Abstract
Osteosarcomas are common bone malignancies in children and adolescents. LDOC1 (leucine zipper, down-regulated in cancer 1), a tumor suppressor, is down-regulated in many cancers. In this study, we investigated the role of LDOC1 in tumor metastasis and its prognostic significance in osteosarcomas. We established osteosarcoma cells stably expressing LDOC1, driven by an HIV-based lentiviral system. We investigated the impact of LDOC1 on migration and invasion abilities in these cells using a transwell assay. LDOC1-associated changes in expression of metastasis-promoting genes were analyzed with a quantitative real-time polymerase chain reaction primer array. A xenograft tumor model (n = 7 mice/group) was used to assess the effect of LDOC1 on osteosarcoma metastasis in vivo. The overall survival and disease-free survival of osteosarcoma patients (n = 74) were analyzed retrospectively based on immunohistochemical analysis of LDOC1 levels in tumors and Kaplan–Meier analysis. LDOC1-expressing osteosarcoma cells displayed decreased migration and invasion in vitro. The quantitative real-time polymerase chain reaction primer array data showed that increased LDOC1 expression up-regulated many metastasis-suppressor genes. In the xenograft model, micro-computed tomography imaging data indicated that increased LDOC1 expression is associated with weaker lung metastasis ability. The Wnt5a signaling pathway promotes osteosarcoma metastasis; LDOC1 expression decreased Wnt5a levels in osteosarcoma cells. Kaplan–Meier analysis showed that higher LDOC1 expression was associated with improved osteosarcoma patient overall survival and disease free survival (p = 0.022). Our data show that LDOC1 is a tumor suppressor in osteosarcoma, and that it regulates metastasis of osteosarcoma cells. Furthermore, LDOC1 might be a valuable prognostic marker in osteosarcomas.
Introduction
Osteosarcoma is the most common bone malignancy in children and adolescents, but the etiology of the disease remains unknown.1–3 The estimated annual incidence of osteosarcoma worldwide is four cases per million people, and approximately 60% of cases occur in patients below 20 years of age. 2 Recent advances in osteosarcoma treatment combine chemotherapy and surgical resection. However, the 5-year rate of disease-free survival (DFS) is only 60%, and regardless of treatment, as many as 30%–40% of patients experience relapse within 3 years after treatment. 4 Moreover, many of the relapsed patients have lung metastases and the survival rate is less than 20%. 4 Given these sobering statistics, there is a need to identify novel treatment targets and prognostic biomarkers to improve the survival rate for osteosarcoma patients.
Osteosarcoma is associated with numerous aberrant oncogenic signaling pathways, including the wingless-type mouse mammary tumor virus integration site family (Wnt) signaling pathway. Activation of the Wnt signaling pathway is associated with oncogenic outcomes. 2 In fact, Wnt antagonists can serve as tumor suppressors and increase the efficacy of chemotherapy drugs. 2 The Wnt5a signaling pathway is involved in metastasis regulation in many tumor types, including osteosarcoma.5–10
The LDOC1 (leucine zipper, down-regulated in cancer 1) protein has been shown to localize to the nucleus, and its expression is down-regulated in pancreatic and gastric cancer cells, compared to the corresponding normal human tissue.11,12 These observations led to the hypothesis that LDOC1 functions as a tumor suppressor. Consistent with this hypothesis, LDOC1 has been shown to induce apoptosis, a form of programmed cell death, in several tumor cell lines.13,14 Reduced LDOC1 expression is also reported to be associated with increased sensitivity to radiotherapy in esophageal cancer cell lines. 15 Therefore, LDOC1 is a potential biomarker and putative therapeutic target for osteosarcoma. Consistent with its role as a tumor suppressor gene, decreased expression of LDOC1 has been shown in several other types of cancers, including prostate cancer, 16 cervical cancer, 17 ovarian cancer, 18 oral squamous cell carcinoma, 19 and thyroid cancer. 13 LDOC1 has been reported to be involved in regulation of proliferation and apoptosisin cancer. However, the significance of LDOC1 expression in tumor metastasis and cancer progression has not yet been investigated. In this study, we investigated the role of LDOC1 in cancer metastasis and tumor pathophysiology and its prognostic significance in osteosarcomas.
