Abstract
Osteosarcoma is the most frequent primary bone tumor that affects adolescents and children. However, diagnostic and prognostic biomarkers for osteosarcoma remain lacking. (Tim-3) T-cell immunoglobulin domain and mucin domain-3, which negatively regulates T cell helper (Th1) cells and affects cytokine expression, has attracted increasing attention due to its critical role in regulating both adaptive and innate immune cells. In this study, we evaluated serum soluble Tim-3 level in osteosarcoma patients to explore its diagnostic and prognostic value for this particular malignancy. Serum soluble Tim-3 level was measured with enzyme-linked immunosorbent assay in 120 osteosarcoma patients, 120 benign bone tumors patients and 120 healthy controls, followed by analysis of the correlation with clinic pathological characteristics. Receiver operating curves, Kaplan-Meier curves, and log-rank analyses as well as Cox proportional hazard models were used to evaluate the diagnostic and prognostic significance. Serum solubleTim-3 level was remarkably elevated in osteosarcoma patients. Osteosarcoma patients with larger tumor size, late stages and distant metastases were accompanied with higher levels of Tim-3. ROC/AUC analysis indicated thatTim-3 served as a reliable marker to distinguish healthy participants from Tim-3 patients. Osteosarcoma patients with higher Tim-3 had relatively lower survival. Multivariate analyses for overall survival revealed that high serum soluble Tim-3 level was an independent prognostic factor for osteosarcoma. Furthermore, Tim-3 levels of CD8+ and CD4+ T cells were elevated in peripheral circulation of osteosarcoma patients. Therefore, It was indicated in our research that elevated serum soluble Tim-3 level might be a novel potential diagnostic and prognostic biomarker for osteosarcoma patients.
Introduction
Osteosarcoma (OS) represents the eighth most commonly occurring cancer with an incidence rate of 4.4 in a million. OS is most prevalent in adolescents and children, occupying 9% of mortality linked with cancer and 5% of malignancies of children. 1 OS arises from osteoid tissues and produces immature bone. 2 Normal bone tissues are often destroyed in this malignancy. 3 Despite the fact that innovative therapies are constantly coming into being, such as adjuvant chemotherapy (ACT), amputation and wide resection of tumors, OS-related mortality, and morbidity of OS are still high.4,5 Disease-free survival (DFS) and 5-year overall survival remains 50%–60%, and almost 40% of OS mortality is caused by lung metastases. 6 Consequently, it is extremely critical to promote novel strategies not only to early diagnosis OS but also to predict its prognosis in order to select the most effective treatment. 7 Therefore, it is clinically vital to identify diagnostic and prognostic biomarkers and to optimize treatment strategies.
As a negative regulatory molecule, T cell immunoglobulin and mucin domain-3 (Tim-3) plays a crucial role in the tumor immunological tolerance. Tim-3 belongs to a family of receptors involved in immune-checkpoint functions. 8 Tim-3 expression was initially identified in CD4+ interferon gamma (IFN-γ) producing cells and in cytotoxic CD8+ lymphocytes and is constitutively expressed on T helper cells (Th1)/cytotoxic T cells (Tc1) cells in mice and human. 9 Tim-3 expression was correlated with T cell dysfunction, blockade of interaction between Tim-3 and its ligand restores IFN-γ production in response to hepatitis C virus (HCV)-specific antigens. 10 Tim-3 is also expressed in a sub-population of Treg that has been especially enriched in tumor-infiltrating lymphocytes and shows immunosuppressive capacity with a correlation of poor prognosis in cancer patients. 11 These data indicate a reliable role of Tim-3 as an innovative biomarker for patients with OS.
In this study, we examined soluble Tim-3 level using enzyme-linked immunosorbent assay (ELISA) and detected Tim-3 expression in CD8+ and CD4+ in peripheral circulation by flow cytometry. The correlation between Tim-3 and tumor features was also investigated. Our results suggest that Tim-3 is an innovative non-invasive predictor of prognosis and diagnosis of OS.
