Abstract
To evaluate the association between severe pulmonary embolism events and bevacizumab, we conducted the first meta-analysis evaluating the incidence and risk of pulmonary embolism associated with bevacizumab-based therapy. We searched PubMed, EMBASE, Cochrane Library, and ClinicalTrials.gov up to September 2016 for randomized controlled trials comparing bevacizumab with no bevacizumab on cancer patients. Incidence rates, relative risks, and 95% confidence intervals were calculated using fixed- or random-effects models. The primary end point was the association of bevacizumab with pulmonary embolism. Subgroup analyses were performed according to tumor type, dose, and publication status. In total, 23 randomized controlled trials were included. For patients receiving bevacizumab, the overall incidence of severe pulmonary embolism events was 1.76% (95% confidence interval = 1.25%–2.27%). Cancer patients treated with bevacizumab did not increase the risk of pulmonary embolism events (relative risk = 1.00, 95% confidence interval = 0.80–1.25). No significant differences in pulmonary embolism incidence or risk among subgroup analyses were observed. No evidence of publication bias was observed. This study suggested that bevacizumab may not increase the risk of pulmonary embolism in cancer patients.
Introduction
Venous thromboembolic event (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), is a common cause of premature death and morbidity. 1 Patients with cancer have higher risk of VTE than age-matched controls. 2 It has been reported that several cancer treatments are associated with increased risk of VTE and PE.3–5 Importantly, patients with cancer who experience VTE and PE have worse prognosis and an increased risk of death. 6
Several meta-analyses refering to the association between bevacizumab and thromboembolic events have been published. One meta-analysis identified five trials suggesting that treatment with bevacizumab increased risk of arterial thromboembolic event (ATE) but not VTE. 7 A second meta-analysis including studies in which ATE and VTE were not distinguished reported that treatment with bevacizumab increased risk of both all-grade and high-grade VTE. 8 A third meta-analysis investigating individual patient data from 10 trials concluded that bevacizumab combined with chemotherapy did not significantly increase the risk of VTE compared with chemotherapy alone. 9 However, none of the meta-analyses investigated the incidence and risk of PE associated with bevacizumab in patients with cancer.
Compared to other VTE, PE has different pathogenesis and performance and is even more life threatening. As the indications for use of bevacizumab are increasing, we conducted a comprehensive systematic review and combined the published and unpublished data from randomized controlled trials (RCTs) for a meta-analysis to quantify the association between severe PE events and bevacizumab therapy in patients with cancer.
Materials and methods
Following the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement, 10 we searched PubMed, EMBASE, Cochrane Library, and ClinicalTrials.gov from January 1960 to September 2016. Keywords used were “bevacizumab,” “Avastin,” and “cancer,” and the search was limited to RCTs. The PubMed search strategy is contained in S1 Table, and this search strategy was translated as appropriate for the search interfaces used for each database. Additional relevant articles in the reference lists of recent meta-analyses that evaluated bevacizumab in patients with cancer were also searched. In order to avoid duplication, only the most complete, recent, and updated report of each clinical trial was included.
Eligibility criteria
Trials were included if they fulfilled the following criteria—(1) participants: cancer patients, (2) interventions: bevacizumab-containing therapy or bevacizumab alone, (3) comparisons: non-bevacizumab therapy, (4) outcomes: number of patients with PE (grade 3–5 or severe) according to study group were available, and (5) study design: prospective RCTs.
Data extraction
Two reviewers independently collected data from included trials, with discrepancies resolved by consulting a third reviewer. Data collected from studies included trial phase, first author, publishing year, treatment arms, sample size, tumor type, dosage of bevacizumab, median follow-up, and number of patients with PE. Two independent researchers accessed the quality of included studies by using the Jadad scores, 11 with discrepancies resolved by consulting a third reviewer. Jadad scores ranged from 0 to 5, where 5 indicates the highest quality. In addition, for studies in which data of PE had not been published, we abstracted the relevant numbers from ClinicalTrials.gov databases of serious adverse events.