Materials and methods
Cell lines
The 143B human osteosarcoma cell line was obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). Unless otherwise specified, the 143B cell line was maintained in Dulbecco’s modified Eagle medium (DMEM; Mediatech Inc., Herndon, VA, USA) supplemented with 10% fetal bovine serum (FBS), 0.015 mg/mL 5-bromo-2′-deoxyuridine, 100 U/mL penicillin, 100 µg/mL streptomycin, and 250 ng/mL amphotericin (antibiotic-antimycotic; Life technologies, Carlsbad, CA, USA) at 37°C in an atmosphere of 5% CO2.
Western blotting
Total protein was extracted from tumor cells or tissues using radio-immunoprecipitation assay buffer (Beyotime Institute of Biotechnology, Shanghai, China) as per the manufacturer’s protocol and stored at −20°C. A total of 40 µg of protein from each sample was separated on a 12% sodium dodecyl sulfate (SDS)-polyacrylamide gel by electrophoresis and transferred onto nitrocellulose membranes (Merck Millipore, Darmstadt, Germany). Blots were immunostained with the primary antibody, and then with secondary antibody. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) served as the loading control. The primary antibody for LDOC1 was purchased from Abcam (Cambridge, UK) and used at a 1:2000 dilution. The other primary antibodies used were anti-Dvl2 (Cell Signaling Technology, USA), anti-GAPDH (CWBIO, China), and anti-Wnt5a (Cell Signaling Technology, USA).
Construction of cells stably expressing LDOC1
First, we constructed the retroviral vector. The pLV.EX3d.P/neo-EF1A > LDOC1 > IRES/eGFP shuttling plasmid was constructed using Gateway™ recombination cloning technology (Thermo Fisher Scientific, Waltham, MA USA). Briefly, overlap extension quantitative real-time polymerase chain reaction (qPCR) was used to amplify the attB1-Kozak-LDOC1-attB2 sequence. Primer sequences were as follows: attB1-K-LDOC1, 5′-GGGGACAAGTTTGTACAAAAAAGCAGGCTGCCACCATGGTGGATGAGTTGGTGCTG-3′; attB2-LDOC1, 5′- GGGGACCACTTTGTACAAGAAAGCTGGGTCTAATAATCATCCTCCTCTTCTTC-3′. Subsequently, pDown-LDOC1 plasmid and pLV.EX3d.P/neo-EF1A > LDOC1 > IRES/eGFP plasmid were constructed. The positive clone was screened using colony PCR, and the positive plasmids were further confirmed by sequence analysis. The LDOC1 expression vector was stably expressed in 143B cells using the Lenti-Pac HIV Expression Packaging System (GeneCopoeia, Rockville, MD, USA). The eGFP was inserted into the same sites, as a negative control, according to the manufacturer’s instructions. Successful transfection was confirmed by detecting eGFP expression using an inverted fluorescence microscope (IX70, Olympus, Germany). Puromycin resistance markers were included in the vector; therefore, stable tranfectants were selected by adding 1 µg/mL puromycin to the cell culture media.
Cell migration and invasion assays
The invasion abilities of cells were assessed using growth-factor-reduced (GFR) Matrigel™ Invasion Chambers (BD Biosciences, Bedford, MA, USA). To obtain the supernatant containing the chemotactic factors, cells were cultured in serum-free DMEM for 24 h, harvested, and pelleted to collect the supernatant. Fibronectin (2.5 mg/L) was added to the supernatant, which was placed in the lower chamber of the transwell plate. The chambers were separated by a Makrolon microporous membrane with a diameter of 13 µm and bore diameter of 8 µm. The top chamber contained 143B cells stably expressing either LDOC1 or eGFP that had been cultured to the logarithmic growth phase (approximately 48 h). The cells were then treated with trypsin and pelleted by centrifugation. The collected cells were washed three times using phosphate-buffered saline (PBS) and cell viability was assessed in a serum-free DMEM suspension containing 5 × 105 cells/mL using trypan blue. Only preparations where cell viability exceeded 95% were used in the migration and invasion assays. The cell suspension (1 × 105 cells in 0.2 mL) was added into each upper chamber in the transwell plate. The cells were cultured in the transwell for 6 h. Migration assays were performed using the same procedure, except that the transwells were not coated with Matrigel.
