Abstract
Breast cancer is one of the leading malignant tumors that endanger women’s health worldwide. Despite the rapid progress on the therapies, including chemotherapy, surgical resection, and other auxiliary methods, there were still numerous people died of breast cancer, which promoted the researchers to concentrate on the prognostic factor of breast cancer. In recent years, an increasing number of studies have been focused on the prognostic value of pretreatment neutrophil-to-lymphocyte ratio in breast cancer. This article is a brief review of the associations between neutrophil-to-lymphocyte ratio and the prognosis of breast cancer patients, which may give a greater insight into the development of breast cancer and enable clinicians to cure it completely.
Introduction
Breast cancer is one kind of multifactorial disease without certain pathogeny. It was reported that personal and family history of breast cancer, age, overweight, menstrual history, genetics, radiation to chest or face before age 30, pregnancy/breast feeding, race/ethnicity, hormone replacement therapy, alcohol consumption, dense breast, lack of exercise, and smoking are contributing factors of breast cancer. 1 What’s more, the morbidity of breast cancer was 12.5%, 2 while the female diagnosed with breast cancer before 40 years accounted for 5%–7% of the total female population.3–6 According to a epidemiological investigation, the morbidity and mortality of breast cancer in China have been very high recently and the urban (78%) has surpassed the rural (58%) in terms of the 5-year survival rate.7,8 However, the International Agency for Research on Cancer (IARC) 9 estimated that the cases of female breast cancer patients in China would reach 234,000 in 2030 and the growth rate was 31.15% greater than the cases in 2008, while the fatality due to breast cancer would reach 70,000 cases and the growth rate was 47.94%. The American Cancer Society (ACS) also predicted that 29% of all new malignant diseases is breast cancer and it is 16% cause of deaths. 10
In spite of the improvement in therapy, the number of deaths is still sufficiently great. Consequently, it is extremely urgent to discovering one kind of substance that can reflect the occurrence and development of tumor, and even the prognosis of it. Recently, increasing studies have investigated the function of the immune system in the growth progression or cessation of tumors.11–13 Of course, the breast cancer was also included. Tumor-associated inflammation was considered as the prognostic factor in tumor and the neutrophils and lymphocytes were the main inflammatory cells. Neutrophil-to-lymphocyte ratio (NLR), just as its name implies, is the ratio between neutrophils and lymphocytes. Its value reduction accompanies by the relatively decreasing neutrophils level or increasing lymphocytes level. And we may gain the value from routine blood parameters. Mounting studies indicate that NLR relates to the prognosis of breast cancer. Subsequently, we will summarize the tumor-associated functions of the neutrophils and the lymphocytes, discuss recent studies of the associations between breast cancer and NLR, and describe the characteristics of NLR and other tumor markers in breast cancer.
The neutrophils
Tumor microenvironment is regarded as a chronic inflammatory environment, which is infiltrated with immunocytes and inflammatory cytokines.14–18 Of these, neutrophils, a factor of tumor prognosis, play an active role in the development of tumor. Some studies 19,20 reported that the degranulation capacity of neutrophils reduced in tumor-bearing mice, and the mechanism was that the highly activated signal transducer and activators of transcription 3 (STAT3) on account of the high-level granulocyte colony-stimulating factor (G-CSF) and interleukin-6 (IL-6) led the normal neutrophils to tumorigenic cells. Furthermore, the tumorigenic gene expression was upgraded and the suppression gene expression was degraded (Figure 1).

The effect of the STAT3 on the tumorigenic gene expression of the neutrophils. The highly activated STAT3 on account of the high-level G-CSF and IL-6 plays a critical role in the expression patterns of tumorigenic genes in neutrophils. It is via epigenetic modification, promoting cancer stem cells self-renewal and differentiation, regulating epithelial–mesenchymal transition-related transcription factors, and adjusting tumor microenvironment.