Materials and methods
Participants
In total, 120 patients with primary OS in orthopedics department of Rizhao People’s Hospital from April 2011 to November 2014 were enrolled. OS diagnoses were based on clinical and histological examination of resected specimens from primary OS. All patients had no history of other cancers and underwent no other prior treatment including chemotherapy (CT) or radiotherapy (RT) when first diagnosed with primary OS. In total, 120 patients with benign tumors in bones, such as ossifying fibromas, chondromas, osteochondromas, and bone cysts, were enrolled in our study. The benign tumors were diagnosed depending on pathology in the same way. Sex- and age-matched 120 controls, unrelated to OS patients, were recruited and had no orthopedic disease or cancer. This study was approved by the Ethics Committee, and the written informed consent was obtained from all participants.
Data collection and follow-up
Patient demographic, pathological type, tumor location, metastasis, and clinical pathology data were collected from medical records upon diagnosis. OS patients underwent standard therapeutic procedures such as neoadjuvant chemotherapy (NACT) and ACT subsequent to radical surgery. Response to CT was categorized as either good (>90% tumor necrosis) or poor (<90% tumor necrosis) depending on histological pathology of cancer samples. After surgery, all OS patients were recruited for follow-up monitoring. On behalf of progression of the illness, DFS referred to the period of time from surgery to tumor relapse or decease or the aftermost follow-up. The latest follow-up was on December 2016. Median follow-up aiming at overall survival lasted for 37.1 months (range: 7–80 months).
ELISA of serum soluble Tim-3
Venous blood samples were obtained from each participant when first diagnosed with OS. Blood samples underwent coagulation within tubes separating serum. Serum specimens were then centrifuged and stored in tubes at −80°C prior to ELISA of Tim-3. Tim-3 concentration was detected in ELISA kit (Sigma) according to the manufacturer’s instructions. Briefly, anti-human Tim-3 antibody was utilized to pre-coat the 96-well plate. Recombinant human Tim-3 and serum specimens were deliquated using buffer, subsequently mixed into wells, and Incubated at room temperature for 1 h. Tim-3 bound to localized antibodies in the wells which were subsequently washed three times. The anti-Tim-3 antibody was biotinylated and placed into wells. The incubation required room temperature and lasted for 1 h. Unbound antibodies were eliminated by washing. Streptavidin conjugated to horseradish peroxidase (HRP) was added. Subsequent to one more washing, tetramethylbenzidine solution and stop solution were added. Finally, the optical density (OD) at 450 nm was detected with the help of a microplate reader. Human Tim-3 was deliquated into a series of concentration, on which standard curve was based. Consequently, Tim-3 concentration was obtained.
Quantitative real-time polymerase chain reaction
Excised tumor tissues and the relevant non-tumor tissues of bones were obtained and stored at −80°C. Total RNA was extracted from frozen tissues using TRIzol reagent (Invitrogen), following the manufacturer’s instructions. Then, 1 µg of total RNA from each sample was used for complementary DNA (cDNA) synthesis using a Reverse Transcription Kit (TaKaRa). After obtaining cDNA, quantitative real-time polymerase chain reaction (qRT-PCR) was performed, under the following conditions: 95°C for 30 s, 60°C for 15 s, and 72°C for 30 s, for 35 cycles.
Western blotting
Tumor tissues and adjacent tissues were homogenized on ice with a glass homogenizer and lysed in radioimmunoprecipitation assay (RIPA) buffer containing protease inhibitor (Sigma) and phenylmethylsulfonyl fluoride (PMSF). Lysates were sonicated and centrifuged at 12,000 r/min at 4°C for 5 min. Then the samples were precipitated, and the supernatants were collected. Extracted proteins were subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred onto polyvinylidene difluoride (PVDF) membranes (Millipore). The PVDF membrane was blocked with TBST (Tris-buffered saline (TBS) buffer with 0.05% Tween-20) containing 5% skim milk and incubated with human Tim-3 antibody (Abcam) and β-actin antibody (Santa Cruz Biotechnology). Subsequently, samples were incubated with HRP-conjugated secondary antibody (Santa Cruz Biotechnology).
