Abstract
Colorectal cancer is one of the most common cancers in the world, and liver metastasis is the leading direct cause of cancer-related deaths in colorectal cancer. MicroRNA is involved in tumor metastasis in many aspects; mounting studies have shown that microRNAs play important roles in colorectal liver metastasis. Although lots of reviews about the association between microRNAs and colorectal cancer metastasis have been published, the reviews specifically focusing on microRNAs and colorectal liver metastasis are still lacking in the literature. To address this issue, here, we summarize the underlying mechanisms of microRNAs in colorectal liver metastasis and explore their potential clinical applications in this aspect.
Introduction
Colorectal cancer (CRC) is the third most commonly diagnosed cancer in males and the second in females. 1 During the recent 30 years, thanks to the achievement of new chemotherapeutic drugs and radiotherapeutic techniques, great improvement has been made in the CRC treatment. Despite of these, there are still at least 50% CRC patients who develop liver metastasis. 2 Liver is the most commonly metastasized organ in CRC. When the primary tumor gets diagnosed, synchronous liver metastasis can be found in up to 25% of the patients, and a further 40%–50% of patients will develop metachronous liver metastasis even after radical resection of the primary tumor.3,4 Colorectal liver metastasis (CRLM) can be considered as the major direct cause of cancer-related deaths in CRC. Curative resection is the most effective treatment of CRLM 4 but is not applicable in more than 70% of patients due to the size, number, location, and extent of involvement of liver metastatic lesions. 5 Therefore, the 5-year survival rate of metastatic CRC remains at about 15%, and the clinical benefit rate (CBR) remains at low level, too. 6 For CRLM, researchers need to develop new therapeutic methods, while a better understanding of the biological mechanisms underlined this process will facilitate this goal.
MicroRNA (miRNA) is a class of endogenous non-encoding RNA molecules of 20–25 nucleotides; they are involved in the post-transcriptional regulation of gene expression in wide physiological and pathological processes, such as cell differentiation, proliferation, and apoptosis, and so on. The biogenesis and function of miRNA in cells are shown in Figure 1. 7 In recent years, substantial evidences have shown that miRNAs are widely involved in human carcinogenesis, and more than 50% of miRNAs are located in the tumor-related genomic regions or fragile sites. 8 So far, more than 1000 miRNAs have been identified in human genome; they actually manipulate many cellular functions and can act as “oncomiRs” or “tumor suppressor miRs.” It is involved in the regulation of expression of 30% of human genes, for example, miR-21 is regarded as a typical oncomir. 9 At present, the clinical applications of many miRNAs have been evaluated and significant progression has been achieved. For CRC, aberrant expression of miRNAs accompanying the progression of tumor is summarized in many reviews in the literature. However, these reviews normally focus on miRNAs in primary cancer or general colorectal metastasis, without putting specific emphasis in liver metastasis. A review to summarize the roles and potential clinical applications of miRNAs specifically in liver metastasis of CRC is still lacking. We herein review the specific studies of miRNA in CRLM and summarize their relationship as well as the potential clinical applications, including early diagnosis, treatment, and prognostic prediction.
