Abstract
Mounting evidence has demonstrated that Bit1 has been investigated as an etiological factor for certain cancers, including esophageal squamous cell carcinoma reported in our previous study, but data regarding possible roles of Bit1 in esophageal squamous cell carcinoma and esophageal adenocarcinoma remain to be elucidated. The purpose of this study was to examine whether Bit1 can be a novel diagnostic marker for the patients with esophageal squamous cell carcinoma and esophageal adenocarcinoma. The results revealed that Bit1 level in esophageal squamous cell carcinoma was significantly higher than that in esophageal adenocarcinoma tissues (
Keywords
Introduction
Esophageal carcinoma (EC) is one of the most common malignancies worldwide, especially in East Asia including China1,2. EC ranks the sixth cause of cancer-related mortality with the global statistics of almost 400,000 new patients diagnosed annually. 3 In terms of histological classification, EC is divided into several subtypes, of which esophageal squamous cell carcinoma (ESCC) and esophageal adenocarcinoma (EA) are the most common histological types and represent more than 90% of all EC histological types.4–6 ESCC is mainly distributed in Asia,7–9 such as China, India, and Iran, whereas a large number of EA cases are found in Western countries.10,11 Recently, there has been a dramatic rise in the incidence and mortality in EA and ESCC in the worldwide, but there was no significant difference in incidence between two histological types.12,13 Currently, due to the different therapy pattern of the patients with ESCC and EA in clinic, it is very important to identify the subtypes of ESCC and EA. However, the effective biomarkers to identify the two histological types are currently unavailable; therefore, it is imperative to identify early diagnostic markers with high sensitivity and specificity for judging ESCC and EA types.
It is well documented that Bcl-2 inhibitor of transcription 1 (Bit1) protein, encoding 179 amino acids, harbors a known structure for its putative active site. 14 Bit1, as the precursor of mitochondrial peptidyl-tRNA hydrolase 2, evokes cell apoptosis under the circumstance of being released from the mitochondria into the cytosol, 14 indicating that Bit1 plays an important role in mediating cell apoptosis. Recently, large amounts of conflicting data revealing Bit1 functions as an oncogene or tumor suppressor in different tumors may be mainly dependent on tumor types,15,16 our previous study revealed Bit1 functions as oncogene in ESCC,17,18 but its expression pattern in EA remains elusive, which will impel us to investigate the differential expression profiles of Bit1 in ESCC and EA. Bcl-2 family proteins, whose members have both anti-apoptotic and pro-apoptotic activity, have a central role in the control of mitochondria-induced apoptosis. 19 Our previous study revealed that Bit1 protein expression was positively correlated with Bcl-2 expression in ESCC, 17 suggesting tight associations of Bit1 with Bcl-2 play an essential role in ESCC, but related functions in EA remain to be elucidated. The migration of tumor cells depends on the degradation of the extracellular matrix (ECM), which can be regulated by complex control of the expression and activity of matrix metalloproteinases (MMPs), a family of zinc-dependent endopeptidases.20–22 It was proved that matrix metalloproteinase 2 (MMP2) is involved in tumor invasion and metastasis in various tumor types, including breast, gastric, pancreatic, and colorectal cancers.23–26 It was also suggested that MMP2 might play an important role in carcinogenesis of EC and could act as a biological marker of invasion and lymph node metastasis in this type of tumor. However, to date, the functions and intrinsic correlations of Bit1, MMP2, and Bcl-2 in ESCC and EA remain elusive; therefore, in this study, we investigated the Bit1, MMP2, and Bcl-2 levels in ESCC and EA tissues, analyzed the correlations between Bit1 levels and clinicopathological features, and further investigated the associations of Bit1 with MMP2 and Bcl-2 in ESCC and EA tissues. These findings may provide novel evidence for Bit1 as new diagnostic biomarker to distinguish ESCC from EA.