To assess migration and invasion, the microporous membrane was removed and the cells remaining on the upper surface were wiped with a cotton ball soaked in 100% methyl alcohol, and then methyl alcohol and hematoxylin stained. The number of cells penetrating the membrane was determined by counting under a microscope. The numbers of cells that underwent migration or invasion were determined from five independent samples in each group (LDOC1- or eGFP-expressing). For each replicate, the tumor cells in 10 randomly selected fields were counted, and the mean counts were calculated.
qPCR Primer Array
LDOC1-associated changes in the expression of genes associated with metastasis were determined in 143B cells stably expressing LDOC1 or eGFP. The Human Tumor Metastasis I 96 All-in-One™ qPCR Primer Array (Cat. No. HAQPA-203-01; GeneCopoeia) 96-well qPCR plate was used. Each array is a panel of validated, optimized qPCR primers for 92 mRNAs closely associated with tumor metastasis, and the housekeeping genes
Xenograft osteosarcoma model
All experimental procedures were approved by the Ethics Committee of the First Affiliated Hospital of Sun Yat-Sen University. Male NOD/SCID mice (14-week-old) were randomly allocated into two groups (n = 7 mice/group). 143B cells (1 × 106 cells in 0.2 mL PBS) stably expressing LDOC1 or eGFP (as the negative control) was injected into the right tibia plateau of each mouse. Tumor volume was measured every week for 5 weeks. The tumor volume was calculated using the following equation: Tumor volume = Length × Width2 × 0.52.
Micro-computed tomography imaging
Micro-computed tomography (CT) imaging was performed 2 weeks after the tumor cells were injected into the mice. Images were acquired using an Inveon Micro-CT scanner (Siemens Preclinical Solution, Knoxville, TN, USA). The animals were anesthetized with 2% isoflurane during the scan. The mice were placed in a prone position on the bed of the scanner, and two bed positions were acquired. The imaging program was a 5-min CT scan. Whole body images were obtained using the micro-CT scanner. The composite micro-CT images were generated using an Inveon Research Workplace (Siemens Preclinical Solution). For image reconstruction, the standard Ordered Subsets Expectation Maximization technique was used.
All of the mice were sacrificed 5 weeks after injection. The tumors and lungs were excised and embedded in paraffin for hematoxylin and eosin (H&E) staining.
Patients
All patients were recruited from the First Affiliated Hospital of Sun Yat-Sen University in Guangzhou, China. This study was approved by the ethics committee of the First Affiliated Hospital of Sun Yat-Sen University. Osteosarcoma patients (n = 74) were retrospectively enrolled for follow-up. The paraffin-embedded tissue sections from the retrospectively enrolled osteosarcoma patients were obtained for immunohistochemistry staining as previously described. 20 All of the enrolled patients were between 10–20 years of age and had a confirmed diagnosis of osteosarcoma based on imaging and pathology findings. Patients who had secondary osteosarcoma accompanied by other tumors were excluded from the study.
