Abstract
Apatinib is a novel tyrosine kinase inhibitor targeting vascular endothelial growth factor receptor 2. This study aimed to evaluate the efficacy and safety of apatinib in metastatic breast cancer (MBC) under non-clinical trial setting, and to study the impact of previous antiangiogenic treatment to the efficacy of apatinib. 52 MBC patients treated with apatinib under non-clinical trial setting in Fudan University Shanghai Cancer Center between January 1st 2015 and October 1st 2016 were included. All patients were included in time-to-treatment failure (TTF) analysis, while 45 patients were enrolled for progression-free survival (PFS) and overall survival (OS) analysis because 7 of the patients with treatment discontinuation due to intolerable toxicities had too short time for efficacy assessment. Impact of previous exposure to antiangiogenic treatment and other factors to patients’ survival were analyzed by Log-rank analysis and Cox multivariate analysis. The median PFS, median OS, and median TTF were 4.90 (95% confidence interval [CI] 3.44 – 6.36), 10.3 (unable to calculate 95% CI), and 3.93 (95% CI 1.96 – 5.90) months, respectively. Previous treatment of bevacizumab did not affect the efficacy of apatinib. Previous exposure to anthracycline, age of 60 years or older and palmar-plantar erythrodysesthesia syndrome were independent predictors for prolonged PFS. Discontinuation of treatment was more common in age group of 60 years or older than that in younger group, although the difference was not significant. Although toxicities were generally managable, a previously unrecorded grade 3~4 adverse event of dyspnea has been observed. This study confirmed the encouraging efficacy and manageable safety of apatinib on pretreated MBC patients in non-clinical trial setting. For the first time to our knowledge, this study found that previous treatment of bevacizumab did not affect the efficacy of apatinib, and reported an undocumented severe adverse effect of dyspnea.
Introduction
Breast cancer (BC) remains one of the top leading causes of cancer-related deaths. 1 Prognosis for patients with metastatic breast cancer (MBC) who fail second-line treatment or are beyond line of standard therapy remains poor, with a median overall survival (OS) of about 1 year,2–8 thus giving rise to an urgent need for developing novel strategies and drugs.
Anti-angiogenesis is an important anti-cancer strategy.9,10 Bevacizumab, an anti-vascular endothelial growth factor (VEGF) monoclonal antibody, has shown encouraging benefits to prolong progression-free survival (PFS) in MBC, but it fails to significantly extend OS.11–14 Anti-VEGF receptor (VEGFR) tyrosine kinase inhibitors (TKIs), such as sunitinib and sorafenib, demonstrate limited clinical benefits and thus they are not used in routine clinical practice.2,15–18
Recently, apatinib, a novel TKI targeting VEGFR-2, has shown satisfying efficacy on various types of cancers with acceptable toxicities.4–6,19–23 Apatinib first made a name of itself for its remarkable survival benefits in patients with metastatic gastric cancer (GC) in a phase III clinical trial, and thus, it has been approved by the China Food and Drug Administration for the third-line treatment of GC. 20 However, due to some imperfections in the design of clinical trial, concerns about exaggeration of survival benefits and patients’ tolerability of apatinib have arisen. 24 In addition, in an era when anti-angiogenic treatment is widely used, question that whether previous exposure to anti-angiogenic reagents affects the efficacy of apatinib has been raised. 24
Hu et al.5,6 conducted two single-arm phase II studies of apatinib in MBC and reported a median PFS and OS of 3.3 and 10.6 months respectively for triple-negative breast cancer (TNBC) patients, while 4.0 and 10.3 months respectively for non-TNBC patients. Based on these results, apatinib has become a preferred choice in the real world for heavily pretreated MBC patients who have no available standard treatment options and fail to attend any clinical trial. However, patients included in these two clinical trials were of respectively younger age than those in the real world, with the median ages being 49–52 years old,5,6 and only 11 patients being 60 years or older (data not published). Although the mean age at diagnosis for BC patients in China is younger (44–55 years old) than that for Western women, an increasing shift toward an older median age has been observed. 25 In more developed cities such as Shanghai and Beijing, 70–74 years old was a second age peak of BC incidence. 25 In addition, unlike patients in clinical trial settings, real-world patients may have various health conditions and different previous treatments. Therefore, efficacy and toxicities of apatinib need better evaluation to guide real-world clinical practice, especially in patients with advanced age. Moreover, the study of TNBC excluded patients previously treated with bevacizumab or any VEGFR TKIs. Currently, bevacizumab remains an important option and the only anti-angiogenic agent for heavily pretreated BC patients in China. Whether patients with previous exposure to bevacizumab could benefit from apatinib treatment as a second anti-angiogenic agent remains unknown.
