Abstract
Metastatic or recurrent cervical cancer has limited treatment options and a high rate of mortality. Although anti-vascular endothelial growth factor drugs have shown great promise as a therapeutic target for treatment of advanced cervical cancer, drug resistance and class-specific side effects negate long-term benefits. The identification of alternative anti-angiogenic factors will be critical for future drug development for advanced or recurrent cervical cancer. In this study, we found that angiopoietins and Tie receptors were highly expressed in cervical cancer cells. Tie-2 expression in tumor cells predicted poorer prognosis. Wound closure assay and Transwell assay showed that upregulated or downregulated Ang-1 and Ang-2 expression promoted or reduced cervical cancer cell lines migration and invasion, respectively. In subcutaneous xenograft models of cervical cancer, downregulation of Ang-1 and Ang-2 attenuated tumor growth. The expression of vimentin and endomucin and microvessel density were all significantly decreased in the siAng-1 group and siAng-2 group relative to the infection control group. Our data support that dual inhibition of Ang-1 and Ang-2 may be an alternative target for anti-angiogenic adjuvant therapy in advanced or recurrent cervical squamous cell cancer.
Introduction
Cervical cancer is the third most common type of cancer in women worldwide. 1 While early-stage and locally advanced cancers can often be cured with the current standard treatment, metastatic or recurrent cervical cancer has limited treatment options and a high rate of mortality. 2 Specifically, the recurrent/metastatic rate for stage I–II disease is approximately 15%–30% and increases to 40%–60% for women with stage III neoplasms. Only 10% of patients with recurrent disease will be alive at 5 years.1,3 Angiogenesis is essential for tumor progression and has shown great promise as a therapeutic target for treatment of advanced cervical cancer.4,5 For example, bevacizumab, which antagonizes vascular endothelial growth factors (VEGFs), reduces mortality by 30% in patients with recurrent, persistent, or metastatic cervical cancer when combined with chemotherapy compared with those who receive chemotherapy alone. 6 However, drug resistance and class-specific side effects negate the long-term benefits of anti-VEGF drugs.7,8 Therefore, the identification of alternative anti-angiogenesic factors will be critical for future drug development.
The angiopoietin (Ang) family is a series of angiogenic factors that bind to tyrosine kinase receptor Tie-2 (TEK) and participate in physiological and pathological angiogenesis. Perivascular cells that coat mature vessels express Ang-1, while Ang-2 is expressed by endothelial cells (EC), and both play an essential role in controlling blood vessel development and stability.9 –11 Angiopoietins have recently been found to be expressed in tumor cells, such as breast cancer, melanoma, gastric carcinoma, and hepatocellular carcinoma. The high expression of angiopoietins together with VEGF promotes the initiation of angiogenesis and maturation of new vessels involved in tumor progression.9,12,13 Ang-1 is a Tie-2 agonist, whereas Ang-2 can function either as an antagonist or a partial agonist of Tie-2 depending on the specific context. Ang-1 and Ang-2 have been shown to also either promote or inhibit tumorigenesis and tumor growth depending on tumor type.9,10,12,13 In cervical cancer, Shim et al.14,15 found that the HeLa cervical cancer cell line stably expressed Ang-1, upregulated or downregulated Ang-1 expression, and promoted or reduced tumor angiogenesis in vivo. However, the expression levels of Ang-1 and -2 in human cervical cancer tissues and their role in cervical cancer progression remain unclear.
In this study, we detected the expression of angiopoietins and Ties in human cervical squamous cell cancer specimens as well as three cervical cancer cell lines. The prognosis value of angiopoietins and Tie expression in cervical cancer tissues was evaluated. In addition, we investigated the role of tumor-derived Ang-1 and -2 in regulating proliferation and invasion of cervical cancer cells and tumor angiogenesis in vitro and in vivo.
Materials and methods
Patients’ histological specimens
Archived histological specimens obtained from 80 patients with invasive cervical squamous cell cancer (International Federation of Gynecology and Obstetrics (FIGO) stages IA1–IIB) and 19 patients with uterine myoma who received surgery between 2012 and 2014 at Wuhan Union Hospital or between 2010 and 2014 at the First Affiliated Hospital of Shihezi University were studied. Mean age was 49.24 years; age range was 29–76 years. The diagnosis was pathologically confirmed for all patients. Cases with neoadjuvant chemotherapy or radiation therapy were excluded. All patients or their next of kin provided written informed consent for the collection of samples and subsequent research. This study was approved by the Research Ethics Committee of the hospital.
