Abstract
DJ-1 is a novel oncogene that can transform NIH3T3 cells in cooperation with the activated
Preoperative serum DJ-1 levels were measured in 300 lung cancer patients and compared with benign pulmonary disease (n = 44) and healthy volunteers (n = 64). Using tissue microarrays and immunohistochemical analyses, we compared the DJ-1 expression between the primary squamous cell carcinoma tumours and matched metastatic tissues from a lymph node.
The baseline preoperative serum DJ-1 of lung cancer patients was significantly higher than that of benign diseases and healthy controls (p < 0.001). In the early-stage subgroup, the median DJ-1 concentration (ng/mL) was significantly higher than that of the advanced stage (12.90 vs 7.75, p < 0.05). Using immunohistochemistry, we observed that the DJ-1 staining intensity was generally weaker and less common in the metastatic tissues compared with that in the primary tumour (McNemar–Bowker Test, p = 0.008).
DJ-1 was highly expressed in the early stage of lung cancer, and its expression was significantly decreased after metastasis. Therefore, DJ-1 may be a potential biomarker for the early diagnosis and monitoring of lung cancer metastasis.
Introduction
Lung cancer is the leading cause of cancer death in China. In 2015, an estimated 7,333,000 new cases of lung cancer will be diagnosed and 6,102,000 patients will die from it. 1 At present, the 5-year relative survival rate was 54.8% for early-stage lung cancer and 4.2% for advanced-stage cancer. 2 Therefore, early diagnosis and aggressive treatment are essential for improving patient survival.
At present, many tumour markers, such as cytokeratin fragment antigen 21-1 (Cyfra21-1), neuron-specific enzyme (NSE), carcino-embryonic antigen (CEA), glycoprotein 125 (CA125) and squamous cell carcinoma antigen (SCC-Ag), have been used for detecting and monitoring lung cancer. However, due to the low diagnostic value in identifying early lung cancer, they are rarely used for lung cancer screening. Therefore, many studies have been performed to improve the diagnostic performance of biomarkers or find new biomarkers for lung cancer diagnosis.
In our previous study, we compared the expression of free proteins released from lung cancer and adjacent normal bronchial epithelial cells, and we found that DJ-1 was a highly expressed protein in the tumour cell samples.
3
DJ-1 was first reported as an oncogene that can transform NIH3T3 cells in cooperation with the activated
In this study, we aimed to investigate the role of DJ-1 protein in lung cancer diagnosis and compared its diagnostic value with other biomarkers. We measured the serum concentration of DJ-1 in lung cancer patients, benign disease patients and healthy controls. We also assessed DJ-1 expression in primary and metastatic cancer tissues.
Materials and methods
Patient cohort
Patients were recruited at the Department of Thoracic Surgery, Cancer Institute and Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College (CAMS, Beijing, China). All patients underwent surgery and had a pathological diagnosis. The histological classification was made according to the World Health Organization (WHO)/International Association for the Study of Lung Cancer (IASLC) classification criteria for lung cancer. The stage was made according to the seventh edition of the tumour–node–metastasis (TNM) staging classification for lung cancer.
Serum from lung cancer patients, benign pulmonary disease patients and healthy controls were collected for enzyme-linked immunosorbent assay (ELISA) analysis. All serum samples were collected between February 2007 and February 2008 and before the anti-tumour therapy. The diagnosis of benign pulmonary disease was established by tissue biopsy or pathological diagnosis after surgery. The healthy controls were recruited from the health check individuals at the Department of Cancer Prevention of CAMS. The selection criteria were as follows: (1) no individual history of cancer, (2) no lung disease and (3) frequency matching to cases by sex and age. After collection, the venous blood was placed in test tubes without anticoagulants, and the serum was separated by centrifugation at 4000 r/min for 10 min and stored at −70°C until analysis. Repeated thawing and freezing of the samples were avoided.