Flow cytometry analysis
Tim-3 was detection based on previously described methods. Peripheral blood sample was incubated for 30 min at 4°C in a dark room with monoclonal antibodies or isotype-matched control. Pe-Cy5-conjugated anti-human CD3, fluorescein isothiocyanate (FITC)-conjugated anti-human CD4, Pe-Cy5-conjugated anti-human CD8, phycoerythrin (PE)-conjugated rat IgG2a isotype control (Sigma), and monoclonal PE-conjugated anti-human Tim-3 (Invitrogen) were used for flow cytometric analysis. Other experimental procedures were carried out based on the instructions, and stained cells were then subjected to flow cytometry analysis.
Statistical analysis
One-factor analysis of variance was adopted to evaluate the distinction of Tim-3 concentration among patients with benign tumor and OS and healthy participants in control group. Distinction of Tim-3 expression between excised cancer tissues and surrounding tissues was evaluated with the help of paired t test. Effectiveness of OS diagnosis was determined by non-parametric receiver operating characteristic (ROC) curve. Kaplan–Meier analysis was used to assess possibilities of survival. Log-rank test was utilized to determine significance of distinction. Multivariate analysis of prognostic factors was performed using a Cox proportional hazards regression analysis. Prism 5.0 was required to perform the whole analyses. p < 0.05 was considered to be statistically significant.
Results
Clinical characteristics and serum soluble Tim-3 level
Clinical features are presented in Table 1. No significant differences among patient characteristics were found. Serum soluble Tim-3 was higher in patients with OS than those with benign tumors or control group (14.4 ± 2.9 vs 10.3 ± 1.7 or 6.3 ± 1.9; Figure 1). Furthermore, serum soluble Tim-3 level of patients with benign tumors was mildly higher than control group.
Characteristics of study population.

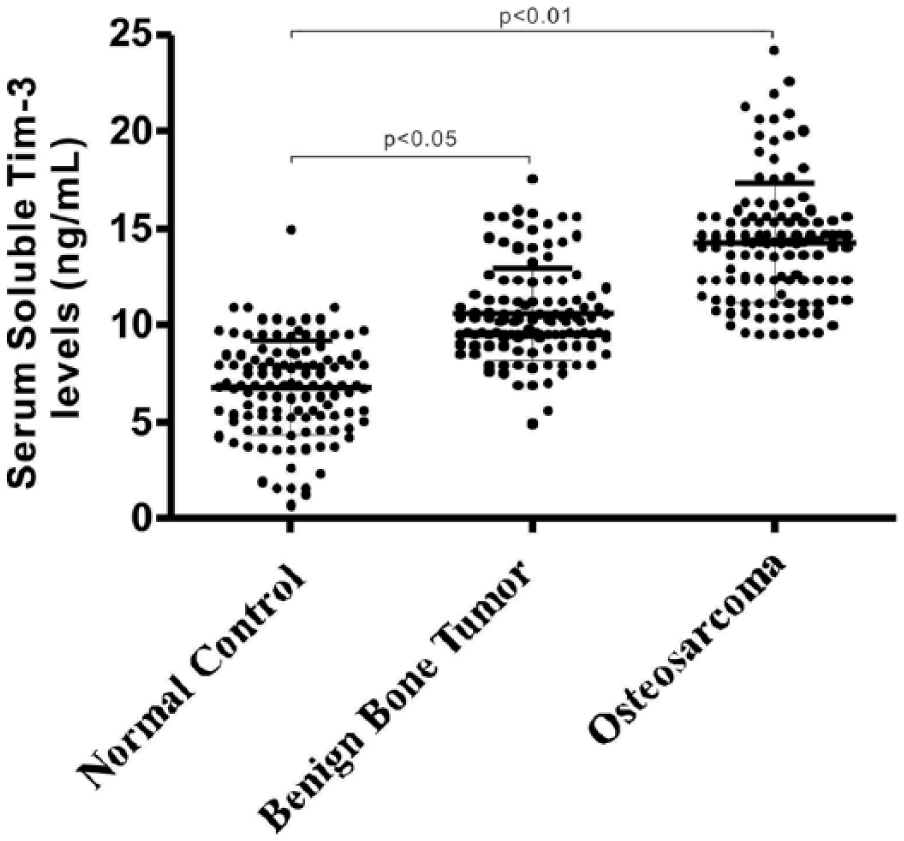
Serum soluble Tim-3 concentration in benign bone tumors, osteosarcoma patients, and the healthy controls.
