Abstract
Progression of breast cancers often depends on hormones among which human growth hormone is prominently involved in breast cancer progression. Earlier studies have reported constitutive activation of nuclear factor-κB, a key regulator of growth hormone receptor–mediated signaling pathway in breast carcinoma, but the precise molecular mechanisms are still elusive. In this study, we investigated the effect of human growth hormone on nuclear factor-κB activation and epithelial–mesenchymal transition in breast carcinoma. Our results explored that autocrine production of human growth hormone enhances cellular proliferation by the activation of nuclear factor-κB (65 kDa) and downregulation of E-cadherin expression. Furthermore, enhanced nuclear factor-κB expression significantly increases cell proliferation and diminishes apoptosis in MCF-7 cell line. Increased expression of nuclear factor-κB significantly enhances mammary carcinoma cell migration and invasion stimulated by autocrine human growth hormone, which results in epithelial–mesenchymal transition of MCF-7 cells. In conclusion, our study revealed the influence of human growth hormone on nuclear factor-κB activity and epithelial–mesenchymal transition in mammary carcinoma. Our findings will help to understand molecular role of “growth hormone–nuclear factor-κB axis” in mammary carcinogenesis which may facilitate the discovery of suitable pathway inhibitors for disease treatment.
Keywords
Introduction
Mammary epithelial carcinoma is one of the leading causes of cancer-related mortality among females in the West predominantly due to a high occurrence of metastasis.1,2 The role of endocrine system in development of mammary gland has been well established. Among the hormones, growth hormone (GH) is an important regulator of mammary gland development3,4 which may promote cell proliferation. 5 However, GH is considered to be true cytokine whose local production directly contributes to tumor progression. 6 In addition, there are reports suggesting the role of growth hormone receptor (GHR)-mediated signaling pathways in development of breast cancer in human either by autocrine or paracrine mechanisms.7,8 Earlier study reported constitutive activation of nuclear factor-κB (NF-κB) and resulting overexpression of anti-apoptotic genes in breast cancer.9–11 However, the precise reasons for activation of NF-κB are not known, but hormonal factors are likely candidates. We hypothesize that human growth hormone (hGH) has been shown to exert anti-apoptotic and proliferative effects through NF-κB. E-cadherin (CDH1) was shown to be aberrantly repressed in many epithelial cancers including breast cancer. 12 Downregulation of E-cadherin expression is a key initiating event in epithelial–mesenchymal transition (EMT),13,14 and transcription factors that repress E-cadherin have often been defined as inducers of EMT. Here, we showed that autocrine production of hGH resulted in increased NF-kB expression and relocalization of E-cadherin to the cytoplasm, leading to loss of cell–cell contacts and decreased cell height. In view of the central role of transcription factor NF-κB in the regulation of EMT, we demonstrate that hGH-dependent constitutive expression of NF-κB confers invasive and metastatic properties of breast cancer cells.
Materials and methods
Cell lines and transfection
Mammary carcinoma cell line MCF-7 was obtained from the American Type Culture Collection (ATCC) and stably transfected with either the wild-type or translation-deficient mutant hGH gene (kindly provided by Dr Peter Lobie, National University of Singapore) using Lipofectamine2000 transfection reagent (Life Technologies, Carlsbad, CA, USA). hGH-MCF-7 and MUT-MCF-7 cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum (FBS), penicillin (100 units/mL), and streptomycin (100 mg/mL) at 37°C in a humidified 5% CO2 incubator.
