Abstract
SOX6 plays important roles in cell proliferation, differentiation, and cell fate determination. It has been confirmed that SOX6 is a tumor suppressor and downregulated in various cancers, including esophageal squamous cell carcinoma, hepatocellular carcinoma, and chronic myeloid leukemia. Netrin-1 is highly expressed in various human cancers and acts as an anti-apoptotic and proangiogenic factor to drive tumorigenesis. The role of SOX6 and netrin-1 in regulating the growth of ovarian tumor cells still remains unclear. Real-time polymerase chain reaction and western blot were used to determine the SOX6 messenger RNA and protein levels, respectively, in ovarian cancer cells and tumor tissues. Stable transfection of SOX6 was conducted to overexpress SOX6 in PA-1 and SW626 cells. Cell viability was measured by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay. Invasion of ovarian cancer cells and migration of human umbilical vein endothelial cells were confirmed by Transwell assays. To overexpress netrin-1, ovarian cancer cells with SOX6 restoration was transduced with netrin-1 lentiviral particles. PA-1 xenografts in a nude mice model were used to conduct in vivo evaluation of the role of SOX6 and its relationship with netrin-1 in tumor growth and angiogenesis. In this study, we found significantly reduced SOX6 levels in PA-1, SW626, SK-OV-3, and CaoV-3 ovarian cancer cell lines and human tumor tissues in comparison with normal human ovarian epithelial cells or matched non-tumor tissues. SOX6 overexpression by stable transfection dramatically inhibited proliferation and invasion of PA-1 and SW626 cells. Also, conditioned medium from PA-1 and SW626 cells with SOX6 restoration exhibited reduced ability to induce human umbilical vein endothelial cells migration and tube formation compared with conditioned medium from the cells with transfection control. Furthermore, an inverse relationship between SOX6 and netrin-1 expression was observed in PA-1 and SW626 cells. Overexpression of netrin-1 in ovarian cancer cells with forced SOX6 expression remarkably abrogated the inhibitory effect of SOX6 on proliferation, invasion of the cells, and tumor xenograft growth and vascularity in vivo. Human umbilical vein endothelial cell migration and tube formation were enhanced in the conditioned medium from the ovarian cancer cells transduced with netrin-1 lentivirus particles. Our observations revealed that SOX6 is a tumor suppressor in ovarian cancer cells, and SOX6 exerts an inhibitory effect on the proliferation, invasion, and tumor cell-induced angiogenesis of ovarian cancer cells, whereas nerin-1 plays an opposite role and its expression is inversely correlated with SOX6. Moreover, our findings suggest a new role of SOX6 and netrin-1 for understanding the progression of ovarian cancer and have the potential for the development of new diagnosis and treatment strategies for ovarian cancer.
Introduction
Ovarian cancer represents the third most common type of cancer. 1 Despite the new treatments and therapies available, ovarian cancer continues to be one of the deadliest cancers among females.1–3 Unfortunately, almost 80% of ovarian cancer patients are diagnosed in the late stages, when the 5-year survival rates drop below 20%. 1 Currently, imaging and cancer antigen-125 (CA-125) test are the most common approaches for ovarian cancer screening tests. However, the two methods remain limited due to low sensitivity and specificity.4,5 Surgery and chemotherapy are the two major treatments currently available for ovarian cancer, whereas the majority of patients with advanced ovarian cancer relapse due to drug resistance. 6 Therefore, there is an urgent need for discovery and development of new diagnostic and treatment strategies for ovarian cancer to ultimately improve survival.
