Abstract
Calcifying nanoparticles have been linked to various types of human disease, but how they contribute to disease processes is unclear. Here, we examined whether and how calcifying nanoparticles isolated from patients with kidney stones are cytotoxic to human bladder cancer cells. Calcifying nanoparticles were isolated from midstream urine of patients with renal calcium oxalate stones and examined by electron microscopy. Human bladder cancer cells (EJ cells) were cultured in the presence of calcifying nanoparticles or nanohydroxyapatites for 12 and 72 h and examined for toxicity using the Cell Counting Kit-8, for autophagy using transmission electron microscopy and confocal microscopy, and for apoptosis using fluorescence microscopy, transmission electron microscopy, and flow cytometry. Changes in protein expression were analyzed by Western blotting. The results showed that the size and shape of the isolated calcifying nanoparticles were as expected. Calcifying nanoparticles were cytotoxic to EJ cells, more so than nanohydroxyapatites, and this was due, at least in part, to the production of intracellular reactive oxygen species. Transmission electron microscopy showed that calcifying nanoparticles were packaged into vesicles and autolysosomes. Calcifying nanoparticles induced greater autophagy and apoptosis than nanohydroxyapatites. Our findings demonstrate that calcifying nanoparticles can trigger bladder cancer cell injury by boosting reactive oxygen species production and stimulating autophagy and apoptosis.
Introduction
Calcifying nanoparticles (CNPs) are spherical and ovoid particles with diameters of 0.05–0.5 µm that can occur in the body and as contaminants in cell culture. 1 CNPs have been shown to present many biological characteristics, including self-proliferation, slow metabolism, coccoid shape, and resistance to acid, heat, and antibiotics. 1 Nevertheless, whether CNPs are the smallest life forms on Earth2,3 or simply inorganic calcium–protein complexes4–6 remains controversial.
Whatever their status, CNPs have been associated with many human diseases, especially diseases of ectopic calcification, such as nephrolithiasis, 7 coronary artery calcification, 8 type III prostatitis, 9 placental calcification, 10 and kidney stones. 11 Indeed, several studies have shown that CNPs can be cultured from the urine of patients with nephrolithiasis.
The cases of bladder tumor with calcification were often seen in the clinical setting, the probability of calcification in bladder tumor is about 0.69%–6.70%; calcification can occur in many types of bladder cancer, for example: transitional cell carcinoma, mucinous carcinoma, and malignant mixed mesodermal tumor. The calcification site on the surface, inside, or around the bladder tumor depends on the different tumor types.12,13 It is generally believed that the calcification of bladder tumor is related to the damage and necrosis of tumor cells, which may lead to local calcium deposition; phosphate is the main component of these calcium salts. Bladder as a storage organ for the urine, urinary CNPs may be involved in the formation of bladder tumor calcification. As we know, CNPs can be cytotoxic to normal cells and several types of cancer cells,14,15 CNPs can induce kidney stone formation by cytotoxicity of renal tubular epithelial cell has been confirmed in our previous work; 16 however, it is unknown whether the CNPs from urine are cytotoxic to bladder cancer and cause tumor calcification. Here, we examined whether and how CNPs isolated from patients with kidney stones are cytotoxic to human bladder cancer cells. We focused on production of reactive oxygen species (ROS), apoptosis, and autophagy as potential mechanisms of cytotoxicity.
Methods
Cell culture
EJ cells were obtained from the KeyGen Biotech (Nanjing, China) and cultured in RPMI-1640 medium at 37°C in an atmosphere of 5% CO2.
CNP culture and morphology analysis
Midstream urine was collected from patients with clinically confirmed renal calcium oxalate stones. The urine was sterile-filtered (0.45 µm) and cultured at 37°C in RPMI-1640 containing 10% fetal bovine serum (FBS) in a humidified atmosphere of 5% CO2. The medium was changed once in every 2 weeks for a total of 6 weeks. CNP concentration was adjusted to 2.0 Meclary turbidity (MCF) immediately before use. CNP morphology was observed using transmission electron microscopy (TEM; H7650; Hitachi, Tokyo, Japan) and scanning electron microscopy (SEM; S3400; Hitachi).