Western blot analysis
Protein extraction and western blot analysis were performed as per the protocols described earlier. 15 Briefly, MUT-MCF-7 and hGH-MCF-7 cells were lysed with hot sample buffer containing 1% sodium dodecyl sulfate (SDS) and then centrifuged for 15 min at room temperature. Protein extracts were separated by 10% SDS–polyacrylamide gel electrophoresis (PAGE) and transferred to polyvinylidene difluoride (PVDF; Hybond C-Extra; Amersham Pharmacia, Aylesbury, UK). After blocking, membranes were incubated with primary anti–mouse monoclonal antibodies (NF-κB (1:1000), E-cadherin (1:1000), P-cadherin (1:1000), and matrix metalloproteinase-9 (MMP-9; 1:1000) obtained from Cell Signaling Technology, Beverly, MA, USA)) for 1 h. The membranes were then washed and incubated with horseradish peroxidase–conjugated goat anti-mouse secondary antibody (1:10,000; Upstate Biotechnology, Lake Placid, NY, USA). Blots were stripped and reprobed using anti-actin antibody (Sigma Aldrich, St. Louis, MO, USA). Protein bands were detected by the Phototype horseradish peroxidase western blot detection system (Cell Signaling Technology).
Luciferase reporter assay
The hGH and MUT hGH vectors were transfected in MCF-7 cells (2 × 105 cells/well in six-well plate) together with luciferase reporter plasmids (2 μg/well) and an internal control pGL4.32 (luc2P/NF-κB-RE/Hygro) vector (Promega, Madison, WI, USA; 2 μg/well) expressing the Renilla luciferase. Transfections were performed using FuGENE 6 (Roche Applied Science, Indianapolis, IN, USA). Following a brief freeze-thawing cycle, the insoluble debris was removed by centrifugation at 4°C for 2 min at 14,000g. A volume of 20-μL aliquots of the supernatant were then immediately processed for sequential quantification of both firefly and Renilla luciferase activity (dual-luciferase assay system). The activity of the co-transfected Renilla reporter plasmid was used for normalization of transfection efficiency. All reporter assays were performed in triplicates.
Flow cytometry
Asynchronously growing MCF-7 clones (hGH-MCF-7) and its mutants (MUT-MCF-7) were treated with hypotonic buffer (1 mg/mL sodium citrate and 0.3% Igepal) for 20 min. The detached nuclei were gently pipetted for a minute and were collected for green fluorescence acquisition. Similarly, 105 cells of each clone were plated and serum starved for 24 h. Briefly, cells were incubated with fluorescein isothiocyanate (FITC)-Annexin V and propidium iodide (PI; BD Biosciences) in binding buffer for 15 min in dark. Stained cells were subjected to flow cytometry analysis using FACSCanto II flow cytometer (BD Biosciences, San Jose, CA, USA). The data were acquired with flow cytometer (Guava 8 HT EasyCyte; Millipore, Billerica, MA, USA); 50,000 events were acquired with flow rate of 0.59 mL/s. Data were analyzed by FlowJo software. Experiments were done three times in duplicates.
Immunofluorescence microscopy
hGH-MCF-7 and MUT-MCF-7 cells were adhered on glass cover slips and fixed with 4% paraformaldehyde for 20 min, washed with phosphate-buffered saline (PBS), and then permeabilized in 0.1% Triton X-100/PBS. After blocking in PBS containing 1% bovine serum albumin (BSA) for 1 h at room temperature, the cells were incubated with primary antibody (E-cadherin) overnight at 4°C followed by incubation with Alexa 488-FITC for 1 h at room temperature within a moist chamber. Following washing with PBS, the cover slips were mounted on glass slides with DAPI Fluoromount G (SouthernBiotech, Birmingham, AL, USA). Images were acquired with Confocal Laser Scanning microscope Lieca SPE Biosystem.
Cell viability assay
To determine cell growth of transfected clones, 2000 cells of each clone were plated in triplicate in 96-well plates and cell viability was assessed by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. MTT reagent (diluted from a 4 mg/mL solution in PBS) was added to all the wells at a final concentration of 0.4 mg/mL, and the cells were further incubated for 4 h at 37°C. The reaction was terminated by adding 200 μL/well dimethyl sulfoxide (DMSO), and absorbance was read at 570 nm in an enzyme-linked immunosorbent assay (ELISA) plate reader. Wells containing complete media alone were used as blank, and the average values were subtracted from the average values of all clones. The assay was repeated three times and analyzed using two-sided Student’s t-test. Data points represent the mean ± SD and P < 0.05 was considered significant.