This study aims to further study the efficacy and safety of apatinib in MBC patients in non-clinical trial setting and to explore the impact of previous anti-angiogenic treatment of bevacizumab to the efficacy of apatinib, which derived from the clinical experience of Fudan University Shanghai Cancer Center (FUSCC).
Patients and methods
Patients and eligibility criteria
The inclusion criteria were shown as follows: (1) female patients with histologically confirmed MBC, who were treated with apatinib in FUSCC under non-clinical trial settings from 1 January 2015 to 1 October 2016; (2) patients who failed in all the standard treatments before apatinib treatment according to National Comprehensive Cancer Network (NCCN) guideline, or were considered intolerant for standard treatments, or willingly chose not to undergo certain standard treatments; and (3) patients with at least one measurable lesion, which was defined by Response Evaluation Criteria in Solid Tumors (RESIST) criteria (1.1). For patients who have been treated with bevacizumab, there were no limits regarding the time since last treatment of bevacizumab before apatinib treatment.
Status of tumor hormone receptor (HR), including estrogen receptor (ER) and progesterone receptor (PR), was determined using immunohistochemistry (IHC), as previously reported. 6 Human epidermal growth factor receptor (HER2)/Neu positivity was defined by IHC and/or chromogenic/fluorescent in situ hybridization (FISH) as previously reported. 6 For patients who underwent biopsies in the metastatic sites, HR and HER2 status were determined based on the latest pathological test before apatinib treatment, in the case of changed molecular characteristics.
Treatment and dose modification
Patients were prescribed with apatinib mesylate manufactured by Jiangsu HengRui Medicine Co. Ltd, and the starting dose, dose modification, and discontinuation for them were determined by clinicians based on data of previous clinical trials as well as the general health status and toxicities of patients.
Efficacy and safety assessments
Patients were followed up until death, loss to follow-up, discontinuation of treatment due to intolerable toxicity, or till the cutoff date of 15 February 2017.
Tumor changes were assessed by computed tomography (CT) and magnetic resonance imaging (MRI), and tumor responses were evaluated by both radiologists and our oncologists based on the RESIST criteria (1.1). Image assessments of tumor responses and physical examinations in routine clinical practice were conducted approximately every 1½ months, or whenever necessary. Best tumor responses were documented. Blood counts and other assessments of toxicity were recommended at various intervals. Adverse events (AEs) were assessed according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE, 4.03). Information of AEs as objective symptoms was collected based on a patient self-report system or by reviewing biochemical test results. AEs occurring during whole treatment as well as their most severe levels were documented.
Primary end point was PFS, which was defined as the time period from initiating apatinib treatment to disease progression or death, regardless of whichever would occur first. Secondary end points included OS and time-to-treatment failure (TTF). OS was defined as the time period from initiating apatinib treatment to the date of death of any cause or last follow-up visit, while TTF was defined as the time period from initiating apatinib to the date of disease progression, treatment discontinuation or death, regardless of whichever would occur first.
