Abstract
New reliable approaches to stratify patients with endometrial cancer into risk categories are highly needed. We have recently demonstrated that DJ-1 is overexpressed in endometrial cancer, showing significantly higher levels both in serum and tissue of patients with high-risk endometrial cancer compared with low-risk endometrial cancer. In this experimental study, we further extended our observation, evaluating the role of DJ-1 as an accurate serum biomarker for high-risk endometrial cancer. A total of 101 endometrial cancer patients and 44 healthy subjects were prospectively recruited. DJ-1 serum levels were evaluated comparing cases and controls and, among endometrial cancer patients, between high- and low-risk patients. The results demonstrate that DJ-1 levels are significantly higher in cases versus controls and in high- versus low-risk patients. The receiver operating characteristic curve analysis shows that DJ-1 has a very good diagnostic accuracy in discriminating endometrial cancer patients versus controls and an excellent accuracy in distinguishing, among endometrial cancer patients, low- from high-risk cases. DJ-1 sensitivity and specificity are the highest when high- and low-risk patients are compared, reaching the value of 95% and 99%, respectively. Moreover, DJ-1 serum levels seem to be correlated with worsening of the endometrial cancer grade and histotype, making it a reliable tool in the preoperative decision-making process.
Keywords
Introduction
Worldwide, the incidence of endometrial cancer (EC) is steadily increasing, making it the most frequent gynecological tumor, particularly in developed countries; it is the fourth most common cancer in women, and the seventh leading cause of cancer-related mortality.1–3
The majority of women presented with low-risk disease, that is, early stage, low grade (G1), endometrioid histology, and a good prognosis. However, a growth in incidence is expected to lead to an increased number of high-risk cases at the time of diagnosis. High-risk ECs include a group of tumors characterized by high grade (G3), advanced stage, non-endometrioid histology, such as serous EC (SEC) or clear cell EC (CCEC), deep myometrial invasion (MI), and lymphovascular space invasion (LVSI). Moreover, high-risk ECs are related to a worse prognosis because they are more clinically and biologically aggressive. 4 Therefore, an extensive surgical staging has a crucial role in the management of affected patients.
Currently, information about grade and histology are derived in most cases from endometrial biopsies performed before surgery. However, significant grade and histological changes between the preoperative biopsy and the definitive surgical specimen have been observed. Particularly, a disagreement of classification has been reported in one third of high-risk ECs.5–7 When this happens, a surgical decision based on the preoperative diagnosis could be inappropriate, leading to a worse outcome. 6 In medical institutions with sufficient gynecologic pathology expertise, frozen section (FS) seems to be highly reliable to provide guidance for the intraoperative decisions of surgical staging. 8 However, in many centers worldwide, the use of FS is still limited. 9
Moreover, both preoperative and intraoperative assessment do not meet one of the major issues regarding EC management, that is, they do not provide any information about molecular differences between tumors. 10
The most comprehensive molecular study has been provided from the The Cancer Genome Atlas (TCGA) that allows to classify EC in four groups with respect to specific mutations (Polymerase (POLE) ultramutation, microsatellite instability (MSI) hypermutation, low copy number, and high copy number) related to clinical outcome. 11 Today, the molecular characterization of EC subtypes seems to have a pivotal role, in guiding and refining EC treatment.
We have recently demonstrated, by means of proteomic approach, that DJ-1 is differentially expressed in EC low- and high-risk sample tissues; the different tissue expression is also reflected at serum level, suggesting its possible role as a clinical biomarker. 12 DJ-1, at first identified as Parkinson’s disease–associated protein 7 (PARK7), is a 20-kDa glycase belonging to the peptidase C56 family. It seems to play an important role in cancer pathogenesis and progression, 13 and an overexpression has been reported in several cancers such as melanoma,14,15 pancreatic, 16 breast 17 and, more recently, in EC. 18 In this experimental study, we evaluated the performance of preoperative serum levels of DJ-1 as predictors of high-risk EC.
Materials and methods
A total of 101 consecutive women with a newly diagnosed EC who were treated at the Unit of Gynaecologic Oncology, Magna Graecia University, Germaneto, and Pugliese-Ciaccio Hospital, Catanzaro, Italy, between June 2013 and December 2015 were enrolled in this prospective observational study. Procedures carried out in this study were in accordance with the guidelines of the Helsinki Declaration on human experimentation and good clinical practice (CGP). Approval by the institutional review board was obtained before starting patient’s enrollment.