Correlation between the serum soluble Tim-3 level and clinicopathological features in OS patients
Our results showed that serum soluble Tim-3 level was not correlated with sex, age, or response to CT. OS patients with femur tumors had greater serum soluble Tim-3 level than those with tumors at other sites. However, the distinction was not significant (data not shown). Tumor size analysis results showed that serum soluble Tim-3 level was increased in the tumors larger than 8 cm compared to tumors smaller than 8 cm (13.1 ± 2.1 vs 17.0 ± 2.9; p < 0.05; Figure 2(a)). Serum soluble Tim-3 level was diverse considering various clinical stages of OS. Tim-3 level of stage III was noticeably higher than that of stage IIB or IIA (17.6 ± 3.3 vs 14.3 ± 1.9 or 12.1 ± 1.5; both p < 0.05). Similarly, serum soluble Tim-3 level of stage IIB was remarkably higher than that of IIA (p < 0.05; Figure 2(b)).Serum soluble Tim-3 level was increased in OS patients with a lower grade (15.8 ± 2.8 vs 13.0 ± 2.0; Figure 2(c); p < 0.05) and OS patients with distant metastases (16.9 ± 2.9 vs 12.8 ± 2.1; Figure 2(d); p < 0.05).

Analysis on the correlation between features of osteosarcoma patients and serum soluble Tim-3 levels. Serum soluble Tim-3 levels in osteosarcoma patients which (a) had diverse tumor size, (b) had diverse clinical stage, (c) had diverse tumor grade, and (d) were with or without distant metastases.
ROC analysis of serum soluble Tim-3 level in OS
