Abstract
Many studies have analyzed the relationship between hypoxia inducible factor 1 alpha expression and its relation to differentiation, lymph node metastasis, and other clinicopathological variables of esophageal carcinoma, but the results are still inconsistent. This meta-analysis was carried out to explore hypoxia inducible factor 1 alpha in esophageal carcinoma and its correlation with clinicopathological features and prognosis, in order to provide comprehensive reference for clinic. A total of 18 studies including 1566 patients with esophageal squamous cell carcinoma were enrolled. The results showed that compared with para-carcinoma tissue, the expression of hypoxia inducible factor 1 alpha was significantly enhanced (odds ratio = 0.122, 95% confidence interval = 0.074–0.201, p = 0.000); hypoxia inducible factor 1 alpha was associated with differentiation (odds ratio = 1.458, 95% confidence interval = 1.108–1.920, p = 0.007), T classification (odds ratio = 0.457, 95% confidence interval = 0.265–0.786, p = 0.005), lymph node metastasis (odds ratio = 0.337, 95% confidence interval = 0.185–0.614, p = 0.000), and pathological tumor–node–metastasis stage (odds ratio = 0.362, 95% confidence interval = 0.177–0.740, p = 0.005), whereas there was no relation to histological grade, lymphatic vessel invasion, blood vessel invasion, 3- to 5-year overall survival and disease-free survival. Patients with hypoxia inducible factor 1 alpha overexpression had poor differentiation, increased depth of tumor invasion, more lymph node metastasis, and late pathological tumor–node–metastasis stage. Hypoxia inducible factor 1 alpha could be an indicator for differentiation, T classification, lymph node metastasis, and pathological tumor–node–metastasis stage, and it is worth further study.
Keywords
Introduction
Solid tumors usually have a hypoxic microenvironment, and hypoxia inducible factor 1 alpha (HIF-1α) has been confirmed to play a key role in adaptation to hypoxia. Downstream target genes of HIF-1α have reached more than 100 types, closely related to growth, metabolism, invasion, metastasis, and other malignant biological behaviors. 1 Our previous study showed that HIF-1α expression in esophageal squamous cell carcinoma (ESCC) under hypoxic condition increased significantly, which boosted migration and invasion ability of ESCC obviously. 2 In 2014, Ping et al. 3 published a meta-analysis that discussed about HIF-1α with clinicopathological and prognostic significance in 942 patients with ESCC. However, esophageal adenocarcinoma is also increasingly becoming common in Western countries, and after 3 years, some new related studies have been published, so the number of cases has been further expanded. Therefore, in this study, a meta-analysis was used again to discuss HIF-1α expression in esophageal carcinoma (including squamous and adenocarcinoma) and its correlation with clinicopathological variables and prognosis, so as to provide comprehensive reference for clinic and translational research.
Materials and methods
The source of literature
Literatures about HIF-1α expression and its relation to esophageal carcinoma were collected from Chinese Biomedical Literature Database, China Academic Journal Full-Text Database, the Cochrane Library, PubMed, and EMBASE database. The keywords were “esophageal/oesophageal carcinoma” or “esophageal/oesophageal cancer” and “hypoxia inducible factor 1 alpha,” “HIF-1 alpha,” or “HIF-1α.” The last retrieval time was 30 April 2017.
Inclusion and exclusion criteria
(1) Literatures were about HIF-1α and esophageal carcinoma, so adenocarcinoma of gastroesophageal junction and gastric cancer were excluded. (2) Original material was detailed, and relevant data could be extracted. (3) The objective of study met pathological diagnostic criteria of Union for International Cancer Control (UICC). Clinicopathological data must be from surgical specimens, without undergoing radiochemotherapy or other anti-tumor treatment; (4) Domestic literatures must be published in the national core journals collected at Peking University Library, and foreign ones were published in full-text English. (5) Statistical method was proper, and expression of the results was clear. Odds ratio (OR), hazard ratio (HR), and 95% confidence interval (CI) could be calculated.
Quality assessment
Evaluation guide of case-control study was used for every independent literature to assess whether there was bias and its influence extent, 4 including the following aspects: (1) The object of study was judged pathologically as esophageal carcinoma and whether its baseline characteristics such as gender, age, and tumor location were clear; (2) whether the data were from postoperative specimen and HIF-1α expression was tested by immunohistochemistry; (3) whether statistical methods were proper, such as comparison of HIF-1α expression in carcinoma and para-carcinoma tissue was by chi-square test and correlation test was by spearman rank correlation analysis; (4) whether experimental design was case-control study; and (5) whether the existence of bias in research was discussed. Among the above five items, if one was met, then 1 point was given; ⩾3 points were considered as reliable quality. According to the unified quality standards, two assessors (S.W.J and Q.X.) evaluated literatures independently, extracted data, and then cross-checked each other. In case of disagreement, it was resolved through discussion or a third researcher (J.W.) to help.