In all, 30 patients were followed up to April 2017. The median follow-up time was 42.31 months (range = 14–85 months). Progression-free survival (PFS) was defined as the time from treatment to the first appearance of progressive disease or to the date of last contact. Overall survival (OS) was defined as the time from treatment to death from any cause or to the date of last contact.
Immunohistochemistry and immunofluorescence
Human antibodies for detection of Tie-1, Tie-2, Ang-1, and Ang-2 in cervical tissue and mouse antibody for CD31 were all purchased from R&D Systems (USA); Ki-67, D240, Endomucin, NG2, and α-SMA were from Abcam (USA). The working concentrations of antibodies were 10 µg/mL for Tie-1, Tie-2, Endomucin, NG2, and α-SMA and 5 µg/mL for Ang-1, Ang-2, and CD31; Ki-67 was 1:250.
Paraffin-embedded sections (4 µm) were deparaffinized, hydrated, and heated for 7 min on high power, followed by 8 min on low power in 10 mM sodium citrate buffer (pH 6.0) with a microwave for antigen retrieval. After being treated with 3% H2O2 for 10 min, samples were incubated with primary antibodies overnight at 4°C. For immunohistochemistry (IHC), standard horseradish peroxidase staining using an appropriate biotinylated secondary antibody (ZSGB-BIO, PRC), the elite ABC kit (avidin–biotinylated enzyme complex; ZSGB-BIO), and diaminobenzidine (DAB; ZSGB-BIO) as the chromogen substrate (Vector Laboratories, USA) were performed according to the manufacturers’ instructions. Sections were counterstained with Gill’s hematoxylin (Sigma–Aldrich, USA) to visualize cell nuclei. For immunofluorescence, the sections were incubated with secondary antibodies conjugated with fluorescence at room temperature for 1 h while avoiding light followed by staining with 4′,6-diamidino-2-phenylindole (DAPI).
The expression of Tie-1, Tie-2, and Ang-1 and Ang-2 in squamous epithelial cells (SECs) and ECs in cervical tissues were separately evaluated. Their expression levels were scored as follows: no staining (0), weak staining (1), moderate staining (2), and strong staining (3). Microvascular density (MVD) was assessed using CD31. 16 The vessel maturity index was expressed as the fraction of α-SMA or NG-2-positive periendothelial cell–associated endomucin-positive vessels per cross section.
Cell lines and culture conditions
Human cervical cancer cell lines SiHa, HeLa, and C33A were purchased from China Center for Type Culture Collection. They were cultured in Dulbecco’s Modified Eagle’s Medium/Nutrient Mixture F-12 (DMEM/F12) media supplemented with 10% fetal bovine serum (FBS). Human umbilical vein endothelial cells (HUVECs) purchased from ScienCell Research Laboratories (USA) were maintained in endothelial cell medium (ECM; Cat. No. 1001; ScienCell Research Laboratories). All of these cell lines were grown in a humidified 5% CO2 with a temperature of 37°C.
Real-time polymerase chain reaction
Total RNA was extracted using TRIzol reagent (Invitrogen, USA), and complementary DNA was synthesized using Reverse Transcription Kit (Toyobo, Japan) according to the manufacturer’s protocol. The sequences of primers used were shown in Supplementary Table 1. Polymerase chain reaction (PCR) was performed as described by the manufacturer using the EVAGreen PCR Master Mix (Toyobo, Osaka, Japan). Amplification protocols were as follows: enzyme activation 95°C/30 s; denaturation 95°C/5 s, annealing/extension 60°C/30 s, and melt curve 65°C–95°C. The transcript level of each specific gene was normalized to the CD31 amplification and was calculated using the comparative threshold cycle (Ct) method (2−ΔΔCt).
Enzyme-linked immunosorbent assay
After centrifugation (2500 r/min for 10 min at 4°C), the supernatants of cultured cells were aliquoted and stored at −80°C. Ang-1 and Ang-2 levels were detected by standardized enzyme-linked immunosorbent assay (ELISA Kit; Uscn Life Science Inc., China) according to the manufacturer’s instructions.
Laser microdissection
Sections were cut from normal cervical formalin-fixed, paraffin-embedded (FFPE) tissues and stored at 4°C until use. Laser microdissection (LMD) was performed according to the previous reports. 17 Briefly, sections were deparaffinized immediately before LMD, stained with 1% Cresyl violet for 1 min, and dehydrated using sequential alcohol grades and xylene. Microdissection was performed using an ArcturusXT LCM instrument (Applied Biosystems, USA). An AutoScan™ analysis software module was implemented when using the ArcturusXT LCM instrument, which allowed the user to visually inspect the regions of interest. Approximately 5000 cells were captured per specimen and used for detection of messenger RNA (mRNA) expression of normal cervical epithelial cells.
