Abstract
Enhancing the sensitivity of laryngeal cells to radiation is crucial for improving the efficacy of laryngeal carcinoma. MicroRNAs are known to play a major role in regulating cellular radiosensitivity. This study was designed to explore the effect and the molecular basis of miR-503 in the radiosensitivity of laryngeal carcinoma cells. Quantitative real-time polymerase chain reaction analysis showed that miR-503 expression was decreased in human laryngeal carcinoma cell lines Hep-2 and TU212, and the downregulation of miR-503 was also observed after irradiation. Upregulation of miR-503 by pre-miR-503 transfection restrained proliferation, promoted progression of Hep-2 and TU212 cells through the cell cycle after irradiation, and sensitized cells to radiation. Dual-Luciferase Reporter Assay verified a direct interaction between miR-503 and the WEE1 messenger RNA 3’-untranslated region. The overexpression of miR-503 significantly decreased WEE1 expression at the messenger RNA and protein levels, whereas the inhibition of miR-503 upregulated the expression of WEE1. WEE1 knockdown by WEE1 small interfering RNA apparently abrogated the inhibitory effect of anti-miR-503 on radiosensitivity. In conclusion, miR-503 could function as an enhancer of radiation responses in laryngeal carcinoma cells by inhibiting WEE1, which may be a potential novel radiosensitizing strategy for laryngeal carcinoma.
Introduction
Laryngeal carcinoma is the second most common malignant tumor of the head and neck 1 and is the sixth most common tumor worldwide, 2 with a 5-year survival rate of approximately 50%. 3 Its incidence has been increasing in recent years. Radiotherapy is the standard treatment modality for patients with laryngeal carcinoma and can allow better larynx preservation than surgery. 4 Although a satisfactory effect has been reported in most patients with laryngeal carcinoma, poor outcome due to radiation resistance occurs in a few patients. 5 It is reported that almost 50% of patients with laryngeal carcinoma do not respond to radiotherapy. 6 Therefore, it is important to investigate the mechanism of laryngeal carcinoma and promote the sensitivity of laryngeal carcinoma cells to radiotherapy.
WEE1, a protein kinase, is a tyrosine kinase that regulates the G2/M checkpoint that precedes entry into mitosis in response to DNA damage. 7 WEE1 is a rare G2/M checkpoint inhibitory protein in human tumor cells and is highly selective for tumor cells without affecting normal cells. 8 Numerous studies have identified WEE1 as a good target for cancer therapy. WEE1 overexpression was observed in multiple malignant tumors and was shown to correlate with poor disease-free survival, 9 including in neuroblastoma, 10 vulvar squamous cell carcinoma, 11 glioblastoma, 12 HER-2-positive breast cancers, 13 among others. WEE1 is also found to be frequently expressed at high levels in many patients with head and neck cancer. 14 Moreover, it has been reported that WEE1 inhibition could enhance the radiation sensitization of osteosarcoma 15 and diffuse intrinsic pontine gliomas. 16 The role of WEE1 in laryngeal carcinoma has not been determined.
MicroRNAs (miRNAs) are a family of small non-coding RNAs that regulate gene expression via special sites within the 3’-untranslated region (UTR) of messenger RNA (mRNA) of the target genes, playing important roles in the physiological and pathological processes of cancer. 17 Various studies have indicated that miRNA could be responsible for radiotherapy response in cancer. MiR-18a and miR-218 modulated the radiosensitivity of cervical cancer cells by promoting radiation-induced apoptosis.18,19 MiR-378g could enhance the radiosensitivity of nasopharyngeal carcinoma cells partially by regulating Src homology region 2 domain-containing phosphatase-1. 20 MiR-216a enhanced the radiosensitivity of pancreatic cancer cells by suppressing autophagy. 21 Recently, miR-503, an intragenic miRNA located at chromosomal location Xq26.3, was found to act as a tumor suppressor in several human cancers. 22 Li et al. 23 found that miR-503 suppressed metastasis of hepatocellular carcinoma cell by targeting PRMT1. Polioudakis et al. 24 revealed that miR-503 may act as a tumor suppressor in breast cancer. MiR-503 was also reported to suppress proliferation of human breast cancer cells by regulating CCND1 expression. 25 Studies have also revealed that miR-503 expression was downregulated in oral cancer. 26 Furthermore, Wagner-Ecker et al. 27 observed that radiation downregulated miR-503 expression in human endothelial cells. MiR-503 was also found to be inversely correlated with expression of Chk1 in radioresistant human gastric adenocarcinoma cells. 28 However, the expression of miR-503 and its role in the radiosensitivity of laryngeal carcinoma cells remain unknown. Therefore, the aim of this study was to investigate the potential role and underlying mechanism of miR-503 in laryngeal carcinoma cells, providing a potential therapeutic option for laryngeal carcinoma.