SOX6, a protein that binds to DNA through a high-mobility group domain that is highly conserved among species, belongs to the D subfamily of sex-determining region y–related transcription factors.7–9 SOX6 has been proven to play important roles in cell proliferation, differentiation, and cell fate determination. Studies have confirmed that SOX6 is a tumor suppressor and downregulated in various cancers, including esophageal squamous cell carcinoma, hepatocellular carcinoma, and chronic myeloid leukemia. However, the role of SOX6 in ovarian cancer and the molecular mechanisms still remains uncertain.10–12
Netrin family members, including netrin-1, prototype of netrin family, are secreted proteins that promote axon outgrowth and act as axonal guidance molecules during neural development. 13 Beside its role in regulating development of central nervous system, recent studies have established that netrin-1 is highly expressed in various human cancers, exerts anti-apoptotic effects, and has been presented as a diagnostic biomarker.14–16 There is also evidence showing that netrin-1 acts as a proangiogenic factor through Unc-5 Netrin Receptor B (UNC5B) and extracellular-signal-regulated kinase (ERK)-1/2-endothelial nitric oxide synthase (eNOS) mechanism to enhance tumor angiogenesis, thereby driving tumorigenesis.14–19 Papanastasiou et al. 20 have demonstrated that netrin-1 is strongly expressed in ovarian malignant tumors but not in non-tumor tissues, suggesting that netrin-1 may represent a new biomarker for ovarian cancer, whereas how netrin-1 regulates the growth of ovarian cancer remains uncertain. In this study, we sought to investigate the role of SOX6 and netrin-1 in regulating the biological properties of ovarian cancer cells and the relationship between SOX6 and netrin-1 expression in the cells.
Materials and methods
Cell culture and human tissues
PA-1, SW626, SK-OV-3, and CaoV-3 ovarian cancer cells were obtained from American Type Culture Collection (ATCC). PA-1 and SW626 cells were grown in Dulbecco’s Modified Eagle’s Medium (DMEM) with 10% fetal bovine serum (FBS) and antibiotic-antimycotic (Gibco). The SK-OV-3 cells were cultured in McCoy’s 5 a medium (Lonza) supplemented with 10% FBS and antibiotic-antimycotic. The CaoV-3 cultures were maintained in MEM medium (Sigma Aldrich) with 10% FBS, sodium pyruvate, and antibiotic-antimycotic. Normal human ovarian epithelial cells from Innoprot were cultured in OEpiCM. Human umbilical vein endothelial cells (HUVECs) were purchased from Lonza and cultured in Endothelial Cell Growth Medium 2 (EGM-2; Lonza). Human high-grade serous and endometrioid ovarian epithelial tumor tissues and the matched adjacent non-cancerous ovarian epithelial tissues were obtained from 26 patients at the Second People’s Hospital of Liaocheng City, Shandong, China. Human samples were handled with approval by the Ethic Commission at the Second People’s Hospital of Liaocheng City, Shandong, China.
Western blot
A volume of 100 µg of protein isolated from cells or human tissues were subjected to 8% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). Transfer was then done onto a polyvinylidene difluoride (PVDF) membrane (Millipore), which was blocked in with 5% non-fat milk and incubated with the anti-SOX6 (1: 250 dilution; R&D Systems), anti-netrin-1 (1: 5000 dilution; Abcam), or anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH; 1:30,000 dilution; Sigma Aldrich) antibodies. The blots were then washed with Tris buffered saline with Tween 20 (TBST) buffer (20 mM TRIS-HCl pH 7.6, 150 mM NaCl, and 0.1% Tween 20) and incubated with horseradish peroxidase (HRP)-conjugated anti-mouse or anti-rabbit IgG antibodies (1:10,000 dilution; Abcam). After being washed, the proteins were visualized with the enhanced chemiluminescence (ECL) western blotting substrate (Millipore).
RNA isolation and polymerase chain reaction analysis
RNA was extracted from whole cell lysates or human tissues using RNeasy Mini Kit (Qiagen), and reverse transcription was performed using a QuantiTect Reverse Transcription Kit (Qiagen). The complementary DNA (cDNA) was used for quantitative real-time polymerase chain reaction (RT-qPCR) with gene primers designed using Primer3Plus. The primers are as follows: Sox6 forward: 5′-TAAGCAACTGA TGAGGTCT C-3′, reverse: 5′-AGGCGATGGTGTGGTAGTT-3′; and GAPDH forward: 5′-ACCCACTCCTCCACCTTTGA-3′, reverse: 5′-CTGTTGCTGTAGC CAAATTCGT-3′. Real-time PCR was conducted using a MyiQ PCR detection system and SYBR mix (Bio-Rad). The relative expression levels of Sox6 transcript were quantified with the Ct method, and GAPDH was used as an internal control.