Statistical methods
Stata 11.0 statistical software provided by the Cochrane collaboration was used for this meta-analysis. As effect sizes, OR, HR, and their 95% CIs were calculated, Q test was applied for heterogeneity test. There was heterogeneity if p ⩽ 0.05, and it needed to use random-effects model; otherwise, if p > 0.05, meant no heterogeneity, and fixed-effects model was used. Z test was used as significance test for combined OR and HR. Publication bias was assessed through funnel chart whether symmetric or not. It may exist if funnel chart was asymmetric. Egger’s linear regression was used for publication bias test.
Results
The retrieval results and quality evaluation
A total of 175 literatures were found initially, after reading the title or abstract, 140 cell or animal researches and reviews were excluded. Within the remaining 35 literatures, 5 had repeated published data, 6 included gastroesophageal junction or gastric carcinoma, 4 were excluded for its clinicopathological data from microscopy specimen, and 2 were excluded for its incomplete data, so eventually a total of 18 studies including 1566 patients were involved in this meta-analysis.5–22 All of them discussed HIF-1α in ESCC. A flow diagram showing how we screened relevant eligible studies is depicted in Figure 1. Positive expression rate of HIF-1α was 30.8%–90.0%. The baseline characteristics of included studies are shown in Table 1.

Flow chart of the meta-analysis.