Materials and methods
Cell culture
Human laryngeal carcinoma cells Hep-2 and TU212 (The Cell Bank of Type Culture Collection of Chinese Academy of Sciences, Shanghai, China) and human embryonic kidney 293 cells (HEK293; American Type Culture Collection, Manassas, VA, USA) were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; Invitrogen, Carlsbad, CA, USA) containing 10% fetal bovine serum (FBS; Gibco, Carlsbad, CA, USA). Human normal bronchial epithelial cells BEAS-2B (American Type Culture Collection) were grown in DMEM/F12 (Gibco) with 5% FBS. All cells were incubated at 37°C with 5% CO2. The cells were harvested when they reached 90% confluence.
Cell transfection
The miR-503 precursor (pre-miR-503) and the negative control (pre-miR-Ctrl) and miR-503 inhibitors (anti-miR-503) and the corresponding negative controls (anti-miR-Ctrl) were purchased from GenePharma (Shanghai, China). Recombinant lentivirus-associated vectors were constructed by Biowit Technologies (Shenzhen, China). Hep-2 and TU212 cells (1 × 105 cells/well) were seeded in six-well plates and cultured for 24 h. Recombinant lentivirus-associated vectors carrying the pre-miR-503, pre-miR-Ctrl, anti-miR-503, or anti-miR-Ctrl were transduced into the Hep-2 and TU212 cells, respectively, using the Lipofectamine 2000 (Invitrogen) according to the supplier’s instructions and incubated for 48 h. Hep-2 and TU212 cells stably expressing the miR-503 or anti-miR-503 were selected using puromycin dihydrochloride (Santa Cruz Biotechnology, Santa Cruz, CA, USA). To knockdown the expression of WEE1, cells were infected with WEE1 small interfering RNA (siRNA; siWEE1; GenePharma) using Lipofectamine 2000 (Invitrogen) according to the manufacturer’s protocol for 48 h.
Radiation exposure
The cells were seeded into 96-well plates and irradiated in a Gammacell 220 Research Irradiator (MDS Nordion, Ottawa, ON, Canada) at doses of 2, 4, 6, and 8 Gy (dose rate: 1 Gy/min).
Colony formation
The colony formation assay was carried out to assess the radiosensitivity of laryngeal carcinoma cells. Hep-2 and TU212 cells (1 × 105 cells/well) were seeded in six-well plates and transfected with pre-miR-503 or anti-miR-503. After 48 h, the plates were irradiated with different dose of X-rays (0, 2, 4, 6, and 8 Gy). After incubation at 37°C for 13 days, the cells were washed with phosphate-buffered saline (PBS), fixed with 100% ethanol, and stained with 0.1% crystal violet for 1 min. Colonies (≥50 cells) were counted. Clone formation efficiency = (number of colonies/number of cells seeded for non-irradiated controls) × 100%. Surviving fraction = colonies counted/(cells plated 6 plating efficiency). GraphPad Prism 5.0 (GraphPad Software, LaJolla, CA, USA) was used to fit the cell survival curve.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was performed to determine the proliferation of cells. Cells (5 × 104 cells/well) were plated in 96-well plates with six replicate wells and incubated overnight. Then, cells were irradiated and allowed to grow for 48 h. Then, 20 μL of MTT (5 mg/mL; Sigma, St. Louis, MO, USA) was added into each well on three consecutive days and the cells were incubated for an additional 4 h at 37°C. Then, the supernatant was discarded and 200 μL of dimethyl sulfoxide (DMSO) was added for 5 min to dissolve the precipitate. Quantification was measured performed at 490 nm and the data, which were derived from triplicate samples of at least three independent experiments, were expressed as a percentage of the control.
