. After 14 days, the values evaluated as 23.26% ± 3.2%, 28.1% ± 3.71%, and 12.29% ± 1.96%, respectively. HIF-1α expression was markedly decreased in group 1 (p < 0.001), and the value in group 1 became similar to that in the control group. Group 2 still had the minimum positive rate. As shown in Figure 3(c), 4 days after the last treatment, VEGFA expression in group 1 did not significantly differ from that in the control group (22.24% ± 4.01% vs 19.5% ± 2.81%; p > 0.05), but it was higher than that in group 2 (vs 10.21% ± 1.65%; p < 0.001). After 14 days, the expression did not significantly change between the control group and group 2; however, it obviously increased in group 1 (vs 56.9% ± 5.63%; p < 0.001)

(a) Immunohistochemical detection of hypoxia-inducible factor-1 α (HIF-1α) and vascular endothelial growth factor A (VEGFA) expression in the control group, group 1, and group 2 at 4 and 14 days after the last LFUM treatment. The cytoplasm of positive cells was stained brown or tan (magnification: 20×; scale bar: 20 μm). The expression level of (b) HIF-1 α and (c) VEGFA protein in the control group, group 1, and group 2 at 4 and 14 days after the last LFUM treatment (*p < 0.05).
Western blot analysis of the HIF-1α and VEGFA expression in tumor tissues
The HIF-1α and VEGFA protein levels were further confirmed via western blot analysis. The HIF-1α protein expression (Figure 4(a)) remarkably increased in group 1 but was the lowest in group 2 (p < 0.001) at 4 days after the last treatment. At 10 days after treatment, although it was still high, the value dramatically declined in group 1 (p < 0.001), and there was no significant change in the control group and group 2 (both p > 0.05). As shown in Figure 4(b), at 4 days after the last treatment, VEGFA expression did not significantly differ between group 1 and the control (p > 0.05) but clearly decreased in group 2 relative to the control group (p < 0.001). After 10 days, the VEGFA levels decreased slightly both in the control group and in group 2 and rapidly increased in group 1 (vs each other; p < 0.001).

HIF-1α and VEGFA protein expression in the control group, group 1, and group 2 at (a) 4 and (b) 14 days after the last LFUM treatment and (c and d) relative band density (*p < 0.05).
Tumor volume calculation
Tumor volumes were calculated at 14 days after the last treatment. Compared to the control group (92.7 ± 15.05 mm3), tumors grew the fastest in group 1 (183.8 ± 38.06 mm3; p < 0.001) and the slowest in group 2 (49.6 ± 12.09 mm3; p < 0.001; Figure 5).