Wound-healing assay
The hGH-MCF-7 and MUT-MCF-7 cells (5 × 104 each) were plated in a six-well plate in triplicates and allowed to grow to confluency. Cells were washed and left in serum-free media for 5 h. A scratch was made and media was added that was either only serum. Images were captured at 100× magnification using BestScope microscope at 48 h. The assay was repeated three times.
Results and discussion
Autocrine hGH increases breast cancer cell number, cell proliferation, cell viability, and migration
Autocrine hGH was previously shown to function as a mammary epithelial oncogene.16,17 To determine whether autocrine hGH modulates mammary carcinoma cell behavior, we stably transfected MCF-7 cells with an expression plasmid encoding the hGH gene (hGH-MCF-7). As a control, MCF-7 cells were stably transfected with the (MUT-MCF-7). Autocrine expression of hGH significantly increased cell proliferation, as determined by MTT assay in hGH-MCF-7 cells (Figure 1). A significant increase in MCF-hGH than that of the MUT-MCF-7 was observed indicating an increase in cell proliferation and/or a decrease in cell death. In addition, when cell motility was examined by wound-healing assay, we observed that autocrine hGH-MCF-7 did stimulate cell migration with a more rapid closing of the wound than observed with MCF-MUT cells (Figure 2). This result strengthen the fact that hGH-MCF-7 facilitates migration of breast cancer cells.
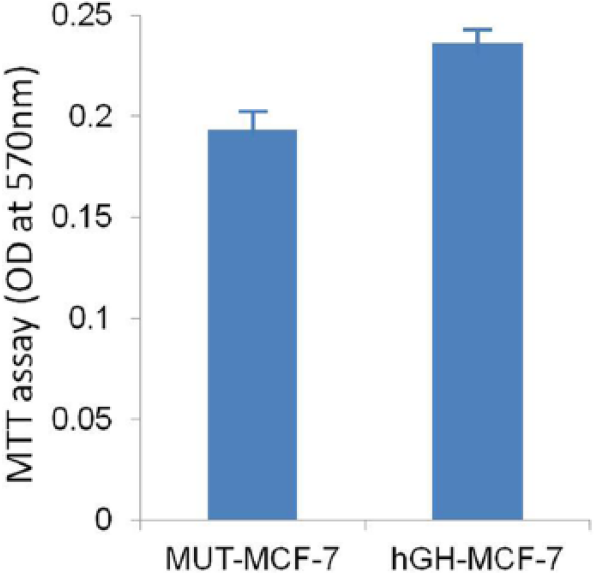
Autocrine hGH increases breast cancer cell proliferation and survival: cell growth/viability test (MTT assay) showed that hGH-MCF-7 cells have higher viability than MUT-MCF-7 cells after 48 h of transient transfection. Each column represents the mean (±SD) of OD at 570 nm.