Cell cycle and apoptosis analysis
Flow cytometry was used to examine the effects of miR-503 on cell cycle and apoptosis in laryngeal carcinoma cells. Briefly, cells were harvested and washed twice with cold PBS. Then, they were fixed in ice cold 70% ethanol overnight at 4°C. After washing with PBS twice, the cells were suspended in PBS containing 50 μg/mL RNase A (Sigma) at room temperature. After 30 min, 2 mg/mL propidium iodide (PI; Sigma) was added at 4°C for an additional 30 min in the dark. Cell cycle and apoptosis were measured using a flow cytometer (Becton Dickinson, San Jose, CA, USA), and the data were analyzed using Cell Quest Software 3.3 (Becton Dickinson). The experiment was performed in triplicate.
Quantitative real-time polymerase chain reaction
Total RNA was extracted from the cells with TRIzol reagent (Invitrogen). Reverse transcription to complementary DNA (cDNA) was carried out using M-MLV reverse transcriptase (Clontech Laboratories, Palo Alto, CA, USA) or the One Step PrimeScript miRNA cDNA Synthesis Kit (TaKaRa, Dalian, China) for mRNA detection or miRNA detection, respectively. Quantitative real-time polymerase chain reaction (qPCR) was carried out in triplicate using SYBR Green Premix (TaKaRa) according to standard methods. The primers for qPCR were as follows: WEE1, 5’-GATGTGCGACAGACTCCTCA-3’ (forward), 5’-CAAAGCGTTCTGCTCATCAA-3’ (reverse); glyceraldehyde 3-phosphate dehydrogenase (GAPDH), 5’-GACTCATGACCACAGTCCATGC-3’ (forward) and 5’-AGAGGCAGGGATGATGTTCTG-3’ (reverse). The relative expression of the target gene was normalized to GAPDH and U6, for mRNA and miRNA, respectively. Relative quantification was calculated using the 2−ΔΔCt method.
Western blotting assays
Protein samples were separated on a sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and electrotransferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA). After being blocked with 3% bovine serum albumin for 2 h, the membranes were sequentially incubated overnight at 4°C with primary antibodies against WEE1 (1:500; Abcam, Cambridge, UK), followed by incubation with horseradish peroxidase–conjugated anti-rabbit IgG secondary antibody (1:2000; Abcam). Proteins were visualized using an enhanced chemiluminescence system (Pierce Biotechnology, Rockford, IL, USA). B-actin (1:5000; Abcam) was used as a control.
Dual-luciferase reporter assay
HEK293T cells (5 × 105) were cultured in 96-well plates and cultured for 24 h. The WEE1 3’-UTR containing the predicted miR-503-binding sequences was amplified and subcloned into pGL3 vector (Promega, Madison, WI, USA). The pGL3 vector containing WEE1 3’-UTR or WEE1 3’-UTR mutated forms (3’-UTR-mut) was co-transfected with pre-miR-503 or controls into HEK293 cells using Lipofectamine 2000 (Invitrogen) for 48 h. Then, cells were harvested and lysed using the lysis buffer (Promega). The luciferase activity was measured using the Dual-Luciferase Reporter Assay System (Promega) according to the manufacturer’s protocols.
Statistical analysis
All data are presented as mean ± SD and were analyzed by one-way analysis of variance (ANOVA) with subsequent Student’s t-test; p < 0.05 was considered statistically significant. Statistical analysis was performed using GraphPad Prism 5.0 software (GraphPad Software, Inc., San Diego, CA, USA).