Immunohistochemistry
For the immunohistochemical analyses, the LDOC1 antibody was purchased from Abcam and used at a 1:500 dilution. Tissues from patients with Osteofibrous dysplasia (n = 19) served as the negative control for LCOC1 staining. The paraffin-embedded osteosarcoma specimens were deparaffinized in xylene and rehydrated in graded alcohol. The samples were treated with boiling citrate buffer (10 mmol/L, pH 6.0) for 20 min and then with 3% hydrogen peroxide in water. The nonspecific staining was blocked by incubation with 5% bovine serum albumin for 1 h at room temperature. After incubation with the LDOC1 antibody for 30 min at room temperature followed by thorough washing with PBS thrice, the slides were incubated with a horseradish peroxidase/Fab polymer conjugate for another 30 min. The sites of peroxidase activity were visualized using diaminobenzidine as the substrate and counterstained with Mayer’s hematoxylin. The stained sections were evaluated independently by two pathologists blinded to the specimen identity and scored based on the intensity and extent of the LDOC1 staining. Randomly selected fields containing at least 100 cells (n = 10) were chosen from each section and examined microscopically. First, the extent of the LDOC1 staining was evaluated based on the amount of area that stained positive for LDOC1 using the following scale: 0% (score 0), <10% (1), 11%–25% (2), 26%–50% (3), 51%–75% (4), and >75% (5). The intensity of the staining was then scored based on the color using the following scale: no cellular stain (score 0), yellow (1), tan (2), and brown (3). The final score was calculated by multiplying the intensity and extent scores. The results were stratified into different groups depending on the straining: negative “−” (score: 0–1), weak positive “+” (score: 2–4), moderate positive “++” (score: 5–9), and strong positive “+++” (score: 10–15). The patients were stratified based on the final staining score for further analyses.
Statistical analysis
The differences in gene expression and migration/invasion abilities of osteosarcoma cells were compared using the independent Student’s t-test. The survival curves of patients were estimated by the Kaplan–Meier method, and the differences between survival curves were compared using the log-rank test. All tests were two-tailed and were considered to be statistically significant for values of p < 0.05. Statistical tests were performed using the PASW Statistics 18.0.0 software.
Results
Increased LDOC1 expression decreases the migration and invasion abilities of osteosarcoma cells
We evaluated the expression of LDOC1 in cultured osteosarcoma cells, a mouse model of human osteosarcoma, and primary osteosarcoma samples from patients to better characterize the role of LDOC1 in osteosarcoma. First, we constructed a pLV.EX3d.P/neo-EF1A > LDOC1 > IRES/eGFP vector to overexpress LDOC1 in vitro in 143B osteosarcoma cells. Osteosarcoma cells were transfected with either the plasmid containing the

Increased LDOC1 expression decreases the migration and invasion abilities of osteosarcoma cells. Osteosarcoma cells (143B) were engineered to stably express either LDOC1 or eGFP (negative control) to assess the effects of LDOC1 overexpression in vitro. (a) Representative example of the western blotting results showing increased LDOC1 protein expression in osteosarcoma cells. The levels of LDOC1 in whole cell lysates were analyzed by immunoblotting with anti-LDOC1 antibodies. GAPDH served as the internal control. (b, c) The abilities of 143B/LDOC1- or 143B/eGFP-expressing cells to migrate (b) and invade (c) were measured using a transwell assay. Representative images of the results are shown. The bar graphs indicate the number of invading cells (mean ± standard deviation).
Increased LDOC1 expression up-regulates the expression of many genes involved in tumor metastasis suppression
Next, we conducted a qPCR array analysis to characterize the changes in gene expression in LDOC1-overexpressing 143B cells, compared to the control 143B cells. The results of the qPCR array are shown in the form of a heat map in Figure 2(a). Of the known metastasis-involved genes, the qPCR array indicated that several were up-regulated significantly (p < 0.05, t-test) in the LDOC1-overexpressing osteosarcoma cells. The overexpressed genes include many tumor suppressors involved in cancer metastasis regulation, including those encoding adenomatous polyposis coli (APC), CTNNA1 (αE-catenin), HIV-1 Tat interactive protein 2(HTATIP2), KiSS-1 metastasis-suppressor (KISS1), phosphatase and tensin homolog (PTEN), SMAD (mothers against decapentaplegic (MAD) and the Caenorhabditis elegans protein SMA) family members 2 and 4 (SMAD2 and SMAD4), tissue inhibitor of metalloproteinases-3 (TIMP3), and tumor protein p53 (TP53) (Figure 2(b)). These results further indicate that LDOC1 regulates the metastatic potential of osteosarcoma cells.