ROC/area under the curve analysis was applied to determine the specificity and sensitivity of different serum soluble Tim-3 levels. It was presented by ROC curve that serum soluble Tim-3 levels could serve as a reliable marker to distinguish patients confronting OS from healthy participants in control group (Figure 3(a)) and to differentiate patients confronting OS from patients merely suffering from benign tumors (Figure 3(b)). As shown in Figure 3(c), serum soluble Tim-3 levels could differentiate terminal cancer (IIB/III) from early clinical stages (IIA). The above results suggested that serum soluble Tim-3 could serve as a reliable marker for discriminating patients with OS.
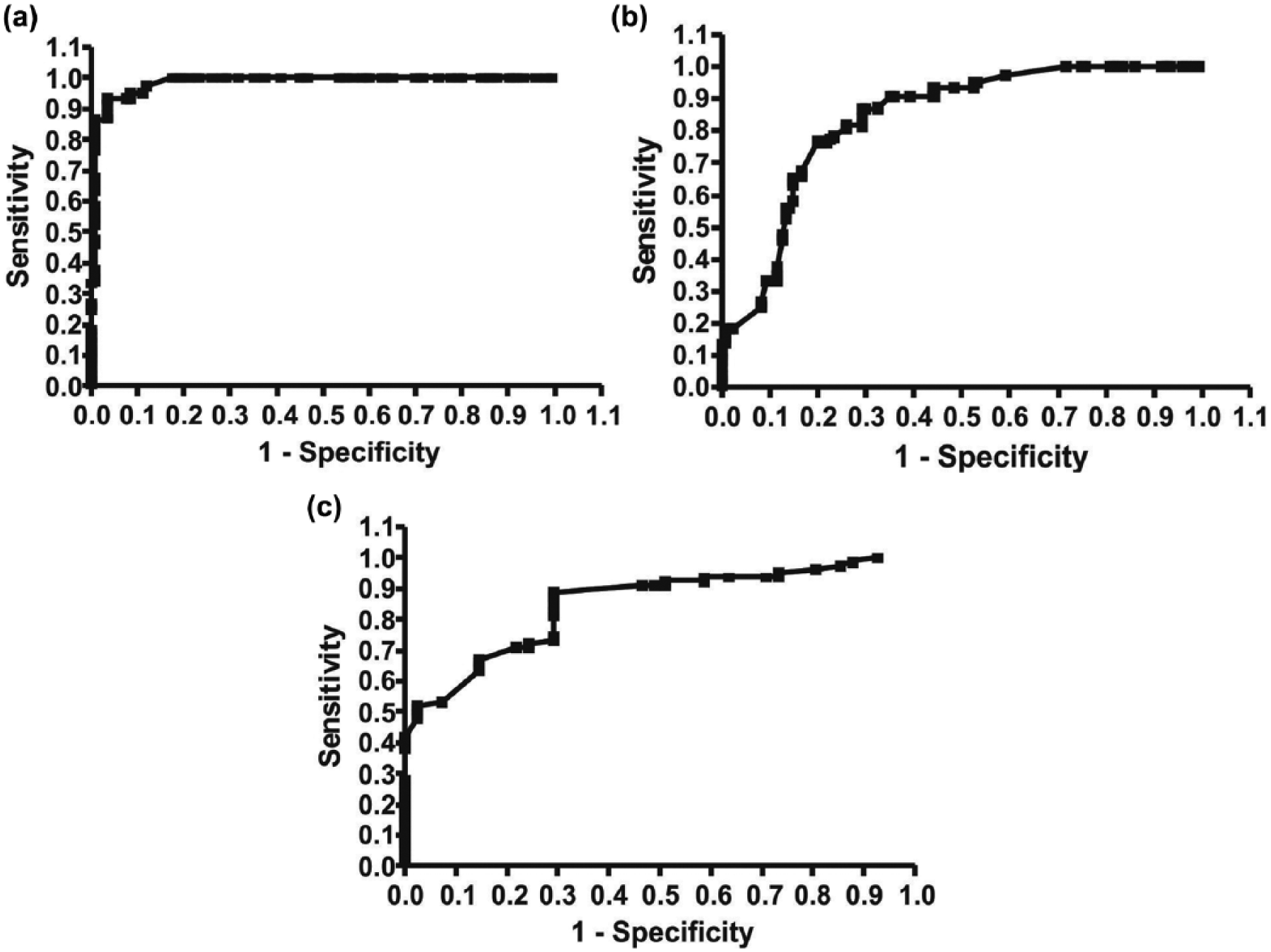
ROC analysis on serum soluble Tim-3 in order to distinguish osteosarcoma from healthy controls or benign bone tumors. ROC for the serum soluble Tim-3 levels which differentiated osteosarcoma from (a) the control and (b) benign bone tumors. (c) Osteosarcoma patients of stage IIB/III were distinguished from those of stage IIA.
Correlation analysis between serum soluble Tim-3 levels and OS prognosis
All the 120 patients with OS in the experimental group received standard treatment and completed follow-up. OS patients were stratified by average serum soluble Tim-3 values. Both the low groups and high groups were investigated. Significant differences in overall survival were detected between the low-expression group and high-expression group of Tim-3 (Figure 4). Patients with higher levels of serum soluble Tim-3 level tended to have shorter overall survival and DFS in comparison with those expressing lower levels of Tim-3. Cox multivariate and univariate regression analysis were used to determine whether Tim-3 could serve as a reliable predictor for OS prognosis. The results demonstrated that elevated Tim-3 concentration alone could predict poor prognosis (Table 2).