Baseline characteristics of included studies.
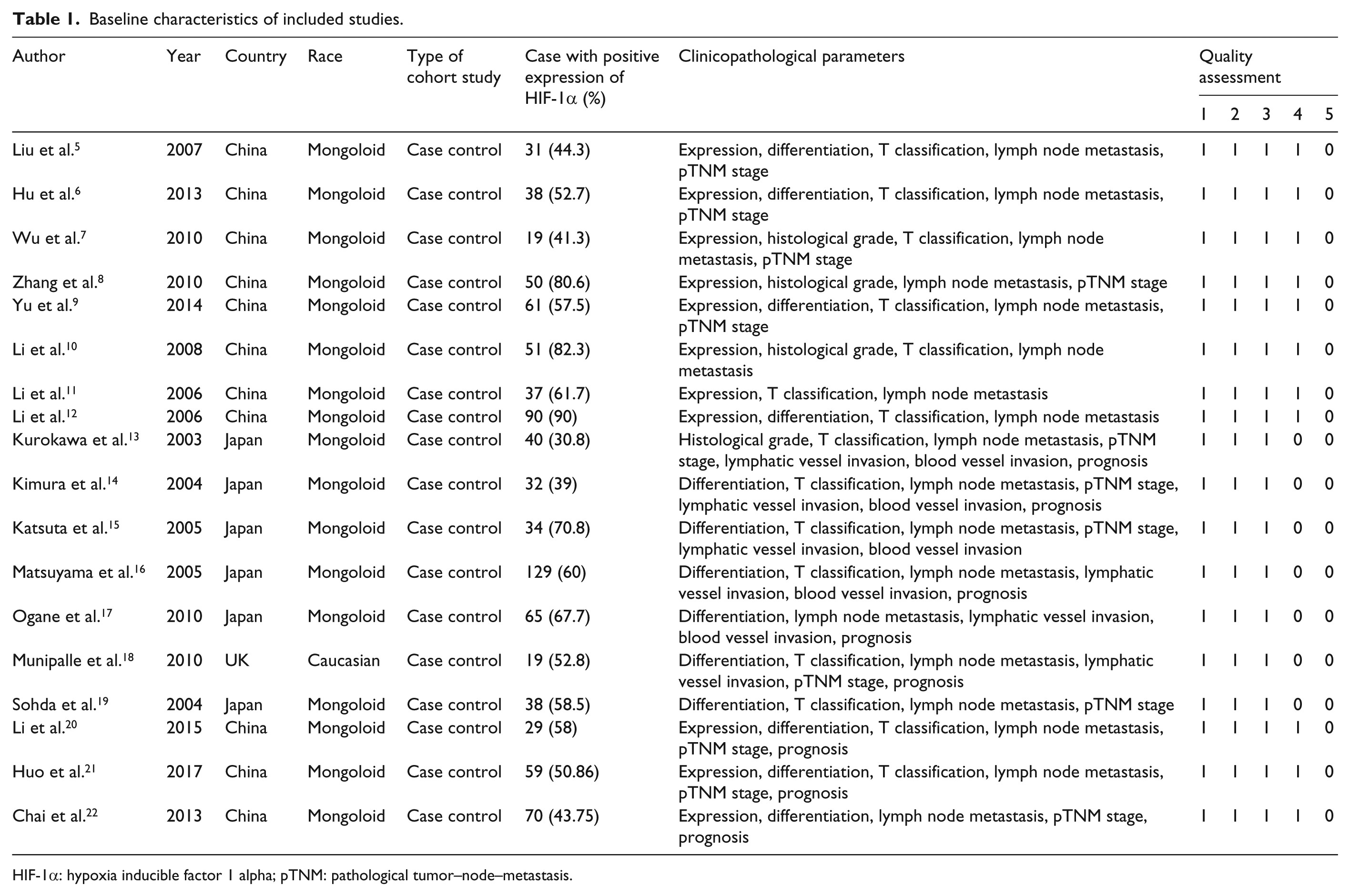
HIF-1α: hypoxia inducible factor 1 alpha; pTNM: pathological tumor–node–metastasis.
HIF-1α expression in ESCC
A total of 11 literatures analyzed HIF-1α expression in ESCC and para-carcinoma tissue. Because of statistical heterogeneity between the two groups (p = 0.010 < 0.05), random-effects model was used. It showed that there was significant difference between ESCC and para-carcinoma tissue (OR = 0.122, 95% CI = 0.074–0.201, p = 0.000), indicating that HIF-1α expression in ESCC was much enhanced than that in para-carcinoma tissue (Figure 2).

Forest plot of HIF-1α expression in ESCC.
Relationship between HIF-1α and clinicopathological parameters of ESCC
HIF-1α and differentiation
A total of 13 literatures were selected, including 367 cases of poor differentiation and 942 of moderate and well differentiation. There was no significant heterogeneity between the two groups (p = 0.207 > 0.05), so fixed-effects model was used. The result showed that there was statistical significance (OR = 1.458, 95% CI = 1.108–1.920, p = 0.007), indicating that with the decrease of tumor differentiation, HIF-1α expression increased (Figure 3).
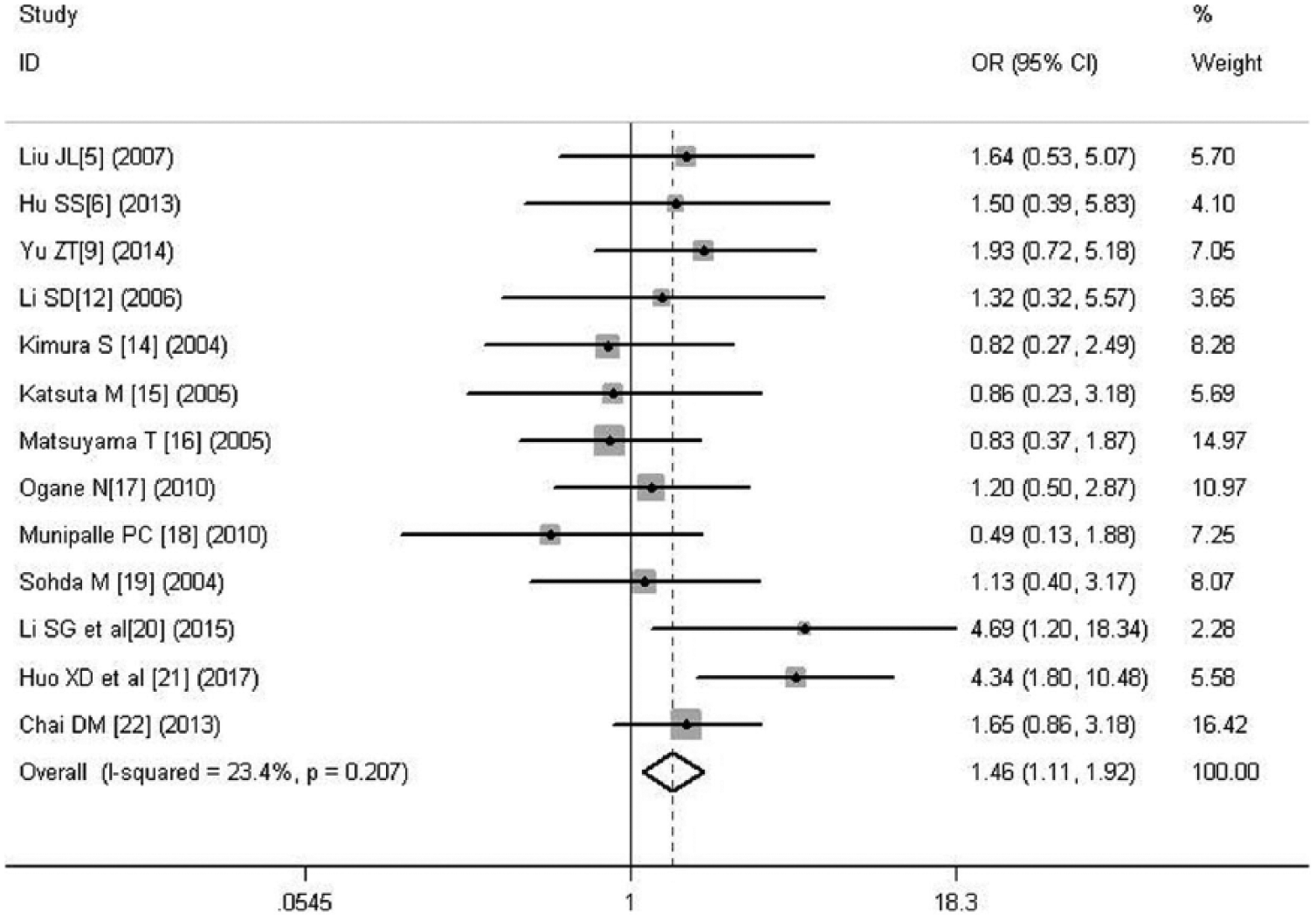
Forest plot of the relation between HIF-1α and differentiation.
HIF-1α and histological grade