Statistical analysis
Median durations of PFS, OS, and TTF were calculated using Kaplan–Meier method, and inter-group comparisons were carried out by log-rank test. Pearson χ2 test was used to analyze categorical variables. Cox regression analysis was used to analyze the correlations between factors and prognosis. All statistical tests were two-tailed, with the significant level being set at p < 0.05. Statistical Package for the Social Sciences Software (SPSS) version 19 was used for all statistical analyses.
Results
Baseline characteristics
A total of 53 female BC patients were treated with apatinib under non-clinical settings in FUSCC from 1 January 2015 to 1 October 2016. One patient was excluded because of lack of measurable lesions. Consequently, altogether 52 patients were included in the final analysis. Patient characteristics at baseline are summarized in Table 1. In total, 13 (25%) patients were 60 years or older while receiving apatinib treatment, with the oldest being 85 years old. All patients had received at least one line of systemic therapy, 42 among whom (80.8%) were heavily treated and underwent more than three lines of systemic therapies. Overall, 10 (19.2%) patients have received previous anti-angiogenic treatment of bevacizumab, with the median time since last treatment being 3 weeks (range: 1.5–107 weeks).
Patient characteristics at baseline.
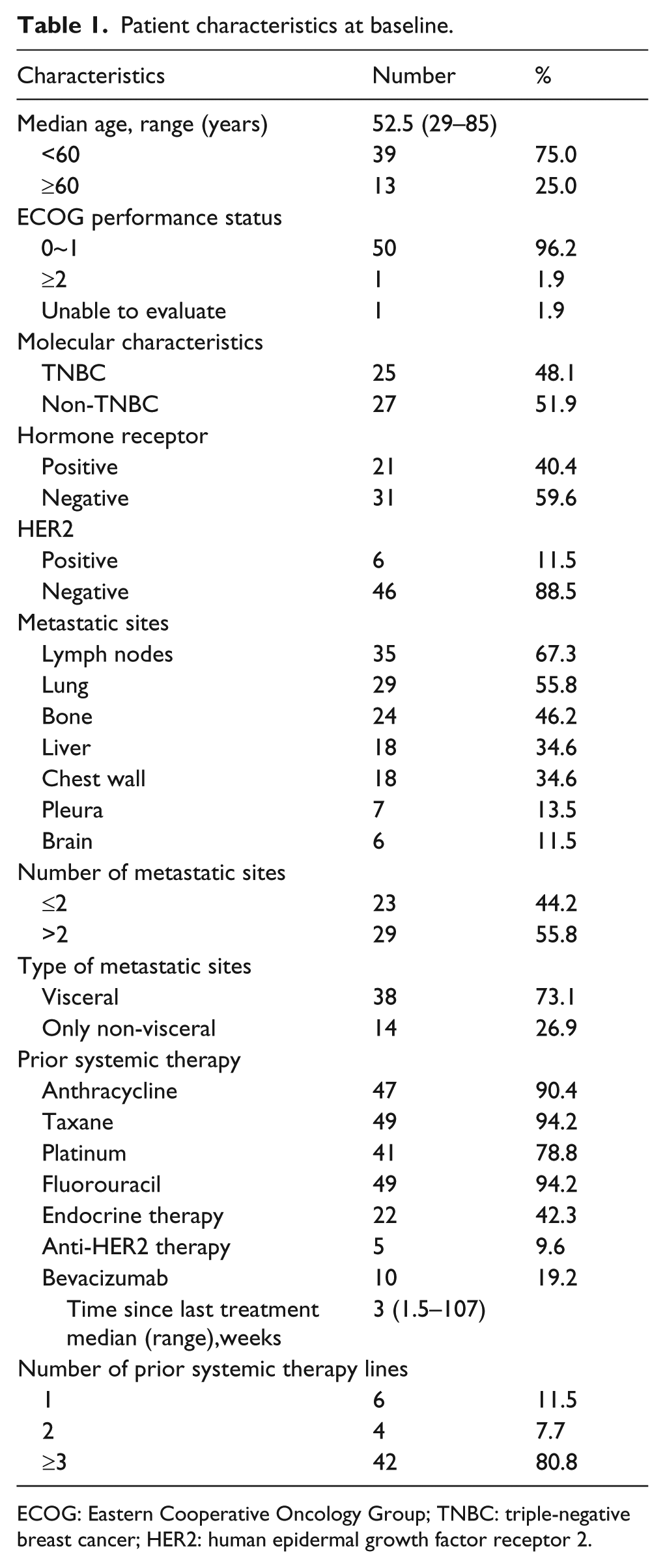
ECOG: Eastern Cooperative Oncology Group; TNBC: triple-negative breast cancer; HER2: human epidermal growth factor receptor 2.