Stable transfection and transduction
PA-1 and SW626 cells were transfected with pcDNA3.1(+) or pcDNA3.1(+)-Sox6 (gift from Dr Hong Wang, Hong Kong University, China) by Lipofectamine 2000 (Thermo Fisher Scientific) as per the manufacturer’s instructions. The transfected cells were cultured with selection medium containing 500 µg/mL G418 for 8 weeks from the second day of transfection, and the selected positive colonies were expanded. To overexpress netrin-1, 1 × 106 PA-1 and SW626 cells stably transfected with Sox6 were infected with 10 µL netrin-1 lentiviral particles (Cat. no. LH804240) and control lentiviruses (cat. no. CV10002) from Vigene Biosciences according to the manufacturer’s instructions.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
PA-1 and SW626 ovarian cancer cells were seeded in 96-well culture plate (Thermo Fisher Scientific) at a density of 5 × 103 cells/well and cultured for 24 and 48 h. At the end of incubation times, the culture medium was replaced with 100 µL fresh medium containing 500 µg/mL 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma Aldrich) and incubated for 3 h at 37°C. The medium was discarded, and 100 µL dimethyl sulfoxide (DMSO) was added into each well to dissolve the formazan produced in the cells. Cell viability was determined by the colorimetric comparison of optical density (OD) at 570 nm.
Cell invasion assay
A volume of 8-µm pore Transwell inserts (Millipore) were put in each well in 24-well plates (Corning). The inserts were coated with 100 µL Matrigel (BD Biosciences; 1:1 diluted with phosphate-buffered saline (PBS)). In the inserts, 1 × 105 PA-1 or SW626 cells were placed. The lower chambers were filled with DMEM with 5% FBS. For the measurement of invasion, the cells in the inserts were removed with cotton swabs and cells that traversed the Matrigel to the lower surface of the insert were fixed with 10% formalin, stained with crystal violet and counted under a light microscope (Olympus).
HUVEC migration assay
HUVECs (1 × 105 cell per well) were seeded onto Millipore Transwell inserts with 8 µm pore size in a 24-well plate. DMEM without FBS and conditioned medium harvested from ovarian cancer cells were added to the lower wells. The cells were incubated and allowed to migrate for 4 or 8 h. The cells in the inserts were removed with cotton swabs and the cells transmigrating to the reverse side of the Transwell membrane were fixed, stained, and counted under a light microscope in 10 random fields.
Tube formation
The formation of capillary-like endothelial tubes was determined by plating primary endothelial cells on growth factor–reduced Matrigel (BD) according to manufacturer’s instructions. HUVECs were suspended in conditioned medium harvested from ovarian cancer cells. An amount of 2 × 104 HUVECs in 50 µL were added into each well coated with Matrigel and incubated for 20 h at 37°C in 5% CO2 atmosphere. Following incubation, cells were labeled by adding 50 µL/well of 8 µg/mL calcein AM in Hank’s Balanced Salt Solution (HBSS) and incubate plates for 40 min. Images were taken using a fluorescent microscope (Olympus) and quantified by ImageJ.
Tumor xenografts
Animal studies were carried out following the procedures approved by the Animal Care and Use Committee at Second People’s Hospital of Liaocheng City, Shandong, China. PA-1 cells overexpressing SOX6 or both SOX6 and Netrin-1 were injected (5 × 106 cells/mouse) subcutaneously into flanks of female nude mice at the age of 6 weeks. Tumor size was measured twice a week for 4 weeks. Animals were then sacrificed, and tumors were removed. Optimal cutting temperature (OCT) compound-embedded frozen tumor tissues were prepared to make cryosections for CD31 staining.
Statistical analysis
Data were presented as mean ± standard deviation and were representative of at least three independent experiments. Student’s paired t-tests were performed on means, and p values were calculated using GraphPad Prism; *p < 0.05 and **p < 0.01 were considered significantly different.