Furthermore, a written consent was obtained from all patients before processing their data from the time of hospitalization, even if data did not include any personal identifying information.
All patients were scheduled for a hysterectomy, bilateral salpingo-oophorectomy (BSO), and peritoneal cytology. A pelvic lymphadenectomy, para-aortic lymphadenectomy, and omentectomy were performed when indicated. Preoperative serum samples were collected on the day preceding the operation.
Exclusion criteria were as follows: Previous or concurrent cancer located in other sites, major comorbidities (such as patients with previous heart failure, renal disease, etc.), genetic susceptibility to gynecologic or non-gynecologic cancers (BRCA1-2 carriers, associated polyposis conditions (APC), Fanconi syndrome), neoadjuvant chemotherapy or hormonal treatment before the hysterectomy, neurodegenerative diseases, discordance in grade and/or histology between endometrial biopsy, and definitive surgical pathology.
To simplify the classification, we have considered high-risk group, patients with non-endometrioid or high-grade endometrioid EC (EEC), and low-risk group, patients with well and moderately differentiated EEC. Patients were subgrouped based on definitive surgical pathology.
The control group consisted of 44 healthy women coming from the blood donors department. Clinical information was obtained by routine laboratory analysis, history, and physical examination. To each control, a gynecological ultrasound, to exclude any uterine or ovarian pathology, and a cancer antigen 125 (CA125) and human epididymis protein 4 (HE4) determination were offered.
DJ-1 serum levels were evaluated in cases and controls. Within the EC patients, DJ-1 levels were compared in low- versus high-risk group and in G1 or moderate-grade (G2) EEC versus EEC-G3 and non-EEC.
Among the low-risk group, we further extended the analysis by evaluating DJ-1 levels in patients with MI < 50% (really low risk) with those of patients with MI ≥ 50% (intermediate risk). Moreover, we evaluated DJ-1 serum levels in all patients subgrouped according to the stage (early (I/II) vs advantages (III/IV) stages).
Analytical methods
Blood samples were collected into Vacutainer tubes by venipuncture after the subjects have been fasting for at least 12 h. The drawn blood was allowed to coagulate at room temperature and centrifuged at 3000g for 10 min. The sera were divided into 300 µL of several aliquots and stored at −80°C until analyzed. Only one thawing was performed per sample in order to avoid protein degradation.
Determination of DJ-1 serum levels
The concentration of serum DJ-1 was analyzed using specific immunoassays (CircuLex Human DJ-1/PARK7 ELISA Kit; CycLex Co., Ltd. Japan). Enzyme-linked immunosorbent assay (ELISA) was performed on duplicate samples according to the manufacturer’s instructions.
Determination of HE4 and CA125 serum levels
Serum levels of CA125 and HE4 were measured using electro-chemiluminescence immunoassay (ECLIA) on a Cobas e601 apparatus (Roche Diagnostics, Switzerland). All assays were performed according to the manufacturer’s processing instructions.
Statistical analysis
Data were analyzed statistically using R software. 19 DJ-1 protein levels were compared using the non-parametric Kruskal–Wallis test to identify statistical differences between groups. p ≤ 0.05 was considered to be significant. The receiver operating characteristic (ROC) was generated for each marker using serum samples. The area under the curve (AUC) values and 99% confidence intervals (CIs) were calculated to determine the specificity and sensitivity. ROC curves were generated using ROCR package. 20 The t test was used to compare continuous variables.
Results
DJ-1, CA125, and HE4 serum levels in EC patients
Of 101 enrolled patients, 56 were categorized as low-risk EC (22 EEC-G1 and 34 EEC-G2) and 45 as high-risk EC patients (25 EEC-G3, 20 G3 non-EEC of which 16 were SEC and 4 were CCEC). The clinicopathological features (age, body mass index (BMI), American Society of Anesthesiologists (ASA) score, comorbidities, histotype, grading, myometrial infiltration, LVSI, and International Federation of Gynecology and Obstetrics (FIGO) stage) and surgical and medical data are provided in Table 1. The control group consisted of 44 healthy blood donors. The two groups did not significantly differ in age and in BMI (Table 1).
Clinical, pathological, and surgical characteristics of patients with endometrial cancer and clinical data of healthy subjects.