Treatment administration
Profile of treatment administration and dose modification is shown in Table 2. Most patients initiated at the dose of 500 mg per day, which had never been increased. Two started at the dose of 750 mg per day but both experienced dose reduction to 500 mg per day and maintained that dosage. Only one managed to escalate to and stayed at the dose of 750 mg per day, with mild toxicities. Overall, 26/52 (50%) patients experienced interruption of treatment. Most common causes for treatment interruption were hypertension (12/26, 46.2%), proteinuria (7/26, 26.9%), palmar–plantar erythrodysesthesia syndrome (PPES; 4/26, 15.4%), and thrombocytopenia (3/26, 11.5%). In total, 19 (36.5%) underwent dose reduction once, with no one having a second reduction, and 8 (15.4%) discontinued apatinib treatment due to intolerable toxicities. Two patients with HER2-enriched BC underwent combined treatment of apatinib with trastruzumab. Inter-group analysis of patients in different age groups showed that discontinuation of treatment due to toxicities was more common in patients of 60 years or older than that in younger group, although the difference was not significant (p = 0.076).
Treatment administration.
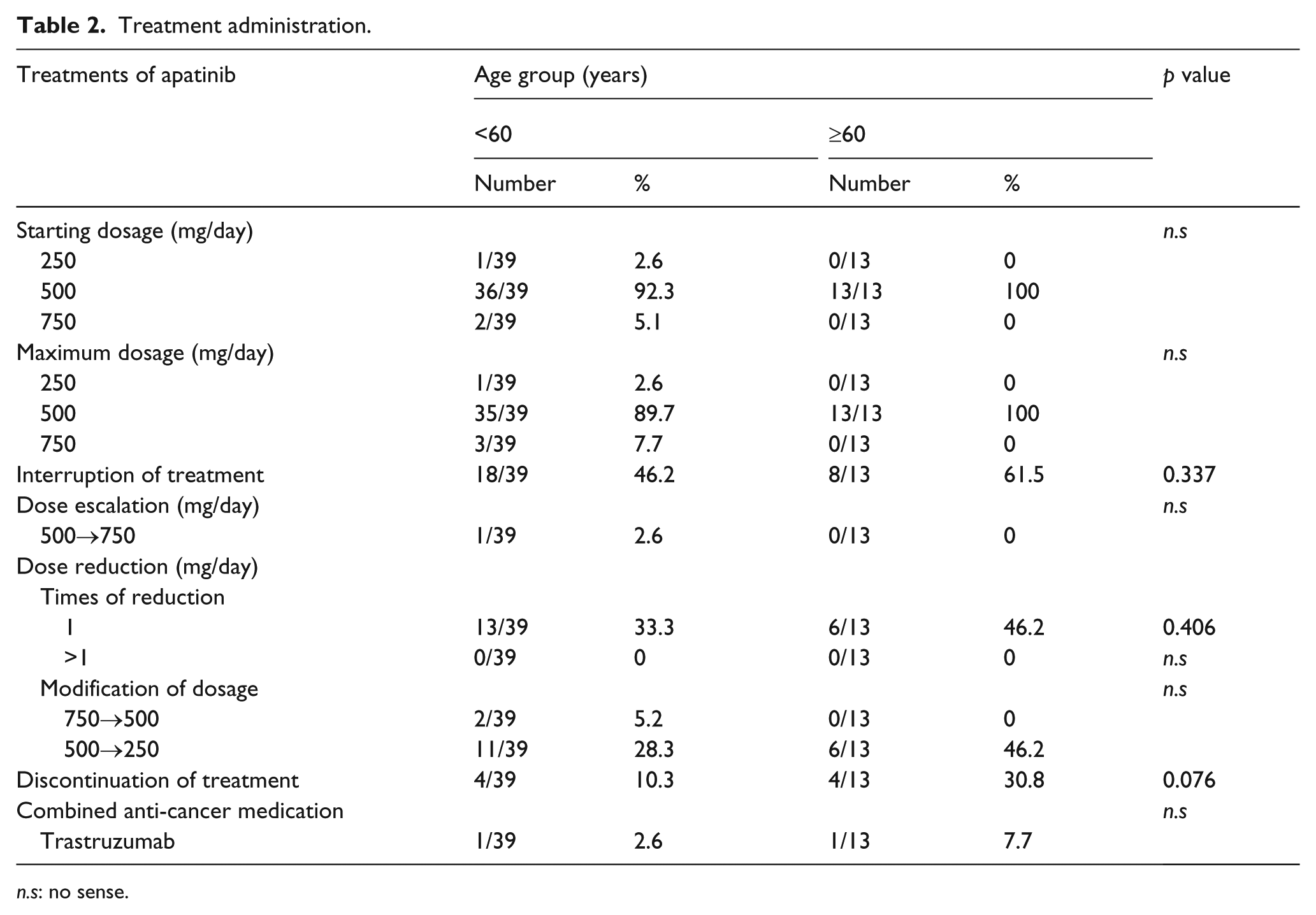
n.s: no sense.
Efficacy
Overall, 8 of the 52 patients experienced treatment discontinuation because of intolerable toxicities, 7 of whom had too short medication time for efficacy assessment. Therefore, altogether 45 patients were enrolled in the analyses of PFS, OS, and clinical responses, while all 52 were included in TTF analysis. The median PFS (mPFS), median OS (mOS), and median TTF (mTTF) were 4.90 (95% confidence interval (CI): 3.44–6.36), 10.3 (unable to calculate 95% CI), and 3.93 (95% CI: 1.96–5.90) months, respectively (Figure 1(a)–(c)). The best clinical responses are listed in Table 3, which revealed that 10 (22.2%) and 21 (46.7%) patients had a best response of partial response (PR) and stable disease (SD), respectively, but complete response (CR) could not be achieved in all the patients (Table 3).

Kaplan–Meier curves of progression-free survival (PFS), overall survival (OS), and time-to-treatment failure (TTF) for patients receiving apatinib treatment. (a) Kaplan–Meier curve of PFS, which indicated the median PFS of 4.90 (95% CI: 3.44–6.36) months. (b) Kaplan–Meier curve of OS, which suggested the median OS of 10.3 (unable to calculate 95% CI) months. (c) Kaplan–Meier curve of TTF, which showed the median TTF of 3.93 (95% CI: 1.96–5.90) months.
Best clinical response.

CR: complete response; PR: partial response; SD: stable disease; PD: progression disease.
Inter-group analysis found no significant differences in PFS and OS between patients previously treated with and without bevacizumab (Figure 2(a) and (b) and Table 4; p = 0.413 and p = 0.736, respectively). Multivariate Cox analysis of factors associated with survival showed that previous treatment of bevacizumab was neither associated with PFS nor OS (Table 4). The relations between other characteristics and survivals were also explored to identify any predictive biomarker (Table 4). Previous treatment of anthracycline was significantly correlated with prolonged PFS and was an independent predictor but was not correlated with OS (Figure 2(c) and (d) and Table 4). In addition, although no correlations between age or PPES and survival were found in log-rank analysis, older age group and PPES were independent predictors for prolonged PFS in Cox multivariate analysis (p = 0.023 and p = 0.020; Table 4).