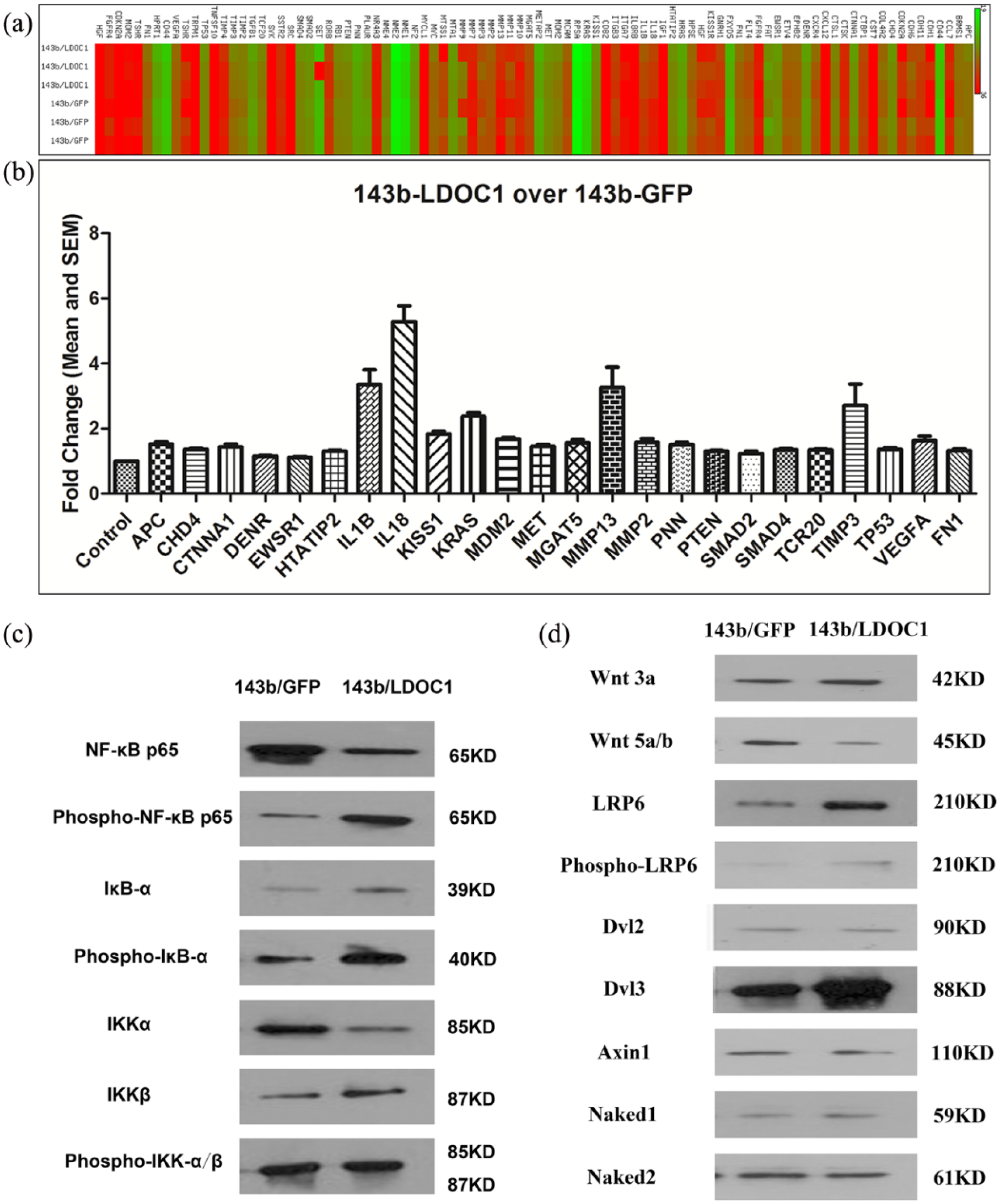
Increased LDOC1 expression up-regulates the expression of many genes involved in tumor metastasis suppression. The expression levels of genes associated with suppression of tumor metastasis was assayed in LDOC1-overexpressing 143B cells and compared to that in control eGFP-expressing 143B cells using a commercially available human tumor metastasis qPCR Primer Array as described in section “Materials and methods.”