Data analysis
The overall incidences of PE in the two groups were our primary outcome. The PE incidences were presented as risk ratio (RR) with 95% confidence intervals (CI) and combined using random-effects model. For studies that reported zero events, we applied the classic half-integer correction to calculate the RR and variance. 12 Heterogeneity is quantified using I-square and Cochran’s Q test statistics. We considered I2 value larger than 50% as the significant heterogeneity, and a random-effects model was used; otherwise (I2 < 25%), we used the fixed-effects model. 13 We used the Begg’s and Egger’s tests to assess the presence of publication bias regarding our primary outcomes. 14 A statistical test with a p < 0.05 indicated statistical significance. All statistical analyses were performed by using Stata software (Version 12.0).
We performed three subgroup analyses, stratified by tumor type, bevacizumab dosages (2.5 mg/kg/week vs 5 mg/kg/week), and publication status of PE data (data published in original article vs collected from ClinicalTrials.gov). All subgroup analyses followed the same analysis procedure.
Results
Literature collection
A total of 1933 articles were found in our initial search; 1807 of which were excluded by screening the titles and abstracts. Further 103 articles were excluded after reviewing full text. Overall, 23 RCTs 15–37 were included in the final analysis. The trial flow summary is reported in Figure 1.
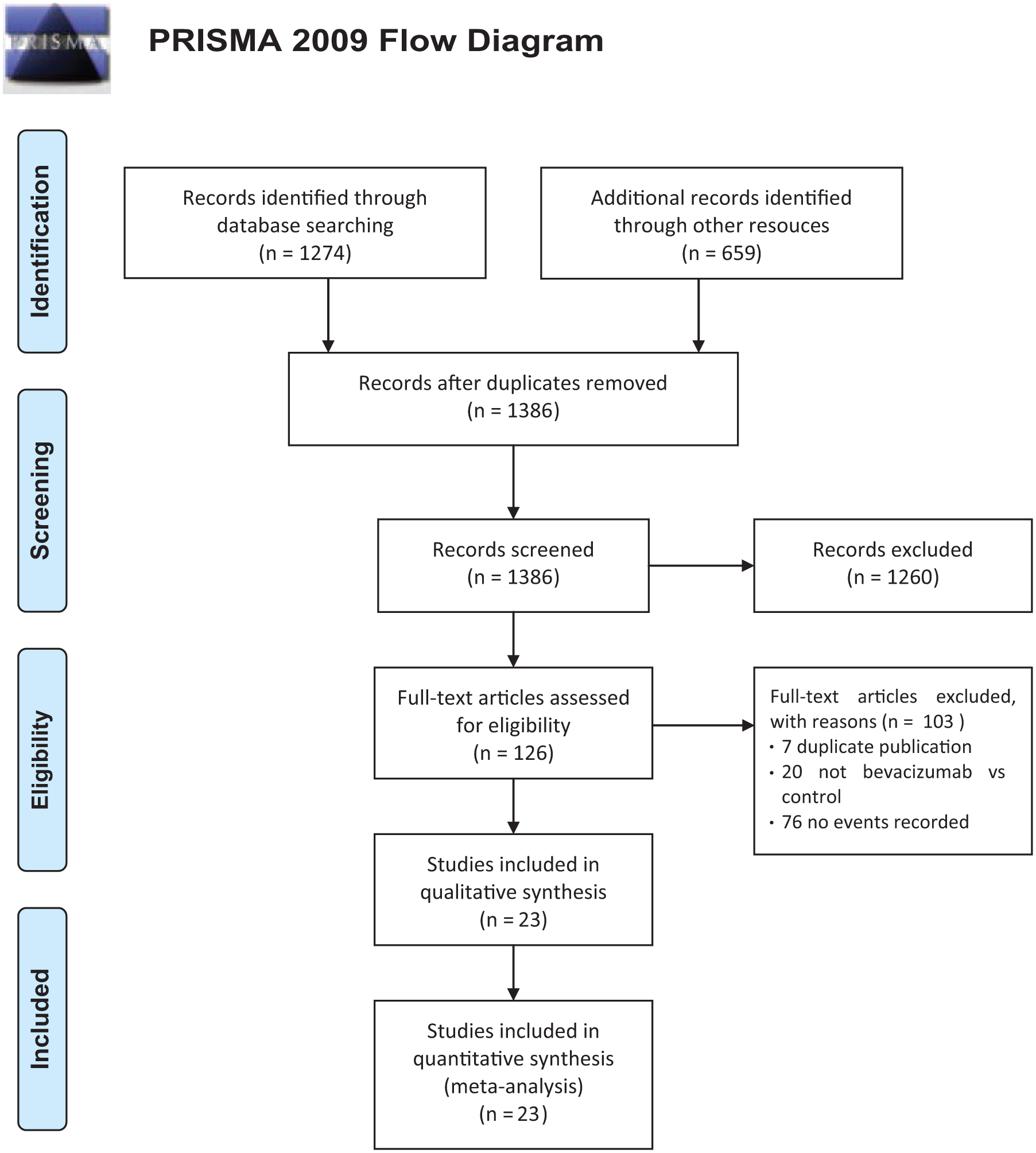
Study flow diagram.