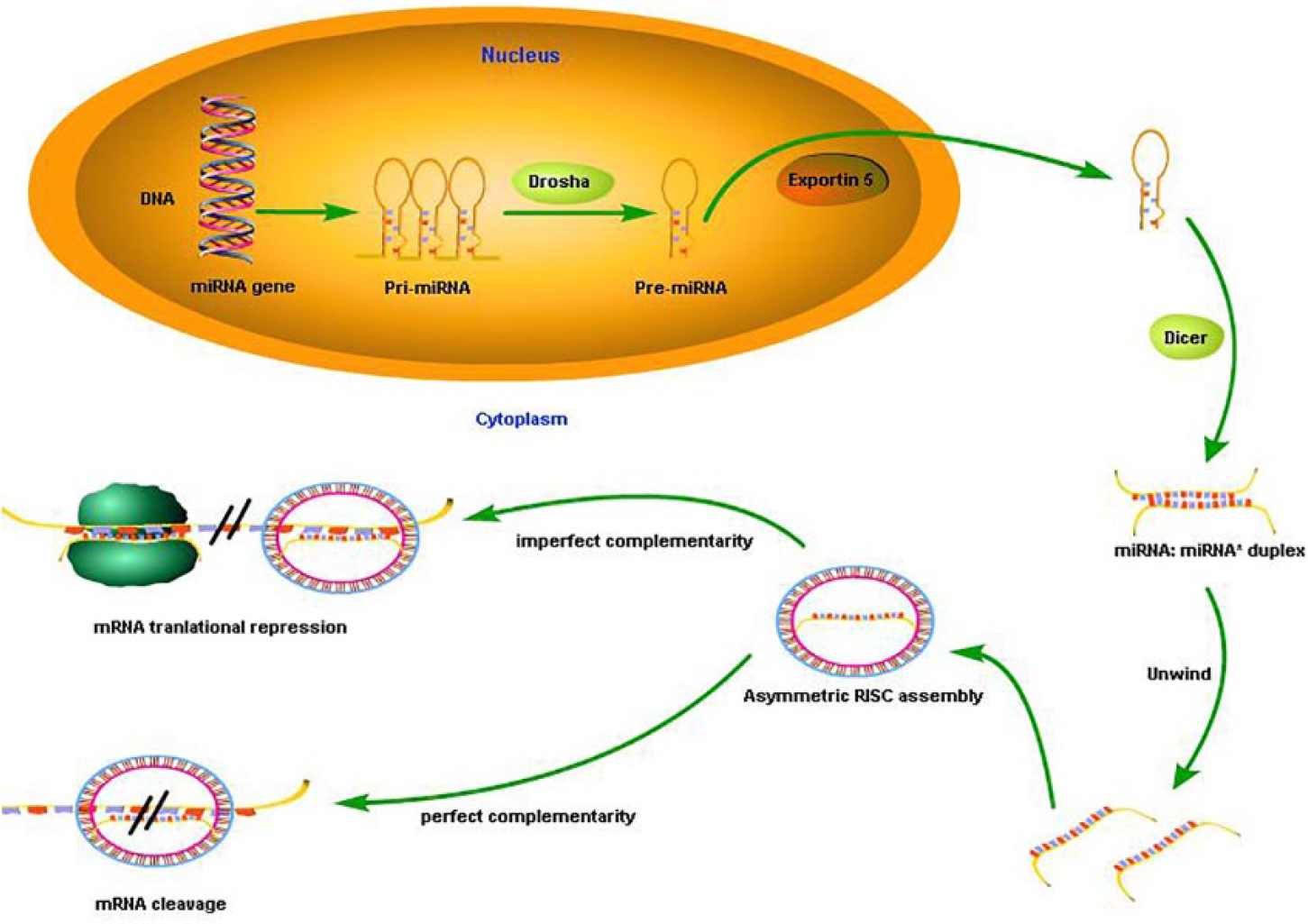
The biogenesis and function of miRNA. First, in the nucleus, miRNA gene was transcribed into pri-miRNA, the pri-miRNA is processed by the nuclear ribonuclease enzyme drosha resulting in the formation of pre-miRNA. Then, the pre-miRNA is transported into the cytoplasm through the protein exportin-5. In the cytoplasm, the pre-miRNA is processed into the mature double-stranded miRNA by the Dicer enzyme. Finally, the mature miRNA was assembled into the RNA-induced silencing complex (RISC) by means of complementary base pairing of target mRNA. Then, the silencing complex of target mRNA is degraded or inhibited to translate through different degree of complementarity.
MiRNAs participate in the cascade of CRLM
In 2007, Ma et al. 10 first reported that miR-10 regulates cell invasion and metastasis in breast cancer; then, more and more studies demonstrated the involvement of a group of miRNAs in tumor metastasis. They have been shown to have anti- or pro-metastatic effects, and thus were named “metastamiRs” (metastasis associated miRNAs). 11 MetastamiRs widely impact the metastatic cascade of cancer, such as apoptosis, epithelial–mesenchymal transition (EMT), evasion, migration, invasion, proliferation, and angiogenesis. 8 Colorectal carcinogenesis represents a sequential progression from normal colonic mucosa to adenoma, then carcinoma, and finally metastasis; the cascade includes the proliferation of primary tumor, invasion of tumor cells, survivival of the tumor cells during migration in the peripheral blood, transferring into the remote organs, and finally further growth by the proliferative and angiogenic mechanisms. Here, we summarize the miRNAs that are involved in the process of CRLM. Figure 2 demonstrates the overview of the process.