Preparation of nanohydroxyapatites
Nanohydroxyapatites (nHAP) was purchased from DK nano Technology (Beijing, China), dissolved in phosphate-buffered saline (PBS), and sonicated for 8 h. Then, it was diluted to 2.0 MCF before use.
Hoechst nuclear staining
EJ cells were seeded onto circular coverslips in six-well plates (1 × 105 cells/well) and allowed to grow until they covered approximately 75% of the coverslip. Then CNP suspension (2.0 MCF), nHAP (2.0 MCF), or PBS was added to the wells (200 µL/well). After culturing for 72 h, the coverslips were processed and stained using the Hoechst 33258 Fluorescence Staining Kit (Solarbio Bioscience & Technology, Beijing, China). Stained coverslips were examined under a fluorescence microscope at an excitation wavelength of 350 nm.
ROS assay
EJ cells were seeded on six-well plates (1 × 105 cells/well) and incubated for 24 h. Then, the cells were exposed to either nHAP (2.0 MCF) or CNPs (2.0 MCF) and incubated for another 72 h. Then, the cultures were processed using the Reactive Oxygen Species Assay Kit (DCFH-DA; Beyotime Biotechnology, Shanghai, China) according to the manufacturer’s instructions, and intracellular ROS levels were analyzed using flow cytometry (BD Biosciences, San Jose, CA, USA).
Cytotoxicity assay
EJ cells were seeded onto 96-well plates (1 × 104 cells/well, 100 µL/well) and cultured overnight. Cultures were exposed to PBS, nHAP (2.0 MCF), or CNPs (2.0 MCF) for 72 h and then processed using the Cell Counting Kit-8 (CCK-8; Beyotime Biotechnology) according to the manufacturer’s instructions. Optical density (OD) was measured at 450 nm using a microplate spectrophotometer. Cell viability was calculated as follows:Cell viability (%) = 100% × (average ODnHAP or CNPs/average ODPBS).
Apoptosis assay
EJ cells were plated onto six-well plates (1 × 105 cells/well) and incubated for 24 h. Then, the cells were incubated with CNPs for 72 h, trypsinized, washed three times with PBS, resuspended in binding buffer, and incubated in the dark for 15 min at room temperature with fluorescein isothiocyanate (FITC)–annexin V and propidium iodide (PI; BD Biosciences). Stained cells were analyzed immediately using fluorescence-activated cell sorting on a flow cytometer (BD Biosciences).
Autophagy assay
Ad-mRFP-GFP-LC3 adenovirus, which carries genes encoding modified red fluorescent protein (mRFP) and green fluorescent protein (GFP), was purchased from Hanbio Biotechnology (Shanghai, China) and transfected into EJ cells according to the manufacturer’s instructions. Transfected cells were treated for 12 h with PBS, nHAP (2.0 MCF), or CNPs (2.0 MCF). Using fluorescence microscopy, we followed two colors (red and green) to indicate successful transduction: loss of green color and increase red spots to indicate fusion of lysosomes and autophagosomes and appearance of yellow color spots (merge of red and green) to indicate formation of autolysosomes. Counting the spots of different colors allowed us to assess the autophagy levels.
Cell morphology
EJ cells were plated on six-well dishes and exposed to CNPs (2.0 MCF) for 12 and 72 h. TEM was used to examine cell morphology, internal structure, and intracellular localization of CNPs.