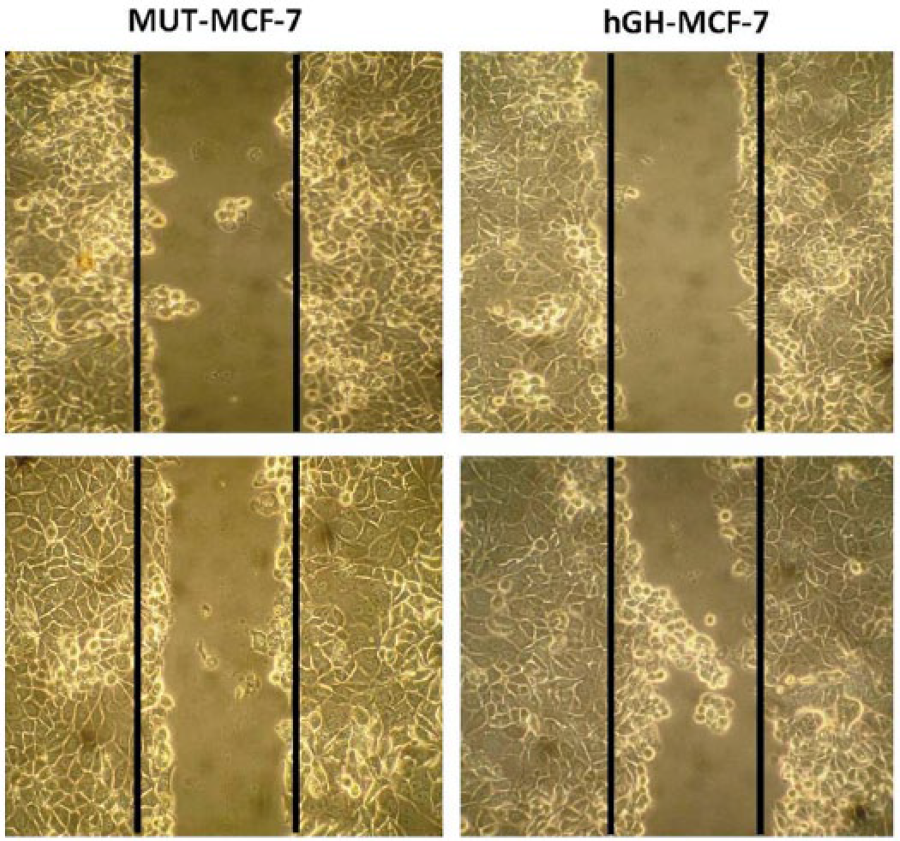
Increased cell migration in hGH-MCF-7 cells over MUT-MCF-7 cells: autocrine production of hGH in human mammary carcinoma cells results in increased cell motility and acquisition of invasiveness. After transient transfection with hGH and MUT hGH, scratch wound-healing assay was performed. The hGH-MCF-7 cells (right panel) show increased cell migration when compared to MUT-MCF-7 cells (left panel). The wounded areas were examined under the 100× magnification.
GH increases NF-κB activity in breast cancer
We investigated potential effects of autocrine hGH on NF-κB activity in MCF-7 cells by co-transfection of hGH-MCF-7 and MUT-MCF-7 cells with luciferase reporter plasmids (luc2P/NF-κB-RE/Hygro). NF-κB activity was increased in hGH-MCF-7 cells but not in MUT-MCF-7 cells (Figure 3).

GH increases NF-kB activity in MCF-7 cells: the hGH-MCF-7 and MUT-MCF-7 cells (2 × 105 cells/well in six-well plate) were co-transfected with luciferase reporter plasmids (2 μg/well) and an internal control, pGL4.32 (luc2P/NF-κB-RE/Hygro) vector (Promega; 2 μg/well) expressing the Renilla luciferase. The hGH-MCF-7 cells showed increased NF-kB expression compared with MUT-MCF-7 cells. Each column represents the mean (±SD) of luciferase fold induction.
GH–dependent activation of NF-κB resulted in the inhibition of epithelial markers
EMT involves specific morphological and molecular alterations, including the loss of E-cadherin-mediated cell adhesion.18,19 E-cadherin, a hemophilic Ca2+-dependent transmembrane adhesion protein, and P-cadherin are essential for ductal mammary branching. 20 To identify NF-κB targets that cause the induction of EMT, we analyzed the expression of E-cadherin and P-cadherin, known transcriptional regulators involved in EMT. Our results revealed that both E-cadherin and P-cadherin expressions were decreased in human hGH-MCF-7 (Figure 4). In addition, expression of E-cadherin was studied by confocal microscopy. Compared with control cells (MUT-MCF-7), decreased expression of E-cadherin was observed in hGH-MCF-7 cells. Immunofluorescence revealed a significantly less number of internalized E-cadherin particles in hGH-MCF-7 cells than that of MUT-MCF-7 cells (Figure 5).

GH-dependent inhibition of epithelial markers: increased expression of NF-κB is associated with the repression of epithelial cell markers. (a) Representative immunoblots for NF-κB and MMP-9. (b) Representative immunoblots for E-cadherin and P-cadherin.

