Abstract
Gallbladder carcinoma is the most common and aggressive malignancy of the biliary tree and highly expresses CD147, which is closely related to disease prognosis in a variety of human cancers. Doxycycline exhibited anti-tumor properties in many cancer cells. CD147 antagonist peptide-9 is a polypeptide and can specifically bind to CD147. The effect of these two drugs on gallbladder cancer cells has not been studied. The aim of this study is to investigate the effect of doxycycline and antagonist peptide-9 on gallbladder carcinoma cells and the possible mechanism of inhibition on cancer cell of doxycycline. To investigate the effects of doxycycline and antagonist peptide-9 on gallbladder carcinoma cells (GBC-SD and SGC-996), cell proliferation, CD147 expression, and early-stage apoptosis rate were measured after treated with doxycycline. Matrix metalloproteinase-2 and matrix metalloproteinase-9 activities were measured after treated with different concentrations of doxycycline, antagonist peptide-9, and their combination. The results demonstrated that doxycycline inhibited cell proliferation, reduced CD147 expression level, and induced an early-stage apoptosis response in GBC-SD and SGC-996 cells. The matrix metalloproteinase-2 and matrix metalloproteinase-9 activities were inhibited by antagonist peptide-9 and doxycycline, and the inhibitory effects were enhanced by combined drugs in gallbladder carcinoma cell lines. Taken together, doxycycline showed inhibitory effects on gallbladder carcinoma cell lines and reduced the expression of CD147, and this may be the mechanism by which doxycycline inhibits cancer cells. This study provides new information and tries to implement the design of adjuvant therapy method for gallbladder carcinoma.
Introduction
Gallbladder carcinoma (GBC) is the most common malignancy of the biliary tract and the fifth most common neoplasm of the digestive tract. 1 GBC prevalence was extraordinarily high in Chile, Bolivia, and other South American countries. 2 GBC is characterized by rapid progression, early metastasis, low cure rate, and poor prognosis. The overall 5-year survival rate is less than 5%. 3 Surgical resection remains the mainstay of therapy and is the only way to achieve long-term survival for GBC patients. Because of the lack of early signs, symptoms, and sensitive screening tests, most patients were diagnosed at the advanced stage. A retrospective study showed that stage tumor–node–metastasis (TNM) IV accounted for 55.1% in 2379 GBC patients in the northwest of China; 4 however, long-term survival is an exception in such advanced cases. The majority of GBC patients lost the chance of radical resection at the time of diagnosis. 5 Unfortunately, systemic chemotherapy, external beam radiotherapy traditionally, and other adjuvant therapies had no obvious effect on survival of either unresectable or resectable GBC patients.6,7 Therefore, new adjuvant therapies represent the new options in treating these GBC patients with advanced stage. 8 CD147 found on the surface of the cancer cells is a highly glycosylated transmembrane protein of the immunoglobulin super family and functions as an upstream modulation of matrix metalloproteinases (MMPs). CD147 can enhance the activity of MMPs, which can digest the basement membrane and components of the extracellular matrix, from where cancer cells pass through and transfer. Recent studies have shown that CD147 is detected mainly in GBC tissues rather than in chronic cholecystitis tissues, and the expression rate is significantly associated with histology types, distant metastasis, and Nevin stages of GBC,9,10 and it therefore may be a potential drug target for GBC.
Doxycycline (DOX), an analogue of tetracycline derived from oxytetracycline or methacycline, has been widely used in the treatment of several infectious diseases with tolerable side effects. With the development of research, DOX showed the ability that affects many cellular functions, such as inhibition of the activity of several MMPs, cytotoxic activity against tumor origins, anti-angiogenesis, and apoptotic effects.11–13 CD147 antagonist peptide-9 (AP-9) is screened from the 12-mer phage random peptide library and its amino acid sequence is Tyr Lys Leu Pro Gly His His His His Tyr Arg Pro. It has been shown to have the ability to inhibit CD147, and therefore, it reduces MMP activity.14,15
In order to investigate the effect of these two drugs on GBC cell, two GBC cell lines (GBC-SD, SGC-996) were purchased. Then, we detected the cell proliferation, apoptosis, and CD147 expression after treated with DOX and the MMP-2 and MMP-9 activity after treated with DOX and AP-9.