Log-rank analysis of predictive factors for survival. Kaplan–Meier curves of PFS and OS for patients (a and b) with or without bevacizumab treatment and (c and d) those with or without anthracycline treatment. p values by log-rank test are displayed.
Log-rank and Cox multivariate analysis of factors associated with prognosis.
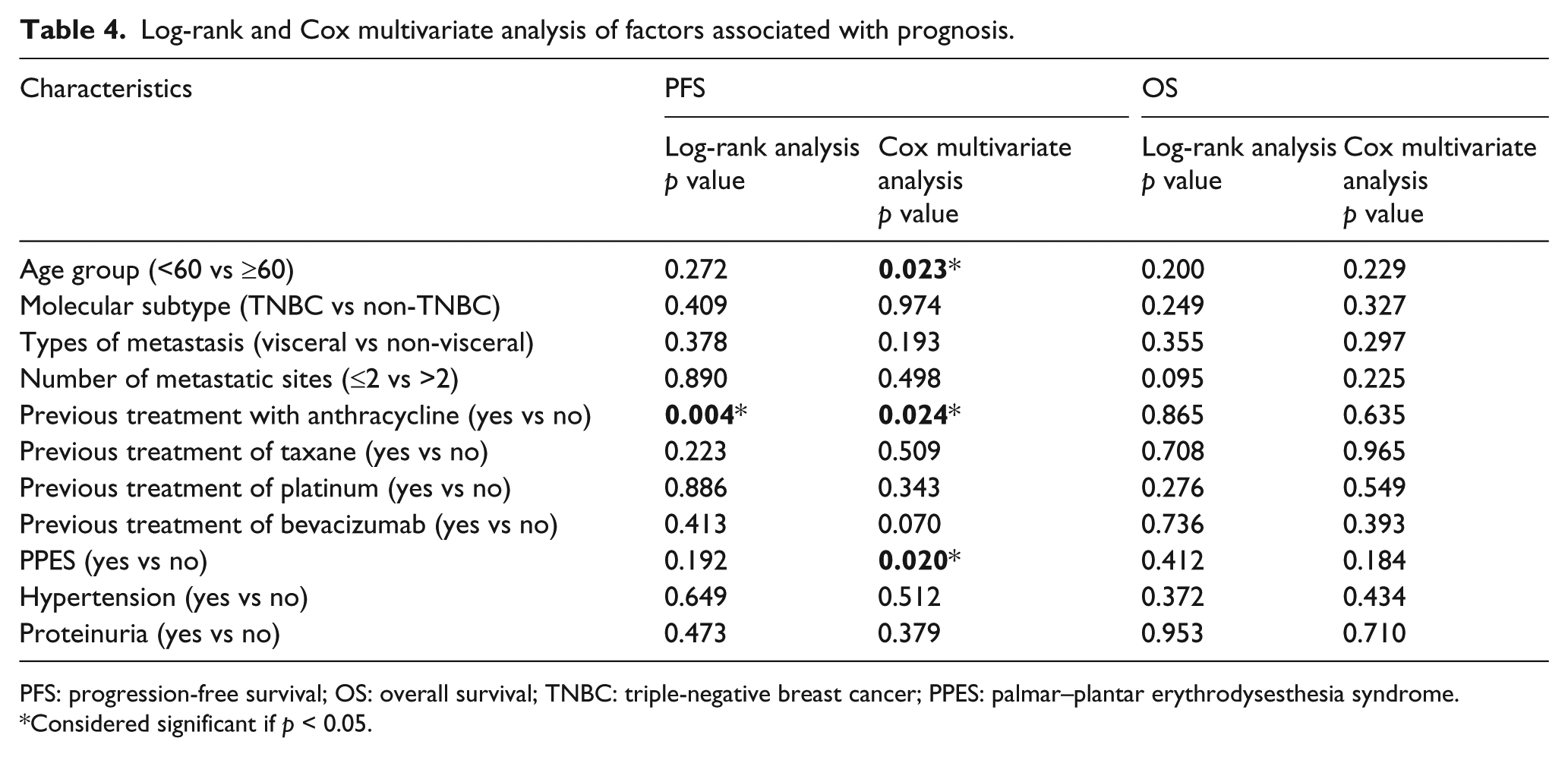
PFS: progression-free survival; OS: overall survival; TNBC: triple-negative breast cancer; PPES: palmar–plantar erythrodysesthesia syndrome.