Results
Reduced SOX6 in ovarian cancer cells
The levels of SOX6 in PA-1, SW626, SK-OV-3, and CaoV-3 ovarian cancer cells and human normal ovarian epithelial cells (NOECs) were first determined. As shown in Figure 1(a) and (b), there were reduced SOX6 protein and messenger RNA (mRNA) expression in the cancer cell lines in comparison to that in normal cells. We then examined SOX6 expression in 26 high-grade serous and endometrioid ovarian epithelial tumor tissues and the matched non-tumor tissues and found that SOX6 was profoundly lower in 69% tumor tissues than that in the normal tissues (Figure 1(c) and (d)).
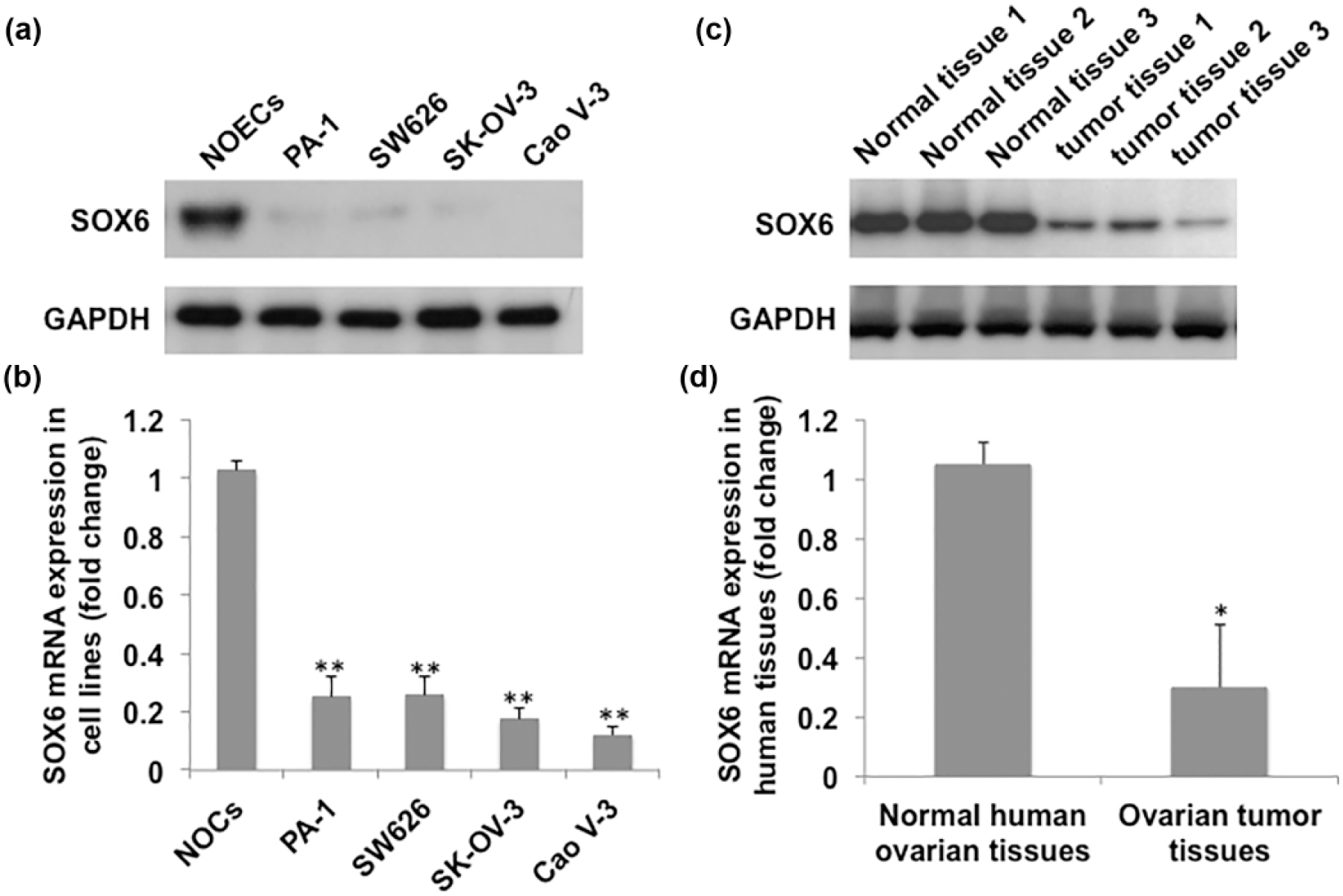
Expression of SOX6 in normal human ovarian epithelial cells; human ovarian cancer cell lines PA-1, SW626, SK-OV-3, and CaoV-3; ovarian carcinoma tissues; and the matched non-tumor tissues. (a) Western blot analysis of SOX6 in human normal ovarian epithelial cells (NOECs) and PA-1, SW626, SK-OV-3, and CaoV-3 ovarian cancer cell lines. (b) SOX6 mRNA levels (shown in fold change) in NOECs and the four ovarian cancer cell line. (c) SOX6 protein level and (d) mRNA level in ovarian carcinoma tissues and matched non-tumor tissues. GAPDH was set as internal control and relative gene expression normalized to levels of GAPDH mRNA was graphed and shown in fold change. Data represent mean ± SD of three independent experiments (*p < 0.05 and **p < 0.01 vs NOECs or matched non-tumor tissues).