Small interfering RNA
Validated control small interfering RNA (siRNA) or human Ang-1 and -2 siRNA (Santa Cruz Biotechnology) were transfected using Oligofectamine reagent (Invitrogen) following the manufacturer’s instructions. Efficacy of gene silencing was controlled by real-time polymerase chain reaction (RT-PCR) for all indicated time points and transfections. Each experiment was performed in triplicate and repeated three times.
Wound-healing assay
Cells were seeded on 6-well plates (40 × 104) and transfected for 48 h with either Ang-1 siRNA or negative control siRNA, followed by the creation of an artificial, homogeneous scratch wound on a confluent monolayer culture of cells with a 200-µL pipette tip. Serum-free medium was added for a further 24 h of incubation, and the cells were imaged at four time points (0, 12, 36, and 48 h). Percent of wound closure was calculated with ImageJ 1.47 software. Each experiment was performed in triplicate and repeated three times.
Matrigel invasion and migration assay
After being transfected with either Ang-1 siRNA or negative control siRNA for 48 h, cells (5 × 104) were plated onto a Matrigel-coated membrane in the upper chamber of a 24-well insert (8 mm pore size) containing serum-free media. The bottom chamber contained DMEM media with 10% FBS. Cells were incubated at 37°C with 5% CO2 for 36 h after plating, after which the bottom of the chamber insert was fixed with methanol and stained with crystal violet (Sigma–Aldrich). Cells that remained in the upper chamber were removed with a cotton swab. The number of cells that had migrated through the pores to the lower surface was quantified by counting five random fields per well using a microscope (Olympus, Japan) at 200× magnification. Transwell migration assays were performed and analyzed in a similar manner with the exception that no Matrigel-coated membrane was placed in the upper chamber.
The assays were also performed utilizing 600 ng/mL rhAng-1 and -2 to 100 µL cultured cell suspensions. Each experiment was performed in triplicate and repeated three times.
Colony formation assay
Cells were plated in six-well plates (500 cells per well) and maintained in DMEM/F12 containing 10% FBS for about 14 days, replacing the medium every 3 days. In the experimental group, 600 ng/mL rhAng-1 and -2 were added into the cultured cell suspensions. The colonies were then fixed with methanol and stained with 0.1% crystal violet. The colony formation was determined by counting the number of stained colonies. Each experiment was performed in triplicate and repeated three times.
Ki-67 cell proliferation assay
Cells (5 × 104) were placed in 96-well plates and maintained in DMEM/F12 containing 10% FBS until cell fusion reached 70%. The cells were washed and fixed with 4% methanol for 30 min and then added 0.1% Triton for 30 min. A similar protocol to IHC was utilized for staining. The ratio of Ki-67 positive cells to the total number of cells was calculated. Each experiment was performed in triplicate. The experiments were repeated three times. Each experiment was performed in triplicate and repeated three times. Each experiment was performed in triplicate and repeated three times.
Terminal deoxynucleotidyl transferase dUTP nick end labeling assay
The tumors were fixed in 4% paraformaldehyde, and the paraffin-embedded sections (4 µm) were deparaffinized and hydrated in alcohol. After being treated with 3% H2O2 in methanol for 15 min, samples were incubated with proteinase K (20 µg/mL in 10 mM Tris/HCl, pH 7.4–8.0; Exiqon, Denmark) for 20 min. The sections were washed in phosphate-buffered saline (PBS) and incubated with terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) solution, using an apoptosis detection kit (Roche Applied Science, USA) and according to the manufacture’s instruction. After washing three times in PBS, the sections were stained with DAB (ZSGB-BIO) for 5 min. The sections were counterstained with hematoxylin and observed under light microscope. Positive staining resulted in a brown reaction product.
Subcutaneous xenografts in nude mice
The effects of HeLa-derived Ang-1 and -2 in vivo were examined in a murine xenograft model. For the model, 4- to 6-week-old male and female immunodeficient BALB/c (nu/nu) mice, weighing 18–22 g, were purchased from Beijing HFK Bioscience Co., Ltd. (China). In all, 24 mice received subcutaneous injection of 3 × 106 HeLa cells and were randomly divided into four groups after 12 days of inoculation. The mice were treated as follows: 10% glucose (solvent control), in vivo-jetPEI (transfection control), Ang-1siRNA + in vivo-jetPEI (siAng-1 group), or Ang-2 siRNA + in vivo-jetPEI (siAng-2 group). All agents were administered intratumorally every 2 days for a total of six times. The tumor size was monitored every 3 days by vernier caliper until 40 days after the last injection, when mice were sacrificed. Tumor samples were embedded with paraffin and were analyzed.
