Abstract
HIF-α may play an important role in the process of tumorigenesis as well as tumor progression. Although a number of investigations have established the significance of HIF-1α in several human tumors, there is still little information available on the clinical significance of HIF-2α expression in non-small cell lung cancer (NSCLC). In present study, immunohistologic expression of HIF-1α/ HIF-2α was studied in a tissue microarray of 140 Stage I-III NSCLCs and correlated with clinicopathologic parameters and clinical outcome. We found that HIF-1α/ HIF-2α showed a cytoplasmic pattern of expression in tumor cells while normal lung components showed negative or weak cytoplasmic staining. High HIF-1α and HIF-2α expression was noted in 49/140 (35.0%) and in 64/140 (45.7%) of the cases respectively. There was no direct correlation between HIF-1α and HIF-2α expression (p = 0.200). The high HIF-2α expression was associated with histology (squamous cell carcinoma vs. adenocarcinomas) in these patients (p = 0.001). Patients in advanced tumor stage had frequent high expression of HIF-2α (p = 0.007), and the similar high expression was also observed in advanced T or N stage (p = 0.030 and 0.043, respectively). HIF-1α showed a marginal association with T stage (p = 0.084), which showed a higher expression in early tumor stage. Univariate analysis of the overall survival demonstrates that HIF-2α expression but not HIF-1α was related to poor outcome (p = 0.005) and it retained significance in multivariate analysis (p = 0.046). In conclusion, HIF-2α expression was related to tumor size, lymph node metastasis, tumor stage and histology. We also found a positive prognostic value of HIF-2α protein expression. HIF-2α might serve as a potential prognosis biomarker in evaluating progression and prognosis of NSCLC. We believe that our study will be of great benefit to the clinical treatment and prognostic evaluation of NSCLC.
Introduction
Although the incidence and mortality rates are declining, lung cancer retains its status as the leading cause of cancer-related deaths in the world, among which non–small cell lung cancer (NSCLC) accounts for about 80% of lung cancer patients with a low 5-year survival rate.1–4 Although significant advances have been achieved in conventional therapies, poor prognosis and short survival of patients, as well as the limited value of any sort of conventional therapy, are factors all demanding novel and more effective therapy. Therefore, it is very important to understand the molecular mechanisms underlying the carcinogenesis.
Region of hypoxia is a common microenvironment during development of many solid tumors. 5 Hypoxia is related to therapy resistance, enhanced metastatic potential, and poor prognosis in numbers of cancers. 6 At the heart of the hypoxia regulatory system is hypoxia-inducible factors (HIFs), which are heterodimeric complex composed of a regulated subunit (e.g. HIF-1α and HIF-2α) and a constitutive subunit (HIF-1β), also known as aryl hydrocarbon nuclear translocator (ARNT). 7 The HIF-α subunit is regulated in an oxygen-dependent manner which determines the HIF system activity. Under normoxic conditions, there is only trace expression of HIF-α, which is rapidly degraded by a process involving its proline residue hydroxylation, interaction with the von Hippel–Lindau (VHL) E3 ubiquitin ligase complex, and rapid subsequent proteasomal degradation. 8 This process is mediated by prolyl hydroxylase domain–containing (PHD) enzymes, such as PHD1, PHD2, and PHD3. Although under hypoxic condition, this oxygen-requiring hydroxylation is inhibited, HIF-α protein is accumulated in cells, which heterodimerizes with HIF-β and then enters the nucleus where it binds to hypoxia-responsible elements (HREs) and activates a network of genes transcription that control several aspects of tumor biology such as energy metabolism, angiogenesis, cellular growth, apoptosis, invasion and metastasis, and chemotherapy resistance.
There are three isoforms of the HIF-α subunit (HIF-1α, HIF-2α, and HIF-3α), of which HIF-1α and HIF-2α have been the most extensively studied.9,10 HIF-1α and HIF-2α subunits are structurally similar in their DNA-binding and dimerization domains which share certain overlapping functions by regulating common hypoxia-inducible genes. 11 However, it has recently become clear that, through independent regulation of distinct target genes or unique protein–protein interactions, HIF-1α and HIF-2α can also mediate divergent functions when expressed in the same cell type and especially in the context of cancer. 7 It is reported that HIF-1α, which is directly or indirectly related to the regulation of development and progression of cancers, is overexpressed in many human cancers.12,13 However, the corresponding role of HIF-2α, however, is less clear, especially in NSCLC.