Materials and methods
Cell culture and drug preparation
Human GBC cell line GBC-SD was purchased from the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China) and SGC-996 was purchased from Genomeditech (Shanghai, China). Either cell line was conventionally cultured in RPMI 1640 medium (Gibco, Grand Island, NY, USA) with 10% fetal bovine serum (Gibco) and antibiotics (100 units/mL penicillin and 100 µg/mL streptomycin (Gibco)) and maintained at 37°C in humidified atmosphere containing 5% CO2. The medium was replaced every 2–3 days. AP-9 and DOX hydrochloride were both purchased from Sangon Biotech (Shanghai, China) and dissolved in culture medium.
Cell proliferation assay
Cell proliferation assay was determined by the Cell Counting Kit-8 (CCK-8; Dojindo Laboratories, kumamoto, Japan). The cells were seeded at the rate of 5 × 103 cells per well with 100 µL of media into 96-well plates. The cells were treated with DOX (5, 10, 20, and 40 µg/mL) for 48 h. After that, CCK-8 (5 µL) for quantitation of viable cell number in proliferation and cytotoxicity assays was added to each well and incubated for 70 min. The absorbance was determined at 450 nm using microplate reader. Experiments were performed in triplets and repeated at least three times.
Protein preparation and western blot assay
Cells were seeded at the rate of 5 × 105 cells per well with 2 mL of media into six-well plates, treated with DOX (10 and 20 µg/mL) for 24 h, and lysed with radioimmunoprecipitation assay (RIPA) buffer (Sangon Biotech) for 30 min on ice. An equal amount (20 µg) of cellular proteins was separated by SDS-PAGE and transferred onto PVDF membranes. The membranes were blocked with 5% non-fat dry milk in TBST buffer (10 mM Tris-HCl, pH7.5, 150 mM NaCl and 1% Tween-20) for 2 h at room temperature with constant shaking and then incubated overnight at 4°C with the following primary antibodies: rabbit anti-human CD147 antibody (1:1000; Abcam, Cambridge, UK) and rabbit anti-human GAPDH antibody (1:5000; Abcam). Membranes were washed with TBST three times for 30 min and incubated with horseradish peroxidase–conjugated secondary antibody (donkey anti rabbit, 1:5000; Abcam) for 2 h at room temperature with constant shaking. Finally, membranes were exposed to film after enhanced chemiluminescence with ECL kit (Solarbio, Beijing, China).
Gelatin zymography
After the treatment with AP-9 (200 µg/mL), DOX (20 µg/mL), and combinations of these inhibitors (AP-9 200 µg/mL and DOX 20 µg/mL) for 24 h, media was changed to RPMI 1640 without FBS and incubated for 24 h. Then, the media was collected and subjected to SDS-PAGE containing 1.5 mg/mL gelatin (Sangon Biotech). After electrophoresis, gels were washed with 2.5% Triton X-100 three times for 45 min with shaking to remove SDS and incubated with a buffer (50 mM Tris, pH 7.6, 150 mM NaCl, 5 mM CaCl2, 1 mM ZnCl2, and 0.02% Brij-35) for 48 h at 37°C. The gels were stained with Coomassie Brilliant Blue R-250 for 3 h and destained with water containing acetic and methanol until the target zones associated with MMP-9 activity were clear.
Early-stage apoptosis analysis
Early-stage apoptosis analysis was determined by Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) staining using the Annexin V-FITC Apoptosis Detection Kit (BD Biosciences, Franklin Lakes, NJ, USA). Cells were seeded at the rate of 5 × 105 cells per well with 2 mL of media into six-well plates, treated with DOX (20 µg/mL) for 12 h, and then washed with ice-cold PBS for three times. According to the protocol, the cells were incubated with 10 µL Annexin V and 200 µL binding buffer for 15 min at room temperature in the dark and then incubated with 5 µL PI. Immediately, samples were quantified by flow cytometry (BD Biosciences).
Statistical analysis
Data were expressed as mean ± standard deviation (SD), and comparisons of means of two groups were conducted using student’s test (version 6.0; GraphPad Prism, GraphPad Software, La Jolla, CA, USA). The p value less than 0.5 was considered to be statistically significant.
Results
Effects of DOX on the proliferation of GBC cell lines
After the treatments with DOX (0, 5, 10, 20, and 40 µg/mL) for 48 h, significant effects were observed in both GBC-SD and SGC-996 cells (Figure 1). GBC-SD showed rows and polygons observed under a microscope. After treated with 20 µg/mL DOX for 48 h, the number of GBC-SD was significantly lower than that in the control group, and part of the cell membranes was incomplete (Figure 1(a)). In GBC-SD cell group, DOX inhibited the cell proliferation compared with control groups (p < 0.01), and the inhibitory effect was more obvious with the increasing concentration, and similar phenomena can be carried over in SGC-996 group.