EC: endometrial cancer; BMI: body mass index; ASA: American Society of Anesthesiologists; FIGO: International Federation of Gynecology and Obstetrics; EEC: endometrioid EC; G1: low grade; G2: moderate grade; G3: high grade; SEC: serous endometrial carcinoma; CCEC: clear cell endometrial cancer; LVSI: lymphovascular space invasion; SD: standard deviation; BSO: bilateral salpingo-oophorectomy.
Data are expressed as mean±SD, number, and percentage. In extensive surgery, peritoneal cytology, hysterectomy, BSO, omentectomy, peritoneal biopsies, and pelvic and para-aortic lymphadenectomy were performed. LVSI was not always described in the pathologic response. It was available for 85 cases.
As shown by the box plot in Figure 1(a), the median serum levels of DJ-1 in patients with EC was 730 pg/mL, significantly higher than found in control subjects (224 pg/mL; p ≤ 0.05). Moreover, DJ-1 levels were significantly higher in high-risk EC versus low-risk EC (1073 vs 444 pg/mL; p ≤ 0.05; Supplementary Figure 1).

(a, c, and e) Box–whisker plots of DJ-1, CA125, and HE4 in serum of 44 healthy subjects and 101 endometrial cancer patients. (b, d, and f) Box–whisker plots of DJ-1, CA125, and HE4 in serum of healthy subjects and patients with endometrial cancer subtypes (EEC-G1: 22 patients), (EEC-G2: 34 patients), (EEC-G3: 25 patients), (SEC-CCEC: 20 patients). Each box and whisker plot illustrates the median, quartiles and extreme values (.). The Y axis is a logarithmic scale (p ≤ 0.05; Kruskal–Wallis test).
In order to evaluate whether DJ-1 serum expression changes according to the grading and histology, we further analyzed serum DJ-1 levels in EEC-G1, EEC-G2, EEC-G3, and in non-EEC (Figure 1(b)).
In the low-risk EEC-G1, the median serum levels of DJ-1 were similar to those found in the control group (289 vs 224 pg/mL). Conversely, DJ-1 levels were significantly higher in the EEC-G2 (586 pg/mL), EEC-G3 (1015 pg/mL), and in the non-EEC (SEC/CCEC) patients (1395 pg/mL) compared to the EEC-G1 group and to the healthy subjects (p ≤ 0.05).
The difference of DJ-1 serum levels remains statistically significant when we compared EEC-G3 and SEC/CCEC versus EEC-G2 (p ≤ 0.05). Finally, DJ-1 levels were not significantly different between EEC-G3 and SEC/CCEC. Among the low-risk group, DJ-1 serum levels were significantly higher in patients with MI ≥ 50% compared to patients with MI < 50% (350 ± 76 vs 602 ± 86 pg/mL; p ≤ 0.05).
By evaluating changes of DJ-1 according to the increase of the surgical stage, we found that serum levels were significantly higher in the advanced stage (III/IV) than in early stage (I/II; 1148 ± 371 vs 873 ± 350 pg/mL; p ≤ 0.05).
As expected, CA125 levels were significantly higher in EC patients compared with healthy subjects (36 vs 13 UI/L; p ≤ 0.05; Figure 1(c)). The difference in CA125 levels remains statistically significant for each EC subtype (EEC-G1 = 36 UI/L, EEC-G2 = 24 UI/L, EEC-G3 = 35 UI/L, and SEC/CCEC = 34 UI/L) compared to controls (p ≤ 0.05), while no significant differences were found within each EC subtype (Figure 1(d)).
Analogously, HE4 levels were significantly different in EC patients versus healthy controls (77 vs 49 pmol/mL; p ≤ 0.05; Figure 1(e)) as well as when each EC subtype (EEC-G1 = 77 pmol/mL, EEC-G2 = 73 pmol/mL, EEC-G3=92pmol/mL, and SEC/CCEC=79 pmol/mL) was compared to controls (p ≤ 0.05); on the contrary, statistical significance is again lost when comparing the single subtypes among them (Figure 1(f)). The DJ-1, CA125, and HE4 median values and interquartile ranges are provided in Supplementary Table 1.
Interestingly, although sample size and follow-up time are limited, patients who recurred or died have higher DJ-1 serum levels compared to individuals with the same EC grade and histotype (data not shown).