In our previous NSCLC tissue microarray (TMA) analysis, we investigated the prognostic impact of HIF-1α and HIF-2α. Overall, we found high tumor cell HIF-2α expression but not HIF-1α to be an independent indicator of poor prognosis. 14 However, with a median follow-up of 31 months, our observation time seems too short to develop an accurate picture of the HIF-2α impaction on the prognosis of NSCLC. So, in this report, a reanalysis of the original study with long-term follow-up was carried out for final conclusions about the prognostic relevance of the HIF-2α expression in NSCLC.
Materials and methods
Patients
For this retrospective study, 140 tissue samples were obtained from anonymized patients, whose illness was diagnosed as NSCLC, undergoing operation at the Thoracic Surgical Division of Zhongshan Hospital, Fudan University (Shanghai, China) in year 2005. All cases were staged according to the international staging system and graded according to the recent 2004 World Health Organization criteria. In this study, no subjects received adjuvant or neoadjuvant chemo- or radiation treatment prior to surgery and no other malignancy within 5 years prior to diagnosis was present. The study protocol was reviewed and approved by the Research Ethics Committee of Zhongshan Hospital, Fudan University. This study was reviewed and approved by the Research Ethics Committee of Zhongshan Hospital, Fudan University. Written informed consent was obtained from all the patients. All specimens were handled and made anonymous based on the ethical and legal standards.
Construction of TMA
For the construction of the TMA, fixed and paraffin wax–embedded tissues from 140 patients’ samples were used. The TMA was constructed as described previously by Kononen et al. 15 Briefly, paraffin blocks containing representative tumor areas were identified on corresponding hematoxylin and eosin (H&E)-stained sections. Areas of interest were identified and marked on the source block. All cases had intact tumor cores (devoid of significant necrosis and containing sufficient tumor) for analysis. The source block was cored and transferred to the recipient block using a 1.0-mm-diameter precision punch (Beecher Instruments, Silver Spring, MD, USA). To overcome tumor heterogeneity, we used two 1.0 mm cores, selected to be as representative as possible after reviewing all the original sections of the tumor. Tissue samples from 140 primary tumor cases and 10 normal lung tissues (taken from normal lung tissue of patients whose illness was diagnosed as benign lung cancer undergoing operation at the Thoracic Surgical Division of Zhongshan Hospital, Fudan University in year 2005) were arranged in rows and columns to construct a TMA. A spreadsheet was elaborated to depict the core locations. The TMA was cut into 4-µm sections and placed on glass slides using an adhesive tape transfer system. We used the Manual Tissue Arrayer I (MTA-I; Beecher Instruments, Sun Prairie, WI, USA). Sections in 30 slides were stained with H&E to confirm the presence of the tumor by light microscopy and to ensure that all the cores were present at the same depth. After drying for 16 h at 60°C, the slides were used for immunohistochemistry (IHC).
Immunohistochemical analysis
Standard indirect immunoperoxidase procedures were used for IHC (Envision Plus; Dako, Carpinteria, CA, USA). TMA sections were deparaffinized in xylene, followed by rehydration in a descending concentrations of ethanol (absolute, 96%, 70%, and 40% for 5 min each) ending with pure water. Next, endogenous peroxidase was quenched in 0.3% hydrogen peroxidase for 20 min. Antigen retrieval was carried out as described in Table 1. Then, the slides were incubated in 10 mM Tris-buffered saline (TBS) with 4% normal goat serum for 1 h and then with the primary antibodies at appropriate conditions. The characteristics of the antibodies used in this study are summarized in Table 1. After washing with TBS, the biotin-free horseradish peroxidase enzyme–labeled polymer of the Envision plus detection system (Dako, Carpinteria, CA, USA) was used as a secondary antibody according to the manufacturer’s instructions. The reaction products were visualized by exposing the sections to 3,3′-diaminobenzidine (Dako). Sections were weakly counterstained with hematoxylin, dehydrated, and coverslipped. We used breast cancer tissue as a strongly positive control for HIF-1α and macrophage for HIF-2α as reported previously. 16 Phosphate-buffered saline (PBS) was substituted for primary antibody as the negative control. For each antibody, all TMA staining procedures were performed in one single experiment.