Murdoch et al. 21 and Okuturlar et al. 22 indicated that the neutrophils had close relationships with the angiogenesis of tumor and the poor prognosis of patients. According to one meta-analysis, 23 the highly expressed tumor-associated neutrophils as a new prognostic factor in cancer had significant statistical correlation with the rate of recurrence-free survival (RFS), cancer-specific survival, and overall survival (OS). In recent years, some scholars put forward the concept of neutrophil extracellular traps (NETs) with respect to the tumorigenic mechanism of the neutrophils. Chromatin and antibacterial protein or peptide constituted NETs,24,25 which included matrix metalloprotein-9 (MMP-9), Cathepsin-G (CG), neutrophil elastase (NE), and so on. It was confirmed that NETs could trap and kill microorganism at the earliest time and played a positive role in the occurrence and development of tumor. Researchers found that the function of MMP-9 was promoting cell proliferation, inhibiting cell apoptosis, facilitating angiogenesis, and boosting the distant metastasis of the tumor cells by degrading extracellular matrix;26–29 CG was able to stimulate the expression of vascular endothelial growth factor (VEGF) messenger RNA (mRNA) and protein and also could facilitate angiogenesis and boost the distant metastasis of the tumor cells by degrading extracellular matrix as well; 30 NE had the capability of promoting cell proliferation and boosting the distant metastasis of the tumor cells directly. 31
The lymphocytes
Inflammatory markers have long been linked with malignancy. Virchow first discovered leukocytes existing in neoplastic tissue in 1863, producing the hypothesis that inflammation plays a critical role in the development of malignant disease. 32 In the mid of 1900s, the relationship of the tumor-infiltrating lymphocytes (TILs) and the patients prognosis in medullary carcinoma of breast was first reported by Moore. 33
Recently, numerous researches34,35 about TILs had carried out and discovered36,37 that T-lymphocytes had effective functions of anti-tumor. To our knowledge, there were two ways to identify and kill tumor cells for T-lymphocytes. 38 One kind was antigen presentation between major histocompatibility complex-1 (MHC-1) molecules and CD8+ T cells, as a result, the cytotoxic T-lymphocytes (CTLs) would be activated, and then the activated tumor necrosis factor (TNF) ligand and perforin (PRF) or granzyme (Gzms) prompted the apoptosis of tumor cells; the other kind was antigen presentation between MHC-2 molecules and CD4+ T cells; consequently, the increasing activated CD4+ T cells improved the function of immune system by recognizing tumor cells and boosted the function of CTL recognition by influencing MHC-1 molecules (Figure 2).
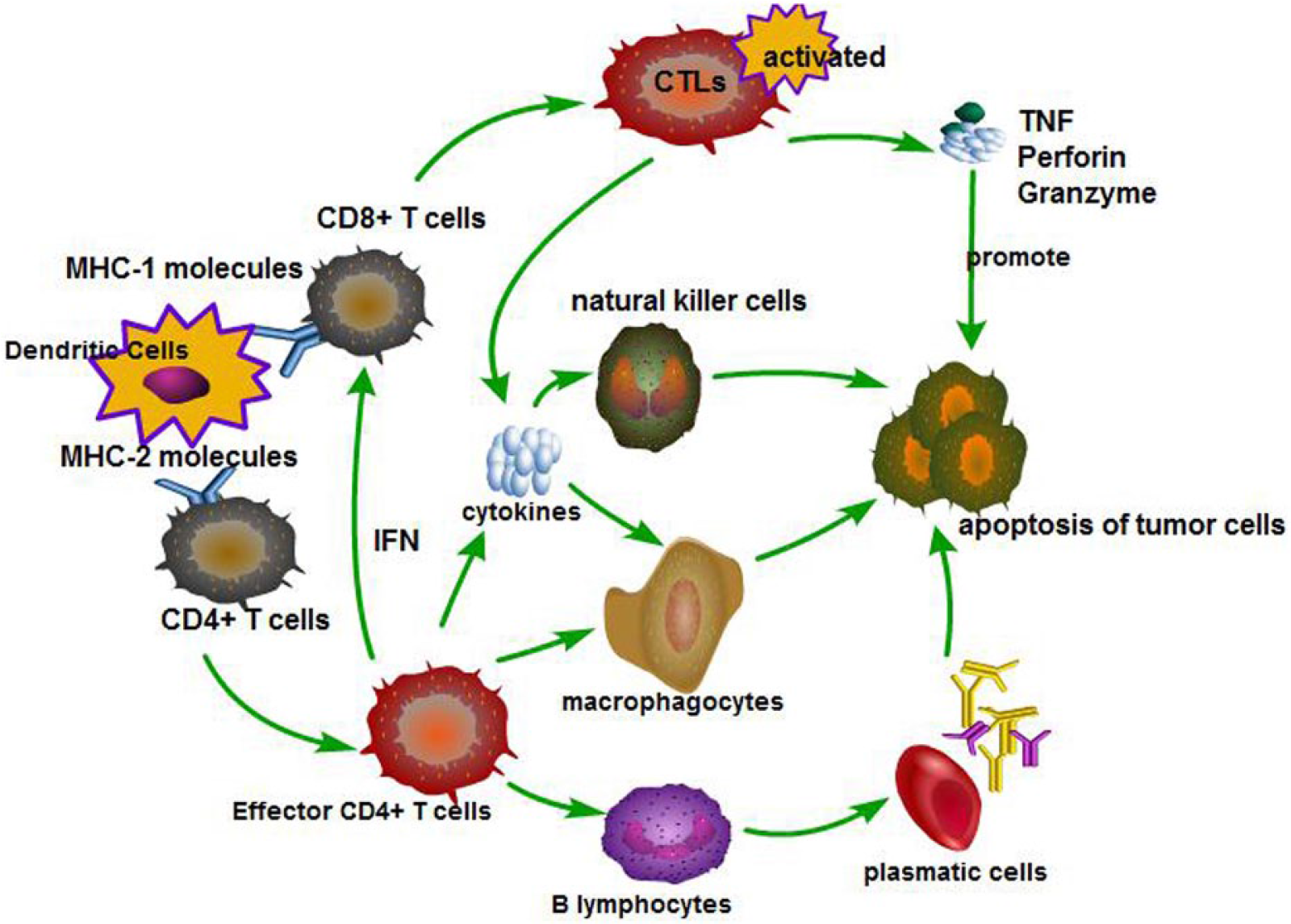