Statistical analysis
Statistical analysis was carried out with SPSS 13.0 statistic software (SPSS, Chicago, IL, USA). Unpaired, two-tailed Student’s t-tests were used for comparisons between two groups. Factor expression was analyzed by non-parametric tests (Mann–Whitney U-test). Rates were compared by chi-squared test. Correlations were analyzed with Spearman’s correlation statistics. Survival analyses were performed and Kaplan–Meier survival curves were generated; the survival distributions were compared by log-rank test. All tests were two-tailed, and a p value of less than 0.05 was considered to be statistically significant.
Results
Expression and prognosis of angiopoietins and Tie receptors in cervical cancer
The expression of angiopoietins and Tie receptors was detected in 80 invasive cervical cancer tissues and 19 normal cervix tissues by IHC. Given that the expression patterns and functions of angiopoietins and Tie receptors in epithelia may be different from vascular ECs, the IHC staining signals in epithelia and endothelia were evaluated separately. Ang-1, Ang-2, Tie-1, and Tie-2 were highly expressed in cervical cancer epithelial cells, while normal cervical epithelia exhibited negative or weakly positive staining (Figure 1(a)–(d)). In tumor-related endothelial cells (TRECs), Tie-2 expression was increased compared with ECs in normal cervical tissues. The expression of Ang-1, Ang-2, and Tie-1 in ECs did not differ between tumor and normal cervix (Figure 1(a)–(d)).
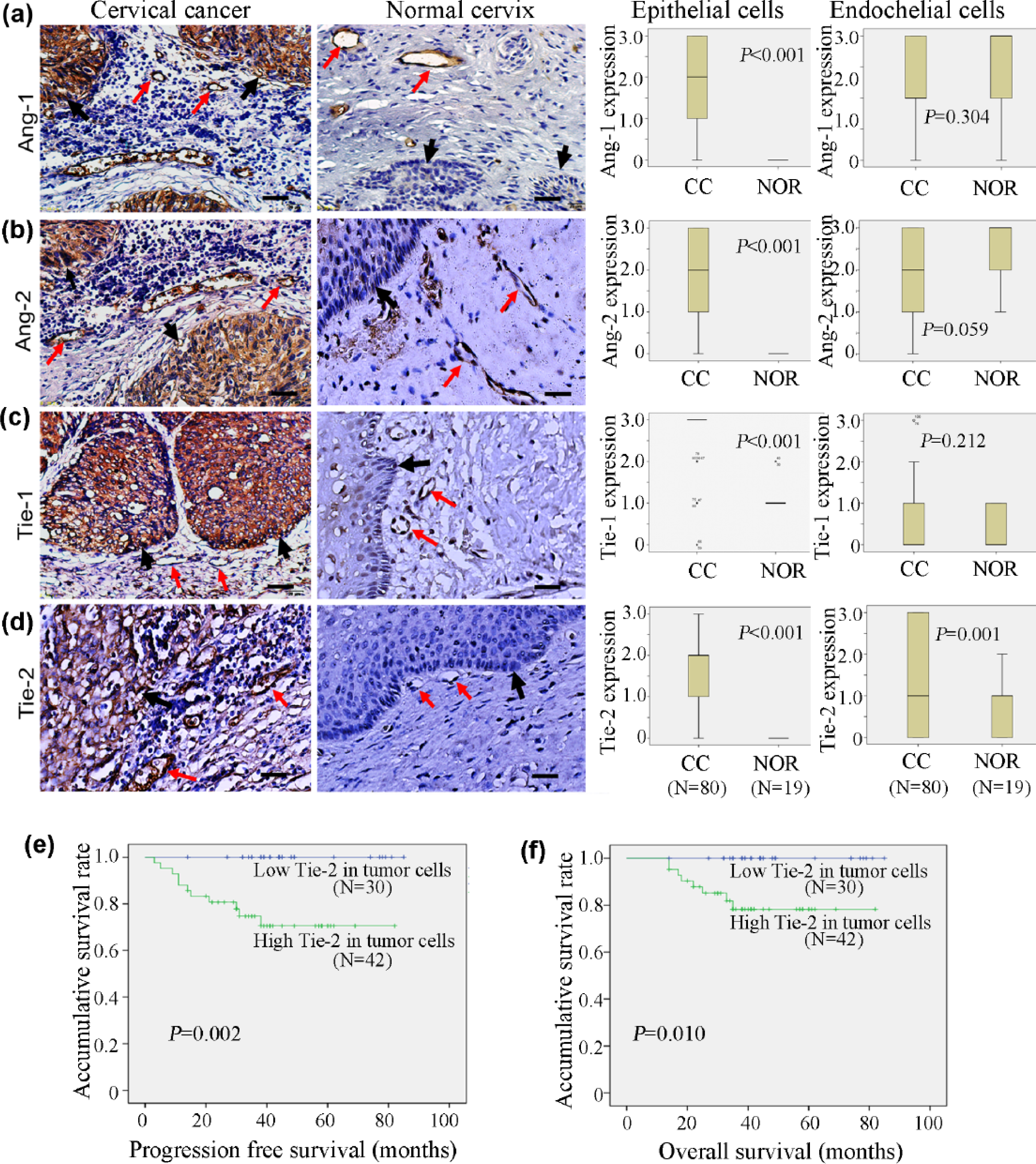
Angiopoietin and Tie receptor protein expression is elevated in cervical cancer tumor cells with Tie-2 predicting poorer prognosis. Representative immunohistochemical staining and quantification of (a) Ang-1, (b) Ang-2, (c) Tie-1, and (d) Tie-2 in cervical cancer cells (first column) versus normal cervical cells (second column). Black arrows denote positively stained epithelial cells, whereas red arrows denote positively stained endothelial cells, all appearing brown (scale bar = 50°µm). Quantification of staining intensity of epithelial (third column) and endothelial (fourth column) cells (0 = no staining; 1 = weak staining; 