Abstract
NUCKS (nuclear, casein kinase, and cyclin-dependent kinase substrate) is implicated in the tumorigenesis of several human malignancies, but its role in ovarian cancer remains unknown. We aim to investigate NUCKS expression and its clinical significance in ovarian cancer. The messenger RNA expression of NUCKS was determined in normal and malignant ovarian tissues using quantitative polymerase chain reaction assay. Immunohistochemistry was applied to detect the status of NUCKS protein expression in 121 ovarian cancer tissues. NUCKS protein high expression was detected in 52 (43.0%) of 121 patients. NUCKS messenger RNA expression was gradually upregulated in non-metastatic ovarian cancers (
Introduction
Ovarian cancer (OC) is the most lethal gynecologic malignancy. 1 Due to a lacking of methods for early detection, nearly 70% of patients present with advanced stage at diagnosis, with a result of poor prognosis. Despite advances in surgery and new chemotherapy regimens during the last few decades, the reported 5-year survival rate for patients with OC was still 25%–30% or even less.2,3 Identification of new prognostic factors to select patients with good or bad outcome might help improve treatment. However, clinicopathological indicators alone do not enable clinicians to make precise prognoses for individual OC patients. The advent of molecularly targeted therapies, however, could dramatically alter clinical outcome. Thus, novel prognostic and predictive markers are needed to better assess patient outcome and guide individual therapies.
The NUCKS (nuclear, casein kinase, and cyclin-dependent kinase substrate) gene, a vertebrate specific gene, is located on human chromosome 1q32.1 and is a highly phosphorylated protein present in a number of mammalian cell types and tissues.4–6 NUCKS1 is a chromatin-associated protein with a role in the DNA damage response and in homologous recombination (HR), a DNA repair pathway critical for tumor suppression. 7 And it was proved to involve in cell-cycle control and DNA repair with regard to the identification of CK2 and PIKK kinases as kinases phosphorylating NUCKS in vivo. 8 Furthermore, NUCKS is implicated in tumorigenesis, proliferation, invasion, and prognosis of many human malignancies.9–17 Our previous study had reported that NUCKS overexpressed in endometrial cancer and played a role in predicting prognosis and recurrence. 18 However, up to date, there have been no published reports evaluating its expression in OC tissues and the clinical significance in OC patients.
This study was performed to investigate the expression of NUCKS in OC and to analyze its relationship to various clinicopathological features, including patient outcome.
Materials and methods
Samples and patients
In the current retrospective study, 121 patients with OC who were surgically treated in the Department of Gynecologic Oncology (Harbin Medical University Cancer Hospital, Harbin, China) from January 2008 to December 2009 were enrolled. Fresh tissues from 10 normal ovaries, 20 non-metastatic OCs, 20 metastatic OCs, and their matched metastatic lesions (
All the paraffin-embedded tissues and fresh tissues were taken with the consent from each patient. This study was approved by the Medical Ethics Committee of Harbin Medical University. Clinicopathological features of the OC cohort are outlined in Table 1. Histological typing was carried out according to World Health Organization (WHO) classification standards. 19 The grading and staging of tumors were assigned according to the Federation of Gynecology and Obstetrics (FIGO) rules. 20 The status of residual disease was divided into R0 (no residual tumor nodule), R1 (the largest residual tumor nodule measuring 1 cm or less), and R2 (the largest residual tumor nodule measuring more than 1 cm). This definition is the most widely accepted among gynecologic oncologists.
Relationships of NUCKS protein expression status and clinicopathological variables in OC.