DJ-1, CA125, and HE4 diagnostic performances
The diagnostic accuracy of DJ-1, CA125, and HE4 was evaluated in the total EC cohort compared to controls by ROC curve analysis. All three markers displayed a very good diagnostic accuracy in discriminating EC patients and controls, being the AUC of DJ-1 the highest (0.89), followed by that of HE4 (0.80) and by that of CA125 (0.71; Figure 2(a)). Among the three, DJ-1 still shows a better diagnostic accuracy in discriminating EEC-G2 (AUC 0.91), EEC-G3 (AUC 1), and SEC/CCEC (AUC 1) from controls with the unique exception of EEC-G1 versus healthy subjects (AUC 0.58). For CA125, AUC values were 0.71 in EEC-G1, 0.67 in EEC-G2, 0.76 in EEC-G3, and 0.69 in SEC/CCEC; for HE4, AUC values were 0.79 in EEC-G1, 0.74 in EEC-G2, 0.84 in EEC-G3, and 0.83 in SEC/CCEC (Figure 2(b–e)).

Receiver operating characteristic (ROC) curve analysis using DJ-1, CA125, and HE4 as serum marker. (a) EC patients versus healthy subjects. (b) EEC-G1 versus healthy subjects. (c) EEC-G2 versus healthy subjects. (d) EEC-G3 versus healthy subjects. (e) SEC/CCEC versus healthy subjects.
The cutoff values, sensitivity, specificity, and positive and negative predictive values, based on ROC analysis, are provided in Table 2.
Diagnostic performance of DJ-1, CA125, and HE4.

CA125: cancer antigen 125; HE4: human epididymis protein 4; EC: endometrial cancer; EEC: endometrioid EC; G1: low grade; G2: moderate grade; G3: high grade; SEC: serous endometrial carcinoma; CCEC: clear cell endometrial cancer; NPV: negative predicted value; PPV: positive predicted value; AUC: area under the curve.
Finally, DJ-1, CA125, and HE4 were analyzed by subdividing the patient’s cohort according to the EC classification mainly used in the clinical practice, that is, low- versus high-risk patients. Results of the ROC curve analysis are summarized in Table 3. From this analysis, it appears that DJ-1 has an excellent accuracy to distinguish between the low-risk EC (EEC-G1/G2) and the EEC-G3 (AUC 0.986), while a poor diagnostic accuracy was found for CA125 (AUC 0.553) and HE4 (AUC 0.596). Noteworthy, for a cutoff value of 751 pg/mL, DJ-1 shows a sensitivity of 95% and a specificity of 91%. The comparison of the low-risk EC (EEC-G1/G2) with the non-endometrioid high-risk EC (SEC/CCEC) gives an AUC value of 0.993, with a sensitivity of 95% and a specificity of 99% at cutoff value of 910 pg/mL. DJ-1 has an excellent accuracy (AUC 0.990) in discriminating the low- (EEC-G1/G2) from all the high-risk ECs (EEC-G3 and SEC/CCEC) maintaining a sensitivity of 98% and a specificity of 93%, at cutoff value of 772 pg/mL. Conversely, both CA125 and HE4 failed in discriminating low- and high-risk EC (AUC 0.530 and 0.596, respectively; Supplementary Figure 2).
Diagnostic performance of preoperative serum DJ-1 levels for discriminating low- and high-risk EC.