In the cascade of CRLM, miRNA is involved in various aspects of metastasis. Here, we summarize the miRNAs involved in metastasis regulation, as well as the related target genes, signaling pathways, and regulatory proteins, from the primary colorectal lesions to the liver.
Initiation of metastasis
It is well known that invasion and migration of the tumor cells are the key steps of initial metastasis, which happen in the primary lesion. They are the start of hepatic metastasis and induce the proliferation and invasion of tumor cells to spread to liver. Specifically, the process includes activation of metastasis-related enzymes, EMT, and translocation of tumor cells.
In the extracellular matrix (ECM) and tumor microenvironment (TME), transglutaminase 2 (TG2) is an important cross-linked enzyme which is closely related to the regulation of tumor migration. By comparing primary colorectal tumor and paired liver metastasis, Cellura et al. 12 found that miR-19 significantly inhibits the expression of TG2 and then change the invasive ability of CRC cells.
EMT is a key molecular step in the process of metastasis. Study showed that miR-30b directly targets and inhibits SIX1 gene to promote CRC metastasis. In the clinical samples, the expression of miR-30b in primary CRC is different from liver metastasis, and inversely correlated with SIX1 expression. 13 Similarly, when compared the metastasis with primary tumor tissues, the expression of miR-200c in CRC tissues without liver metastasis is significantly increased. Then, further studies showed that miR-200 family (miR-200a and miR-200c) decreases the expression of zinc finger E-box-binding homeobox 1/2 (ZEB1/2) in EMT, and ZEB1/2 upregulates vimentin and deregulates E-cadherin.14,15 Also, manganese superoxide dismutase (MnSOD) was found to promote EMT in the process of CRLM. Overexpression of miR-212 inhibits liver and lung metastasis by targeting MnSOD and prevents tumor progression. 16
Phosphatase of regenerating liver 3 (PRL-3) is a proto-oncogene known to promote tumor metastasis, especially in CRC. Zhang et al. showed that PRL-3 gene activates signal transducer and activator of transcription 3 (STAT3)-induced expression of miR-21, miR-17, and miR-19a. The experimental overexpression of these miRNAs leads to the proliferation and invasion of colon cancer cells. Then, the results were confirmed in the clinical samples of matched metastatic lesion and primary CRC tissues. 17
Comparing normal colorectal mucosa and pure liver tissue, Feiersinger et al. found that miR-21 is upregulated in primary CRC and liver metastasis. When compared primary CRC with liver metastasis, the latter showed relatively low miR-21. This indicates that miR-21 may be involved in the initiation of CRC but not in the metastasis. 18
MiRNAs drive migration, invasion, and metastasis
The process that cancer cells invade the blood vessels and spread through the circulation system is an important part of CRC metastasis, which is named as “intravasation.” Then, they extravasate out of the blood vessels and invade into the target organs and settle down. In CRC, circulating tumor cells predominantly settle in liver, but relatively rare in the bone or brain. This is mainly due to the portal vein system that connects colon and rectum to liver. In this process, it is generally believed that miRNAs play important roles in tumor cell intravasation, extravasation, and evasion of immune attack. Asangani et al. discovered that in CRC progression, programmed cell death 4 (PDCD4) is negatively correlated with miR-21 expression. Downregulation of PDCD4 gene suppresses tumor transformation and invasion. Further study confirmed that miR-21 regulates PDCD4 which increases the invasion, intravasation, and metastasis of CRC cells. 19 Epidermal growth factor–like domain 7 (EGFL7) is an important angiogenic protein. In the process of pathological angiogenesis, EGFL7 is secreted into the ECM and regulates vascular sprouting. At the same time, EGFL7 can inhibit the adhesion of vascular endothelial cells, loosen the vessel, and promote cancer cell intravasation. In stage III/IV CRC, Hansen et al. 20 found that expression of miR-126 and EGFL7 is closely related with each other and speculated that miR-126 may be involved in angiogenesis and intravasation process.