Summary of the characteristics of the antibodies used in this study.

HIF: hypoxia-inducible factor; EDTA: ethylenediaminetetraacetic acid.
Immunohistochemical evaluation
Evaluation of immunostaining was independently performed by three investigators without any knowledge of the clinical data. Morphologically, histopathological features and immunohistochemical staining were evaluated independently. In many cores, there are a mixture of stromal cells and tumor cells. However, by morphological criteria only staining intensity and density of tumor cells were scored. Both HIF-1α and HIF-2α expression were assessed using the grading system established in previous studies.14,17 The intensity of cytoplasmic staining was scored as absent, weak, moderate, or strong. The extent of cytoplasmic staining was expressed as the percentage of positive cancer cells from 0% to 100%. Tumors were scored according to the intensity and extent of staining—score 1: tumors with absent or weak cytoplasmic reactivity and no nuclear reactivity, score 2: tumors with moderate/strong cytoplasmic reactivity in a percentage of cancer cells lower than the mean value and no nuclear reactivity, score 3: tumors with moderate/strong cytoplasmic reactivity in a percentage of cancer cells higher than the mean value, score 4: tumors with a clear nuclear reactivity with or without cytoplasmic reactivity regardless of the intensity. Tumors with score 1/2 were grouped as low HIF-α expression, while tumors with score 3/4 as high HIF-α expression.
Statistical analyses
Statistical analysis and graph plotting were performed using SPSS 15.0 software (SPSS Inc, Chicago, IL, USA). The correlation between HIF-1α/HIF-2α expression and various clinicopathological parameters or other immunohistochemical proteins was determined by χ2 and Student’s t tests as appropriate. Overall survival (OS) times were defined as the period from primary surgery until the death of the patient or the latest follow-up. Kaplan–Meier survival and Cox proportional hazard analyses were used to determine survival differences and identify independent prognostic variables. In all tests, a two tailed p < 0.05 was considered to be significant.
Results
The characteristics of the 140 patients
An overview of clinicopathological parameters is given in Table 2. The mean age of patients was 60 years, with a range between 26 and 79 years. Most NSCLC cases were in males. More than half of the tumors were squamous carcinoma (57%) and approximately two fifth of the tumors were poorly differentiated (42%). Clinical follow-up was recorded until July 2013. The median duration of follow-up for this cohort was 45 months (range: 3–101 months) at the time of analysis. By the end of follow-up, there were 84 local recurrences or distant metastases and 91 deaths.
Clinical and histological features of 140 patients with NSCLC.

NSCLC: non–small cell lung cancer.
Median (range).
Immunohistochemical expression of HIF-1α/HIF-2α
Both HIF-1α and HIF-2α showed a negative or weak cytoplasmic reactivity in control cores from normal lung tissue. The HIF-1α and HIF-2α staining were present mainly in the cytoplasm. Nuclear staining was observed only sparsely (Figure 1). The extent of staining varied among tumors. In some tumor cores, a subset of cells morphologically identified as macrophages showed abundant cytoplasmic HIF-2α immunoreactivity, and they were excluded in evaluation of HIF-2α staining in tumor cells. The percentage of cancer cells with HIF-1α moderate/strong cytoplasmic reactivity ranged from 0% to 100% (median: 35%, mean: 52%). The percentage of cancer cells with HIF-2α moderate/strong cytoplasmic reactivity ranged from 0% to 92% (median: 31%, mean: 37%). Using the grading system according to the intensity and extent of staining as proposed in the Materials and methods section, the tumors were subdivided into low (n = 91) and high (n = 49) groups according to the HIF-1α expression, and also low (n = 76) and high (n = 64) groups according to the HIF-2α expression. Both HIF-1α and HIF-2α were low in 55 cases, and both were high in 24 cases. There was no direct correlation between HIF-1α and HIF-2α expression (p = 0.200).