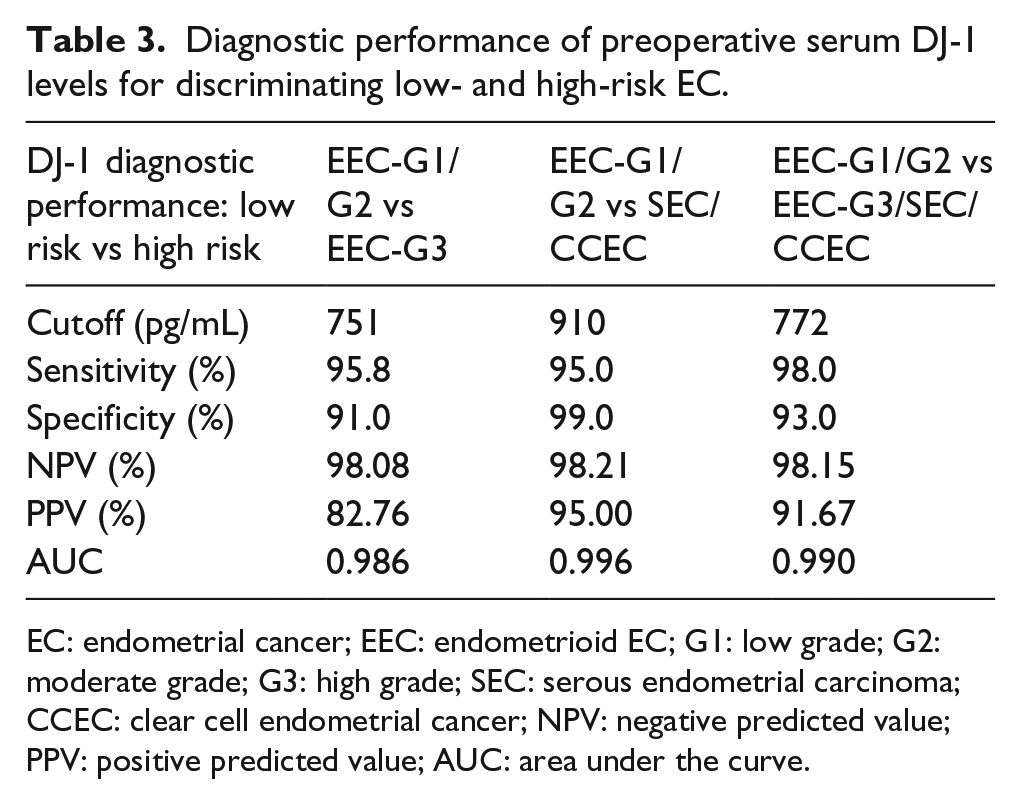
EC: endometrial cancer; EEC: endometrioid EC; G1: low grade; G2: moderate grade; G3: high grade; SEC: serous endometrial carcinoma; CCEC: clear cell endometrial cancer; NPV: negative predicted value; PPV: positive predicted value; AUC: area under the curve.
Discussion
The accurate preoperative characterization of EC as well as the appropriate management of the affected patients still remains a clinical challenge; therefore, the identification of new, specific, and reliable biomarkers to discriminate between high- and low-risk patients, or even better, between low-, intermediate-, and high-risk patients is eagerly needed.
Currently, the potentially useful serum biomarkers available for EC diagnosis and management are CA125 and HE4. The former is only useful as a marker of spreading of the disease already outside the uterus, 21 while for the latter there are growing data in the literature about its potential utilization.22–24
We have recently identified DJ-1, by a proteomic approach, in a group of proteins differentially expressed in EEC and SEC tissues when compared to healthy controls and suggested its possible role as biomarker to improve differential diagnosis between EC subtypes. 12 DJ-1 is a multifunctional, ubiquitous, 20-kDa protein mainly involved in the protection of the cell from oxidative damage. 13 Along with this function, DJ-1 affects several processes such as cell migration and adhesion, chemotaxis, proliferation, and apoptosis. 13 DJ-1 altered expression has been reported in breast, 17 lung, 25 ovarian,26,27 esophageal, 28 and pancreatic cancer tissues. 16 At least in some of these tumors, the increased expression of DJ-1 in neoplastic cells is also reflected at the serum level.29,30
In our first experimental study, we have shown that DJ-1 tissue expression was significantly higher in SEC compared with EEC-G1 and EEC-G2. Interestingly, we observed that more than one third of EEC-G3 had a DJ-1 expression more similar to SEC compared to that of the correspondent endometrioid histotype. 12 Moreover, we found that tissue overexpression of DJ-1 was also reflected at serum level using sera of 10 patients with EEC, 5 patients with SEC, and 20 healthy women by Western Blot assay. 12
To validate the potential role of DJ-1 as a serum biomarker, in this prospective observational study, we have determined DJ-1 levels in the sera of a large cohort of patients with EC; the analysis was performed on the total cohort of affected patients, composed of 101 individuals, as well as by subdividing the patients on the basis of the EC grading and histotype. Even though these are preliminary data and a prospective multi-centric study is still ongoing for clinical validation, present results indicate that DJ-1 might be confidently proposed as new and accurate serum biomarker for EC.
Moreover, since its expression seems to be correlated with the worsening of the EC grade and histotype, DJ-1 may also be considered as a marker for redefining EC risk classification according to the molecular subtype specific profile.