Results
MiR-503 is expressed at low levels in human laryngeal carcinoma cells
We first investigated the expression of miR-503 in two human laryngeal carcinoma cell lines Hep-2 and TU212 and human normal bronchial epithelial cells BEAS-2B by qPCR. Significantly lower levels of miR-503 were observed in both Hep-2 and TU212 cells compared with BEAS-2B cells (p < 0.05, Figure 1(a)). To investigate whether miR-503 was involved in the radiosensitivity of laryngeal carcinoma cells, we analyzed the expression of miR-503 in Hep-2 and TU212 cells at different time points (0, 4, 8, 12, and 24 h) post-irradiation with 2 Gy or at 4 h post-irradiation with different doses (0, 2, 4, 6, and 8 Gy). As shown in Figure 1(b) and (c), miR-503 expression was reduced at all time and dose points, indicating that miR-503 may play an important role in radiation sensitivity of laryngeal carcinoma cells.
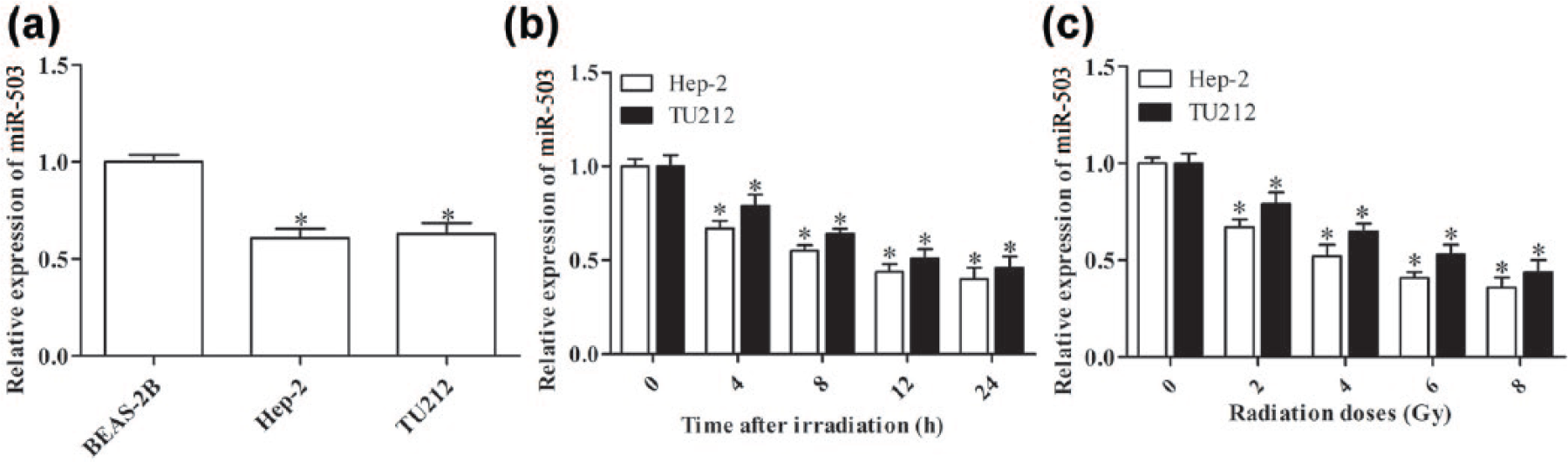
qPCR analysis of miR-503 expression. (a) The miR-503 level in BEAS-2B, Hep-2, and TU212 cells. (b) The relative expression of miR-503 in Hep-2 and TU212 cells treated with 2 Gy radiation and cultured for 0, 4, 8, 12, and 24 h. (c) The relative expression of miR-503 in Hep-2 and TU212 cells at 4 h post-irradiation with 0, 2, 4, 6, or 8 Gy (*p < 0.05 vs BEAS-2B cells; *p < 0.05 vs 0 h timepoint or 0 Gy).