Increased LDOC1 expression decreases Wnt5a levels in osteosarcoma cells
The Wnt5a signaling pathway has been reported to regulate the metastasis of osteosarcomas. 21 Therefore, we next evaluated whether increased LDOC1 expression in osteosarcoma cells alters Wnt5a expression. Western blotting analysis showed that LDOC1 overexpression in 143B osteosarcoma cells did not affect the expression of disheveled segment polarity protein 2 (Dvl2), an intracellular effect of multiple Wnt signaling pathways. Nevertheless, LDOC1 overexpression resulted in decreased expression of Wnt5a protein (Figure 3). Since the Wnt5a signaling pathway has been shown to promote metastasis of osteosarcoma, these data indicate that LDOC1 expression might down-regulate the Wnt5a signaling pathway and thereby inhibit the metastasis of osteosarcoma cells.

Increased LDOC1 expression decreases Wnt5a levels in osteosarcoma cells. The expression levels of Dvl2 and Wnt5a in cell lysates prepared from the 143B/LDOC1- or 143B/eGFP-expressing cells were analyzed by immunoblotting with anti-Dvl2 antibodies and anti-Wnt5a antibodies. GAPDH served as the internal control. Data shown are representative of the results from three independent trials.
Increased LDOC1 expression decreases lung metastasis in an osteosarcoma xenograft model
The in vitro evidence from the LDOC1-overexpressing osteosarcoma cells suggested that LDOC1 expression regulates the metastatic potential of osteosarcoma cells. To test the role of LDOC1 in osteosarcoma metastasis and progression in vivo, we used a xenograft model of osteosarcoma in NOD/SCID mice. 143B osteosarcoma cells stably overexpressing LDOC1 or eGFP were injected into male NOD/SCID mice. The representative examples of the xenograft osteosarcoma tumors at the study endpoint are shown in Figure 4(a). Tumor metastasis was monitored with micro-CT imaging (Figure 4(b)). Many of the relapsed osteosarcoma patients have tumors metastasize to the lungs. 4 We quantified lung metastases in our mouse model using micro-CT imaging 2 weeks after the injection of tumor cells. The results showed that in mice that received LDOC1-overexpressing osteosarcoma cells, there were fewer lung metastases compared to those in mice injected with the control 143B cells (Figure 4(c)). Our data showed that increased LDOC1 expression significantly decreased pulmonary metastases of osteosarcoma cells (by 23%; p < 0.05, t-test). The number of pulmonary tumors in mice injected with LDOC1-overexpressing osteosarcoma cells (10 ± 3 tumors per mouse) was significantly less than the number in mice injected with the control cells (13 ± 4 tumors per mouse; p < 0.05, t-test). These results indicate that increased LDOC1 expression in osteosarcoma cells might attenuate the metastatic potential of osteosarcoma cells in vivo, and that LDOC1 functions as a tumor suppressor gene in the xenograft mouse model.

Increased LDOC1 expression decreases lung metastasis in an osteosarcoma xenograft model. NOD/SCID mice were injected with either 143B/LDOC1- or 143B/eGFP-expressing cells, and lung metastasis was monitored by micro-CT imaging 2 weeks post the injection of tumor cells (n = 7 mice/group). (a) Representative examples of primary tumor formation in the mice. (b) Micro-CT imaging was used to monitor the presence and number of lung metastases 2 weeks after tumor injection. Representative images from mice injected with LDOC1-expressing 143B cells are shown. (c) The number of lung metastases in mice with tumors originating from LDOC1-overexpressing (143B/LDOC1) versus control (143B/eGFP) cells was counted at the study endpoint (5 weeks post the injection of tumor cells).