Materials and methods
Tissue samples
A total of 33 tumor tissues and paired normal tissues were obtained from surgically resected EC from Linzhou Tumor Hospital (Henan, China). Histologial type of EC was confirmed by pathological examination. Fresh samples were dissected manually to remove connective tissues and stored immediately at −80°C until analysis. Tumor tissues were obtained from poorly, moderately, and well-differentiated squamous epithelial cell carcinomas or adenocarcinoma. The corresponding normal tissues were obtained at 5 cm or more away from malignant tissues. Data were recorded regarding the clinicopathological parameters of tumors, including age, sex, stage, grade, and lymph node metastasis. The mean age was 61.5 years (range: 44–74 years). None of the patients in this study had undergone chemotherapy or immunomodulatory therapy before surgery. Six cases of adenocarcinoma were found with lymph node metastases. All the specimens were obtained with full ethical approval from the host institutions.
Antibodies and reagents
Mouse monoclonal immunoglobulin G (IgG) to PTRH2 (Bit1) and horseradish peroxidase (HRP)-linked affinity-purified goat anti-IgG was purchased, from Abcam (Hong Kong, China) and Zhongshan Golden Bridge (Guangdong, China), respectively; mouse monoclonal IgG2b to Bcl-2 and β-actin was obtained from Sanying (Wuhan, China); rabbit polyclonal IgG to MMP2 and HRP-linked antibodies was purchased from Abgent (Suzhou, China) and Boster (Wuhan, China), respectively.
Protein extraction and Western blot analysis
Total proteins were extracted from 100 mg of corresponding specimens of primary EC and normal esophageal epithelial tissues with radioimmunoprecipitation assay (RIPA; Boster) buffer according to the manufacturer’s protocol. Protein concentration was determined using Bradford reagent (Sigma, Shanghai, China), and bovine serum albumin (BSA) was utilized as a standard. Subcellular protein was added to 5× protein sample buffer, heated at 100°C for 2 min, separated on 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) with 3 µL of pre-stained protein molecular weight marker (Fermentas, Waltham, USA) as a standard at 120 V for 2–3 h, transferred to a polyvinylidene fluoride membrane, followed by blocking with 5% milk/Tris buffered saline with Tween 20 (TBST; 25 mM Tris (pH 7.5), 150 mM NaCl, and 0.1% Tween 20) at room temperature for 1–3 h. The membrane was primarily incubated overnight at 4°C with antibodies against Bit1 (1:10,000), Bcl-2 (1:2000), MMP2 (1:1000), and β-actin (1:10,000) diluted in 0.5% skimmed milk. The membrane was rinsed three times in TBST and then incubated with peroxidase-conjugated anti-mouse or anti-rabbit IgG (1:5000 or 1:2000 in 5% milk/TBST) for 2 h at 37°C. The activity of membrane-bound peroxidase was detected by enhanced chemiluminescent detection kit (ECL system, Beyotime, Shanghai, China). Thereafter, the densitometric quantitation of protein bands was performed using National Institutes of Health (NIH) image software. Protein expression was quantified as the ratio of a specific band to the corresponding β-actin value in the same lane.
Statistical analysis
All data analyses were performed by SPSS 17.0 statistical software package (SPSS, Chicago, IL, USA). The results were expressed as mean ± standard deviation (SD). Differences among the multiple groups were tested by one-way analysis of variance (ANOVA). The relationships between protein expression in ESCC and EA were calculated using Pearson or Spearman rank correlation test.
Results
Clinical data of the patients with EC
Clinical data of the patients with EC were summarized in Table 1, which included a sample with a total of 33 cases, with 11 cases of EA and 22 cases of ESCC. All samples comprised 24 males and 9 females, who did not receive the chemotherapy prior surgery.
Patients’ clinical data of department of chest surgery.

ESCC: esophageal squamous cell carcinoma; EA: esophageal adenocarcinoma.