Expression of HIF-1α and HIF-2α in normal and neoplastic lungs. (a) HIF-1α is not expressed in normal lung tissue. (b) HIF-2α is not expressed in normal lung tissue. (c) Weak cytoplasmic expression of HIF-1α in lung tumor tissue. (d) Weak cytoplasmic expression of HIF-2α in lung tumor tissue. (e) Strong cytoplasmic expression of HIF-1α in lung tumor tissue. (f) Strong cytoplasmic expression of HIF-2α in lung tumor tissue.
HIF-1α/HIF-2α expression and clinicopathological parameters
For correlation with various clinicopathological features, HIF-α expression was categorized as low and high as previously described. The associations between HIF-1α/HIF-2α expression and clinicopathological variables are shown in Table 3. No statistically significant correlations were found between HIF-1α and any of the clinicopathological parameters as evaluated in this study. However, HIF-2α expression increased with the development of the T stage (p = 0.030) and the N stage (p = 0.043). There was also a significant correlation between the HIF-2α expression and tumor–node–metastasis (TNM) stage (p = 0.007). In addition, a significantly higher level of HIF-2α expression was observed in squamous cell carcinomas than in adenocarcinomas (p = 0.001).
The relationship between HIF-1α/HIF-2α and clinicopathological factors.
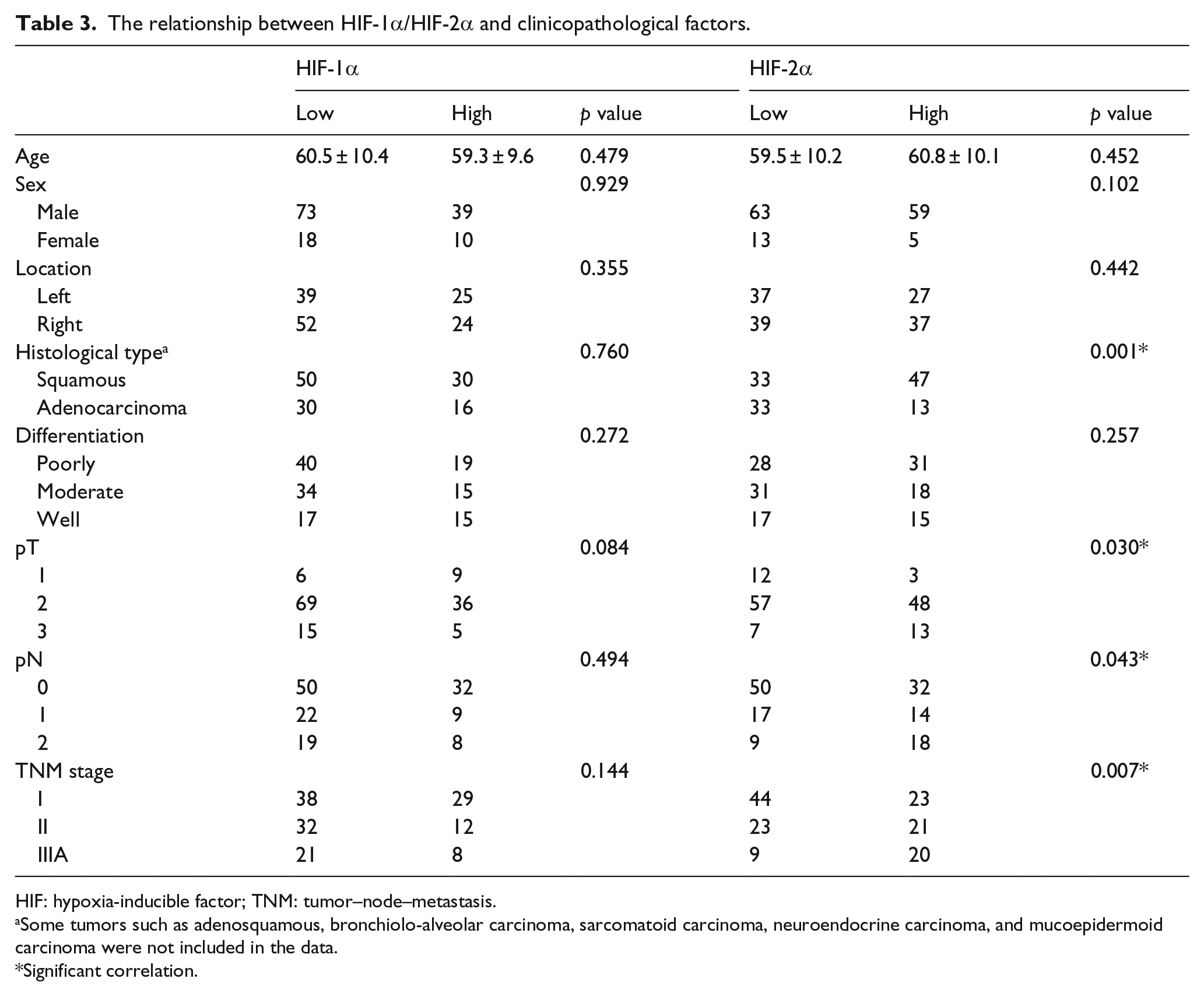
HIF: hypoxia-inducible factor; TNM: tumor–node–metastasis.
Some tumors such as adenosquamous, bronchiolo-alveolar carcinoma, sarcomatoid carcinoma, neuroendocrine carcinoma, and mucoepidermoid carcinoma were not included in the data.
Significant correlation.
HIF-1α/HIF-2α expression and patients’ OS
Figures 2 and 3 show the Kaplan–Meier survival curves plotted for HIF-1α/HIF-2α expression. In 140, Stage I–III patients, the survival analysis revealed that high levels of HIF-2α were correlated strongly with shortened OS (p = 0.005, log-rank test; Figure 2(b)), while there was no significant difference in OS prognosis with