Overexpression of SOX6 inhibits growth and invasiveness of ovarian cancer cells
We then sought to investigate the function of SOX6 in the ovarian cancer cells by restoration of SOX6 in PA-1 and SW626 cells. PA-1 and SW626 cells stably transfected with pcDNA3.1-Sox6 showed almost five-fold increase in SOX6 protein level compared with the cells transfected with the control vector (Figure 2(a)). SOX6 overexpression significantly inhibited growth (Figure 2(b)) and invasion (Figure 2(c)) of PA-1 and SW626 cells.

Introduction of SOX6 in PA-1 and SW626 cells significantly inhibited proliferation and invasion of PA-1 and SW626 ovarian cancer cells. (a) Overexpression of SOX6 in PA-1 and SW626 cells by stable transfection. (b) Cell proliferation of PA-1 and SW626 cells were determined by MTT assay. (c) Effect of SOX6 overexpression on invasion of PA-1 and SW626 cells. Cells transfected with control plasmid were set as control. The data were shown in fold change and represent mean ± SD of three independent experiments (*p < 0.05 vs PA-1 cells transfected with control vector).
SOX6 suppresses migration and tube formation of endothelial cells induced by the conditioned medium from ovarian cancer cells
Tumor growth and progression rely on angiogenesis initiated by cancer cells for the delivery of nutrients, oxygen, and growth factors. Angiogenesis allows the tumor cells to invade nearby tissue and to gain access to systemic circulation. To investigate whether SOX6 restoration in ovarian cancer cells has effect on angiogenesis induced by the cancer cells, HUVEC migration and tube formation were performed. As shown in Figure 3(a) and (b), the conditioned medium from PA-1 and SW626 cells with restoration of SOX6 exhibited reduced ability to induce HUVEC migration and tube formation compared with the cells transfected with control vector.