A reclassification of risk based on the molecular features is strongly needed for EC subgroups, such as the high-grade (G3) EEC, that have never been clearly classified according to dualistic model of EC.31–34
Over the last 3 years, few groups11,35–38 have investigated the prognostic value of different molecular alterations involved in endometrial carcinogenesis, demonstrating that the genomic features of ECs allow for a more reproducible and accurate risk reclassification, which may help in choosing the most appropriate surgical and adjuvant treatment for women with aggressive tumors.
The Atlas group showed that not only SEC but almost 25% of EEC-G3 have extensive copy number alterations, few DNA methylation changes, low estrogen receptor/progesterone receptor (ER/PR) levels, and frequent TP53 mutations that are hallmarks of a more aggressive biological profile. In these cases, a more extensive surgical staging, including pelvic and para-aortic lymphadenectomy and, perhaps, omentectomy, should be performed. 11
Furthermore, among patients defined for histology and graded as low-risk EC patients (those that might benefit from a non-surgical treatment or a conservative surgery without lymphadenectomy or ovarian preservation), there is a subset showing, instead, biological behavior and clinical prognosis more similar to high-risk tumors. 33
Even though the next-generation sequencing (NGS) technology has significantly lowered the costs, molecular and genetic testing is unlikely to be widely applied in the clinical practice. Therefore, the need for reliable and cost-saving alternative approaches to stratify patients into risk categories is quite high.
Today, some medical institutions propose the use of FS on uterus because the concordance rate of different variables between FS and definitive surgical pathology in EC has been reported as high with respect to histological subtype, grade, MI, and tumor size. However, data are institution-dependent and limited to centers with high gynecologic pathology expertise. 9
In most institutions, FS are used to evaluate MI, in cases in which the preoperative MI is inconclusive in order to decide whether a lymphadenectomy should be performed on pelvic lymph nodes, and if they are removed, to guide the decision about para-aortic lymphadenectomy.
From this point of view, a biomarker differentially expressed in different EC subtypes both in serum (as confirmed by this study) and in tissue (as we have shown by immunohistochemistry (IHC) in previous study), 12 is, of course, more user-friendly and cost-saving tool compared to genomic testing and FS assessment.
The DJ-1 specificity, sensitivity, and accuracy are comparable to those of HE4 or even slightly better. Both these markers show a diagnostic efficiency significantly greater than that of CA125. Even more interesting is the comparison of DJ-1 with HE4/CA125 in the population study when subdivided for grading and histotype. DJ-1 fails in the identification of the G1 patients, where HE4 appears to be the most suitable in this regard. The discriminating ability in distinguishing among healthy and affected individuals of HE4 is not paralleled, on the other hand, by an equal ability of this marker in discriminating among endometrial cancers different histotypes and grades. DJ-1, instead, has an excellent accuracy in categorizing the low- and high-risk EC patients, making this finding extremely relevant for clinicians at the time of surgery.
The EEC-G3 and the non-EEC, also identified as high-risk EEC, are biologically more aggressive than the low-risk EEC-G1/G2 tumors, thus requiring a different and substantially more extensive surgical treatment. 6 Since a discrepancy has been described between preoperative and definitive histological diagnosis in at least one of three of cases, DJ-1 determination appears even more significant clinically.
Recently, Shu et al. 18 have demonstrated an increased expression of DJ-1 in EC tissues, without a significant correlation with the pathological type or tumor stage. The presence of increased serum levels of DJ-1 found in EC patients most probably reflects an enhanced intracellular expression of this gene, according to our previous results. 12 In this study, we demonstrate that serum DJ-1 amounts increase significantly in parallel with the worsening of the neoplastic grade and histotype. At the moment, we do not have a clear explanation for the discrepancy between Shu’s findings and data reported in this study; we suggest that the different sensitivity of the techniques used for DJ-1 detection (protein measurement by ELISA versus reverse transcriptase polymerase chain reaction (RT-PCR) in tissue specimens) or to the different classification of the population study might be taken into account. Interestingly, our results are in agreement with those of Pylväs-Eerola et al. 30 in endometrioid-type epithelial ovarian cancer, where the high DJ-1 serum levels are associated with the histological grade.
In conclusion, our data strongly suggest that DJ-1 blood testing might be an informative and feasible measurement in EC. However, the direct implementation of DJ-1 testing into clinical practice deserves further studies aimed, among others, at increasing patients number, establishing inter-laboratory standardization using appropriate restricted-criteria order set (e.g. exclusion of patients with major comorbidities such as previous heart failure).39–41
Footnotes
Acknowledgements
The authors thank Caterina Alessi for editing the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