Expression and immunolocalization analysis of E-cadherin in hGH-MCF-7 and MUT-MCF-7 cells: (a–c) and (d–f) confocal laser scanning micrographs of hGH-MCF-7 and MUT-MCF-7 cells, respectively. After transfection (48 h), cells were stained by FITC-conjugated anti-E-cadherin antibody (green) and counterstained by DAPI (blue; scale bar = 10 μm).
Effect of autocrine hGH on apoptosis of MCF-7 cells
We then investigated whether the hGH-induced growth of MCF-7 cells was due to a decrease in apoptosis. Our results showed apoptosis in 86.79% of MUT-MCF-7 cells, while only 56.70% of hGH-MCF-7 cells stained positively for Annexin V. A slight increase in the Annexin V+ PI+ population was also observed in MUT-MCF-7 cells, which indicate late apoptotic or dead cells. There was an increase in viable and non-apoptotic cell populations (Annexin V-PI–) in hGH-MCF-7 cells to MUT-MCF-7 cells (6.80% to 35.60% Annexin V- PI–, respectively; Figure 6(a)). The apoptosis-impede effect of hGH on MCF-7 cells was determined by DAPI staining. In contrast to hGH-MCF-7 cells, which had very little condensed or fragmented chromatin, the majority of MUT-MCF-7-transfected cells displayed apoptotic features, including condensed nuclei (Figure 6(b)).
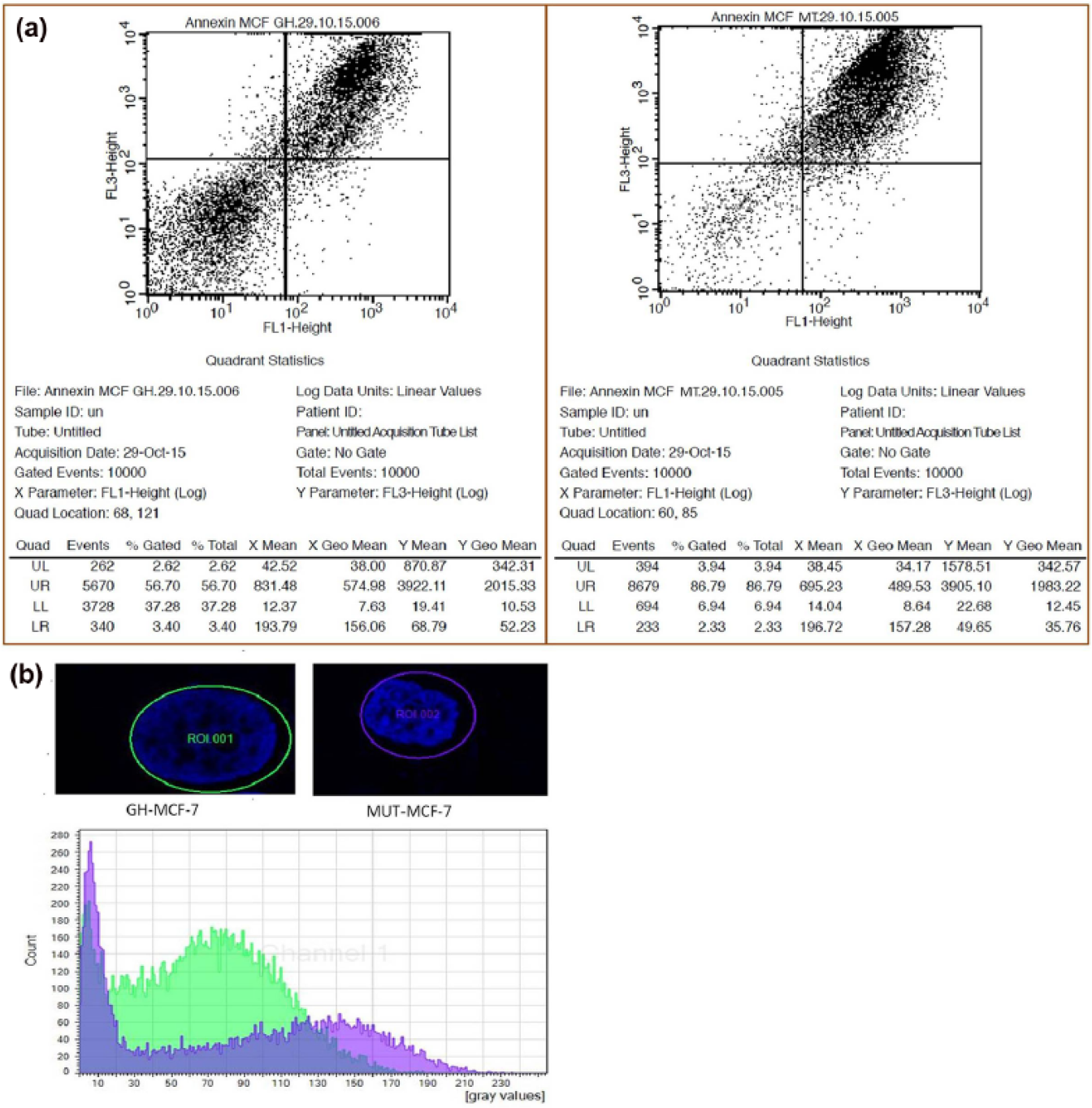
Flow cytometric analysis of hGH-MCF-7 and MUT-MCF-7 breast cancer cells: (a) dot plots show that 86.79% of MUT-MCF-7 cells displayed apoptosis, whereas in the hGH-MCF-7, 56.70% of cells stained positive for Annexin V aggregates. The percentage of events in each quadrant was shown. (b) Cross-sectional analysis along the flattened MCF cell’s nucleus. DAPI staining shows a width of cell fluorescent intensity histogram visualized using confocal microscopy. Data are presented as merge of green and violet channel (green and violet indicates GH-MCF-7 and MUT-MCF-7 cells, respectively).