Western blotting
EJ cells were plated on six-well dishes, cultured for 24 h, and then treated with CNPs or nHAP for 12 and 72 h. The culture supernatant was aspirated and the cells were washed three times with ice-cold PBS, trypsinized, and then lysed with lysis solution (100 µL/well) for 30 min at 4°C. Total cell lysates were prepared and probed on Western blots using rabbit monoclonal antibodies against Bax, Bcl-2, Beclin-1, LC3-B, and glyceraldehyde 3-phosphate dehydrogenase. All the antibodies were obtained from Cell Signaling Technology (Boston, USA) and diluted to 1:1000 ratio.
Statistical analysis
Data were analyzed statistically using SPSS 16.0 (IBM, Chicago, IL, USA). All experiments in this study were repeated at least three times, and the data shown are mean ± standard deviation. Inter-group comparisons were assessed for significance using Student’s t test (two groups) or one-way analysis of variance (ANOVA; more than two groups), the statistical analysis is two-sided. p < 0.05 was defined as the threshold of significance.
Results
CNP morphology by electron microscopy
Based on SEM and TEM, the CNPs that we cultured showed morphological properties that were similar to those previously reported (Figure 1), namely, a coccoid form and diameters ranging from 100 to 400 nm. In addition, CNPs were caught in the act of dividing by binary fission. 1 TEM revealed internal cavities and inclusions, and both types of electron microscopy showed a dense, needle-filled outer shell on the particles.

(a–c) Transmission electron micrographs of CNPs. The arrow indicates apparent CNP division. (d and e) Ultrathin-section transmission electron micrographs of CNPs. The arrow indicates mineralized outer membrane of CNPs. (f) Scanning electron micrograph of CNPs.
Cytotoxicity of CNPs
CCK-8 assay was used to evaluate the cytotoxicity. We detected the survival rate of EJ cells and reflected the cell metabolism activity. After 72 h of intervention, based on the CCK-8 assay, CNPs were significantly more toxic to EJ cells than nHAP, while both treatments were more toxic than the control (all p < 0.01; Figure 2).
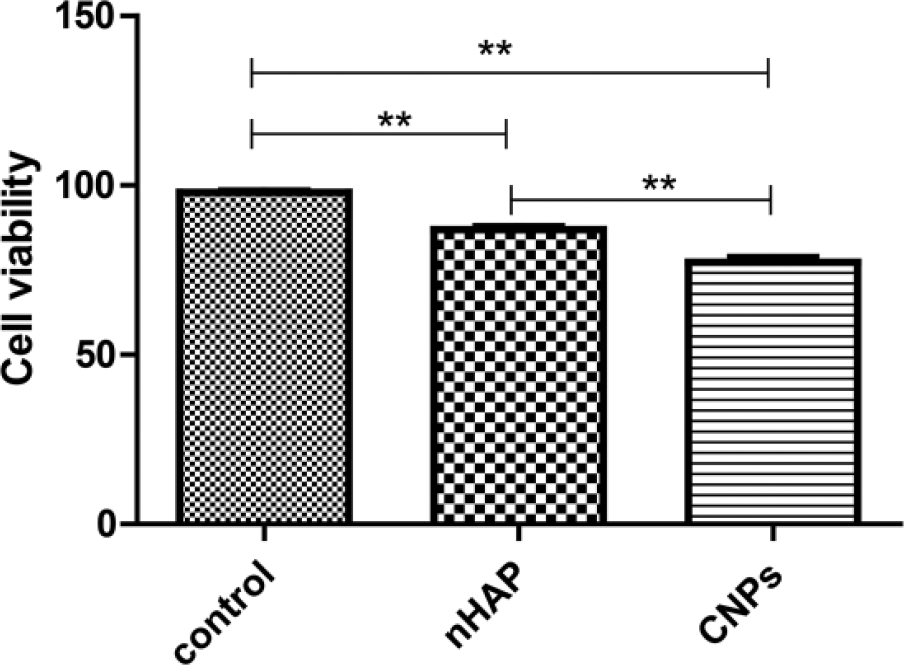
Viability of EJ cells after 72-h incubation with PBS (control), nHAP, or CNPs (**p < 0.01).