Almost all the ESCC and EA tissues were positive for Bit1, MMP2, and Bcl-2 expression
We used western blot analysis to detect the expression patterns of three proteins in 22 cases of various differentiated ESCC tissues and adjacent tumor-free esophageal tissues. In addition, we also examined them in 11 cases of EA tissues and corresponding normal esophageal tissues. The results revealed that the three genes were expressed in almost all the 33 cases of esophageal epithelial tissues (Figures 1 and 2).
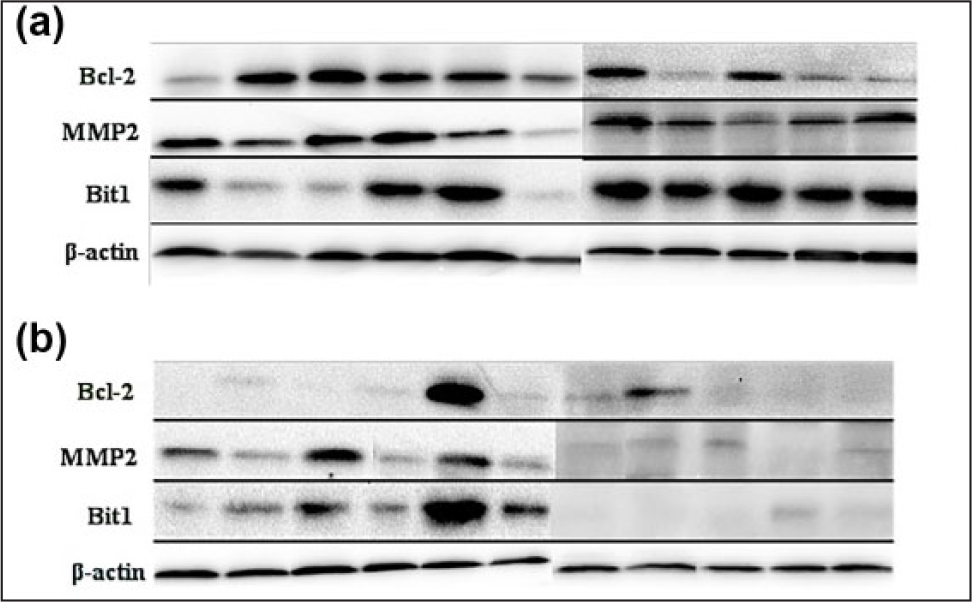
The expression of Bit1, MMP2, and Bcl-2 in (b) EA (11 cases) and (a) corresponding normal esophageal tissues. Total proteins were isolated from different esophageal tissues, protein concentrations were determined by Bradford reagent, and Western blot analysis was utilized to detect the Bit1, MMP2, and Bcl-2 protein level using anti-Bit1, anti-MMP2, and anti-Bcl-2 antibody in which β-actin was used as an internal control.

The expression of Bit1, MMP2, and Bcl-2 in (b) poorly (5 cases), (d) moderately (11 cases), and (f) well-differentiated (6 cases) ESCC tissues and (a, c, and e) their corresponding normal esophageal tissues, respectively. Total proteins were isolated from different esophageal tissues, protein concentrations were determined by Bradford reagent, and Western blot analysis was utilized to detect the Bit1, MMP2, and Bcl-2 protein level using anti-Bit1, anti-MMP2, and anti-Bcl-2 antibody in which β-actin was used as an internal control.