In tumor progression, extravasation and transendothelial migration (TEM) are important steps. E-selectin, which is an inducible member of the selectin adhesion receptor family, plays an important part in the process of leukocyte extravasation. But some studies show that by binding to E-selectin, tumor cells hijack the inflammatory process and enhance the ability to adhere to endothelial cells. 21 Recently, in colon cancer cells, Zhong et al. found that interleukin-1β (IL-β) induced the transcription of miR-31, and miR-31 directly inhibited the expression of E-selectin by targeting the 3′ untranslated region (3′UTR) of E-selectin messenger RNA (mRNA). Therefore, they clarified the important role of miR-31 in the process of tumor cell extravasation. 22
Metastatic colonization in liver
The final step of cascade in CRLM is metastatic colonization in liver. Studies have shown that miR-26b expression is reduced in embryonic stem cells (ESCs) and LoVo cells. At the same time, it was proved that overexpression of miR-26b inhibits the activity of CRC cells. Therefore, it is suggested that miR-26b may be involved in the regulation of tumor stem cells, leading to the growth of tumor cells in metastasized organ. 23
The successful establishment of new metastatic foci depends mainly on the proliferation of tumor cells and the ability to adapt to the new environment. Several studies demonstrated in CRC cells that the high expression of miR-103/107 in the presence of hypoxia targeted tumor metastasis suppressor gene DAPK (death-associated protein kinase) and KLF4 (Kruppel-like factor 4) promoting cell viability and decreasing intercellular adhesion. Then, they confirmed that miR-103/107 mediates the downregulation of DAPK and KLF4 enabling the colonization of CRC cells at metastatic site, and in the mouse model of CRC, the high expression of miR-103/107 also enhances local tumor invasion and liver metastasis. 24 By microarray miRNA screening, Larsen et al. found that miR-551a and miR-483 suppress liver colonization and metastasis. 25
It is well known that metastatic foci in the new organ need to induce new blood vessels to supply the increased demand of oxygen and nutrients. Angiogenesis is an essential component for CRC remote metastasis; 26 it promotes the colonization and the further growth of tumor cells to metastasize into liver. It has been found that specific subset of myeloid cells can promote the tumor angiogenesis through downregulation of angiopoietin-like 7. 27 Whether miRNA is involved in the process is not yet known. However, it has been reported that miR-195 directly targets vascular endothelial growth factor (VEGF) to inhibit the growth of hepatocellular carcinoma. 28
Clinical applications of miRNA in CRLM
Discovery of miRNA establishes a new field in cancer research and provides new potential clinical applications. MiRNA can be tried as a novel therapeutic target due to its ability to regulate relevant genes and signaling pathways.
So far, global miRNA expression profiling is of increasing importance as a useful clinical tool. Its clinical applications mainly focus on working as biomarker, prognostic prediction, and potential target therapy.
At present, in CRLM, many studies compare the relative expression of miRNA in metastatic lesions to the adjacent normal tissues. Therefore, to better understand the deregulated miRNAs involved in CRLM, researchers compared miRNA expression profiles in primary and metastatic tissues from the same patient, and simutaneously compared miRNA expression in primary CRC with or without metastasis. 29 For example, Hur et al. compared primary CRC tissue and liver metastasis tissue from same patients and verified by in situ hybridization that let-7i, miR-10b, miR-221, and miR-320a are downregulated in the liver metastasis and the expression of miR-885-5p in colorectal tumor tissue increased. The low expression of let-7i in primary colorectal tissue indicates a poor prognosis and distant metastasis. The high expression of miR-10b independently predicts distant metastasis. Interestingly, high expression of miR-885-5p in serum predicts prognosis, lymph node involvement, and distant metastasis, but its expression was not found in the tissue. 30
MiRNAs biomarkers in CRLM
MiRNAs are remarkably stable and are even resistant to RNase activity, thus showing a consistent expression level. 31 They have almost all the requirements of an ideal of biomarker in cancer. In addition, miRNAs can be collected in non-invasive ways and show satisfied sensitivity and specificity. 32 It is notable that miRNAs are highly stable in different conditions of both healthy and diseased patients because most of the circulating miRNAs are combined in lipid or lipoprotein complexes, such as apoptosis bodies, microvesicles, or exosomes. 33
At present, the studies that test miRNAs as biomarkers are mainly from two origins: tissue and circulating fluid, including plasma (or serum) and feces. 32 Perilli et al. investigated miR-182 expression in a series of 20 stage IV CRC, he found that miR-182 was significantly overexpressed in both primary CRCs and liver metastasis compared to normal tissues. But when compared within them (liver metastasis vs primary CRC), no significant difference was found. Then, they measured plasma miR-182 levels before and 30 days after radical liver metastasectomy. They observed that miR-182 level in plasma was significantly reduced 1 month after surgery. Thus, this study revealed the significance of miRNA in clinical implications. First, the specific involvement of miR-182 in CRC indicates its potential role as a diagnostic marker; second, miR-182 alone or in combination with its target genes (ENTPD5, TSP-1, and PDCD4) may serve as prognostic markers to monitor the relapse in CRC patients. Surely, this hypothesis would need to be studied in a prospective, randomized controlled trial. Finally, high miR-182 expression in advanced CRCs suggests further functional study whether it can become a candidate target for CRC therapy. 34
Although miRNA has the potential to serve as a marker for identifying patients at risk more accurately, independent large patient cohort studies are still needed before determining miRNA as a clinical marker. In the Table 1, we summarize some of the miRNAs that are associated with CRLM diagnosis and prognosis.