The tumour tissues were collected between October 2000 and October 2002. The inclusion criteria were as follows: (1) the pathological diagnosis was primary squamous cell carcinoma of the lung, (2) the patient underwent radical resection of the tumour and corresponding regional lymph node dissection and (3) no anti-cancer therapy was received before the surgery. For immunohistochemical analyses, formalin-fixed, paraffin-embedded tissue specimens derived from 68 patients were obtained as tissue microarrays (TMAs). The tissue samples consisted of the primary tumour tissues and matched metastatic tissues from lymph nodes.
This study was approved by the Chinese Academy of Medical Sciences Cancer Hospital Ethnic Committee (NCC2013-028). All participants provided their written informed consent to participate in this study.
Quantitative ELISA for DJ-1 protein
ELISA was performed using 96-well microtiter plates. All incubations were performed at room temperature and followed by washing seven times with 0.01 PBS with 0.05% Tween-20 (PBST, pH 7.4) unless indicated otherwise.
Microtiter plates were coated with 50 µL of mouse anti-DJ-1 IgG (D2943; Sigma, St. Louis, MO, USA) diluted in coating buffer (0.05 M Na2CO3/NaHCO3, pH 9.6, diluted 1:1000). After overnight incubation at 4°C, the wells were washed three times. To each well, 300 µL of 2% bovine serum albumin (BSA) in PBST was added to block the non-specific binding sites and then samples were incubated for 4 h. Afterwards, the wells were washed three times. DJ-1 standard protein (diluted with PBS) or serum (50 µL) was added and incubated for 1 h, and samples were washed afterwards.
During the following detection, antibodies were diluted in PBST containing 1% BSA (antibody dilution). Plates were treated with 50 µL of rabbit anti-mouse IgG (P9373, diluted 1:2000; Sigma, St. Louis, MO, USA) and incubated for 1 h. After washing, horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (ZB2301, diluted 1:8000; Sigma, St. Louis, MO, USA) solution was added and incubated for 0.5 h. The enzyme activity of HRP was assessed by the addition of 3,3′,5,5′-tetramethylbenzidine (TMB) substrate (Cell Signaling Technology, Danvers, MA, USA). After 30 min, the reaction was stopped with the addition of 100 µL of 2 M H2SO4 per well. The optical density (OD) was measured at 450/570 nm using a microtiter plate reader (Bio-Rad 680, Hercules, CA).
In general, we detected the serum DJ-1 concentrations of 300 lung cancer patients, including 236 cases detected once and 64 cases randomly selected for a repeated detection. The coefficient of variation was 3.7% within the plate and 8.4% between different plates.
Quantitative analysis for biomarkers
The serum levels of NSE, CEA, CA125 and Cyfra21-1 were determined on a Cobas e411 analyser with Elecsys kits (Roche Diagnostics Corp., Indianapolis, IN, USA). The assays utilize the electrochemiluminescence immunoassay (ECLIA) method, and the unit of measurement is units per millilitre (U/mL). The serum level of SCC-Ag was determined using an ARCHITECT I2000SR immune analyser system (Abbott, Santa Clara, CA, USA). The assay utilizes the chemiluminescent microparticle immunoassay (CMIA; Abbott Japan Co., Ltd., Tokyo, Japan). Each test included a standard control (coefficient of variation (CV) < 5%).
Immunohistochemical staining and scoring
In all, 5-µm-thick sections of each TMA were immunohistochemically stained according to the streptavidin/peroxidase standard protocols. After dewaxing, rehydration and antigen retrieval, the sections were immunostained with rabbit anti-human polyclonal DJ-1 antibody (1:200, HPA004190; Sigma, St. Louis, MO, USA) overnight at 4°C. Slides were incubated with biotinylated secondary antibody (rabbit anti-goat IgG) for 30 min and then treated by an SA/HRP kit (ZSGB-Bio, Beijing, China) for 30 min at room temperature; they were subsequently developed with diaminobenzidine (DAB) for visualization.
The TMAs were read by a pathologist blinded to the identity of the tissue sections and clinicopathological parameters of the patients. The staining intensity in any given tissue section received a grade of 0, 1, 2 or 3. A higher grade indicates a higher intensity of staining.