2 = moderate staining; 3 = strong staining) shown as box plot with p value inlaid (n = 35 (cervical cancer) and 19 (normal cervical)). Kaplan–Meier survival analysis of (e) progression-free survival and (f) overall survival among cervical cancer patients with positive or negative Tie-2 expression in tumor cells (scale bar = 500°µm).
The prognostic value of angiopoietins and Tie receptors was evaluated in 72 cervical cancer patients with follow-up data. Of the 72 patients, 11 suffered from recurrence and 8 died. Ang-1, Ang-2, and Tie-1 in tumor cells and TRECs did not show significant prognostic value. However, we found that high Tie-2 expression in tumor cells was significantly associated with shorter PFS (Figure 1(e), p = 0.002) and OS (Figure 1(f), p = 0.010). In Tie-2 positive expression group, 11 of 42 (26.2%) patients developed recurrent disease and 8 (19.1%) patients died; the estimated 3-year PFS and OS were 70.6% and 78.3%, respectively, while no recurrence or death occurred in the negative Tie-2 expression group.
The increased expression of Ang-1, Ang-2, Tie-1, and Tie-2 in cervical cancer cells was also analyzed in cell lines. The mRNA levels of Ang-1, Ang-2, Tie-1, and Tie-2 were significantly elevated in SiHa, HeLa, and C33A cell lines compared with normal cervical epithelial cells obtained from normal cervix tissues by LMD (Figure 2(a)). ELISA assays revealed that similar to HUVECs, SiHa, HeLa, and C33A cells secreted soluble Ang-1 and Ang-2 (Figure 2(b)). The expression pattern of Ang-1, Ang-2, and Tie receptors in cervical squamous cancer cells and TRECs suggests a possible autocrine and paracrine role of angiopoietins in cervical squamous cell cancer.

Angiopoietin and Tie mRNA is increased in multiple cervical cancer cell lines. (a) Quantification of mRNA levels of Ang-1, Ang-2, Tie-1, and Tie-2 in SiHa, HeLa, and C33A cell lines relative to normal cervical epithelial cells obtained from normal cervix tissues by laser microdissection. (b) Secreted Ang-1 and Ang-2 concentrations were determined by ELISA of culture media from HUVECs, SiHa, HeLa, and C33A cells. Data are shown as mean ± SD (n = 3; *p < 0.05, **p < 0.01).