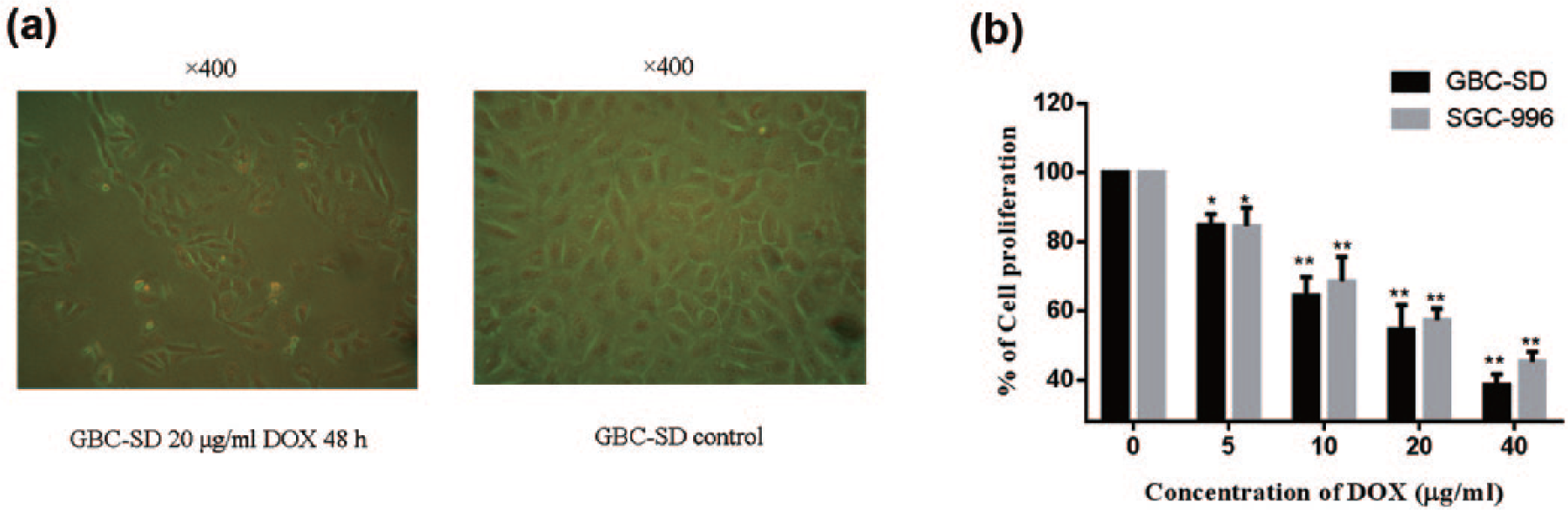
To investigate whether DOX has the ability to inhibit the proliferation of GBC, cell proliferation was detected after treated with DOX (0, 5, 10, 20, and 40 µg/mL) for 48 h. (a) GBC-SD showed rows and polygons observed under a microscope. After treated with 20 µg/mL DOX for 48 h, the number of GBC-SD was significantly lower than that in the control group, and part of the cell membranes was incomplete. (b) Compared with the control groups, cell proliferation rates were reduced in both GBC-SD and SGC-996 (n = 6). With the increase in the concentration of drugs, the inhibition was more obvious. When the concentration was 40 µg/mL, the inhibition effects of DOX on GBC-SD and SGC-996 were over 50% (*p < 0.05 compared with control groups; **p < 0.01 compared with control groups).
Effects of DOX on expression of CD147 in GBC cells
To investigate the possible mechanism of action of DOX, we treated the GBC cells with DOX and then detected the CD147 expression level by western blot (Figure 2). The relative expression levels were normalized to GAPDH (Figure 2(a)). After treatment with DOX, the CD147 levels were significantly decreased compared with control groups in both GBC-SD and SGC-996 cells (p < 0.01), and protein expression was decreased with the increasing concentration.

Expression of CD147 after treatment with DOX (0, 10, and 20 µg/mL) for 24 h. (a) Western blotting was done on cell lysates from GBC-SD and SGC-996 cells treated with DOX (n = 3). The relative expression level was normalized to GAPDH. (b) After treatment with DOX, the CD147 levels were significantly decreased compared with control groups in both GBC-SD and SGC-996 cells, and protein expression was decreased with increasing concentration (**p < 0.01 compared with control group).