Statistical analysis
The statistical analysis was performed with SPSS software (version 16.0), GraphPad prism 4 and MedCalc. Nonparametric methods were performed to analyse the difference in the serum DJ-1 concentration among the various groups. Mann–Whitney tests were used for comparisons between two groups, and Kruskal–Wallis tests were used for multiple groups. Receiver operating characteristic (ROC) curves were used to estimate the diagnostic accuracy of tumour markers. MedCalc was used to compare the differences among the area under the curve (AUC) of the ROC. Statistical differences in the levels of DJ-1 in paired tumour and metastasis tissues were analysed with the McNemar–Bowker Test. Statistically significant differences were defined as comparisons resulting in p
Results
Patient characteristics
Overall, 300 lung cancer patients, 44 benign lung disease patients and 64 healthy controls were enrolled in this study. No significant difference was observed in the sex or age ratio among the three groups. In the case group, the histological types of the tumours were as follows: 151 adenocarcinomas, 104 squamous cell carcinomas, 31 small-cell lung cancer (SCLC) tumours and 14 uncommon pathological types. The demographics and clinical characteristics are shown in Table 1.
Demographics and clinical characteristics of study population.

SCLC: small-cell lung cancer.
The diagnostic value of DJ-1 protein for lung cancer
The comparison of serum DJ-1 concentrations in each studied groups is shown in Table 2. In the case group, the median concentration of DJ-1 was 9.31 ng/mL (range: 0.00–172.00 ng/mL), which is significantly higher than that of the benign disease group (median = 4.67 ng/mL, range: 0.00–54.13 ng/mL, p < 0.001) and healthy control group (median = 4.38 ng/mL, range: 0.00–43.23 ng/mL, p < 0.001). Using the benign disease patients as the negative group and lung cancer patients as the positive group, we drew ROC curves to compare the diagnostic value of DJ-1 protein with other tumour markers (Figure 1). As shown in Table 3, six markers had a similar performance in lung cancer diagnosis (AUC (95% confidence interval (CI)): 0.645 (0.568–0.722) for DJ-1, 0.620 (0.539–0.702) for NSE, 0.640 (0.552–0.728) for CA125, 0.729 (0.663–0.795) for CEA, 0.751 (0.679–0.822) for Cyfra21-1, and 0.618(0.533–0.703) for SCC-Ag).
The mean level of DJ-1 for lung cancer, benign diseases and healthy controls.

ADC: adenocarcinoma; SCC: squamous cell carcinoma; SCLC: small-cell lung cancer.

ROC curves to assess the value of serum DJ-1, NSE, CEA, CA125, Cyfra211 and SCC-Ag levels in lung cancer patients compared to benign lung disease controls.
The area under the ROC curves.