A total of four literatures were involved, including 171 cases of G1 + G2 and 119 of G3 + G4. Because of statistical heterogeneity between the two groups (p = 0.011 < 0.05), random-effects model was used. There was no significant difference between G1 + G2 group and G3 + G4 group (OR = 1.194, 95% CI = 0.307–4.642, p = 0.798 > 0.05), indicating that there was no correlation between HIF-1α and histological grade (Figure 4).

Forest plot of the relation between HIF-1α and histological grade.
HIF-1α and T classification
A total of 16 literatures were selected, including 456 cases of T1 + T2 patients and 888 cases of T3 + T4 patients. For statistical heterogeneity existing between the two groups (p = 0.000 < 0.05), random-effects model was used. There was significant difference between T1 + T2 group and T3 + T4 group (OR = 0.457, 95% CI = 0.265–0.786, p = 0.005). It showed that with enhancement of invasion depth, HIF-1α expression increased (Figure 5).

Forest plot of the relation between HIF-1α and T classification.
HIF-1α and lymph node metastasis
A total of 17 literatures were analyzed, including 759 negative cases and 817 positive cases. Significant heterogeneity was found between the two groups (p = 0.000), so we used random-effects model. The result showed that there was statistical significance between the two groups (OR = 0.337, 95% CI = 0.185–0.614, p = 0.000), indicating that HIF-1α expression in patients with lymph node metastasis was higher than that without lymph node metastasis (Figure 6).