Two ways to identify and kill tumor cells for T-lymphocytes. Dendritic cells (DCs) induce the activation and differentiation of naive CD8+ T cells to cytotoxic T-lymphocytes (CTL) that recognize and kill tumor cells via releasing cytotoxic granules containing perforin and granzymes. In addition, by secreting IFN-γ, cytotoxic CD8+ T cells may also potentiate function of macrophages and NK cells. On the other hand, DCs activate naive CD4+ T cells and promote their differentiation toward effector CD4+ T cells, which contribute to an efficient activation of CD8+ T cells and B cells and also regulate function of innate immune cells as macrophages and NK cells.
Moreover, it was reported39,40 that the Th1/Th2 cytokine type had altered significantly in tumor-bearing body, which manifested as the degradation of Th1-associated cytokine (such as interferon (IFN)-γ, IL-2, TNF-α, and IL-12) and the upgradation of Th2-associated cytokine (such as transforming growth factor (TGF)-β, IL-4, IL-5, IL-6, and IL-10). Furthermore, it was beneficial for tumor evading from immune surveillance and attack under this circumstance. Similar results were also confirmed in non–small cell lung cancer, nasopharyngeal carcinoma, gastrointestinal tract cancer, ovarian cancer, melanoma, osteosarcoma, and lymphoma. Moreover, the change of the type had positive correlation with the degree of malignancy.41,42 Zhang et al. 43 also found that terminal ovarian cancer patients with TILs have longer RFS and OS than the patients without TILs, which seemed to enlighten clinicians to cure cancer patients via TILs transfusion.44,45
NLR and the prognosis of breast cancer patients
Generally, the immune system, especially the neutrophils and lymphocytes, does exercise some influence on tumor cessation and progression. We may conclude that there are some relationships between tumor and NLR. Actually, an increasing number of studies had concentrated on the relationships between NLR and the prognosis of tumor, and the breast cancer were also included.46–53
In a retrospective, longitudinal, cohort study 54 of 437 consecutive female breast cancer patients, Azab et al. concluded that NLR, as an independent predictor of breast cancer mortality, was superior than platelet-to-lymphocyte ratio (PLR). While Liu et al. 55 indicated that both increased NLR and PLR are associated with poor survival in hormone-receptor-negative (HR−) breast cancer, but only NLR is independently correlated with OS and disease-free survival (DFS). Dirican et al. 56 reported that NLR was shown to be better than derived neutrophil/leukocyte–lymphocyte ratio (dNLR) in terms of predicting prognosis in patients with breast cancer and a high pretreatment NLR (NLR > 4) was associated with poor survival (DFS and OS) in patients.