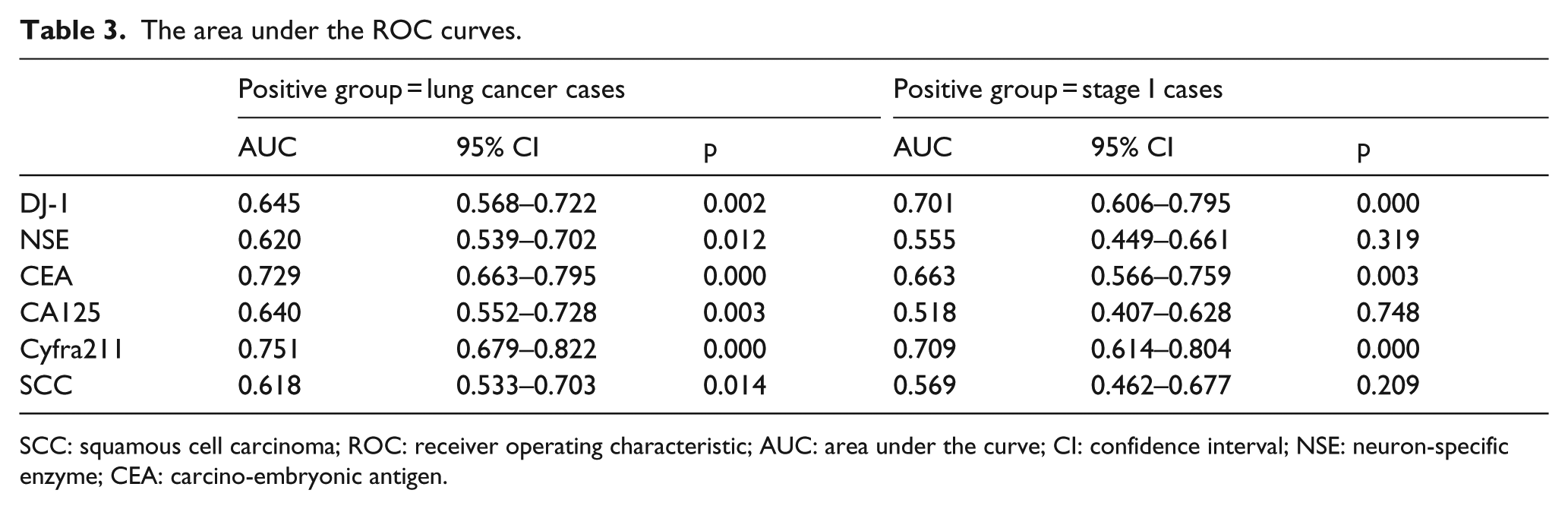
SCC: squamous cell carcinoma; ROC: receiver operating characteristic; AUC: area under the curve; CI: confidence interval; NSE: neuron-specific enzyme; CEA: carcino-embryonic antigen.
Serum DJ-1 concentrations with the clinical stages
The case group was divided into two subgroups according to the clinical stage: early stage (stage I) and advanced stage (stages II–IV). In the early-stage subgroup, the median DJ-1 concentration was 12.90 ng/mL, which is significantly higher than that of the advanced stage (median = 7.75 ng/mL, p < 0.05).
We further compared the diagnostic value of tumour markers to identify early-stage lung cancers using the stage I lung cancer as the positive group (Figure 2). DJ-1 showed better performance (AUC: 0.701 (0.606–0.795)) than using the lung cancer population as the positive group (AUC: 0.645 (0.568–0.722)), while others had worse or similar performance (Table 3). The comparison of ROC curves showed that DJ-1 performed significantly better in identifying early-stage lung cancers compared with NSE and CA125.

ROC curves to assess the value of serum DJ-1, NSE, CEA, CA125, Cyfra211 and SCC-Ag levels in early-stage lung cancer patients compared to benign lung disease controls.
Serum DJ-1 concentrations with tumour metastasis
In addition, based on the metastasis status (lymph node and distant metastasis status), subgroup analyses were performed to study the possible relationship between the serum DJ-1 concentration and metastasis status. The case group was divided into the M0 and M+ subgroups according to the distant metastasis status when diagnosed. The median DJ-1 concentration (mU/mL) was 10.00 in the M0 subgroup, which is significantly higher than that of the M+ subgroup (median = 7.07, p = 0.020, Table 2). According to the lymph node metastasis status, the case group was divided into the N0 and N+ subgroups. The median DJ-1 level (mU/mL) was 10.76 in the N0 subgroup and 8.63 in the N+ subgroup. However, no significant difference was found between the two subgroups (p > 0.05, Table 2).
DJ-1 expression in primary tumour tissues and matched metastatic tissues from lymph nodes
A summary of DJ-1 expression in primary and metastatic tissues is given in Table 4. DJ-1 expression was detected in 98.5% of the primary tissues (67/68; score 1 (50%; 34/68), score 2 (29.4%; 20/68) and score 3 (19.1%; 13/68)), while it was detected in 83.8% of the metastatic tissues (57/68; score 1 (63.4%; 43/68), score 2 (11.8%; 8/68) and score 3 (8.8%; 6/68)). Compared with the DJ-1 staining intensity in the primary tumour, the intensity was generally weaker and sparser in the metastatic tissues (Figure 3). A significant difference was demonstrated between the primary and matched metastatic tissues (McNemar–Bowker Test, p = 0.008).