Ang-1 and Ang-2 have no effects on cervical cancer cell proliferation in vitro
We next pursued siRNA-based Ang-1 and -2 loss-of-function experiments. The mRNA expression of Ang-1 and -2 was significantly downregulated 74% and 72% (HeLa), 79% and 75% (SiHa), and 74% and 76% (C33a), respectively (Supplementary Figure 1(a)). EdU (Supplementary Figure 1(b) and (c)) and Ki-67 cell proliferation assays (Supplementary Figure 1(d) and (e)) revealed that the repression of Ang-1 or Ang-2 had no significant effects on the proliferation of cervical cancer cells. Similarly, we found no significant alteration in cell proliferation after stimulation with rhAng-1 and -2 (Supplementary Figure 1(f)–(h)).
Ang-1 and Ang-2 promote cervical cancer cell migration and invasion in vitro
Wound closure assay showed that downregulation of Ang-1 and -2 with siRNA significantly reduced the distance of HeLa, SiHa, and C33a cell migration at 12 and 24 h relative to controls (NC siRNA; Figure 3(a), Supplementary Figure 2(a), Supplementary Table 2). Transwell migration assay also revealed that decreased Ang-1 and -2 expression significantly attenuated cell migration in three different cervical cancer cell lines, including HeLa, SiHa, and C33A (Figure 3(b), Supplementary Figure 2(b), Supplementary Table 3). Additionally, we studied the contribution of Ang-1 and -2 to cervical cancer cell invasion with Transwell invasion assay. The invasion of Ang-1- and Ang-2-silenced cervical cancer cells was strongly reduced relative to controls in HeLa, SiHa, and C33A (Figure 3(c), Supplementary Figure 2(c), Supplementary Table 3).

Ang-1 and Ang-2 promote cervical cancer cell migration and invasion in vitro. (a) Representative image from the wound-healing assay at times 0, 12, and 24 h and quantitative analysis of migration distance of HeLa (top), SiHa (middle), or C33A (bottom) cervical cancer cell lines transfected with NC, Ang-1, and -2 siRNA. (b) Representative image of Transwell migration assay and quantitative analysis of number of migrated cells after transfected with NC, Ang-1, and -2 siRNA. (c) Representative image of Transwell invasion assay and quantitative analysis of invasion rates of cells transfected with NC, Ang-1, and -2 siRNA. (d) Quantitative analysis of migration distance of cells after treated with 0.5% BSA as a control or stimulation with rhAng-1 and -2 at 0, 12, and 24 h. Quantitative analysis of (e) migrating cells and (f) invasion rate of cells after treated with 0.5% BSA as a control or stimulation with rhAng-1and -2, respectively. Data are shown as mean ± SD (n = 3; scale bar = 500°µm; *p < 0.05, **p < 0.01, and ***p < 0.001).
Conversely, increasing Ang-1 and -2 by administering rhAng-1 or -2 increased cervical cancer cell migration and invasion relative to 0.5% bovine serum albumin (BSA) exposed to cervical cancer cells. Specifically, wound closure assay showed that compared with control, higher Ang-1 and -2 levels increased the HeLa, SiHa, and C33A migration distances at 12 and 24 h (Figure 3(d), Supplementary Figure 2(d), Supplementary Table 4). Transwell migration assay showed similar results (Figure 3(e), Supplementary Figure 2(e), Supplementary Table 5). Transwell invasion assay showed that elevated Ang-1 and -2 both significantly promoted cervical cancer cell invasion compared with respective controls (Figure 3(f), Supplementary Figure 2(f), Supplementary Table 5).
Downregulation of angiopoietin attenuates tumor growth in vivo
In order to explore whether cervical cancer cell–derived Ang-1 and -2 promote tumor growth, we established subcutaneous xenograft models of cervical cancer in nude mice. The expression of Ang-1 and Ang-2 was repressed by in vivo siRNA transfection and confirmed by quantitative real-time polymerase chain reaction (qRT-PCR; Supplementary Figure 3). The growth of cervical cancer xenografts was slowed in both siAng-1 and siAng-2 groups relative to control groups (Figure 4(a)–(c)). Expression of vimentin, an interfilament protein associated with tumor metastasis, was decreased significantly, and TUNEL-positive cells, an apoptotic index, was increased significantly, while the expression of Ki-67 was not affected (Figure 4(d)), consistent with our in vitro experimental results.