HIF-1α expression (p = 0.489, log-rank test; Figure 2(a)). We further analyzed the survival of patients with Stage I NSCLC, a statistically significant survival difference was also observed between those low and high in HIF-2α expression (p = 0.042, log-rank test; Figure 3(b)). In this group, the 5-year survival of patients with high HIF-2α expression was 29.7% versus 49.9% of patients with low HIF-2α expression, as shown in Figure 3(b). However, HIF-1α expression was not found to be correlated to OS in Stage I NSCLC patients (p = 0.638, log-rank test; Figure 3(a)).

Kaplan–Meier survival curves for overall survival stratified by tumor HIF-1α or HIF-2α expression in Stage I–III patients. (a) Univariate survival analysis showed no relationship between HIF-1α and overall survival in patients with Stage I–III NSCLC (p = 0.489, log-rank test). (b) Univariate survival analysis indicated that HIF-2α expression is associated with poorer prognosis in patients with Stage I–III NSCLC (p = 0.005, log-rank test).

Kaplan–Meier survival curves for overall survival stratified by tumor HIF-1α or HIF-2α expression in Stage I patients. (a) Univariate survival analysis showed no relationship between HIF-1α and overall survival in patients with Stage I NSCLC (p = 0.638, log-rank test). (b) Univariate survival analysis indicated that HIF-2α expression is associated with poorer prognosis in patients with Stage I NSCLC (p = 0.042, log-rank test).
Table 4 shows the results obtained from multivariate analysis for potential factors influencing OS, with all factors statistically significant in the univariate analysis entered as covariates. Only HIF-2α, age, and N stage had an independent prognostic meaning (p = 0.046, 0.042, and 0.034, respectively) for the whole group of 140 patients for OS.
Multivariate Cox regression analysis for potential factors influencing overall survival.

OR: odds ratio; CI: confidence interval; HIF: hypoxia-inducible factor.
Significant correlation.