Study characteristics
Studies enrolled 16,458 participants who were randomized to bevacizumab group or control group. Seven studies were conducted in breast cancer patients, six in colorectal cancer patients, two in ovarian cancer patients, one each in pancreatic cancer, gastric cancer, non–small cell lung cancer (NSCLC), extensive-stage small-cell lung cancer (ESCLC), melanoma, angiosarcoma, glioblastoma, and lymphoma (Table 1). In addition, eight studies received bevacizumab at 2.5 mg/kg/week, and 15 trials received bevacizumab at 5 mg/kg/week. PE was defined by the common terminology criteria for adverse events (CTCAE) criteria in all the included trials. Data of PE events were collected from ClinicalTrials.gov in 14 trials.18–21,25–30,33–35,37 All trials were RCTs, and 11 trials were double blinded. The level of evidence for each trial was graded from scores 2 to 5 according to the Jadad score (S1 Table).
Characteristics of studies included in the primary analysis.

NCT: National Clinical Trial; NR: not reported; NSCLC: non–small cell lung cancer; ESCLC: extensive-stage small-cell lung cancer; R-CHOP: rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone.
Number of patients for safety analysis.
Incidence and risk ratio of PE
High-grade (or defined as serious) PE occurred in 154 of 8666 participants randomized to the bevacizumab group and 141 of 7792 participants randomized to the control group. The incidence of PE in bevacizumab group was 1.76% (95% CI = 1.25%–2.27%) and 1.74% (95% CI = 1.28%–2.20%) in patients receiving non-bevacizumab regimens (Table 2). Analysis of 23 trials showed that bevacizumab was not associated with increased risk of PE, the pooled RR was 1.00 (95% CI = 0.80–1.25, p = 0.98) with non-significant heterogeneity (I2 = 0%; Figure 2).
Relative risk of PE by subgroup.

PE: pulmonary embolism; CI: confidence interval; NSCLC: non–small cell lung cancer; NA: not applicable; ESCLC: extensive-stage small-cell lung cancer.

Relative risk of PE in cancer patients’ treatment with bevacizumab compared with control.
Subgroup analysis by dose regimen
We conducted a subgroup analysis according to dose and schedule, with the high-dose group receiving 5 mg/kg/week and the low-dose group receiving 2.5 mg/kg/week. The RR of PE for patients who received low dose of bevacizumab compared with control was 1.08 (95% CI = 0.80–1.45, P = 0.62). Similarly, no significant difference was found between patients treated with high dose of bevacizumab and controls (RR = 0.90, 95% CI = 0.64–1.27, p = 0.62; Table 2).
Subgroup analysis by publication status of PE data
We explored whether the publication status may influence the risk of PE. For PE data reported in nine published articles, the pooled RR of PE was 0.85 (95% CI = 0.59–1.23, p = 0.39). For PE data collected from ClinicalTrials.gov, the pooled RR of PE was 1.09 (95% CI = 0.82–1.45, p = 0.54). No statistically significant difference between subgroups was detected (Table 2).
Subgroup analysis by tumor type
In addition, we also performed a subgroup analysis to clarify whether different tumor type would alter the RR of PE associated with bevacizumab. We found that the majority of the evidence is provided by studies in colorectal cancer and breast cancer patients. Although effect sizes and incidences of PE were variable, no statistically significant differences between types of tumor were observed (Table 2).