Association of LDOC1 staining with the survival rate of patients with osteosarcoma
We next investigated the expression of LDOC1 in patients with osteosarcoma and its clinical prognostic relevance. We examined the expression and localization of LDOC1 protein in primary osteosarcoma samples obtained from 74 patients with osteosarcoma who were retrospectively enrolled in the study and had completed clinical follow-up. The osteosarcoma tissue samples were obtained during surgical resection and then stained for this study using immunohistochemistry. The representative images show examples of strong positive staining, weak positive staining, and negative staining (Figure 5). LDOC1 expression was observed in both the nucleus and cytoplasm of osteosarcoma cells (Figure 5), indicating that LDOC1 may act as a regulator of the Wnt signaling pathway in vivo.

Representative immunohistochemical images of LDOC1 staining in tumor cells in osteosarcoma. Immunohistochemical analysis of LDOC1 staining in samples classified as strong positive, weak positive, or negative staining, as indicated. Original magnification ×100.
The demographic and disease characteristics of the patients are presented in Table 1. The mean age was 17.2 years (range: 6–45 years) and the mean follow-up time was 86 months (range: 6–144 months). LDOC1 staining was detected in 69 cases (93.2%). The level of LDOC1 expression in each osteosarcoma tumor was determined by immunohistochemistry, and the patients were stratified based on LDOC1 levels into two groups: (1) weak or negative staining and (2) strongly positive for LDOC1 staining. Survival analyses were conducted based on overall survival (OS; Figure 6(a)) and DFS (Figure 6(b)). The results showed that there was no statistically significant correlation between LDOC1 immunohistologic staining and OS in osteosarcoma (log-rank p = 0.187; Figure 6(a)). However, the results of the Kaplan–Meier analysis indicate that the survival rates in most high-LDOC1-staining cases were, in general, lower than in those with weak or negative LDOC1 staining (Figure 6(a)). Kaplan–Meier analysis indicated that LDOC1 staining is associated with DFS of patients with osteosarcoma; strong positive signal for LDOC1 immunohistologic staining in the tumors was associated with improved DFS (log-rank p = 0.022; Figure 6(b). We conclude that high LDOC1 expression in osteosarcoma is associated with better OS and DFS of the patients.
Demographic data of patients enrolled in the retrospective survival analysis.

NOS: not otherwise specified.

Kaplan–Meier analysis of DFS in patients with osteosarcoma. (a) OS and (b) DFS data for retrospectively enrolled osteosarcoma patients (n = 74) who had completed clinical follow-up were analyzed using the Kaplan–Meier method. The tumors were stratified based on the intensity of the LDOC1 staining by immunohistochemistry, and patients were classified as strongly positive (green) or weak positive, or negative (blue). Censored data are indicated with a vertical hash mark. DFS was defined as the absence of a metastatic event.
Discussion
Little is known about the role of LDOC1 in cancer metastasis, its contribution to the pathophysiology of osteosarcomas, and prognostic significance, if any. Here, we investigated the role of LDOC1 in metastasis of osteosarcoma cells and evaluated the clinical implications of LDOC1 expression in patients with osteosarcoma. Data from our in vitro analyses showed that increased LDOC1 expression in osteosarcoma cells results in decreased migration and invasion of osteosarcoma cells, concomitant with up-regulated expression of many genes involved in tumor metastasis suppression. In the xenograft osteosarcoma mouse model, increased LDOC1 expression was associated with decreased Wnt5a levels, and reduced the lung metastasis ability of osteosarcoma cells. Finally, higher LDOC1 expression in osteosarcoma was associated with better OS and DFS of patients with osteosarcoma. Our results are the first, to our knowledge, to uncover the involvement of LDOC1 in cancer metastasis regulation and identify it as a potential marker for osteosarcoma prognosis.