Downregulation of angiopoietin attenuates tumor growth in vivo. Subcutaneous cervical cancer xenografts in athymic nude mice after 12 days of HeLa cell (3 × 106) inoculation were treated as follows: 10% glucose (solvent control), in vivo-jet PEI (Glu + PEI, transfection control), and Ang-1 or -2 siRNA + in vivo-jetPEI (siRNA Ang-1 or siRNA Ang-2 group, respectively; four groups, n = 6). All agents were administered intratumorally every 2 days for up to six times. The mice were sacrificed 40 days after injecting medicine. (a) Images of nude mice with subcutaneous tumors at the last day of experiment. (b) Images of tumors excised from the mice of each group. (c) Growth curves of the tumor xenografts. The tumor size was monitored every 3 days by vernier caliper. (d) Representative images of H&E staining and immunohistochemical staining of Ang-1, Ang-2, Ki-67, and vimentin (positive cells all stain brown) from the removed tumor samples in glucose, Glu + PEI, siRNA Ang-1, or siRNA Ang-2 groups. Counterstained with Gill’s hematoxylin (scale bar = 500°µm). (e) Representative images of H&E staining and immunofluorescence staining of endomucin (red) and α-SMA (green) from the removed tumor samples of glucose, Glu + PEI, siRNA Ang-1, or siRNA Ang-2 group. Endomucin and α-SMA co-staining appears yellow (scale bar = 50°µm). Quantitative analysis of the number of (f) endomucin-positive vessels and (g) the cover rate of α-SMA in endomucin-positive microvessels in control and siRNA Ang-1 or siRNA Ang-2 group (n = 3). Representative images of (h) CD31-positive microvessel to determine microvessel density (MVD) in control and (i) siRNA Ang-1 or siRNA Ang-2 groups and quantitative analysis of MVD (n = 3). Red arrows denote positively stained microvessels. Counterstained with Gill’s hematoxylin (scale bar = 500°µm). Data are shown as mean ± SD (*p < 0.05, ***p < 0.001).
We also investigated the alterations in tumor vasculature and periendothelial supporting cells in xenografts. Immunostaining for EC marker endomucin was significantly decreased in the siAng-1 group and siAng-2 group relative to the infection control group (19.67 ± 3.21 vs 33.33 ± 4.93, pAng-1 = 0.0213; 17.33 ± 1.53 vs 33.33 ± 4.93, pAng-2 = 0.0609). However, the cover rate of the muscle cell marker α-SMA in endomucin-positive microvessels had no significant difference (Figure 4(e)–(g)). The density of microvessels was also significantly decreased in the siAng-1 and siAng-2 groups compared with infection control group (15.53 ± 1.5 vs 29.53 ± 2.53, pAng-1 = 0.0104; 13.4 ± 3.02 vs 29.53 ± 2.53, pAng-2 = 0.0021; Figure 4(h) and (i)).
Discussion
Tumor angiogenesis is the most fundamental trait for tumor growth. 18 Tumor nodules cannot derive nutrients through diffusion when greater than 1–2 mm 3 .18,19 Thus, tumors exploit multiple avenues to recruit blood vessels. Over-expressed Ang-1 derived by tumor cells has been shown to stimulate tumor angiogenesis in astrocytomas, glioblastoma, and colorectal cancer, while elevated Ang-2 enhanced vasculature formation and positively correlated with tumor growth in colorectal, gastric, hepatoma, breast cancer, glioblastoma, and malignant melanoma.9,12,13,20 As a result, anti-angiogenic therapies are increasingly being studied as adjuvant therapy for multiple types of cancer. In cervical cancer, anti-VEGF adjuvant therapy has improved survival of some patients affected with advanced or recurrent disease. 6 In this study, we found that cervical cancer cells highly express angiopoietins and Ties, and positive Tie-2 expression in tumor cells predicted poorer prognosis; the tumor cell–derived angiopoietins promote tumor cell migration and invasion in vitro and could therefore be alternate therapeutic targets for treatment of advanced or recurrent cervical cancer.
We found that in addition to ECs, cervical cancer cells also expressed abundant Ang-1, Ang-2, Tie-1, and Tie-2 that contributed to the invasive phenotype promoting tumor progression. Similar results were reported in gastric cancer as 67.4%, 68.5%, 77.5%, 84.3%, and 52.8% of gastric adenocarcinomas showed positive staining in the cytoplasm of carcinoma cells for the Tie-1, Tie-2, Ang-1, Ang-2, and Ang-4, respectively, and Ang-1 and -2 levels were higher in advanced disease than in early-stage disease. 21 Moreover, circulating levels of soluble Ang-2 tightly correlated with tumor progression and shorter survival in melanoma and neuroendocrine tumor patients.22,23 Melanoma cells were Tie-2 positive and secreted Ang-2 that acted as an autocrine regulator of tumor cell migration and invasion. 23 The co-expression of angiopoietins and Tie-2 in cervical cancer cells demonstrated in this study suggests an autocrine angiopoietin/Tie-2 action. Further study to determine the degree to which this autocrine action is involved in cervical cancer cell migration and invasion is warranted.