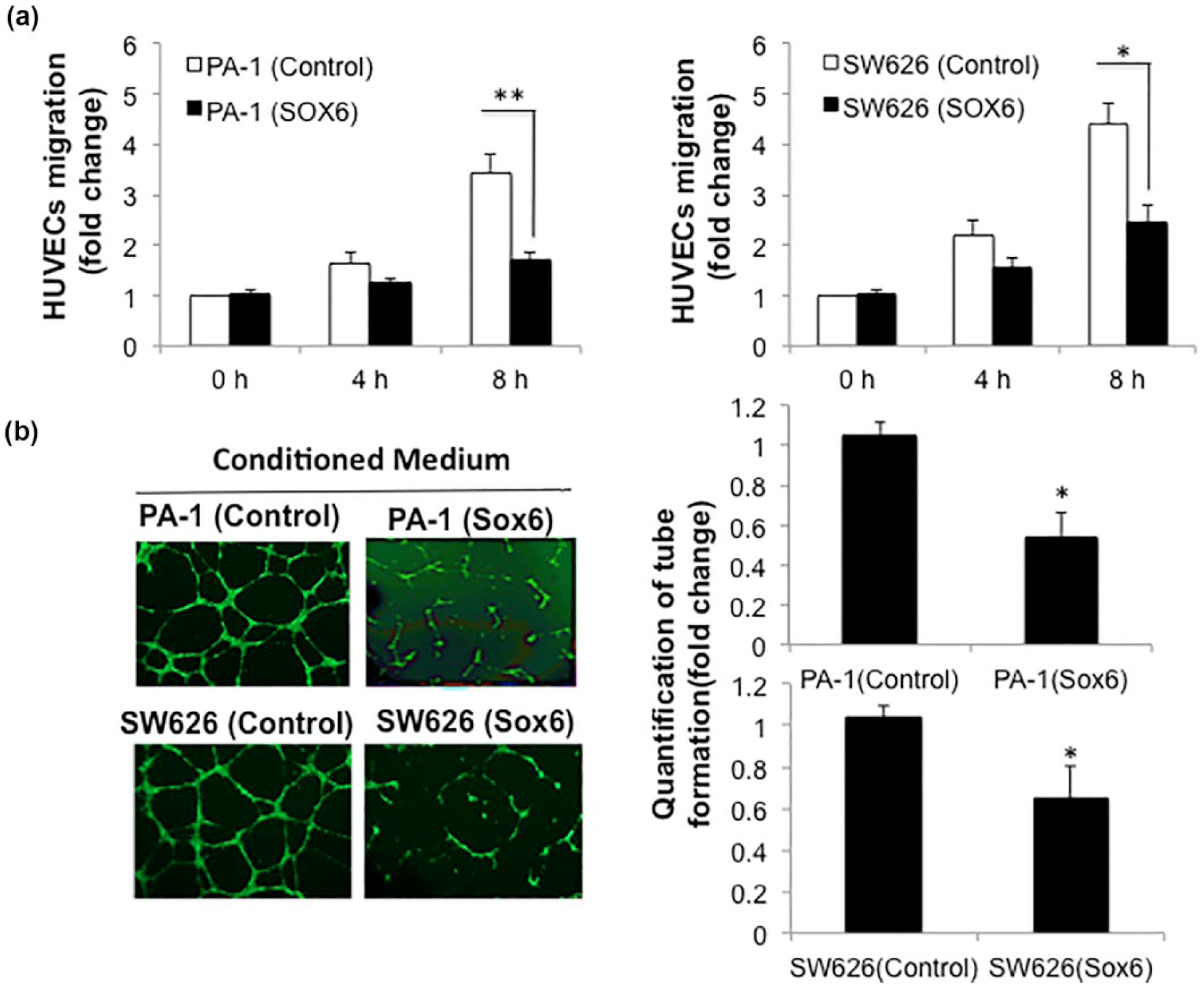
SOX6 expression in PA-1 and SW626 cells results in inhibition of endothelial cell chemotaxis and tube formation. (a) The conditioned medium from PA-1 or SW626 cells with restoration of SOX6 was used as chemoattractant for HUVECs in migration assay. (b) HUVECs were plated on reconstituted basement membrane Matrigel in conditioned medium from PA-1 or SW626 cells overexpressing SOX6 by stable transfection for formation of capillary tubes. Quantification of the results of the tube formation assay was conducted by measuring and summing the length of all tubular structures observed in 10 random fields. The data were shown in fold change and represent mean ± SD of three independent experiments (*p < 0.05 and **p < 0.01 vs cells transfected with control vector).