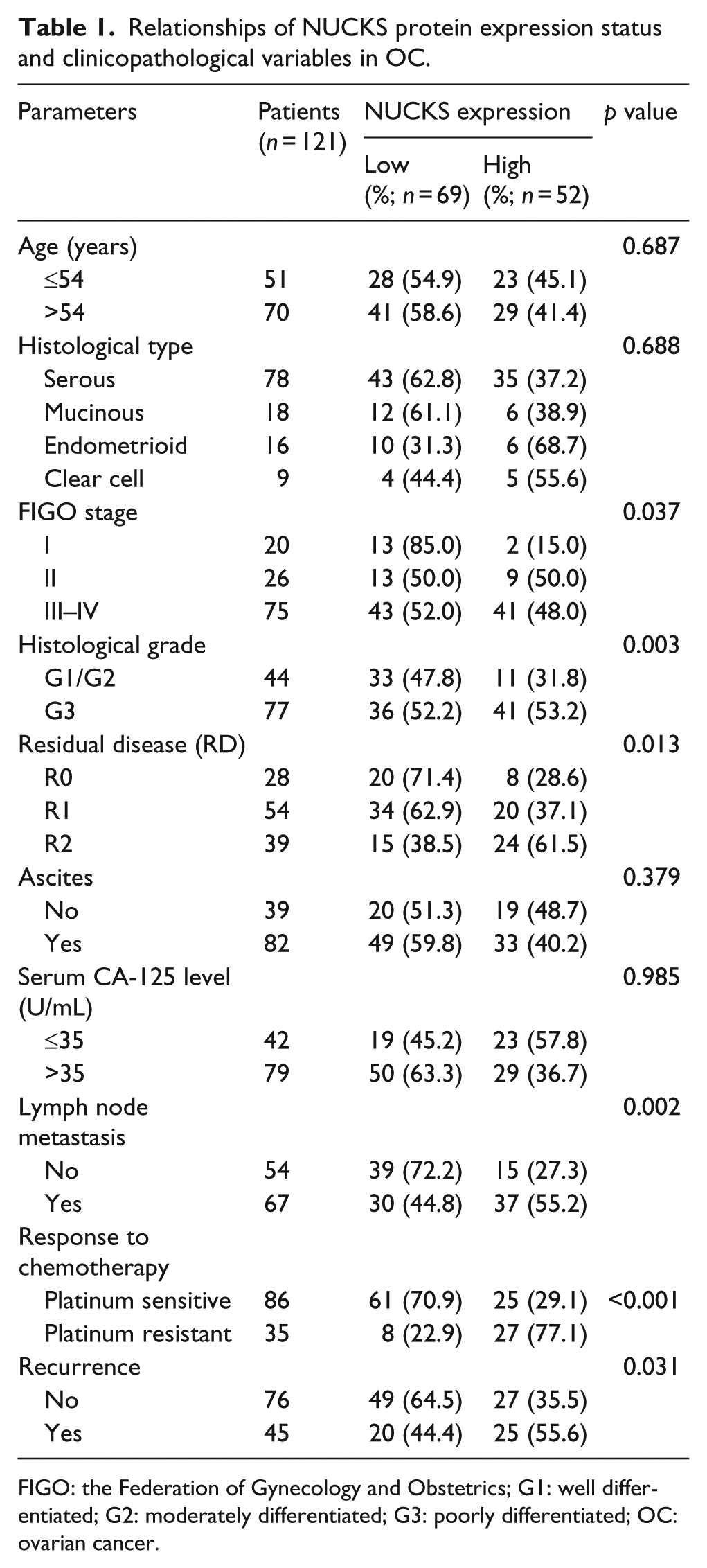
FIGO: the Federation of Gynecology and Obstetrics; G1: well differentiated; G2: moderately differentiated; G3: poorly differentiated; OC: ovarian cancer.
All the patients were followed up on regular basis and the last follow-up was carried out in December 2015, with the median observation period of 44 months (2–92 months) and 79 cancer-related deaths.
Immunohistochemical staining and evaluation
Immunohistochemistry staining was carried out using the Two-Step IHC Detection Reagent Kit (Zhong Shan Golden Bridge Biological Technology Inc., Beijing, China) following the manufacturer’s protocols. Experimental steps had been described in our previous study 18 with a primary antibody specific for NUCKS (dilution 1:700; Abcam, Cambridge, MA, USA). Two independent pathologists blinded to the clinical variables conducted the immunoreactivity score for NUCKS expression. When there was disagreement, a senior pathologist was reached until a consensus. Immunoreactivity was scored by counting the percentage of positive cells as follows: 18 0, fewer than 10% of positive nuclei of epithelial cells were detected; 1, 11%–30% were detected at low staining intensity; 2, more than 30% but less than 60% were detected with moderate staining intensity; and 3, more than 60% of positive nuclei were detected with high staining intensity. For statistical analysis, the score of NUCKS expression was defined as low expression if the overall score was 0–1 and high expression if the sum was 2–3.