ROS generation induced by CNPs
CNPs accumulate in organelles and probably induce ROS production by mitochondrial pathway. CNPs induced the production of significantly higher levels of ROS than nHAP, and both treatments elicited significantly higher production than the control (all p < 0.01; Figure 3).

ROS production by EJ cells following 72-h incubation with (a) PBS, (b) nHAP, or (c) CNPs. (d) Quantitation of flow cytometry results (**p < 0.01).
Cell apoptosis induced by CNPs
Based on a nuclear staining assay in which blue nuclei indicate living cells and white nuclei indicate apoptotic cells, CNPs induced the greatest amount of cellular apoptosis, followed by nHAP and then the PBS control (Figure 4(a)–(c)). These results were confirmed and extended using flow cytometry to detect the cells that were stained with FITC–annexin V and/or PI. CNP exposure led to larger proportions of cells in early apoptosis (annexin V-positive, PI-negative) and late apoptosis (annexin V-positive, PI-positive) than nHAP exposure did (Figure 5).

Hoechst 33258 staining of nuclei in EJ cells treated with (a) PBS, (b) nHAP, or (c) CNPs. White arrows indicate normal cells and red arrows indicate apoptotic cells (magnification: 400×).

Apoptosis in EJ cells incubated with (a) PBS (control), (b) nHAP, or (c) CNPs. Cells were stained with FITC–annexin V and PI. (d) Quantitation of flow cytometric results (**p < 0.01).
Autophagy induced by CNPs
We assess the level of autophagy in the cells by counting the number of red spots (autolysosomes) and yellow spots (autophagosomes). While both CNP and nHAP induced autophagy, CNP exposure was associated with significantly more autophagosomes and autolysosomes (p < 0.01; Figure 6).
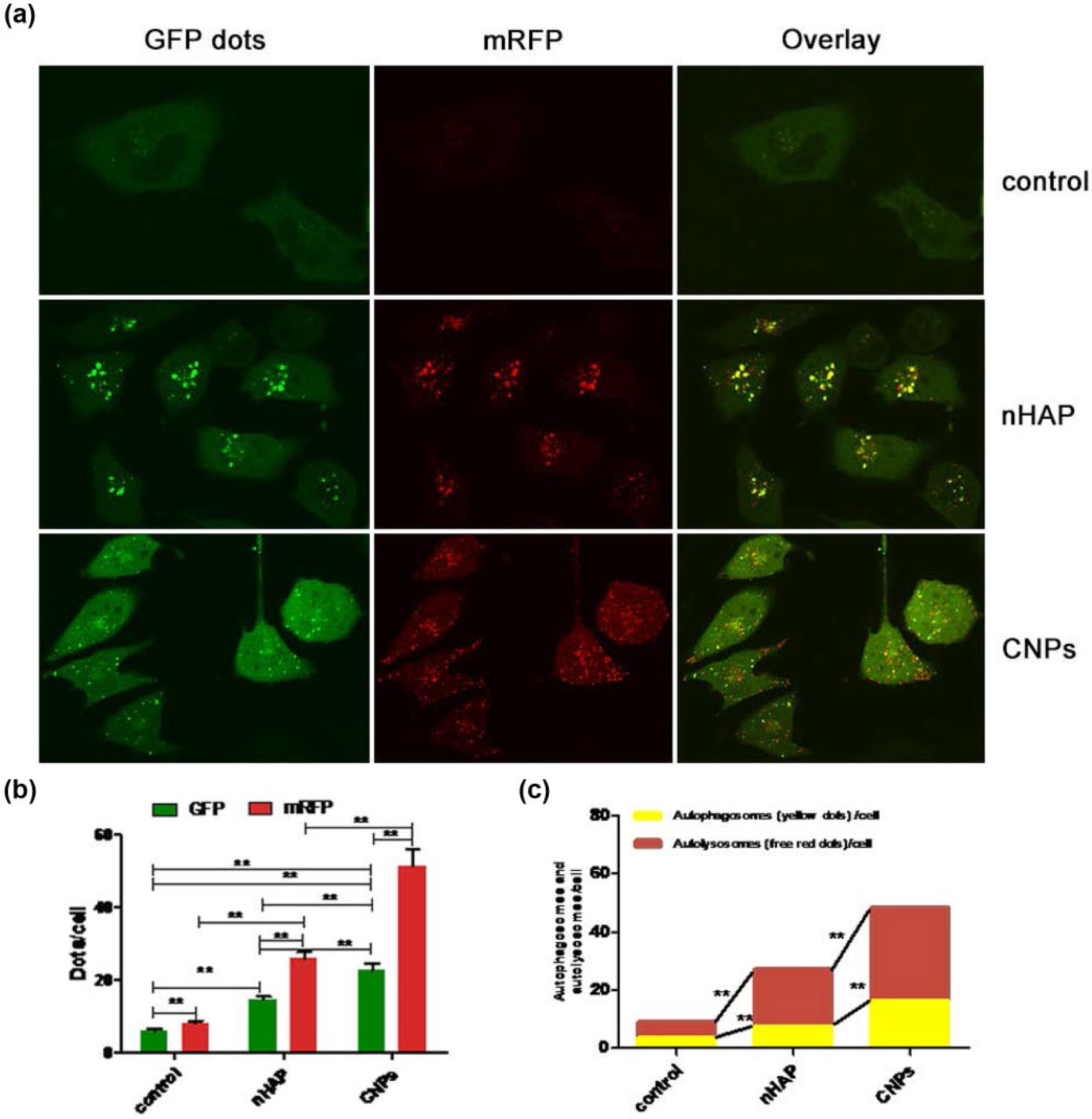