The clinical implication of miRNAs in CRLM.

CRLM: colorectal cancer liver metastasis; miR: microRNA; l-CRC: colorectal cancer with liver metastasis; p-CRC: primary colorectal cancer; CRC: colorectal cancer; LM: liver metastasis.
Hazard evaluation and prediction of liver metastasis in CRC
By comparing the expression of serum miRNA in CRC patients with and without liver metastasis, Wang et al. found that miR-29a expression significantly increased in patients with liver metastasis. They then verified whether the expression of miR-29a can predict liver metastasis, and the results showed that miR-29a had relatively good sensitivity (75%) and specificity (75%). 35
In an experimental study of 224 samples, Yin et al. assessed the expression of circulating miRNAs in a large cohort of patients with localized CRC, synchronous liver metastasis, and other organ metastasis. They confirmed that serum levels of miR-126, miR-141, and miR-21 are closely related to the early onset of CRLM, and the results suggest that these miRNAs can be used as novel markers for clinical diagnosis or prediction of early-stage liver metastatis. 47 Circulating miR-141 is closely associated with stage IV colon cancer. Elevated plasma levels of it also predicts poor prognosis in CRC patients. Meanwhile, miR-141 combined with carcinoembryonic antigen (CEA) improved the diagnostic accuracy of CRC. 48
In addition, the expression levels of miR-21, miR-135a, miR-335, miR-206, and let-7a in primary tumors are closely related to the occurrence of metastasis, which may serve as a potential tool for clinical diagnosis. 49 Although miRNA has reliable stability and is relatively mature in the detection of liver metastasis, high costs and long latency time of serum miRNA are the limitations of miRNA as blood-based diagnostic markers for screening purpose. 50
Treatment of CRLM
The clinical treatment of liver metastasis from CRC is mainly based on the clinical pathological features of tumor–node–metastasis (TNM) staging, histological features, and tumor invasion edge. 51 Although the stage of tumor is the basis for the choice of treatment, but according to the molecular characteristics of the tumor, the response to the same chemotherapy drugs also have significant differences. Therefore, patients with the same stage apply diverse treatments. 52 Interfering with the oncogene, anti-oncogene, or related target gene in its regulatory pathway can effectively inhibit the growth of tumor cells to therapy. 53 While a large number of studies have confirmed that miRNA can be used as an oncogene or tumor suppressor gene involved in tumor occurrence and development. 54 At the same time, stable, efficient control, and other characteristics of miRNA lay the foundation for its gene target treatment. Under current circumstances, the principle of using miRNA as an anti-cancer drug is based on two concepts: one is that the expression of miRNA in tumor tissues is disordered compared with normal tissues; another is that the expression of miRNA can change the cancer phenotype. Therefore, miRNA as anti-tumor drugs is mainly concentrated in a few areas, which includes inactivating the antigenic activities of onco-miRNAs, restoring the expression of tumor suppressive miRNAs, modulation of miRNA processing, and cancer stem cell directed miRNA-therapy. 55 Figure 3 depicts the main strategy of miRNA-based anti-tumor therapy.