Discussion
Tumor hypoxia is well recognized as a major driving factor for tumor growth and resistance to therapy.18,19 In addition to promoting tumor cell survival during hypoxic stress by shifting cells toward anaerobic metabolism, neovascularization, and resistance to apoptosis, the hypoxia response may drive other responses that contribute to tumor aggressiveness such as increased genetic instability, invasion, metastasis, and an undifferentiated phenotype.20,21 HIF-1α and HIF-2α are the pivotal mediators in the process which have been the molecular switch for control of cellular responses to low oxygen conditions. They are usually increased under hypoxic conditions and can activate transcription of many genes that are critical for cellular function under hypoxic conditions.7,22,23
HIF-1α and HIF-2α are structurally and functionally similar but not identical. They have their unique patterns of downstream gene induction. Several reports suggest that HIF-1α and HIF-2α show distinct expression patterns and functions.24,25 Although HIF-1α is the most widely described and most well-known isoform, a great deal of evidence indicates that HIF-2α plays an important role as HIF-1α in cancer. 24 Overexpression of HIF-2α in renal cell carcinomas (RCCs) leads to enhanced angiogenesis and tumor progression even in the absence of HIF-1α. 26 In Hamidian et al.’s study, HIF-2α immunostaining was identified in neuroblastoma tissues which was correlated with clinicopathological features. 27 Also, HIF-2α is related to poor outcome in colorectal carcinoma, 28 hepatocellular carcinoma, 29 and melanoma. 30 These findings raise the possibility of tissue-specific differences in the relative importance of HIF proteins in determining tumor progression.
The prognostic role of HIF-αs (HIF-1α and HIF-2α) in primary NSCLC is also variably reported; however, its role in determining prognosis in NSCLC has not been definitively established. There has been lots of controversy. In some reports, high expression of HIF-1α has been associated with poor prognosis in NSCLC. 31 However, the impact of HIF-1α on the survival is found to be associated with better prognosis in NSCLC in another report. 32 Kim et al. 33 found that HIF-1α expression was associated with a shorter disease-free survival in the univariate analysis; however, its prognostic relevance was not evident in the multivariate analysis including CA IX (carbonic anhydrase IX). Similar correlation was demonstrated by Swinson et al. 34 in 204 consecutive cases of resected Stage I–IIIA NSCLC patients. A study in NSCLC showed that HIF-2α expression was related to a poor outcome whereas HIF-1α expression was not, which was consistent with our results.14,35 However, the median follow-up period of 31 months in our sturdy is too short to draw a definitive conclusion at this time, which is likely to be considerably longer. Further studies are needed to make conclusion. So, we have completed a long-term (>5 years) follow-up of these patients with a median follow-up period of 45 months. In this study also, we made the same conclusion.
A possible explanation for the heterogeneity is that both HIF-1α and HIF-2α can modulate the transcriptional activation of a set of common genes but not all the genes activated by the counterparts. 24 Another finding is that the various hypoxia-inducible genes vary in their sensitivity to HIF-1α and HIF-2α. 36 In our preliminary experiment, we found that HIF-1α protein was transiently increased and reached a peak at 4 h and then it decreased almost to basal line 12 h later when the A549 cells were cultured under hypoxia, while HIF-2α gradually accumulated and persisted at a high level after a long-term hypoxia exposure. 37 These observations agree with some previous reports.38–40 These findings led us to hypothesize that HIF-1α may transiently induce and mainly mediate the response to acute and severe hypoxia, while HIF-2α may induce a longer response and control the response to chronic and moderate hypoxia regulation which occurs later, and its protein level is gradually elevated and maintained in response to the chronic and moderate hypoxia microenvironment in later-stage tumor lesions. 37
In conclusion, this study found a positive prognostic value of immunohistologically determined HIF-2α protein expression and presents HIF-2α as an unfavorable prognosis biomarker in patients with early operable NSCLC. These findings shed light onto the HIF research in NSCLC and may reveal new methods for the prevention, early detection, and molecular-targeted treatment of the disease. Nevertheless, other independent prospective studies are needed to establish the prognostic value of HIF-2α expression.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Basic research project of Changzhou (Grant number: CJ20140041), High-Level Medical Talents Training Project (Grant number: 2016CZBJ042), Jiangsu Planned Projects for Postdoctoral Research Funds, and Jiangsu Provincial Medical Youth Talent (Jiangsu Health Scientific Education (2017) No.3).