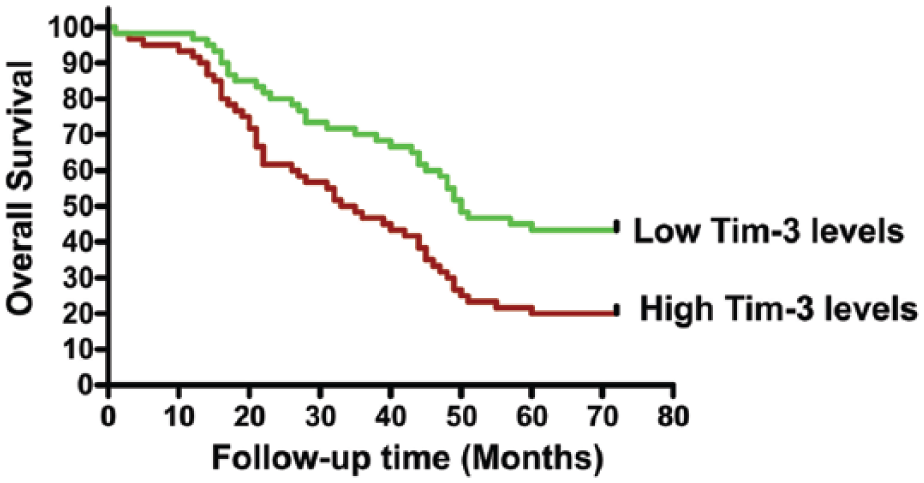
The Kaplan–Meier survival curve. The overall survival of osteosarcoma cases which had high serum soluble Tim-3 levels versus that of those which had low Tim-3 levels.
Univariate and multivariate Cox proportional hazards analysis for overall survival of osteosarcoma patients.
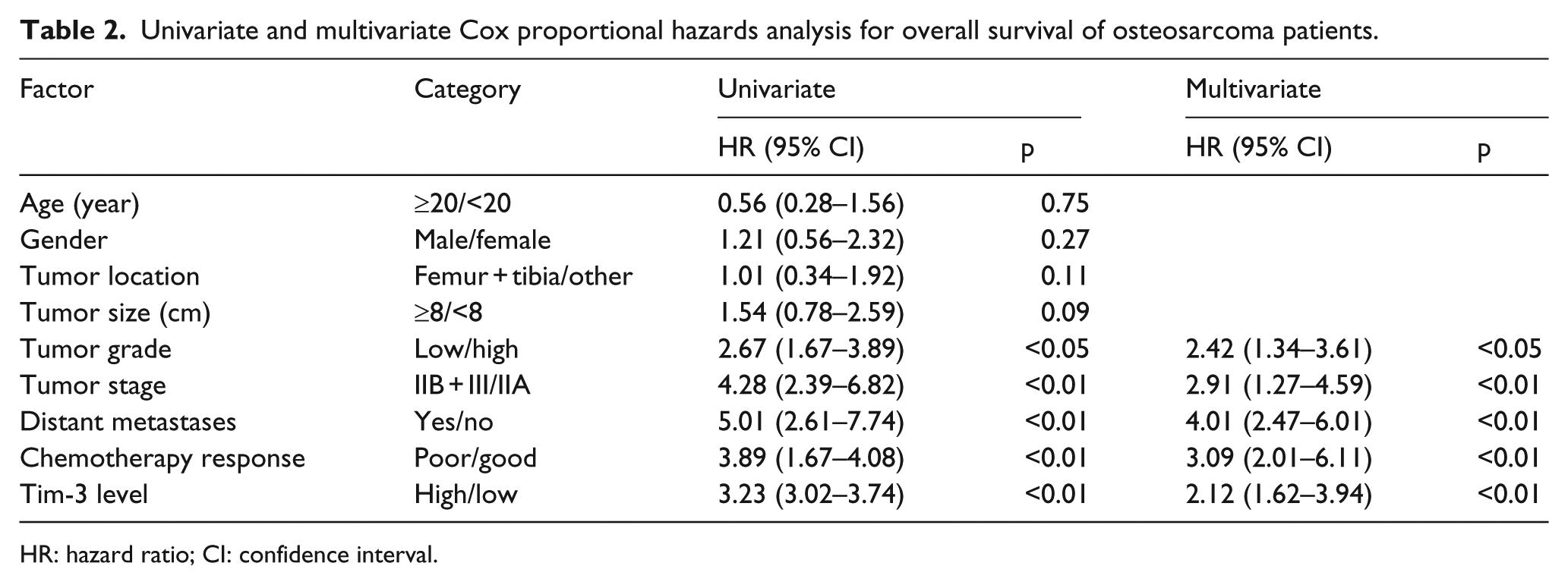
HR: hazard ratio; CI: confidence interval.