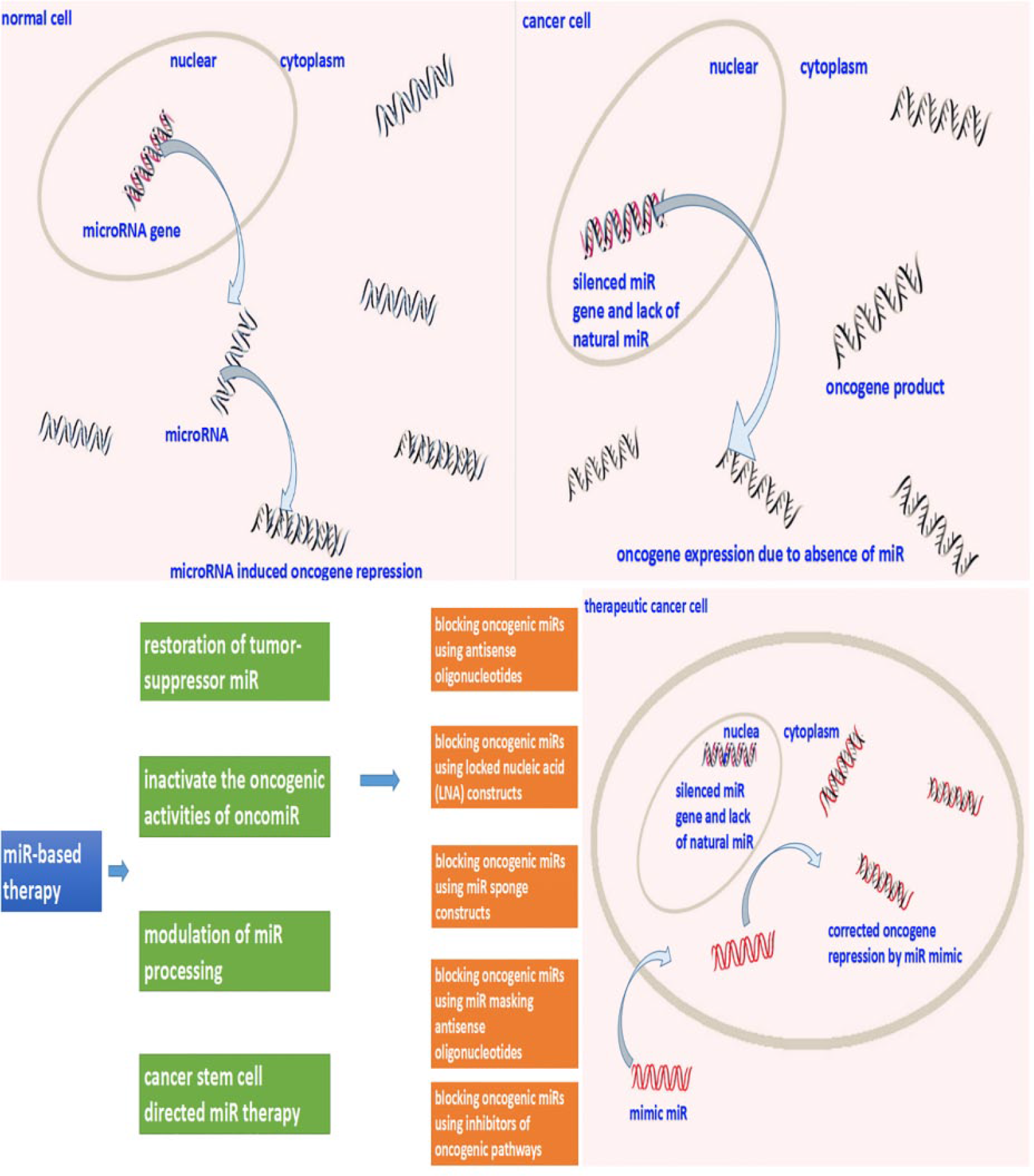
The main treatment strategies based on miRNA function focus on four aspects. The most valuable application of miRNA therapy is to inactivate the oncogenic activities of oncomiR to inhibit the activity of oncogenes.
In addition, due to the emergence of drug resistance, it brings more obstacles of therapy for CRC metastasis. Because of the resistance to treatment, many patients presented great reduction in the efficacy, leading to tumor recurrence or regeneration. But drug targeting specific miRNAs can influence chemo-sensitivity in CRC patients. This is also a direction for miRNA to participate in cancer therapy. 56 MiRNA expression is correlated with CRC therapeutic outcome. Colon tumor patients with high expression of miR-21 receiving adjuvant fluorouracil–based chemotherapy were found to have poor therapeutic outcome. 57
Recently, Qiu et al. 58 found in the mouse model that a combination of miR-497 and bufalin had a synergistic effect on the inhibition of CRC metastasis, suggesting the potential value of miR-497 in clinical therapy for CRC metastasis.