Effects of AP-9, DOX, and combinations of these two inhibitors on the activity of MMP-9 in GBC cells
Compared with the control group, MMP-2 and MMP-9 activity was inhibited in AP-9- and DOX-treated groups (Figure 3). Combination therapy showed an even stronger inhibitory effect as compared with AP-9 or DOX alone (Figure 3).
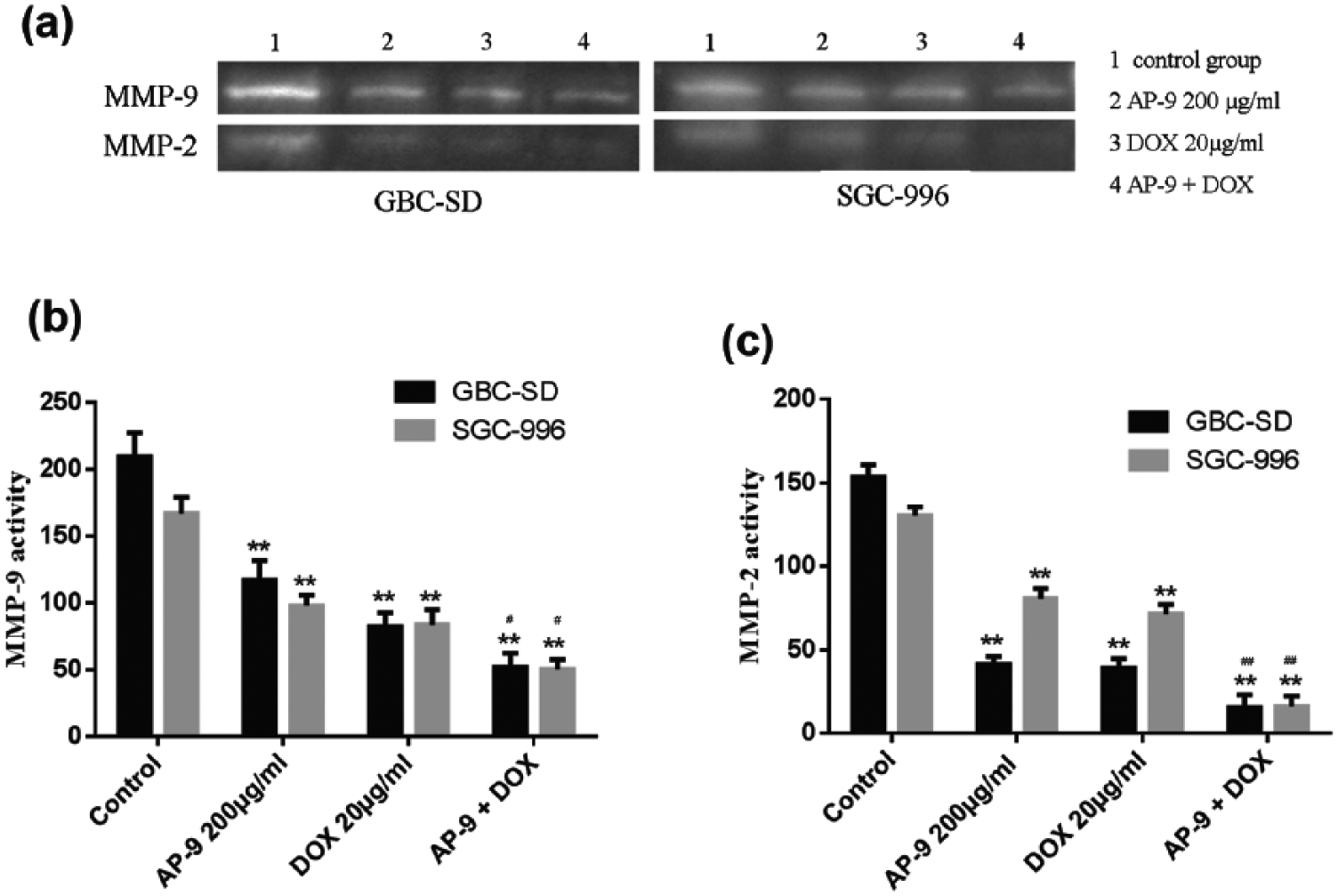
Activity of MMP-2 and MMP-9 after treatment with AP-9 and DOX. (a) The activity of MMP-2 and MMP-9 was detected by gelatin zymography (n = 3). (b) After treatment with 200 µg/mL AP-9 and 20 µg/mL DOX and their combinations (200 µg/mL AP-9 and 20 µg/mL DOX), the activity of MMP-9 was significantly inhibited in both GBC-SD and SGC-996 compared with control groups (p < 0.01). The MMP-9 activity of the combined drug groups was lower than that of the single-drug groups in both cell lines (p < 0.05). (c) The activity of MMP-2 was also significantly inhibited in both GBC-SD and SGC-996 compared with control groups (p < 0.01). The MMP-2 activity of the combined drug groups was lower than that of the single-drug groups in both cell lines (p < 0.01). **p < 0.01 compared with control group; #p < 0.05 compared with AP-9 and DOX group; and ##p < 0.01 compared with AP-9 and DOX group.