The levels of Tim-3 in OS tumor tissues
The levels of Tim-3 in tumor and tumor-adjacent tissues were detected by RT-PCR. As shown in Figure 5(a), messenger RNA (mRNA) concentration of Tim-3 in OS tissues was noticeably elevated in comparison with tumor-adjacent tissues. In compliance with concentration of Tim-3 mRNA, western blotting analysis revealed that Tim-3 protein level was also elevated in the OS tumor tissues (Figure 5(b)). These findings indicated that elevated production of Tim-3 was the potential source of serum Tim-3.

Tim-3 expression levels in osteosarcoma tissues: the expression levels of (a) Tim-3 mRNA and (b) protein in tumor tissues and their adjacent tissues.
The expression level of Tim-3 in CD4+ and CD8+ T cells in peripheral circulation of OS patients
Finally, we examined Tim-3 expression in CD8+ and CD4+ T cells in peripheral circulation from 30 OS patients and 30 healthy controls. Rising ratio of Tim-3 was discovered in CD4+ and CD8+ cells from patients confronting OS compared with participants in control group (6.18 ± 2.88 vs 3.3 ± 1.68, 7.32 ± 2.87 vs 2.94 ± 1.80, respectively; p < 0.05), which is presented in Figures 6(a) and (b), respectively. These findings indicated that Tim-3 might play an essential role in OS pathogenesis by regulating diverse immunocytes. Furthermore, we compared the proportion of Tim-3 in CD4+ and CD8+ cells in peripheral circulation between both genders in control group. No significance in the distinction was found suggesting that sex had nothing to do with Tim-3 expression (data not shown).

Percentage of Tim-3 on CD4+ and CD8+ T cells in (a) osteosarcoma patients and (b) healthy controls. Each dot represented one subject.
Discussion
As the most prevalent primary bone cancer, OS mainly attacks teenagers, young adults, and children, occupying 5% of pediatric cancer and nearly 20% of the whole bone tumors.12,13 Metaphyses of long bones are vulnerable to OS, among which humerus, proximal tibias, and distal femora are the most vulnerable.14,15 In terms of patients who are diagnosed with OS but without metastases, radical surgery combined with RT and CT noticeably promotes 5-year survival. 16 Nevertheless, most OS patients are diagnosed at advanced stages, and long survival after diagnosis of advanced OS is rare. 17 Because of the marked heterogenicity and relatively low incidence rate of OS, mechanisms of OS pathogenesis remain unknown in comparison with other kinds of cancer. 18 Currently, NACT with methotrexate, ifosfamide, cisplatin, and doxorubicin has been extensively used to reduce the recurrence rates and improve survival of the patients with OS.19 –21 Insufficiency of early predictors to detect metastases and predict survival is a challenge worthy of study.
Tim-3 was first found to be expressed specifically on Th1 cells. 22 Expression of Tim-3 has also been discovered on CD8+ T cells, Treg, Th17, natural killer (NK) cells, dendritic cells (DCs), monocytes, lung cancer cells, melanoma, mast cells, and other types of lymphocytes up to now.23,24 Recently, Tim-3 is regarded as a negative regulatory molecule, which plays a crucial role in anti-tumor immunity. 25 Nevertheless, the underlying mechanisms are still undiscovered. Vital to cancer progression, abnormal expression of Tim-3 is reported to be related to various types of cancer. 26 However, the relationship between expression of Tim-3 and OS has not been reported. Thus, we estimated expression level of Tim-3 in OS patients and found that mRNA and protein levels of Tim-3 were remarkably higher in OS tissues than that in tumor-adjacent tissues. We discovered that Tim-3 concentration was remarkably higher in patients with OS, especially OS in lower grades, at more terminal stage, and accompanied with distant metastases. Serum soluble and tissue Tim-3 maybe a diagnostic biomarker to distinguish OS from benign bone tumors and controls and to distinguish advanced from early OS stages. Furthermore, Tim-3 is a more sensitivity biomarker for late-stage than early-stage OS tumors. In addition, elevated serum soluble Tim-3 alone was a reliable predictor of prognosis for patients mentioned above. We are the first to report the utility of Tim-3 as a serum diagnostic biomarker for OS. Consistent with prior research, our findings showed that serum soluble Tim-3 level was correlated with the presence of cancer. In addition, ROC data demonstrated that the sensitivity of Tim-3 in distinguishing OS from healthy controls and benign tumors was low. Sensitivity of other markers was also limited, and the resolution to this problem was challenging. It has been reported that coinstantaneous examination of combination of various biomarkers could promote sensitivity without sacrificing specificity. Recently, the development of protein microarrays and immune bead assays has improved the simultaneous detection of serological biomarkers, and these could overcome low sensitivity issues.