RNA extraction, reverse transcription, and qPCR
Total RNA from fresh tissues was extracted using the TRIzol reagent (Invitrogen, Carlsbad, CA) according to the manufacturer’s instruction. About 2 µg RNA from each sample was used for complementary DNA (cDNA) synthesis with 2× Power Taq PCR MasterMix kit (PR1702; BioTeke, Beijing, China). qPCR was employed to quantify the fold change of NUCKS messenger RNA (mRNA) level by Exicycler 96 real-time reverse transcription polymerase chain reaction (RT-PCR) system (Bioneer, Daejeon, Korea) with SYBR Green Mastermix (SY1020; Solarbio, Beijing, China). Expression data were normalized to the geometric mean by β-actin. The qPCR primers for NUCKS were listed as follows: forward, 5′-TCTGATGATGCAGATGAAGATTA-3′; reverse, 5′-CTGCTGAGTGAGAATCATCC-3′. The primers for β-actin were also listed as follows: forward, 5′-CTTAGTTGCGTTACACCCTTTCTTG-3′; reverse, 5′-CTGTCACCTTCACCGTTCCAGTTT-3′. All genes were tested in triplicate.
Statistical analyses
All statistical analyses were carried out with SPSS software package (Chicago, IL, USA, 17.0). For continuous variables, student’s t-test was performed. The chi-square test and Fisher’s test were used to analyze the differences in categorical variables. Survival curves were plotted by the Kaplan–Meier method and compared by the log-rank test. Multivariate survival analysis was carried out for all the variables that were significant in the univariate analysis using the Cox regression model. A two-sided probability value of
Results
Elevated expression of NUCKS mRNA in OC tissues
To investigate the potential role of NUCKS in the tumorigenesis, qPCR was carried out in normal ovarian tissues (

(a) Comparison of NUCKS mRNA relative expression levels in normal ovary (N = 10), non-metastatic ovarian cancer (OC; nT = 20), metastatic OC (mT = 20), and its matched metastatic lesion (M = 20). (b) Increased trend of NUCKS mRNA expression in metastatic OC and its matched metastatic lesion.
NUCKS expression and its association with the clinicopathological features of OC
NUCKS was localized in the nuclei of tumor cells (Figure 2). In a few slides, weak or moderate NUCKS expression was observed in cytoplasm of tumor cells accompanied by nuclear staining of tumor cells. In the same specimen, nuclear staining intensity in tumor cells is higher than cytoplasm. Of the OC specimens examined, low nuclear NUCKS expression was detected in 69 (57.0%) of 121 patients, whereas high nuclear NUCKS expression was detected in 73 (43.0%) of 121 patients.

The representative images for NUCKS protein expression. (a) Low expression in ovarian cancer (OC). (b) High expression in OC. (c) Low expression in non-metastatic OC. (d) High expression in metastatic OC.
Significant correlation was observed between NUCKS expression level and several prognostic risk factors such as FIGO stage (
Survival analysis for the prognosis of patients with OC
To assess NUCKS as a predictor of survival, the Kaplan–Meier analysis method was used to investigate the correlation between NUCKS expression and overall survival (OS)/disease-free survival (DFS). The log-rank test showed that the 5-year OS and DFS rates were 18.8% and 15.2%, respectively, in patients with a high level of NUCKS expression, compared with the 53.4% and 49.4% in their counterparts (
Univariate survival analysis of OS and DFS in 121 patients with OC.

FIGO: the Federation of Gynecology and Obstetrics; G1: well differentiated; G2: moderately differentiated; G3: poorly differentiated; OS: overall survival; DFS: disease-free survival; OC: ovarian cancer; SE: standard error.

Subgroup analyses for the overall survival (OS) and disease-free survival (DFS) of ovarian cancer (OC) patients with high and low NUCKS expression: (a) OS in all OCs, (b) DFS in all OCs, (c) OS in early-stage OCs, (d) DFS in early-stage OCs, (e) OS in advanced-stage OCs, and (f) DFS in advanced-stage OCs.
Multivariate Cox regression analysis showed that the high NUCKS expression was considered as an independent prognostic marker for both OS and DFS of patients with OC (
Multivariate survival analysis of OS and DFS in 121 patients with OC.

FIGO: the Federation of Gynecology and Obstetrics; OS: overall survival; DFS: disease-free survival; OC: ovarian cancer; HR: hazard ratio; CI: confidence interval.