Differential expression pattern of Bit1 in human ESCC and EA tissues
To verify the differential expression of Bit1 in ESCC and EA, Western blot analysis was employed to detect the expression pattern of Bit1 in 22 cases of ESCC tissues and paired normal esophageal epithelial tissues as well as 11 cases of EA tissues and corresponding normal tissues. The results revealed that relative Bit1 protein level in ESCC tissues (1.49 ± 0.75) was significantly higher than that in EA tissues (0.91 ± 0.48;

The relative level of Bit1 protein in ESCC, EA, and corresponding normal esophageal tissues as well as various differentiated ESCC tissues and their corresponding normal esophageal tissues. Total proteins were isolated from different esophageal tissues, protein concentrations were determined by Bradford reagent, and Western blot analysis was utilized to detect the Bit1 protein level using anti-Bit1 antibody in which β-actin was used as an internal control. The relative protein level was determined according to the ratio of Bit1 protein level to β-actin level (*
Bit1 contributed to maintenance of poorly differentiated ESCC
To further explore the relationship between Bit1 and tumor differentiation, Bit1 expression was detected in different differentiation degree of ESCC. We found that Bit1 expression in poorly differentiated ESCC was significantly higher than that in normal esophageal tissues (
Relationship between Bit1 protein level and clinicopathological features in EC tissues
To elucidate the possible role of Bit1 protein level in the development and progression of esophageal cancer, we performed the investigation regarding the association of Bit1 protein level in esophageal cancer patients with various clinicopathological features, such as gender, age, tumor differentiation, and lymphatic metastasis. Western blot analysis was used to detect the Bit1 protein level in 22/11 cases of ESCC/EA specimens, and 22/11 cases of adjacent tumor-free esophageal specimens matched with the ESCC/EA specimens enrolled in this study. As indicated in Table 2, there was an obvious correlation between Bit1 protein level and the clinicopathological types of esophageal cancer (
Relationships between protein expression of Bit1 and clinicopathological factors in esophageal carcinoma patients.
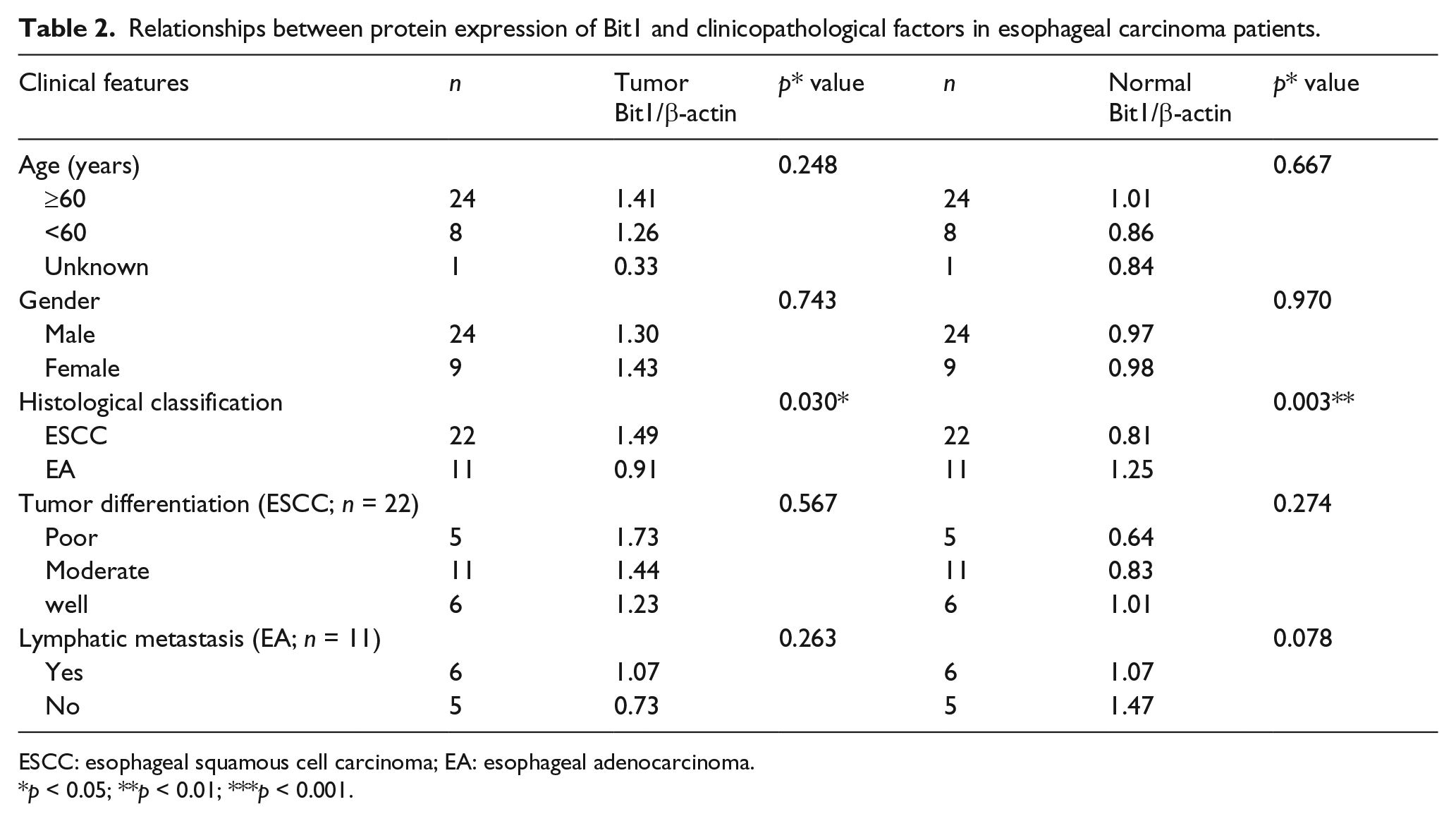
ESCC: esophageal squamous cell carcinoma; EA: esophageal adenocarcinoma.