The inhibitory effect of SOX6 and stimulatory effect of netrin-1 on invasion and angiogenesis in ovarian cancer cells
It has been reported that netrin-1 is strongly expressed in ovarian malignant tumors but not in non-tumor tissues. We first determined the levels of netrin-1 in PA-1, SW626, SK-OV-3, and CaoV-3 ovarian cancer cells and human normal ovarian epithelial cells. We found that netin-1 was highly expressed in ovarian cancer cells but not in the normal ovarian epithelial cells (Figure 4(a)). We then examined the levels of netrin-1 in PA-1 and SW626 cells with forced SOX6 overexpression by transfection. It is interesting that restoration of SOX6 expression in the cells dramatically reduced netrin-1 expression as shown in Figure 4(a). The inverse relationship between SOX6 and netrin-1 expression suggested that the function of SOX6 in ovarian cancer cells might be correlated with netrin-1. To determine whether netrin-1 expression was involved in the regulation of ovarian cancer cell invasion and angiogenesis and its effect on inhibition by SOX6, PA-1 cells stably expressing SOX6 were transduced with lentivirus expressing netrin-1. Transduction of netrin-1 lentivirus significantly increased netrin-1 expression in PA-1 and SW626 cells with forced SOX6 overexpression (Figure 4(a)). The inhibition of proliferation and invasion of PA-1 cells and induced tube formation by PA-1 cells with SOX6 restoration was significantly abrogated by transduction of netrin-1 lentivirus (Figure 4(b)–(d)).

The inhibitory effect of SOX6 on invasion and angiogenesis in ovarian cancer cells is through downregulating netrin-1. (a) Netrin-1 is highly expressed in human and PA-1, SW626, SK-OV-3, and CaoV-3 ovarian cancer cell lines in comparison to normal ovarian epithelial cells (NOECs). Netrin-1 expression was downregulated in PA-1 and SW626 cells with forced SOX6. Restoration of netrin-1 by transduction of netrin-1 lentiviral particles significantly increased netrin-1 levels suppressed by SOX6 in the cells. (b) Netrin-1 overexpression by transduction markedly abrogated the inhibition of cell proliferation and (c) invasiveness by SOX6 in PA-1 cells. Cell proliferation or invasiveness of the cells with forced SOX6 expression only served as control. (d) HUVECs were plated on Matrigel in conditioned medium from PA-1 cells expressing SOX6 by stable transfection or the cells with both transfection of SOX6 and transduction of netrin-1 lentiviruses for formation of capillary tubes. Quantification of the results of the tube formation assay was conducted by measuring and summing the length of all tubular structures observed in 10 random fields and tube formation induced by conditioned medium from PA-1 cells with only SOX6 restoration was set as control. The data were shown in fold change and represent mean ± SD of three independent experiments (*p < 0.05 and **p < 0.01 vs cells with SOX6 transfection).
SOX6 and netrin-1 play opposite roles in is tumor growth in vivo
PA-1 cells stably transfected with SOX6 and transduced with lentivirus expressing netrin-1 were injected subcutaneously into nude mice. Tumor volume was recorded for 4 weeks. PA-1 cells overexpressing SOX6 grow slowly as xenografts in the nude mice in comparison with PA-1 cells transfected with control vector. Transduction of netrin-1 lentivirus attenuated the inhibitory effect of SOX6 restoration in tumor growth (Figure 5(a)). Enhanced tumor angiogenesis demonstrated by anti- CD31 staining in tumors derived from the PA-1 cells co-overexpressing SOX6 and netrin-1 was observed when compared with tumors composed of the PA-1 cells with only SOX6 restoration (Figure 5(b)). These results indicate that SOX6 and netrin-1 are contradictory in controlling ovarian tumor growth and angiogenesis.

SOX6 reduced growth of PA-1 tumors in nude mice through downregulating netrin-1 expression. (a) Quantification of tumor volume from day 0–28 post-engraftment of the PA-1 cells overexpressing SOX6 with or without transduction of netrin-1 lentivirus. (b) Quantification of CD31 staining in tumors derived from PA-1 cells expressing SOX6 or co-overexpressing SOX6 and netrin-1 was conducted by calculating the CD31-positive cells in 10 random fields. The tumor volume and CD31 staining in PA-1 cells with only SOX6 restoration was set as control. The data were shown in fold change and represent mean ± SD of three independent experiments (*p < 0.05; **p < 0.01).
Discussion
The tumor suppressor role of SOX6 has been studied in various cancer types.10–12 However, the function of SOX6 in ovarian cancer has not been fully elucidated. In this study, we found that SOX6 was frequently downregulated in ovarian cancer cell lines and primary ovarian tumor tissues in comparison with the normal ovarian epithelial cells and matched non-tumor tissues, indicating that SOX6 may negatively regulate the growth and progression of ovarian tumor.