(a) The image illustrates the tumor volume at day 14 after the last LFUM treatment: (1) group 1, (2) control group, and (3) group 2. (b) Tumor volume of the above three groups (*p < 0.05).
Discussion
Tumor angiogenesis plays an important role in the growth, invasiveness, and metastasis of PCa, and it has been the focus of several anti-angiogenic studies. US therapy receives more attention due to its non-invasiveness and efficiency. Many studies have proved that low-frequency US combined with microbubbles could induce obvious microvessel damage in tumors mainly due to inertial cavitation, which can cause cell necrosis, apoptosis, and enhanced responses to radiation therapy.18,19,22,29,30 However, these studies used a wide range of US parameters and radiation times. In this study, we evaluated the effect of LFUM on interrupting bleeding under the same conditions and with different radiation times. The results showed that compared to the control group, the tumor blood supply was efficiently blocked immediately after radiation in the experiment groups. However, the blocking effect was obviously durable in group 2, in which the radiation was repeated four times at 3-h intervals on each treatment day. In group 1, the radiation treatment was administered only once per day, and the blood reperfusion recovered to normal levels after 3 h of the treatment.
Hypoxia conditions can upregulate proteins associated with angiogenesis, such as VEGFA, HIF, cyclooxygenase 2 (COX-2), and iNOS, which are correlated with both tissue repair and tumor progression. Contrary to vessel damage of LFUM, many studies have shown that therapeutic US affects angiogenesis. Scheven et al. 27 showed that low-frequency US influenced VEGF production by odontoblast-like cells and promoted dentine repair. Enwemeka et al. 31 reported that low-intensity US might enhance the healing process of the human tendo calcaneus, while high-intensity US was not necessary to augment it. Similarly, Barzelai et al. 32 indicated that despite limb ischemia, low-intensity US could significantly improve tissue angiogenesis and blood flow. Furthermore, it is well established that PCa is exposed to fluctuating oxygen tensions due to a changing environment, and both chronic and acute hypoxia exist in PCa, as HIF-1α and VEGFA protein levels are both increased in PCa compared to benign prostate hyperplasia (BPH).5,15,33 However, no reports have investigated the effect of LFUM on the hypoxia environment of PCa, whose primary effect on the body is limiting the blood supply.
According to our investigation, we found that the tumor hypoxia response to LFUM treatment was opposite under different radiation times, even when the conditions used were identical. Furthermore, the expression levels of HIF-1α and VEGFA, which are the intrinsic markers of hypoxia and angiogenesis, changed constantly with observation time. As radiation was carried out only once each time, HIF-1α expression was the highest at 4 days after treatment, then gradually declined, although it remained higher in group 1 than in the other groups. The VEGFA level in group 1 increased continuously and peaked at 14 days after treatment. When the radiation was repeated four times in each treatment, HIF-1α expression significantly declined and increased slightly thereafter but remained lower than the other two groups. The change in the VEGFA expression was similar.
Immunohistochemical analysis indicated that under the same conditions of LFUM, radiation for a longer time with durable blocking blood effect could inhibit proliferation and promote apoptosis of tumor cells, whereas if the blood flow returned, tumor hypoxia was stimulated, cell proliferation was promoted, and apoptosis was inhibited. These results were confirmed by the analysis of tumor volume: Tumor volume increased obviously in the hypoxia-stimulated group, whereas tumors grew slowly in the hypoxia-suppressed group.
Previous studies on the hypoxia response may give several possible reasons for this: Long-term blockage of blood supply leads to extreme hypoxia that drives cells to death, whereas temporary blockage results in a less extreme hypoxic environment where tumor cells adapt to low oxygen levels and exhibit survival characteristics.9,34 HIF-1 is a major regulator of oxygen homeostasis and mediates cellular hypoxia response by promoting processes, including angiogenesis and glycolysis. 35 The interrupting perfusion effect of LFUM might induce acute hypoxia, which was correlated to upregulate the expression of HIF-1α and VEGF proteins and downregulate differentiation-specific proteins or proteins involved in regulating cell proliferation. 36 Hypoxic enhancement of HIF-1α expression and activity was correlated with both the phosphoinositide 3-kinase (PI3K)/Akt/mechanistic target of rapamycin (mTOR) and p44/42 mitogen-activated protein kinase (MAPK) pathways.34,37
HIF-1α accumulates and stabilizes only at low oxygen tension. When oxygen tension increases to normal levels, HIF-1α protein is rapidly and constantly degraded by ubiquitination and proteasomal degradation.
38
This is consistent with the changing HIF-1α protein levels in group 1. HIF-1α was also correlated with aberrant p53 accumulation, which upregulates VEGF expression, promoting tumor angiogenesis.
11
Huang et al.
16
revealed that HIF-1α regulates the PCa PC3 cell proliferation, apoptosis, and migration to some extent by regulating the expression of target genes such as
In conclusion, this study investigated the impact of different radiation times of LFUM on prostate tumors. We found that when the radiation was repeated four times on each treatment day, the effects of interruption were more durable, and the treatment inhibited cell proliferation and promoted apoptosis. Moreover, when the radiation was carried out once per treatment day, the hypoxia response was stimulated, HIF-1α and VEGFA expression was increased. A number of studies indicated that US-stimulated microbubble radiation provides a physical effect for anti-angiogenic therapy. However, under the same conditions, the effects of LFUM on PCa are opposite when using different radiation times. The effect is at least partly dependent on the influence of the hypoxic environment within the tumor. These results add to the information that the use of LFUM as an anti-tumor method is a complex process and should be further investigated before clinical application.
Footnotes
Acknowledgements
The authors acknowledge the assistance of Prof. Yuanyi Zheng. R.H. and Y.X. are joint first authors. All experimental procedures were in strict accordance with the policy of the Institutional Animal Care and Use Committee (IACUC) of Shanghai Jiao Tong University Affiliated Sixth People’s Hospital, Shanghai, China, and conformed to the Ministry of Science and Technology of Health Guide for Care and Use of Laboratory Animals (Publication No. 398, 2006). The animals in this study were anesthetized by pentobarbital sodium, and all efforts were made to minimize suffering. The IACUC has approved this study.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (Grant No. 81271597) and Construction of Clinical Assistant Department of Hospital in Shanghai (SHDC.22015001).