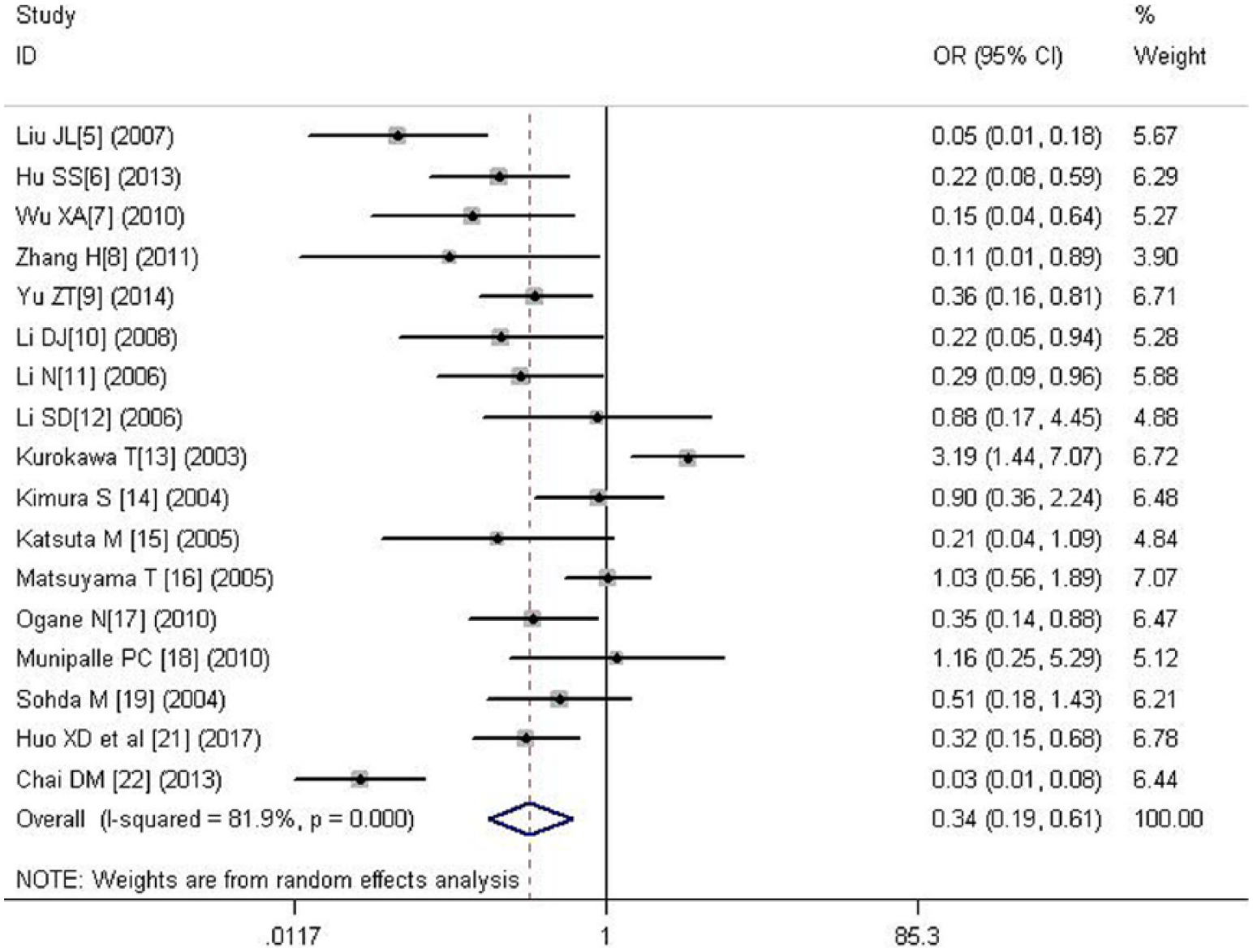
Forest plot of the relation between HIF-1α and lymph node metastasis.
HIF-1α and pathological tumor–node–metastasis stage
A total of 12 literatures were involved, including 454 cases of I + II stages and 490 of III + IV stages. Because of significant heterogeneity between the two groups (p = 0.000), random-effects model was used. There was significant difference (OR = 0.362, 95% CI = 0.177–0.740, p = 0.005), indicating that HIF-1α had correlation with pathological tumor–node–metastasis (pTNM) stage. The later pTNM stage, the higher the HIF-1α expression (Figure 7).

Forest plot of the relation between HIF-1α and pTNM stage.
HIF-1α and lymphatic vessel invasion
A total of five literatures including 171 cases with non-lymphatic vessel invasion and 400 with lymphatic vessel invasion were involved. Because of significant heterogeneity between the two groups (p = 0.005), random-effects model was used. It showed no statistical significance (OR = 0.560, 95% CI = 0.219–1.431, p = 0.226), indicating that there was no correlation between HIF-1α and lymphatic vessel invasion (Figure 8).

Forest plot of the relation between HIF-1α and lymphatic vessel invasion.
HIF-1α and blood vessel invasion
A total of five literatures including 350 cases with non–blood vessel invasion and 221 with blood vessel invasion were involved. There was no significant heterogeneity between the two groups (p = 0.054 > 0.05), so fixed-effects model was used. The result showed no statistical significance between the two groups (OR = 0.971, 95% CI = 0.667–1.413, p = 0.877), indicating that there was no correlation between HIF-1α and blood vessel invasion (Figure 9).