We first examined the function of SOX6 by measuring the cell growth activity in the ovarian cancer cells with forced SOX6 overexpression. Restoration of SOX6 in the cells inhibited cell growth. As SOX6 acts as a transcription factor, it may inhibit the transcription or function of factors involved in cell cycle in ovarian cancer cells. The growth inhibition in ovarian cancer cells might be caused by cell-cycle arrest. G1/S phase transition is a major checkpoint for cell-cycle progression, which is negatively regulated by p21WAF1/CIP1, a cyclin-dependent kinase (CDK) inhibitor, through inhibiting CDK and activity of cyclin D-CDK complex.21–23 Qin et al. 10 revealed that SOX6 was able to upregulate p21WAF1/CIP1 and downregulate cyclin D1/CDK4 complex and cyclin A to inhibit G1-/S-phase transition in esophageal squamous cell carcinoma cancer cells. Recent studies have demonstrated that cyclinD1 has been associated with tumor invasion and metastasis in clinical studies and in vivo experiments, and cyclin D1 regulates invasion and metastasis through the phosphorylation of paxillin.24–26 In addition to the suppression of cell growth by SOX6 in the ovarian cancer cells, inhibitory effect of SOX6 on ovarian cancer cell invasion was also observed in this study, suggesting that SOX6 may prevent metastasis of ovarian tumors. The SOX family of transcription factors has emerged as modulators of canonical Wnt/β-catenin signaling in diverse development and disease contexts. 27 Iwai et al. 28 demonstrated that Wnt-β-catenin pathway is critical for invasion and migration of tumor cells through inducing matrix metalloproteinase-7 (MMP-7) and Rho family-mediated E-cadherin redistribution and actin cytoskeleton rearrangement. Qin et al. 10 and Iguchi et al. 9 have reported that SOX6 suppressed Wnt/β-catenin signaling pathway through downregulating β-catenin expression and binding to β-catenin in tumor cells.
In this study, it is interesting that the conditioned medium from PA-1 and SW626 cells overexpressing SOX6 through stable transfection exhibited reduced ability to induce HUVECs migration and tube formation. These findings indicated that SOX6 participates in the regulation of angiogenesis in the tumor microenvironment. Furthermore, we found that netrin-1 was strongly expressed in the ovarian tumor cells and there was an inverse relationship between SOX6 and netrin-1 expression in the ovarian tumor cells. We postulated that the changes in netrin-1 levels in the tumor cells with SOX6 restoration might contribute to ovarian tumor cell–induced angiogenesis. Previous studies have shown that netrin-1 is highly expressed in various human cancers including ovarian malignancies and has been identified as a biomarker and a proangiogenic factor to indicate the onset and progression of cancer. Netrin-1 enhances tumor initiation, promotion, and progression through suppressing apoptosis and promoting angiogenesis.14–16,29,30 Recently, Huang et al. 31 demonstrated that netrin-1 reduced p21 and p27 expression levels in pancreatic cancer cells. Although we found an inverse relationship between SOX6 and netrin-1 expression and opposite effects exerted by SOX6 and Netrin-1 on the biological properties of ovarian cancer cells, it is still unclear whether netrin-1 is directly regulated by SOX6 or whether the effects of netrin-1 and SOX6 in our study are “true, true, and unrelated.” Further studies need to be carried out in order to confirm whether netrin-1 is a downstream target of SOX6 and directly regulated by SOX6 in ovarian cancer cells.
Taken together, our study showed the evidence that SOX6 is a tumor suppressor in ovarian cancer, and netrin-1 plays a stimulatory role in the growth and progression of ovarian cancer. The inverse correlation between SOX6 and netrin-1 expression in ovarian cancer cells indicates that netrin-1 might be a downstream target of SOX6. A better understanding of the role of SOX6 and netrin-1 will have translational potential for early diagnosis and may lead to the development of new treatment strategy against malignant ovarian tumor.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