Induction of autophagy in EJ cells transduced with adenovirus Ad-mRFP-GFP-LC3 and incubated with PBS (control), nHAP, or CNPs. (a) In the merged image, yellow puncta indicate autophagosomes, while red puncta indicate autolysosomes. (b) Mean numbers of green and red puncta per cell. (c) Mean numbers of autophagosomes and autolysosomes per cell. Data shown are from at least five independent experiments (**p < 0.01).
CNP-induced changes within cells
Ultrathin-section TEM was used to observe the state of cells exposed to CNPs (Figure 7). The particles themselves looked similar to CNPs cultured in the absence of cells. The CNPs was observed mainly in the cytoplasm, most of intracellular CNPs are adjacent to mitochondria. The cells, for their part, showed cytoplasmic and mitochondrial swelling; CNPs were visible inside intracellular vesicles. Signs of apoptosis, autophagy, and necrosis were also observed.

Transmission electron micrographs of EJ cells exposed to (a–g) CNPs or (h and i) nHAP. Panel (b) is a zoomed image of panel (a). Arrows indicate CNPs. In panel (c), arrows indicate typical CNPs in intracellular vesicles. Panel (d) shows swollen mitochondria and CNPs. The arrow in panel (e) indicates an autolysosome. Panel (f) shows an apoptotic cell. Panel (g) shows CNPs and cytolysis.
Changes in protein expression induced by CNPs
Expression of LC3, Beclin-1, and the apoptosis indicator Bax was significantly higher in CNP cultures than in nHAP or control cultures (p < 0.05). Conversely, expression of Bcl-2 was significantly lower in the CNP group than in the control group (p < 0.05; Figure 8).

(a and c) Western blotting of apoptosis-related proteins in EJ cells. (b and d) Quantitation of Western blots (*p < 0.05).