Considered significant if p < 0.05.
Safety
All 52 patients were included in the toxicity analysis. Toxicities were generally well tolerated or could be managed by symptomatic treatments, as was shown in Table 5. The most common non-hematologic AEs were hypertension, proteinuria, PPES, and liver dysfunction, while hematologic AEs, including thrombocytopenia, leukopenia, and neutropenia, also occurred at high rates. Notably, severe dyspnea of grade 3~4, which has not been documented before, was seen and caused treatment discontinuation in two patients, one from age group of less than 60 and one from age group of 60 years or older.
Adverse events.

AE: adverse event; PPES: palmar–plantar erythrodysesthesia syndrome.
Discussion
This study reported the efficacy and safety of apatinib on pretreated MBC patients in non-clinical trial setting, and for the first time to our knowledge, explored the impact of previous anti-angiogenic treatment of bevacizumab to apatinib. In addition, we also reported a grade 3~4 AE of dyspnea, which has not been documented in previous studies.
Apatinib is a highly potent and highly selective small molecular TKI targeting VEGFR-2 that is previously known as YN968D1, 9 which can reverse multi-drug resistance of cancer cells by inhibiting the function of multiple adenosine triphosphate (ATP)-binding cassette transporters, apart from anti-angiogenesis. 26 Although the anti-tumor efficacy of apatinib has been demonstrated in several types of cancers including BC,4–6,19–23 doubts of its efficacy and safety still exist and the impact of previous anti-angiogenic treatment remains unknown.24,27
The results of survival of our observational study are basically consistent with those reported by clinical trials, with the mPFS and mOS of 4.90 and 10.3 months, respectively. Sunitinib and sorafenib monotherapy on MBC patients showed the mPFS of 2.5–3.6 months.2,15–18 The mPFS of apatinib is numerically longer than those of sunitinib and sorafenib, suggesting a more promising therapeutic effect, despite the fact that direct numerical comparison may not be appropriate. In our study, 95% CI of OS could not be calculated, mainly because the number of patients who reached end-point events till the cutoff date was not enough. A longer follow-up time for the cohort would help to collect information for the end-point events and calculate the 95% CI of OS.
Further exploration showed that there was no difference in survival between patients with and without previous bevacizumab treatment. However, few studies have ever explored the efficacy of a second anti-angiogenic agent in patients with MBC. Only Schwartzberg et al. 28 conducted the clinical trial to date in our knowledge to explore the benefits of sorafenib plus chemotherapy in MBC patients who have failed bevacizumab. Results showed a significant improvement in PFS in sorafenib plus chemotherapy group than in placebo plus chemotherapy group (3.4 vs 2.7 months), but the difference in OS was not significant. 28 Besides, results of other studies also provided hints of a potential benefit of a second anti-angiogenic agent. A phase I trial by Mayer et al. 29 investigating the efficacy of tivozanib in MBC showed that no patients achieved response of CR and four patients achieved response of PR. Notably, among the four patients with PR, three were previously exposed to bevacizumab. 29 Yardley et al. 30 explored the benefits of adding ramcirumab to eribulin in MBC patients, and subgroup analysis showed that previous exposure of bevacizumab more than 6 weeks before study did not result in significant difference in PFS. These studies suggested the benefits of second anti-angiogenic therapy. Considering the results of this study and the above reports, we suggest that previous anti-angiogenic treatment is not a contraindication of apatinib treatment. However, due to lack of evidence, we do not know how much time is required for wash out before start of apatinib as second anti-angiogenic treatment.