Ding et al. identified a set of genes that play an important role in medicating CRLM in an orthotopic mouse model of CRC. Then, they further studied whether APOBEC3G functions via the regulation of miRNA. They found that matrix metalloproteinase 2 (MMP2) is suppressed by miR-29; overexpression of APOBEC3G enhances colon cancer cell migration and invasion through restoration of MMP2 from inhibition of miR-29. The findings also get confirmed in human CRC samples. This suggests that APOBEC3G/miR-29/MMP2 pathway add to our understanding the mechanisms of CRLM and may develop new effective therapies in the treatment of advanced colon cancers. 59
Although the theoretical strategy for the treatment of tumors with miRNA is relatively mature, the practical application of miRNA in CRLM treatment remains in the experimental stage. But for some other diseases, several miRNAs have entered clinical practice, such as miR-34. MiR-34 can inhibit the growth of tumor, and regulate the p53 pathway as well as MET, MYC, WNT, and more than 20 oncogenes expression. 60 Daige et al. created a miR-34 mimic molecule, miR-rx34, which induces cell cycle arrest, senescence, and apoptosis of tumor cells. Through intravenous injection, the level of miR-34 increases in multiple organs without immune response. In the animal model of liver tumor, it causes complete tumor regression. Now, the application of miR-34 treatment in liver cancer has entered phase I trials.60,61 In addition, anti-miR-122 has been processed to phase II clinical trials for the treatment of human hepatitis C virus. 62
The applications of these methods in other diseases provide an example for CRLM. However, one single miRNA can mediate the expression of multiple genes, whereas a specific gene can be regulated by several different miRNAs. 63 Therefore, miRNA-based gene therapy is also facing challenges, such as side effects, particularly the potential for off-target miRNA activation that could lead to unwanted toxicities. Except for this, the problem of how to deliver miRNAs to specific target tissues without causing toxic effects also needs to be solved. 55
Prognostic prediction of CRLM
It is widely acknowledged that miRNA can assess the risk of metastasis and predict the hazard of post-operative recurrence and the overall survival of patients. In addition, miRNAs can also be used to evaluate chemotherapy response in CRLM patients and predict the chemo-resistance to specific drugs.
Study showed that miR-181a expression was elevated in CRLM. In the CRC cell line transfected with miR-181a, abilities of migration and invasion were enhanced, and they were decreased after the inhibition of miR-181a expression. In addition, the study confirmed that miR-181a directly targeted Wnt inhibitory factor-1 (WIF-1), which is one of the most important antagonists of Wnt signaling pathway and is closely related to tumorigenesis and development of tumor suppressor genes. Ectopic expression of miR-181a inhibits epithelial marker E-cadherin and β-catenin while enhances mesenchyme marker vimentin. These data suggest that miR-181a expression is associated with metastasis and survival in CRC. MiR-181a is associated with distant metastasis and can serve as an independent prognostic factor for advanced CRC. 41
In addition, by multivariate analysis, the expression of miR-625 was positively correlated with lymph node metastasis, liver metastasis, low survival rate, and poor prognosis in CRC patients. 46 By comparing CRC patients with or without hepatic metastasis, an abnormal expression of miR-214 was found. Further study showed the association between downregulation of miR-214 and increased level of fibroblast growth factor receptor 1(FGFR1), which can lead to the occurrence of liver metastasis. In addition, miR-214 was associated with an unfavorable prognosis and can be used as a potential marker to predict the survival of CRLM. 45 In order to obtain more liver metastasis-related miRNAs, some studies were carried out by comparing the metastatic foci, the invaded stroma, and normal liver tissue. The researchers used laser capture microdissection (LCM) technology to get samples from the center of liver metastasis, liver tissue near the tumor invasion front, and normal liver tissue. Then, they screened the expression of miRNAs in each type of the samples. They found that the expression of miR-19b and miR-194 was upregulated at the liver invasion front, but the expression of let-7 decreased at the tumor invasion front. Univariate analysis demonstrated that these miRNAs are adverse prognostic marker of tumor recurrence and overall survival in CRLM. 42
It is important to monitor the tumor recurrence after radical resection. Kingham et al. studied the expression of miRNAs in pre-operative serum and tumor tissue in 38 patients with CRLM. They found that the expression of miR-203 in patients with shorter survival was significantly higher than that in patients with long-term survival. 44 In addition, miR-203 was significantly higher in the plasma of stage IV CRC patients compared to early-stage CRC. 64 Hur et al. 65 also showed that increased expression of miR-203 in CRLM serum was associated with worse survival.