Discussion
In this study, we showed, for the first time, that the expression of NUCKS was gradually upregulated in non-metastatic OCs, metastatic OCs, and its matched metastatic lesions at mRNA level in comparison with that in normal ovarian tissues. High expression level of NUCKS was significantly associated with FIGO stage, histological grade, residual disease, lymph node metastasis, response to chemotherapy, and recurrence. High expression level of NUCKS served as independent prognostic factors for short OS and DFS rates in patients with OC. Moreover, a stage-stratified analysis showed that NUCKS overexpression correlated with DFS in early-stage OC, OS and DFS in advanced-stage OC. In the light of these findings, we demonstrated that NUCKS has the potential to be a novel predictor of progression as well as survival in OC patients.
Increased NUCKS expression has been reported in several cancers,11–18,21 including OC. 9 However, the dynamic and complex phosphorylation of NUCKS implies that NUCKS has the possibility to interact with several binding partners and hence being involved in multiple biological functions. NUCKS overexpression was observed in the majority of invasive carcinomas, including atypical ductal hyperplasia, ductal carcinoma in situ, and invasive ductal cancer.11,22 Also, it was associated with high-invasive phenotype in mouse lung adenocarcinoma cell strains. 10 Furthermore, increased NUCKS expression was associated with high-invasive factors in cervical cancer 13 and was highly upregulated in colon cancer patients with distant metastasis. 12 A similar observation was also made in gastric cancer tissue. In addition to that NUCKS correlated with high-invasive factors such as FIGO stage, histological grade, residual disease, and lymph node metastasis in OC, we interestingly found that NUCKS mRNA expression gradually increased in tissues as follows: normal ovary, non-metastatic OC, metastatic OC, and its matched metastatic lesion. Furthermore, elevated NUCKS protein expression indicated a suboptimal cytoreduction in OCs with larger residual disease. All these may indicated a significant role of this gene in the neoplastic progression.
NUCKS appears to be important for cell-cycle progression 23 and is involved in facilitating and maintaining the transcription activity of some genes during rapid proliferation. 9 NUCKS as well as p53 is phosphorylated on multiple sites in proliferating cells in vivo.4,24,25 Zduniak et al. 15 emphasized that NUCKS abundance in malignant skin tumors is higher than that of the well-known proliferation marker Ki67, thus suggesting that NUCKS protein as a novel proliferation marker for skin tumor specimens. In gastric cancers, NUCKS expression was associated with Ki67 and the combination of these two makers might further distinguish a subgroup of patients with poor prognosis. 14 Additionally, there is evidence that the NUCKS might be involved in facilitating and maintaining the activity of transcription of some genes.9,21 However, what is the exact underlying mechanism of the interaction of NUCKS with Ki67 remains to be clarified.
To further characterize the clinically prognostic implication of NUCKS in OC, we used stage-stratified analysis to evaluate the prognostic value of NUCKS protein expression. The data suggest that NUCKS provide a prognostic potential in OC especially in advanced OC. NUCKS overexpression was associated with recurrence-related survival in cervical cancer, endometrial cancer, colon cancer, and gastric cancer. Yet, its overexpression served as a predictor of overall survival only in endometrial cancer and gastric cancer in addition to advanced OC.
In our study, we also reported that NUCKS overexpression was associated with response to platinum-based chemotherapy. In accordance with this finding, Shen et al. 17 showed that miR-137 sensitizes chemosensitivity to paclitaxel and cisplatin in lung cancer through inhibiting PI3K/AKT pathways via targeting NUCKS1. The levels of NUCKS correlated with the levels of Bax and activated caspase-3, which are indicative of apoptosis. Moreover, NUCKS was upregulated very early during neuronal apoptosis in vitro. 26 These suggest that NUCKS plays complex roles in cell homeostasis.
Taken together, we believe that this study provides the first clinical evidence that NUCKS is an independent prognostic marker for OS and DFS in OC patients. Our findings suggest that NUCKS is highly associated with progression, response to chemotherapy, and prognosis; is a new biomarker for this disease; and is a potential therapeutic target.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the grants of National Natural Science Foundation of China (81502225), the Postdoctoral Foundation of China (2015M581480), the Postdoctoral Foundation of Heilongjiang Province of China (LBH-Z15123), the Haiyan Foundation of Harbin Medical University Cancer Hospital (JJMS2016-01), the Innovation Foundation for Talents of Harbin Medical University (2016lczx77), and the Youth Elite Training Foundation of Harbin Medical University Cancer Hospital (JY2016-03).