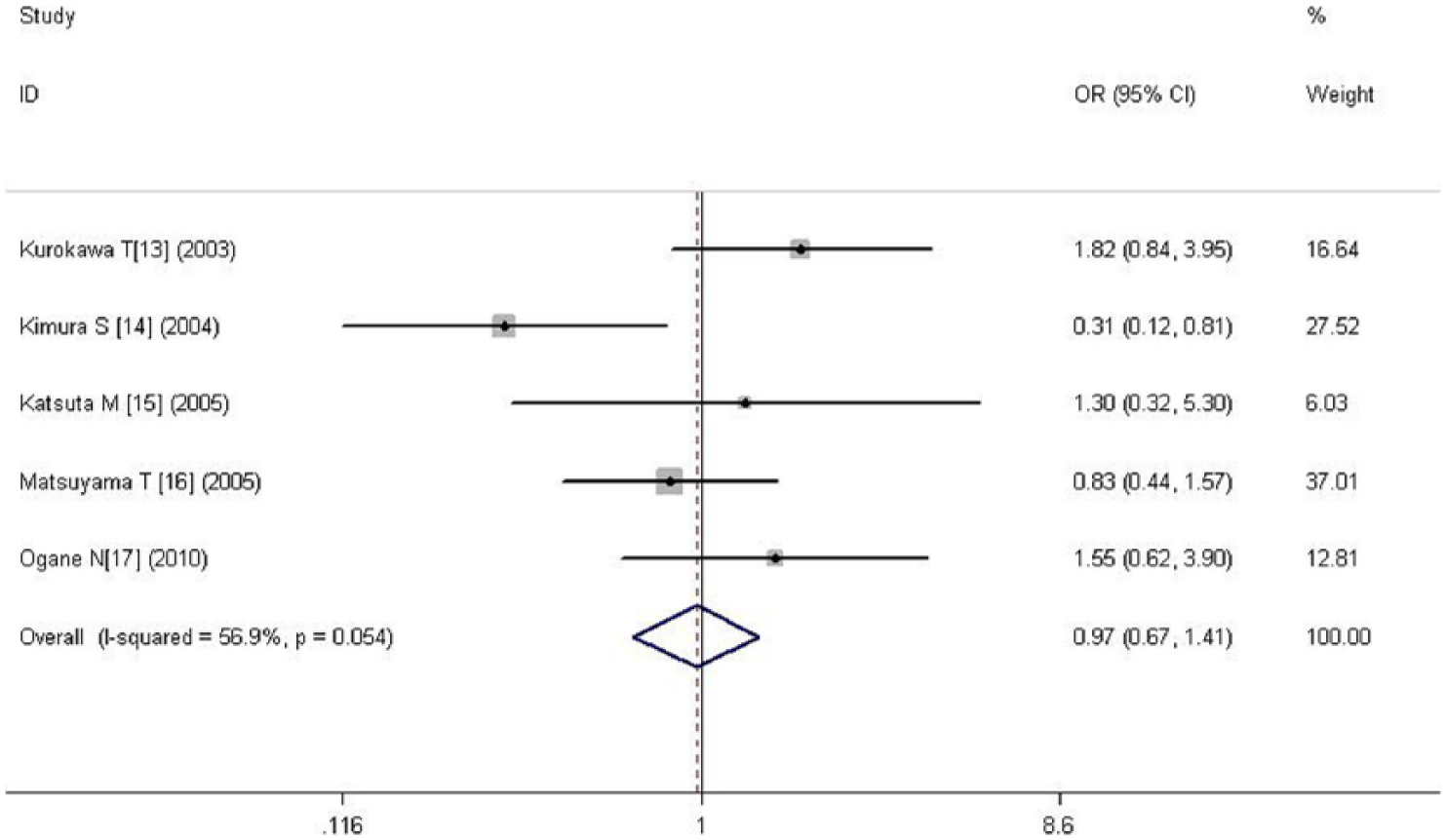
Forest plot of the relation between HIF-1α and blood vessel invasion.
Relationship between HIF-1α and prognosis of ESCC
HIF-1α and 3- to 5-year overall survival
Meta-analysis of three and six studies on 3- to 5-year overall survival (OS). It revealed that HIF-1α overexpression had no correlation with 3- to 5-year OS (HR = 0.823, 0.533–1.272, p = 0.381; HR = 0.840, 0.606–1.165, p = 0.295, respectively; Figures 10 and 11).