The qPCR array analysis showed that increased LDOC1 expression in osteosarcoma cells resulted in up-regulation of many tumor suppressors related to cancer metastasis, including APC, CTNNA1, HTATIP2, KISS1, PTEN, SMAD2, SMAD4, TIMP3, and TP53. APC has been reported to regulate colorectal cancer metastasis;
22
CTNNA1 is an invasion-suppressor gene in human colon cancer cells;
23
up-regulation of HTATIP2 is reported to attenuate metastasis in hepatocellular carcinoma;
24
KISS1 is a metastasis-suppressor gene in many cancers;
25
loss of SMAD4 is reported to predict liver metastasis in human colorectal cancer;
26
PTEN expression can suppress the tumorigenicity and metastasis of tumor cells;
27
reduction in SMAD2/3 signaling suppresses metastasis of breast cancer cells;
28
expression of the TIMP3 gene reduces the metastasis of breast cancer cells;
29
and deletion mutations in
LDOC1 contains two potential interaction domains that are likely key to understand its function: a leucine zipper-like motif and a proline-rich region that contains the SH3 consensus sequence. 11 In other models, protein–protein interactions involving a leucine zipper motif have been shown to have roles in cancer. For example, in breast cancer models, activator protein-1 (AP-1) signaling was inhibited by RNA-binding protein with multiple splicing 1 (RBPMS1) isoforms that bound to the leucine zipper region of c-Jun. 31 The interaction of LDOC1 with the above tumor suppressors warrants further study. In addition, the potential interaction of LDOC1 leucine zipper with proteins involved in cancer metastasis, nuclear LDOC1 binds directly to the verprolin homology domain of WAVE3 and inhibits the degradation of p53. 32 LDOC1 has also been shown to interact with the tumor suppressor protein mammary serine protease inhibitor (MASPIN; Serpin B5), which also interacts with p53. 33 Additional studies are required to elucidate the network of protein interactions surrounding LDOC1 and the clinical impact of those interactions. The fact that increased LDOC1 expression in osteosarcoma cells up-regulates the expression of many tumor metastasis suppressors supports the hypothesis that LDOC1 can regulate the metastatic potential of osteosarcoma cells.
It is well known that the Wnt signaling pathway plays an important role in the metastasis of osteosarcoma. 34 Both Dvl2 and Wnt5a are Wnt signaling pathway proteins and are involved in tumor invasion and metastasis. Dvl2 silencing has been shown to reduce androgen-dependent prostate tumor cell migration. 35 Many studies have reported that Wnt5a can increase the metastasis of osteosarcoma.5–10 One study showed that Wnt5a can promote breast cancer cell migration via the Dvl2/Rab35/Rac1 signaling pathway. 36 In this study, we found that increased LDOC1 expression in osteosarcoma cells specifically decreases Wnt5a, but not Dvl2, levels. Taken together, our data suggest that LDOC1 may regulate the metastasis of osteosarcoma via directly/indirectly interacting with the Wnt signaling pathway.
Immunohistochemical analysis of LDOC1 expression in osteosarcoma showed that the expression of both nuclear and cytoplasmic LDOC1 is decreased, suggesting that the balance of the two forms is likely important for regulating its function in osteosarcoma. Consistent with the data from the mouse model, in osteosarcoma patients, strong positive levels of LDOC1 staining in osteosarcoma tumors correlated with better outcomes, that is, improved OS and DFS. Thus, LDOC1 appears to act as a tumor suppressor gene in osteosarcoma.
Conclusion
Our study is among the first to characterize the role of LDOC1 in cancer metastasis and to evaluate the prognostic potential of LDOC1 in patients with osteosarcoma. The results suggest that LDOC1 is a tumor suppressor in osteosarcoma, and that LDOC1 regulates the metastasis of osteosarcoma cells. Furthermore, LDOC1 may be a valuable prognostic marker and a therapeutic target in osteosarcomas.
Footnotes
Acknowledgements
The authors thank Drs Long-Juan Zhang, Fan Meng, and Guo-Yan Liang for their comments on the “Discussion” section of the manuscript. Bi-Cheng Yong and Jin-Chang Lu contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the 5010 Clinical Research Fund administered by the Sun Yat-Sen University (no. 1800009).