Bcl-2 and MMP2 protein expression was detected in ESCC and EA tissues
To further delineate the expression of Bcl-2 and MMP2 proteins closely related to Bit1 in ESCC and EA tissues, Western blot analysis was utilized to investigate the expression of Bcl-2 and MMP2 proteins in different esophageal tissues. The results revealed that expression of MMP2 (1.46 ± 0.77) and Bcl-2 (1.03 ± 0.72) proteins in ESCC tissues was markedly higher than those in EA tissues (0.69 ± 0.43 and 0.68 ± 0.61, respectively) as well as normal esophageal tissues (

The relative image density of MMP2 and Bcl-2 in ESCC and EA tissues as well as their corresponding normal tissues. Total proteins were isolated from different esophageal tissues, protein concentrations were determined by Bradford reagent, and Western blot analysis was utilized to detect the Bit1 protein level using anti-MMP2 and anti-Bcl-2 antibodies in which β-actin was used as an internal control. The relative protein level was determined according to the ratio of targeted protein level to β-actin level (*
Expression patterns of Bcl-2 and MMP2 in various differentiated ESCC tissues
A large amount of evidence has demonstrated that Bit1 is tightly associated with Bcl-2 and MMP2 expression in multiple tumors. To verify the expression patterns of Bcl-2 and MMP2 in various differentiated ESCC tissues, Western blotting was utilized to investigate Bcl-2 and MMP2 expression. The results demonstrated that MMP2 expression in poorly and moderately differentiated ESCC was significantly higher than those in corresponding normal esophageal tissues (

The relative levels of MMP2 and Bcl-2 proteins in various differentiated ESCC tissues and corresponding normal esophageal tissues. Total proteins were isolated from different esophageal tissues, protein concentrations were determined by Bradford reagent, and Western blot analysis was utilized to detect the MMP2 and Bcl-2 expression level using anti-MMP2 and anti-Bcl-2 antibodies in which β-actin was used as a loading control. The relative protein level was determined according to the ratio of targeted protein level to β-actin level (*
The role of Bit1 is different in ESCC and EA tissues that correlated to the levels of Bcl-2 and MMP2 proteins
It is well documented that Bit1 exerting biological functions is closely associated with Bcl-2 and MMP2 expression in a variety of tumors.