The expression of DJ-1 in the primary tumour tissues and the matched metastatic tissues from lymph node.


Immunohistochemistry analysis of DJ-1 protein in primary and metastatic tissues. Higher expression of DJ-1 protein was detected in primary specimens of lung cancer (a) compared to the matched lymph node metastases (b). Immunohistochemistry confirmed that DJ-1 protein was down-regulated after lymph node metastasis.
Discussion
In this study, we detected the serum concentration of DJ-1 in the lung cancer and control groups and compared its diagnostic value with other tumour markers. We also compared the DJ-1 expression between the primary SCC tumours and matched metastatic tissues from lymph nodes. Our results are summarized as follows. First, DJ-1 could identify the lung cancers and benign diseases and yields a better performance in the diagnosis of early-stage cancer compared with NSE and CA125. Second, the DJ-1 expression has a negative relationship with the lung cancer metastasis status. The patients with distant metastases have lower DJ-1 concentrations. Third, high DJ-1 expression was detected in most primary tumours, while down-regulation or loss of expression was found in the matched metastatic tissues.
The concentration of DJ-1 protein in stage I lung cancers was significantly higher than for advanced stages. When we compared the diagnostic value of lung cancer among DJ-1 and the other biomarkers, all biomarkers could distinguish between lung cancer and benign patients with the similar accuracies. The AUC of the ROC curve ranged from 0.618 to 0.751 (Table 3). Using the benign patients as the negative control group with the cases or early-stage cancer patients as the positive group, the diagnostic performance of the biomarker was compared. Each biomarker, except DJ-1, performed worse for early-stage cancers than for the cases (Table 3). In this sense, DJ-1 may be a better biomarker for differentiating between benign and early-stage lung cancer.
The expression of DJ-1 was negatively correlated with lung cancer metastasis. This conclusion was supported at both the blood and tissue levels. The serum concentrations of DJ-1 were significantly lower in patients with distant metastases than in those with focal lesions. The DJ-1 immunohistochemistry staining intensity in the primary tissues was significantly higher than in the matched metastatic tissues. Therefore, we speculated that the downregulation of the DJ-1 gene might occur during metastasis. In this sense, DJ-1 protein may be a potential biomarker for monitoring metastasis.
Based on the above findings, we supposed that DJ-1 may play an important role in the early stage of lung cancer, and its function is gradually weakened in the course of tumour progression. After metastasis, DJ-1 was no longer the driving force, while other mutations and signalling pathways became dominant, which is consistent with previous findings. Earlier studies have demonstrated that DJ-1 may have the greatest impact in tumourigenesis, before other genetic aberrations tend to dominate in the transformed cells. After the tumourigenesis, its biological function tends to decrease. 15
However, our findings conflict with the results of a previous study. 25 Fan et al. observed that non-small-cell lung cancer (NSCLC) patients with later stage cancer have a significantly higher level of serum DJ-1 than those with early-stage cancer. Considering differences in the study populations between the two studies may affect the results, we removed the SCLC cases and recalculated our findings, but the results did not significantly change (data not shown). Accordingly, a likely explanation for the difference in the results is sampling error caused by the relatively small sample size. Hence, multi-centre studies are needed to gain a more accurate conclusion.
Conclusion
DJ-1 was highly expressed in the early stage of lung cancer, and its expression was significantly decreased after metastasis. Therefore, DJ-1 may be a potential biomarker for early diagnosis and monitoring of lung cancer metastasis. However, multicentre and molecular biology studies are needed to verify its clinical application and to determine its molecular mechanism.
Footnotes
Acknowledgements
B.H. and J.W. have contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CAMS Innovation Fund for Medical Sciences (CIFMS; grant no. 2016-I2M-1-001).