Discussion
Whether CNPs can be considered as organisms remains controversial. In this study, we observed them to have morphological characteristics similar to those already reported, as well as the ability to undergo binary fission, which we consider to be a coincidental mimic of biological behavior. TEM revealed a dense, burr-like outer shell on CNPs, which appears similar to hydroxyapatite shell. 1 Whatever their nature, CNPs were highly toxic to human bladder cancer cells in our study, and this toxicity appears to reflect several simultaneous processes, including ROS production and induction of apoptosis, necrosis, and autophagy. Our results may help shed light on why CNPs appear to be associated with various types of human diseases, as well as how they damage or kill tumor cells.14,15
Our results provide one of the clearest, most detailed decriptions of CNPs inside cells. Our visualization of CNPs within vesicles inside cells suggests that the particles enter cells by some kind of endocytosis. We speculate that CNPs enter cells via the phospholipid bilayer through a process akin to viral packaging, which is the way that many nanoparticles enter cells.17,18 Future work should verify this pathway of “enveloped inclusion” and determine whether it is general to all CNPs.
Our results provide the first evidence that CNPs from the urine of patients with kidney stones are toxic to urinary system tumor cells. This work extends the literature showing similar toxicity to other types of tumor cells. Our results suggest that this toxicity occurs by mechanisms similar to those behind the toxicity of many nanoparticles, which accumulate in organelles such as mitochondria and induce ROS production. These radicals compromise membrane stability and therefore the cell’s existence. The increase in oxidative stress signals triggers cell death signals. 19 At the same time, our results suggest that CNPs also trigger autophagy, which has not been reported for the CNPs affecting other cells. This increase in autophagy was observed directly by microscopy as well as indirectly through upregulation of the autophagy markers LC3 and Beclin-1.20,21
CNP-induced apoptosis was observed not only directly by microscopy and flow cytometry but also indirectly through the simultaneous upregulation of pro-apoptotic Bax and downregulation of anti-apoptotic Bcl-2. The observed induction of apoptosis by CNPs likely reflects increased ROS production and autophagy. While autophagy can be a protective “waste-clearing” process, excessive autophagy can cause autophagic cell death. The autophagy in our experiments may have been triggered by ROS, which have been shown to promote autophagy by activating the C-jun n-terminal kinase (JNK) signaling pathway.22–24 JNK activation leads to phosphorylation of Bcl-2, which releases Beclin-1 from the Beclin-1/Bcl-2 complex, thereby promoting autophagy. 25 Furthermore, cleavage of Beclin-1 induces the release of apoptotic cytokines and promotes mitochondrion-mediated apoptosis. 26 JNK can promote the expression of Bax, Fasl, and tumor necrosis factor (TNF-α) by transcription factor AP-1 (activator protein 1) as well. High expression of pro-apoptotic proteins such as Bax influences the function of mitochondria, mitochondrial damage leads to the release of cytochrome C which can inhibit autophagy and induce the apoptosis of cells. 27
The CNPs from urine could induce autophagy, apoptosis, or necrosis of bladder cancer cells. Continuously proliferating CNPs combine with cell debris, necrotic tissue, and inflammatory exudation eventually leading to sustained local deposition of calcium and phosphorus, gradually forming calcification of bladder cancer. The calcification located on the surface of bladder transitional cell carcinoma may be especially related to the high content of CNPs in urine.
Our results identify several pathways that may mediate CNP-induced injury in human bladder cancer cells. There may be other pathways involved, which we did not examine here. For example, perhaps CNPs cause rapid change of intracellular calcium level, as has been described for certain nanoparticles. 28 This leads to cytoskeletal deformation and inhibits cell proliferation, 29 causing marked changes in cell morphology, proliferation, and surface wrinkles. Future studies should examine this and other possible mechanisms by which CNPs injure cells.
Footnotes
Acknowledgements
The authors are grateful to the members of the Electron Microscopy Group in the Department of Life Science at Guangxi Medical University, Guangxi, People’s Republic of China. J.-H.W. conceived and coordinated the study; designed, performed, and analyzed the experiments; and wrote the article. Y.-L.D., Q.L., J.-C.Y., Y.-L.L., Z.-Q.H., and X.-F.G. carried out the data collection, data analysis, and revised the article. All authors reviewed the results and approved the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81360113).