27
To preliminarily elucidate the possible molecular mechanisms of Bit1-mediated apoptosis, we explored the relationship between Bit1 protein expression and Bcl-2 as well as MMP2 protein expression. We found that expression of Bit1 protein was positively correlated with the expression of MMP2 protein in ESCC and EA tissues (
Discussion
Increasing evidence has demonstrated that many genes have been verified as a potential molecular target for EC,27,28 but identification of different subtypes of EC, including ESCC and EA is more important using new biomarkers. This study demonstrated that Bit1 displayed differential expression profiles in ESCC and EA, and thus, may be a novel judging marker for ESCC and EA. Further investigation revealed the same tendency for Bcl-2 and MMP2 in ESCC and EA. The positive associations of Bit1 with Bcl-2 and MMP2 were found. Therefore, combinations of Bit1, Bcl-2, and MMP2 will be a new strategy for distinguishing the subtypes of ESCC and EA.
Recently, data reported that Bit1 expression level in ovarian carcinoma was markedly higher than that in the normal tissues;
15
in addition, Bit1 expression contributed to the survival of cervical cancer,
29
implying Bit1 functions as an oncogene in the ovarian and cervical cancers. However, Bit1 depletion increased cell adhesion and migration of breast carcinoma, suggesting Bit1 acts as a tumor suppressor in breast carcer.
16
Our previous study performed the investigation regarding the expression of Bit1 in 60 cases of ESCC tissues, 59 cases of dysplasia tissues, and 45 cases of normal esophageal epithelial tissues by immunohistochemistry; the result revealed that compared to normal esophageal epithelial tissues and dysplasia tissues, there was an obvious increase in Bit1 protein level in ESCC tissues.
17
However, detailed expression pattern of Bit1 in EA remains unclear; however, in this study, we found that Bit1 expression level in ESCC tissues was significantly higher than that in EA tissues (
MMPs are involved in the regulation of abundant tumor cellular processes beyond the ECM degradation, such as modification of cell surface receptors, release of growth factors, and regulation of inflammation and immunity cytokines, cell–cell adhesion molecules, and other proteinases.30–32 Bcl-2 also have a significant role in the tumor cells apoptosis.
19
In addition, numerous data have high relevance since the role of MMP2 and Bcl-2 in carcinoma pathogenesis and the association among its expression, tumor invasion, and disease severity were well known.19,23–26,33 As our previous studies have shown the importance of Bcl-2 in the invasive behavior of EC
17
and its association with apoptosis inducing factor (AIF).34,35 Furthermore, the data also revealed that Bit1 and Bcl-2 overexpression was closely associated with tumor differentiation, lymphatic metastasis, and tumor–node–metastasis (TNM) stage, and also suggested that a combination of Bit1 and Bcl-2 may be a novel predictor for ESCC metastasis.
17
Based on the above findings, to further explore the associations of Bit1 with MMP2 and Bcl-2 in ESCC and EA, we also investigated MMP2 and Bcl-2 expression in ESCC and EA and analyzed their correlations in ESCC and EA. Our results showed that MMP2 and Bcl-2 expression in ESCC tissues was significantly higher than those in EA tissues (
Taken altogether, our results demonstrate that Bit1, Bcl-2, and MMP2 levels in ESCC tissues were significantly higher than those in EA tissues, and the positive associations of Bit1 with Bcl-2 and MMP2 are exhibited both in ESCC and EA tissues. These findings highlight the important value of Bit1 in the diagnosis of ESCC and EA; more importantly, combinative detection of Bit1, Bcl-2, and MMP2 will provide more valuable information in clinic. Future directions will further explore its accuracy and sensitivity in diagnosis of the patients with ESCC and EA.
Footnotes
Acknowledgements
J.C. and H.L. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the High School Science and Technology Innovation Talents Project of Henan Province (15HASTIT037), the Foundation and Cutting-edge Technology Projects of Henan Province (152300410052), and the National Natural Science Foundation of China (81071723).