Tim-3 is defined as membrane molecule generated by the sub-category of some types of T cells. 27 It was reported in prior research that Tim-3 could modulate Th1 immune reaction and regulate immune tolerance. 23 Tim-3 is also vital to allergy, immune tolerance, autoimmune illness, and even tumor-resisting immunity. 28 Numerous studies suggested that Tim-3 expression existed in various kinds of cancer tissues, such as glioma, acute myeloid leukemia (AML), prostate cancer, gastric cancer, ovarian cancer, lung cancer, and cervical cancer.23,25,26,29 In our study, flow cytometry was applied to detect expression of Tim-3 in CD8+ and CD4+ cells in OS patients. Serum Tim-3 level was remarkably higher in OS patients than in healthy controls, indicating that Tim-3 was crucial to OS pathogenesis by modulating diverse immunocytes. Our results suggested that Tim-3 level might serve as a reliable predictor of OS pathogenesis and progression through modulating CD4+ cells. It is well known that CD8+ cells are vital to microenvironment of cancer. 27 Our study indicated that the Tim-3 level on CD8+ cells was relevant to OS progression. The possible mechanism might be the depletion of T cells caused by Tim-3 that brought about cancer relapse. A distinct relationship between survival and expression of Tim-3 was presented in our study. It was demonstrated in multivariate analysis that vulnerability to death of patients expressing excessive Tim-3 was noticeably higher than vulnerability of patients expressing limited Tim-3. Various factors can have an effect on prognosis of OS, such as size and location of tumor, cancer stage, CT response, and patient characteristics.21,30 According to our findings, the relationship between serum concentration of Tim-3 and prognosis of OS was independent and significant. In addition, in terms of OS patients, distant metastases, tumor grade, CT response, and clinical stage were also significantly related to poor prognosis of OS.
Detection of serum soluble Tim-3 was performed by blinded pathologists. ELISA was used in our study to quantitatively estimate Tim-3 concentration. Those merits mentioned above guaranteed the credibility of our results and attraction of our analysis clinically. However, this study also has several limitations. The design of our research was cross-sectional, leaving alone any possible relationship between OS vulnerability and serum Tim-3 concentration. Also the source of high serum soluble Tim-3 was not clear, higher soluble Tim-3 was noted in OS tumor tissues, but whether higher serum soluble Tim-3 was totally attributable to soluble Tim-3 overexpression in that tumor tissue requires further exploration.
Conclusion
Our study demonstrated that serum soluble Tim-3 was a reliable predictor of OS diagnosis. Furthermore, it was testified that Tim-3 level was more frequently elevated in OS patients with adverse clinical stage and presence of distant metastasis than those OS patients without adverse clinical stage and presence of distant metastasis. It was revealed in multivariate survival analysis that serum soluble Tim-3 was an independent prognostic factor for both progression survival free and overall survival in OS patients. Our study convectively demonstrated the possibility of serum Tim-3 and expression of Tim-3 on CD8+ and CD4+ cells in peripheral circulation to predict prognosis and diagnosis of OS without invasion. In the future, we will further study the hypothesis extensively and develop the current parameter as a biomarker to diagnose the condition at an early point of time. We will compare the sensitivity and specificity of Tim-3 with other well-known OS biomarkers, such as Hedgehog, CADM1, CD44, and CXC chemokines. Also, we will use Tim-3 in combination with other OS biomarkers to improve the sensitivity of OS diagnosis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