We also found that previous exposure to anthracycline was an independent predictor of longer PFS but not OS. Similar phenomenon could not be observed in patients treated with other cytotoxic drugs such as taxane and platinum. It has been demonstrated in previous study that cytotoxic drugs, such as anthracycline and taxane, contribute to enhancing tumor vascularization in BC. 31 It suggests that tumors exposed to these drugs may be more dependent on angiogenic pathway than others, thus responding better to anti-angiogenic therapy. The reason why only anthracycline but not taxane and platinum affected the efficacy of apatinib remains to be further studied. We should also keep in mind that due to the small sample size of our study, the correlation between previous exposure to anthracycline and PFS might be found by chance, and the negative results of correlation between anthracycline and OS might be caused by the small sample size of the study and loss to follow-up in majority of patients without previous exposure to anthracycline. Study in a larger cohort is needed to further confirm our findings.
Toxicity profile was basically similar to that reported in clinical trials.5,6,19–21 Although most of the toxicities were well tolerated, many patients still experienced interruption, dose reduction, and discontinuation of treatment. Moreover, we call your attention to the AE of severe dyspnea. Although mild dyspnea has been reported in clinical trials, but no events of grade 3 or 4 has been recorded. Clinicians should pay more attention to this side effect, especially in patients with pulmonary or cardiac combinations. Moreover, discontinuation of treatment was more common in patients of 60 years or older than that in younger group, suggesting a lower tolerability of apatinib in this age group. Given that we suggest a more frequent monitoring of AEs and a more aggressive symptomatic treatment for patients of advanced age.
Encouragingly, one patient managed to escalate to 750 mg per day, indicating that dosage and toxicity profile may vary among patients. It has been recently found in a study that pharmacokinetic features of apatinib vary greatly among patients with different types of cancers. 32 Patients with GC exhibited remarkably lower bioavailability than those with other cancers including BC, which might be probably attributed to the fact that gastric surgery has affected apatinib absorption by stomach. 32 Therefore, previous stomach diseases and gastric surgeries should be taken into account while deciding dosage for patients. Interestingly, our patient who tolerated the dose of 750 mg per day did not have a previous history of stomach disease or surgery. Whether there were other factors affecting her tolerance or pharmacokinetic features of apatinib remains to be studied. More pharmacological studies of apatinib should be carried out on patients with different medical conditions, so as to optimize the dosing schedule to achieve maximal benefits and least side effects. Given that better response was correlated with higher drug concentration, 6 physicians can try to increase dosage in patients with mild toxicities and in good performance status as appropriated.
Conclusion
The encouraging efficacy and safety of apatinib on pretreated MBC patients in non-clinical trial setting have been demonstrated in our study. For the first time in our knowledge, we found that previous treatment of bevacizumab did not affect the efficacy of apatinib, and reported a grade 3~4 AE of severe dyspnea, which should be monitored to ensure safety. Our study is a single-center retrospective study with small sample size. More multi-center randomized controlled double-blind clinical trials focusing on apatinib monotherapy or apatinib-based combined therapy are urgently needed, so as to pursue more beneficial treatments for heavily pretreated MBC patients and even for those at earlier disease stage.
Footnotes
Acknowledgements
Y.L. and Z.W. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by FUSCC Ethics Committee and was performed in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards and applicable local regulatory requirements and laws. All patients were informed of the off-label use of apatinib and gave their verbal and/or written consents before treatment in accordance with clinical practice guidelines of FUSCC.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the grant from Shanghai Municipal Commission of Health and Family Planning (No. 201640069) and Shanghai Natural Science Foundation (No. 17ZR1405700).