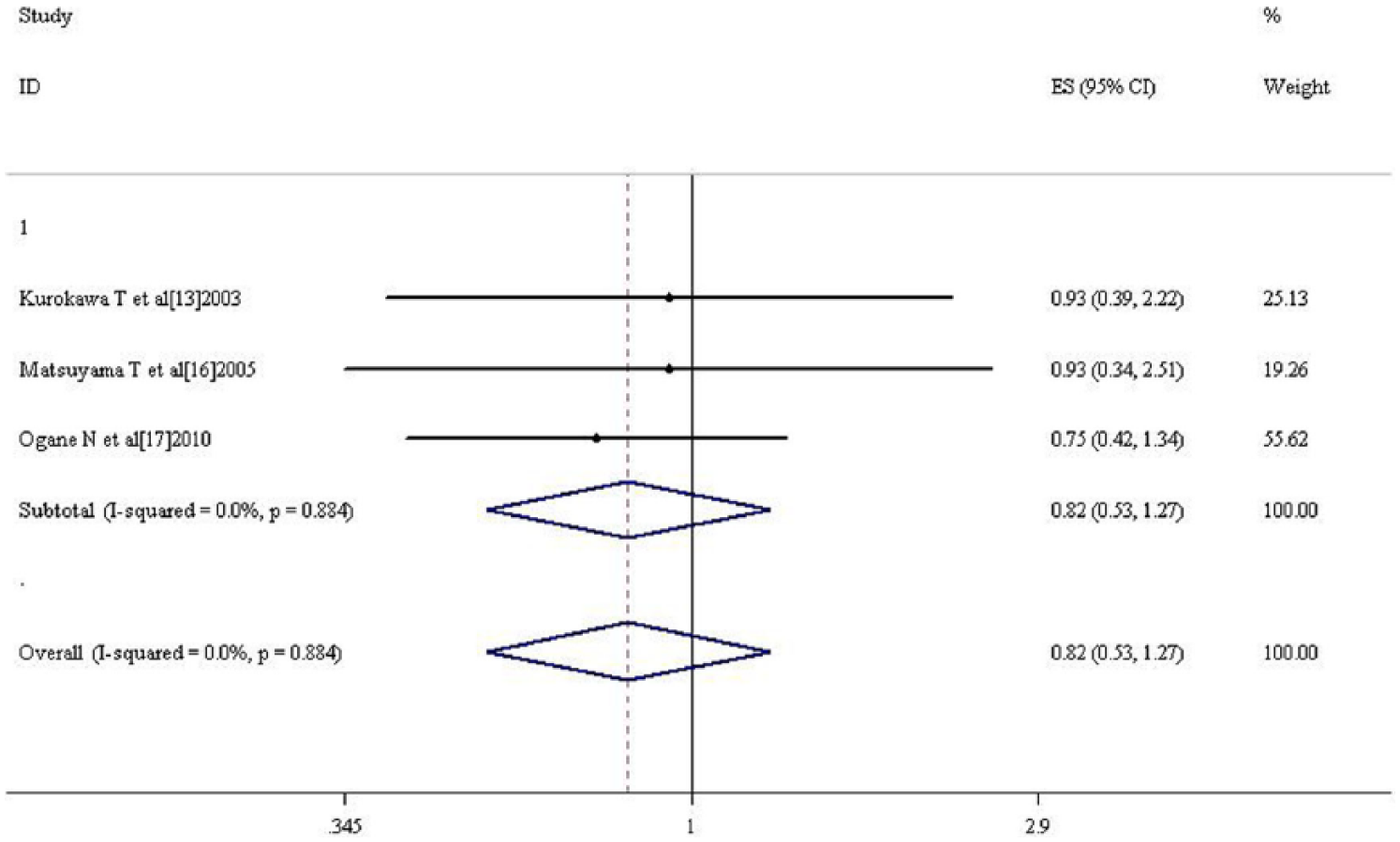
Forest plot of the relation between HIF-1α and 3-year OS.

Forest plot of the relation between HIF-1α and 5-year OS.
HIF-1α and 3- to 5-year disease-free survival
Two studies provided information on disease-free survival (DFS). The pooled HRs estimate for 3- to 5-year DFS for both was 0.790 (95% CI = 0.478–1.306, p = 0.358), indicating that HIF-1α overexpression was not associated with DFS of ESCC (Figures 12 and 13).

Forest plot of the relation between HIF-1α and 3-year DFS.

Forest plot of the relation between HIF-1α and 5-year DFS.