Besides, univariate analysis showed that high expression of hsa-miR-103 was associated with poor prognosis. The results were also confirmed in esophageal cancer carcinoma. 66 However, high expression of hsa-miR-103 also resulted in decreased migration ability of non-malignant tumor cells. 67 This may be illustrated by the fact that some of the miRNAs have a dual role in promoting and suppressing cancer.
Discussion
In general, miRNAs can function as either tumor promoters or suppressors in numerous types of cancer. The ability of miRNAs to target multiple genes and biological signaling pathways has drawn intense interests for their potential clinical utility. At present, there are many ongoing miRNA researches related to CRC. Lots of studies summarize the expression signature of miRNAs in CRC; several miRNAs play important roles in the regulation of specific function, including tumor growth, invasion, angiogenesis, migration, and planting. Some progression has been achieved but much more is needed.
A number of important studies have been conducted in CRC metastasis. In 2011, Lin et al. 68 compared the expression of miRNAs in CRC tissues with or without liver metastasis. In 2015, Hur et al. 30 compared the expression profiles of miRNA between liver metastasis and primary CRC. Therefore, the summary of the relationship between miRNAs and CRLM is urgently needed. At present, liver metastasis, lung metastasis and lymph node metastasis are summarized as CRC metastasis to study, but a separate review of liver metastasis was not found. In terms of metastasis, lung metastasis and liver metastasis are mainly hematogenous metastasis, but lymph node metastasis belongs to the lymphatic metastasis. According to the expression profile of miRNA, there were differences between liver and lung metastasis. Mathieu et al. analyzed metastasis in liver and lung metastasis and found that the expression patterns of miRNAs were significantly different. The expression of miR-199-5 in liver metastasis was significantly lower than that in lungs. At the same time, the expression patterns of miRNAs were different between normal liver tissue and lung tissue. 69 As mentioned above, because liver is an accessory digestive organ, the portal vein system collects blood directly from colon, rectum, and intestine. Therefore, liver is of great significance in the metastasis of CRC, and in clinical terms, liver metastasis accounts for more than 70% of colorectal synchronous metastasis. 70 Altogether, the study of miRNAs in liver metastasis is a critical issue.
We suggest that the aberrant expression miRNA can be considered as a biomarker of liver metastasis from CRC but not as a target for treatment. The establishment of a target should have a complete signaling pathway, as well as influence downstream genes. So in Figure 3, we collated miRNA markers for diagnosis and prognosis. Although many studies have found that miRNA has the potential for liver metastasis diagnosis and prognosis, there was no follow-up analysis of patient survival. In this regard, we only think that the miRNA is a biomarker in CRLM, so these miRNAs are not collected in the Figure 3. At the same time, some studies show that miRNA has a complex function in diagnosis and prognosis. 71 We just try to draw a clear boundary between diagnosis and prognosis. In the diagnosis of CRLM, our inclusion of miRNA was able to potentially predict or differentiate the presence of liver metastasis. The prognosis related miRNA is mainly used to determine the possible outcome of patients with liver metastasis. During treatment, certain therapies reduce the liver metastasis and then patients get the chance of a radical operation. At present, the most typical example of miRNA used in the treatment of cancer is in the clinical stage of miR-34. Its principle is to restore the expression of miR-34 in cancer cells and to inhibit tumor growth. 72 This provides a great reference value for the application of miRNA in CRLM.
Moreover, the clinical impact of miRNAs in CRLM is mostly identified in the proof-of-concept studies in cell lines, but animal models and large sample cohorts has to be confirmed in carefully designed clinical studies. Therefore, we still have a long way to go in the research of miRNA, but the prospects for the future are bright.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by the National Natural Science Fund of China (No. 81272375 and No. 81472713). L.Z. is funded by 2015 Innovation and Creation Fund of Shandong Academy of Medical Science.