Ozyalvacli et al. 57 found that preoperative high NLR was a significant diagnostic predictor of distinction of breast cancer from benign proliferative breast disease and elevated NLR was also an important prognostic marker for primary invasive breast cancer in a randomized controlled trial, and the optimal cutoff for NLR was 2.96. In multivariate analysis, 52 NLR is an independent predictor of short- and long-term mortality in breast cancer patients with NLR >3.3 after adjusting for possible confounder. As for patients undergoing breast cancer surgery, 58 in Center 1, NLR ⩾4 is associated with a higher risk of relapse; in Center 2, NLR ⩾3 is associated with a higher risk of relapse and higher mortality.
Chen et al. 59 demonstrated that patients with NLR ⩾2.06 showed poorer response to neoadjuvant chemotherapy and a lower pathological complete response (pCR) rate than those with NLR <2.06. High NLR was an independent prognostic factor for poor RFS and breast cancer–specific survival (BCSS) in these patients with breast cancer undergoing preoperative chemotherapy. However, Casares et al. 60 discovered that cell death induced by some types of chemotherapy can improve CTL responses. Therefore, it is necessary to consider the stability of NLR in the process of drug therapy. Clinicians may think over what kinds of drugs the patients have taken prior to employing NLR as the prognostic factor of breast cancer. Furthermore, when it comes to the diagnosis of breast cancer in the early stages, it should be noted that at this period the indicators of immune system appear not to be strong enough for the detection, due to a lack of systemic abnormalities in the body. When it comes to [> IIa/b] stages, the value of NLR ratio would rather likely play a predictive role.
Japanese researchers Nakano et al. 61 also reported that preoperative NLR might be an independent prognostic factor for survival in Japanese patients with breast cancer, meanwhile they pointed out that NLR was significantly higher in patients with lower body mass index in multivariate analysis. As for Chinese patients with breast cancer, Yao et al. 62 found that patients with high NLR >2.57 showed a significantly lower OS than those with lower NLR; there was a significant survival difference according to NLR in the luminal A and triple-negative subtypes. Liu et al. 63 reported that NLR is independently correlated with OS and DFS; the cutoff value of NLR (3.0) was consistent with that of most of previous studies.
Characteristics of NLR and other tumor markers
It has been approximately 20 years since researchers investigated tumor markers in breast cancer.64,65 Generally, when it comes to the prognostic factors, there is a need that the characteristics of the tumor markers must be reproducible, cheap, readily performed, and have high quality.
As is well known to us, the number of positive axillary lymph nodes, tumor size, lymphatic and vascular invasion, histological tumor type, and sex steroid receptors are routinely used as prognostic factors in breast cancer. Other pathologic prognostic factors include histological grade and nuclear grade. Moreover, some biological parameters, such as oncogene products (p53, c-erb B-2), carcinoembryonic antigen (CEA), carbohydrate antigen 15.3 (CA 15.3), proteins (pS2), or enzymes (cathepsin D, urokinase) have been incorporated into prognostic factors. Both the pathologic prognostic factors and the biological parameters have not been standardized.66–74
In this review, we introduce a new prognostic factor in breast cancer. The strengths of the indicator does happen to be equal to the necessary characteristics of tumor markers. It is accessible to gain the values of neutrophils and lymphocytes from blood routine tests and cheaper than other tumor markers. Regrettably, we still cannot obtain data from the literature of its sensitivity and specificity owe to limited studies, nor NLR values corresponding to different tumor stages.
Summary
In conclusion, NLR is more likely to be a clinically prognostic factor in breast cancer; the low scores have invariably been shown to be deleterious in terms of tumor progression. Moreover, the neutrophils and lymphocytes are routine detection index and affordable to obtain. However, we still cannot gain the identical cutoff point and the exact mechanism via previous studies. Henceforth, further feasibility studies with multi-central and a large sample size are required before it can be considered for clinical use.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