Sensitivity analysis
In order to ensure that the combined OR and HR values were not severely driven by the trial whose OR and HR were similar to the combined ones, a sensitivity analysis was proceeded by taking them out. After the removal, recombined OR/HR for the remaining trials is shown in Table 2, which were almost consistent with previous results.
Sensitivity analysis through removing relevant literature.

OR: odds ratio; HR: hazard ratio; CI: confidence interval; pTNM: pathological tumor–node–metastasis; OS: overall survival.
Publication bias analysis
Egger’s test was performed to assess the publication bias. Table 3 has shown there was no indication of a publication bias on HIF-1α expression, clinicopathological parameters, and OS. Because the number of studies about DFS was comparatively few, both publication bias test and sensitivity analyses were not performed.
Publication bias between HIF-1α and clinicopathological parameters.

HIF-1α: hypoxia inducible factor 1 alpha; CI: confidence interval; pTNM: pathological tumor–node–metastasis; OS: overall survival.
Discussion
When solid tumor volume exceeds 1 mm3, its blood supply cannot meet the need for growth, and the midst is relative in lack of oxygen environment. 23 Under hypoxia, tumor cell makes some stress response, and many adaptive changes in the gene transcription and expression occur. 24 HIF-1α is the core transcription factor for tumor cells to adapt to hypoxia, and it plays an important role in energy metabolism, invasion, and metastasis. 25
As one of solid tumors, esophageal cancer is a common malignant tumor. Despite advances in screening and multimodal management, its overall survival is still poor. A great need remains for additional information on the biological behavior of esophageal cancer and to identify new markers for prognostic indicators and targets to new therapeutic strategies. There have been many published articles about HIF-1α protein expression in ESCC and its relationship with clinicopathological features, but the results are still inconsistent. Although esophageal adenocarcinoma is also increasingly common in Western countries, study of HIF-1α relation to it is few. In this study, a meta-analysis was used for to analyze HIF-1α expression and its clinical significance in esophageal carcinoma (including squamous and adenocarcinoma), in order to provide comprehensive reference for clinic. According to the inclusion and exclusion criteria, 18 literatures of high quality were involved, all of which discussed HIF-1α in ESCC. From Table 1, we could see that the included studies were all ⩾3 points, which meant higher quality. Positive rate of HIF-1α was between 30.8% and 90.0%, and these researches covered different variables such as differentiation, T classification, lymph node metastasis, and prognosis, which could provide more comprehensive data. The results have shown that HIF-1α protein expression in ESCC was significantly enhanced than that in normal tissues (OR = 0.122, 95% CI = 0.074–0.201, p = 0.000). HIF-1α had correlation with differentiation, T classification, lymph node metastasis, and pTNM stage, while there was no relation to histological grade, lymphatic vessel invasion, blood vessel invasion, 3- to 5-year OS, or 3- to 5-year DFS. HIF-1α could be a critical determinant of differentiation, T classification, lymph node metastasis, and pTNM stage. We also conducted a sensitivity analysis for detecting whether the combined OR/HR was severely driven by the trials whose OR and HR were similar to the combined ones. After taking out the relevant literature, we found that the results were basically the same as before (Table 2).
Because majority of included studies were from China and Japan, all cases were ESCC patients. Only one literature discussed about HIF-1α in esophageal adenocarcinoma. 26 However, it was a pity that this study did not meet our inclusion criteria. Its result showed that HIF-1α did not associate with tumor or nodal status, and perhaps HIF-1α had little effect on the progression of esophageal adenocarcinoma. Of course, it needs more studies to verify.
Considering that original publication bias exists to a certain extent, and it may affect the reliability of meta-analysis results, the 18 included articles were also investigated by Egger’s analysis. It showed that the funnel plot was symmetric (p > 0.05; Table 3), which indicated the bias was small, at least, the potential bias had no substantial influence on the final conclusion, which further increased the reliable conclusion that overexpression of HIF-1α protein increases the risk of ESCC. HIF-1α could be an indicator for differentiation, T classification, lymph node metastasis, and pTNM stage. However, this meta-analysis has limitations from the published literature, because the adopted researches were all openly published, and languages were only in Chinese and English, so the unpublished literatures and existing language bias also affect the results. In addition, despite the expression of HIF-1α was all detected by the immunohistochemical methods, the antibody manufacturer, dilution, and judgment standard were not identical, which may also have influence on the results.
In conclusion, this study showed that HIF-1α expression in ESCC is much enhanced. HIF-1α could be an indicator for differentiation, T classification, lymph node metastasis, and pTNM stage, so it is worth further study.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