Effects of DOX on early-stage apoptosis of GBC cells
To examine the effects of DOX on the early-stage apoptosis of GBC cells, we treated the cells with DOX (10 and 20 µg/mL) for 12 h and then detected by Annexin V/PI staining. Cells treated with DOX showed increasing early-stage apoptotic rate (Figure 4).

Early-stage apoptosis was detected after treatment with several concentrations of DOX (10 and 20 µg/mL) for 12 h by Annexin V/PI staining. After treatment with DOX, the early-stage apoptosis was significantly decreased compared with control groups in both GBC-SD and SGC-996 (**p < 0.01 compared with control group).
Discussion
DOX is a well-known and widely used antibiotic. The bacteriostatic mechanism of DOX inhibits protein synthesis. 16 According to this mechanism, DOX is thought to inhibit protein synthesis in eukaryotic cells and thus be applied in the treatment of malignancies. Recently, DOX has been recognized to have the anti-tumor properties in various cell types of cancers, that is, breast cancer, colon cancer, and melanoma.13,17,18 DOX can inhibit the activity of MMPs,19,20 cytotoxic activity against tumor cells, 17 and induction of apoptosis in various types of cancer cells18,19 and has the anti-angiogenesis ability. 16 This study showed that the rate of cell proliferation and the activity of MMP-2 and MMP-9 were significantly inhibited. The rate of apoptosis was increased after DOX treatment. Recent studies showed the involvement of a ROS-ASK1-JNK pathway in DOX-induced melanoma cell apoptosis. 7 In the breast cancer cells and hepatocellular carcinoma cells, CD147 gene was significantly decreased after DOX treatment.20,21 In this study, CD147 was shown in a significant decreasing trend after treated with DOX, and this supports the conclusion obtained from previous studies.20,21 Previously, a wide array of previous studies have demonstrated that CD147 serves as a pluripotent molecule and is involved in multiple stages of tumor progression with the abilities to promote tumor cell proliferation, invasiveness, metastasis, and VEGF production.21,22 In this study, we are able to show that the DOX induces the significant inhibitory effect on the proliferation and apoptosis of cells, which maybe because DOX reduced the CD147 expression level and thus inhibited GBC cells.
CD147 is expressed in normal and diseased tissues of humans and is particularly enriched on the surface of malignant tumor cells, such as glioma, laryngeal squamous cell, ovarian, renal cell, and skin carcinoma.23–25 CD147 is the core molecule in the process of growth, invasion, proliferation, and metastasis of malignant tumors. 26 Recent study showed that melanoma cell growth was inhibited by CD147 silencing. 27 AP-9 is a specific CD147-binding peptide, which may avoid undesired immune reactivity compared with common antibody-mediated targeting of proteins for its smaller molecules that only include 12 amino acids. In this study, the MMP-2 and MMP-9 activity was inhibited by AP-9 in both GBC-SD and SGC-996 cells. Previous studies have showed that CD147 might facilitate CypA-induced MMP-9 production by activating ERK1/2 and JNK MAPK pathway 14 and AP-9 can block the expression and activation of MMPs and enhanced the invasive ability of THP-1 cells through binding CD147, 15 which is consistent with our present results.
Taken together, in this study, we were able to demonstrate the inhibitory effect of DOX on GBC cells, which could be via the inhibition on cell proliferation, CD147 expression, and the promotion on apoptosis induction, and we also demonstrate that MMP-2 and MMP-9 activity was inhibited after treated with AP-9 and DOX. Our data might provide the important information for the design of new therapeutics for GBC treatment.
Footnotes
Acknowledgements
The authors thank Dr Jun Ma for providing help for the whole experiment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Henan Province Science and Technology Development Plan in 2015 (No. 152102310256)