The mechanisms of Ang-1 and -2 affecting invasion and migration remain elusive. In addition to Tie-2, integrin pathways are involved in the Ang-2-mediated cell migration in both endothelial and non-ECs. In Tie-2-negative ECs, Ang-2 binds to αvβ3, α5β1, and αvβ5 integrins and activates the downstream focal adhesion kinase (FAK), which in turn leads to migration of ECs and sprouting angiogenesis. 24 The Ang-2 expressed by Tie-2-deficient human breast cancer cells promoted tumor metastasis to distant lymph nodes and lung in vivo by stimulating breast cancer cell migration and invasion through the α5β1 integrin–mediated pathway regulating expression of E-cadherin and Snail and phosphorylation of Akt and glycogen synthase kinase-3β (GSK-3β).25,26 However, Tie-2 has stronger affinity than integrin receptors toward Ang-2, and the Ang-2-mediated β1 integrin activation can be inhibited by Tie-2 in a kinase-independent manner. 27 Thus, Ang-2 acting via Tie-dependent or -independent pathways may be dependent on the expression pattern of Tie and integrin receptors on cell membrane. Our HeLa xenograft model showed that inhibiting Ang-1 or -2 resulted in attenuated tumor growth and decreased vimentin expression. In breast cancer, a negative association between Ang-2 and E-cadherin expression levels has been found. 25 Loss of E-cadherin and acquisition of vimentin are hallmarks of epithelial-to-mesenchymal transition (EMT) that are recognized as a critical step in tumor metastasis initiation. These results implicate EMT in angiopoietin-induced tumor progression; we are devoting research for verifying this mechanism.
Previous reports on various types of cancer, such as hepatocellular carcinoma, gastric carcinoma, breast cancer, and melanoma, have shown that elevated Ang-1 or -2 in these tumor cells resulted in vessel destabilization, pericyte dropout, and stimulated tumor angiogenesis.9,12,13 Similarly, our experiments showed that cervical cancer cell–derived Ang-1 or -2 played a critical role in regulating tumor angiogenesis. The molecular mechanism of angiopoietin-involved angiogenesis is complicated. Angiopoietins activate downstream Tie-2 signaling pathway and phosphorylate Tie-2 and then bind and activate several other effectors, including tyrosine kinase–related Dok-related protein (DOKR), endothelial nitric oxide synthase (eNOS), SH2 domain–containing phosphatase (SHP2), growth factor receptor–bound protein 2 (GRB2), and the p85 subunit of PI3K19, ultimately contributing to EC survival, proliferation, migration, and permeability.9,10,12 In addition to the Tie-2 pathway, the integrin-mediated pathway is also involved in Ang-2-induced angiogenesis by direct interaction between Ang-2 and integrins that leads to phosphorylation of the integrin adaptor FAK and subsequent Ras-related C3 botulinum toxin substrate 1 (RAC1) for activation, migration, and sprouting angiogenesis.24,28
Ang-2 has been an attractive target for improved anti-angiogenic tumor therapy, and several Ang2-targeting drugs are in clinical trials. 20 A recent study revealed that antibodies inhibiting the interaction between Ang-1, Ang-2, and Tie-2 receptor, such as AMG 380 and AMG 780, offered greater superiority in inhibiting tumor growth, viable tumor fraction, and tumor EC proliferation for advanced solid tumors compared with antibody solely interrupting the binding of Ang-2 and Tie-2. 29 Moreover, clinical trials for multiple solid tumors indicated that dual inhibition of Ang-1 and -2 had more advantages than only blocking Ang-2/Tie signaling.30,31 We found that Ang-1 and -2 were highly expressed in cervical cancer cells and downregulating Ang-1 and -2 inhibited tumor growth, suggesting that dual inhibition of Ang-1 and -2 may be a potential anti-angiogenic pharmaceutical target for the treatment of cervical cancer.
In conclusion, angiopoietins are highly expressed in cervical cancer cells. The elevated expression stimulates cervical cancer cell migration, invasion, and tumor angiogenesis, promoting cervical cancer progression. Dual inhibition of Ang-1 and Ang-2 may be a promising strategy for anti-angiogenic therapy in advanced or recurrent cervical cancer.
Footnotes
Acknowledgements
P.Y. and N.C. have contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This report was supported by the China Postdoctoral Science Foundation (grant no. 81472443).