One of the most fundamental biological processes in tumor metastasis is the process of EMT. Transcription factors play an important role in EMT. Most of the EMT-modulating transcription factors, namely, Twist, Snail, Slug, Zeb1, and Zeb2 are regulated by NF-κB. 21 Estrogen plays a major role in the initiation and progression of breast cancer.22,23 As evident from studies using estrogen receptor (ER)-positive (MDA-MB-231) and ER-negative (MCF-7) breast cancer cell lines, NF-κB is regulated independent of ER expression. The novel finding of this study using reporter assay and western blot analysis is that hGH regulates NF-κB expression in breast cancer. To gain further understandings on the role of NF-κB in EMT, we performed extensive evaluation of NF-κB activation through hGH in human breast cancer cell line in order to clarify the extent and clinical importance of NF-κB activation in hormone-dependent breast cancer. E-cadherin is a transmembrane glycoprotein involved in calcium-dependent intercellular adhesion and is specifically associated with epithelial cell-to-cell adhesion. MMPs’ procession of E-cadherin contributes to the initiation of EMT. 24 However, the expression of MMP-9 is low in patients with strongly positive estrogen and progesterone receptors, 25 which are associated with poor progression of breast cancer. 26 Based on our findings, we proposed that autocrine hGH promotes NF-κB, which in turn increases the production of MMP-9, whereas decreases the production of E-cadherin either by direct mechanism or by enhanced expression of MMP-9. We have demonstrated for the first time that the autocrine GH-dependent induction of NF-κB in MCF-7 cells results in augmented expression of matrix metalloproteinase-9 (MMP-9) activity and decreased expression of E-cadherins, which leads to EMT. In summary, our study revealed influence of hGH on NF-κB activity and EMT in mammary carcinoma. Our findings will help to understand molecular role of GH-NF-κB axis in mammary carcinogenesis which may facilitate the discovery of suitable pathway inhibitors for disease treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Basic Science Research Programme (UGC New Delhi), Osmania University, Hyderabad-500007, India.