Publication bias
The Begg’s and Egger’s test did not indicate significant publication bias in the meta-analysis for the risk ratio of PE (Begg’s test p = 0.958; Egger’s test p = 0.946).
Discussion
PE has the potential to lead to a number of physiological changes due to obstruction of the pulmonary arteries and may lead to death. Previous meta-analysis investigated the association between bevacizumab and thromboembolic events, but none of these studies evaluated the incidence and risk of PE associated with bevacizumab. Therefore, we performed the first meta-analysis refering to this clinical issue. Based on 23 RCTs, our results demonstrated that bevacizumab was not associated with an increased risk of PE in cancer patients. Results were consistent across subgroups defined by dose, data of publication status, and type of tumor.
The incidence of PE is usually unclassified due to its low incidence. Instead, the incidence of PE is considered to be included into the categories, such as thromboembolic events or thrombosis events. After reviewing all published articles, only nine trials fulfilled the inclusion criteria and reported PE data were included. Therefore, we also searched ClinicalTrials.gov, and additional 14 trials were included. It should be mentioned that PE was reported as “serious adverse events” or “other adverse events” in the ClinicalTrials.gov and presented as adverse events according to CTCAE criteria in the publications. The definition of serious adverse events may differ from high-grade (grade 3–5) adverse events. Subgroup analysis by publication status of PE suggested that bevacizumab was not associated with an increased risk of PE in cancer patients.
In subgroup analysis by tumor type, there was a trend of increased risk of PE in colorectal cancer patients. These may be because that in patients with colorectal cancer, the effect on survival is the greatest and therefore more additional cycles of chemotherapy was used in these patients. The increased risk of VTE might be associated with the bevacizumab-combination therapy and not with treatment with bevacizumab by itself. Most patients among selected articles received concurrent treatment including different medicines; it has been reported that both 5-fluorouracil and platinum increased risk of VTE in cancer patients.4,38 Previous study support the hypothesis that these drugs damage the vascular network and endothelium, induce platelet activation, and upregulate prothrombotic factors, 39 which may result in thrombus formation.
Given the potentially differing dose intensity of bevacizumab may influence the risk of PE, we conducted a subgroup analysis according to dose and schedule with the high-dose group receiving 5 mg/kg/week vs the low-dose group receiving 2.5 mg/kg/week. Results showed that both high and low dose of bevacizumab were not associated with PE. This was in contrast with the study by Nalluri et al., 8 which suggested that the bevacizumab was associated with dose-independent increased risk of VTE. These may be because ATE and VTE were not distinguished in the study by Nalluri et al., 8 and it has been confirmed that bevacizumab was associated with ATE. 7
Our study has several limitations. First, trials included in this study were conducted at various international institutions by different investigators, and patient selection criteria varied between studies. Second, all patients received chemotherapy, which may itself be thrombogenic.4,38 Thus, whether bevacizumab monotherapy would increase the risk of PE cannot be answered by this meta-analysis. Third, PE and other adverse events are generally reportable only up to 30 days following last treatment date. Therefore, events occurring after 30 days following last treatment would not have been captured, thus creating a bias in the analysis. Finally, an important limitation of this study is the lack of information on cancer burden, smoking history, surgical history, body mass index (BMI), proximity to surgery, and other factors addressed in earlier comments. 8
In summary, the current analysis suggests that the use of bevacizumab did not increase the risk of PE among patients with cancer. Whether bevacizumab alone is associated with PE would require trials comparing bevacizumab as a single agent to no therapy, which would be infeasible.
Footnotes
Acknowledgements
J.C. and W.Y. designed the article; M.L., Y.Z., and Z.C. wrote the manuscript, searched the library, and reviewed all articles; Y.Q., Z.P., Z.C., and Y.S. extracted data and evaluated the bias. All authors reviewed the manuscript. M.L. and Y.Z. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the grants from Science and Technology Planning Project of Guangdong Province (No.2016ZC0178) and The Doctoral Scientific Research Foundation of Affiliated Hospital of Guangdong Medical University (No.10301B010012).