MiR-503 sensitizes Hep-2 and TU212 cells to irradiation
To study the effect of miR-503 on the radiosensitivity of laryngeal carcinoma cells, Hep-2 and TU212 cells were transfected with pre-miR-503 or anti-miR-503 for 48 h to increase or decrease the miR-503 level, respectively (p < 0.05, Figure 2(a) and (b)); 4 h post-irradiation with different doses (0, 2, 4, 6, and 8 Gy), MTT and colony formation assays were performed to examine the effect of miR-503 on post-irradiation cell survival. MTT assay results showed that miR-503 overexpression significantly decreased the proliferation of Hep-2 and TU212 cells 4 h post-irradiation with 2 Gy (Figure 2(c) and (d)). Conversely, miR-503 inhibition promoted the proliferation of cells after radiation (p < 0.05; Figure 2(c) and (d)).

MiR-503 is involved in the radiosensitivity of Hep-2 and TU212 cells. qPCR analysis of miR-503 expression in (a) Hep-2 and (b) TU212 cells after transfection with pre-miR-503 or anti-miR-503. MiR-503 modulated the proliferation of (c) Hep-2 and (d) TU212 cells 4 h post-irradiation with 2 Gy, as detected by MTT assay. (e and f) The Hep-2 and TU212 cells transfected with negative controls, pre-miR-503, or anti-miR-503 were exposed to 0, 2, 4, 6, and 8 Gy irradiation and incubated for 13 days, and survival curves were determined using the colony formation assay. MiR-503 disrupted the cell-cycle distribution in (g) Hep-2 and (h) TU212 cells after irradiation, as analyzed by flow cytometry. MiR-503 increased the percentage of apoptotic cells in (i) Hep-2 and (j) TU212 cells 4 h post-irradiation with 2 Gy. (N = 3; *p < 0.05 vs control).
Moreover, we found that Hep-2 and TU212 cells transfected with pre-miR-503 showed reduced survival fraction after irradiation, while cells transfected with anti-miR-503 showed increased survival fraction compared with the control group (p < 0.05, Figure 2(e) and (f)). These results suggested that miR-503 sensitizes laryngeal carcinoma cells to radiotherapy.
MiR-503 inhibits G2/M arrest and enhances apoptosis of Hep-2 and TU212 cells after radiation
To study the mechanism by which miR-503 enhanced radiosensitization, we measured cell cycle and cell apoptosis by flow cytometry. As shown in Figure 2(g) and (h), Hep-2 and TU212 cells transfected with pre-miR-503 showed an obvious increase in the G0/G1 phase and a decrease in the G2/M phase after 4 h post-irradiation with 2 Gy. In addition, we observed that overexpression of miR-503 caused an increase in apoptosis in Hep-2 and TU212 cells after 4 h post-irradiation with 2 Gy (Figure 2(i) and (j)). These results implied that miR-503 induced the sensitivity of Hep-2 and TU212 cells to radiation, possibly due to the abolishment of G2/M and increased cell apoptosis.
MiR-503 targets WEE1 and regulates its expression in laryngeal carcinoma cells
To further explore the possible target gene(s) of miR-503, we identified a potential miR-503 target site in the WEE1 mRNA 3’-UTR region that was revealed by bioinformatics analysis (Figure 3(a)). To confirm the direct interaction between miR-503 and WEE1, luciferase reporter vectors containing miR-503 complementary sequence of the wild-type (Wt) or mutant (Mut) WEE1 3’-UTR were constructed (Figure 3(a)). MiR-503 overexpression significantly repressed the luciferase activity of the Wt WEE1 3’-UTR but not the Mut 3’-UTR (Figure 3(b)). Additionally, both the mRNA and protein level of WEE1 were suppressed in Hep-2 and TU212 cells transfected with pre-miR-503, but upregulated in cells transfected with anti-miR-503 (Figure 3(c)–(e)). These results demonstrated that WEE1 is a direct target of miR-503 in laryngeal carcinoma.

MiR-503 directly targets WEE1 and regulates its expression in Hep-2 and TU212 cells. (a) The putative binding site between miR-503 and WEE1. (b) Dual-Luciferase Reporter Assay was performed to detect the interaction between the miR-503 and WEE1 3’-UTR after transfection for 48 h. Both the mRNA and protein levels of WEE1 were downregulated in Hep-2 and TU212 cells transfected with pre-miR-503, but upregulated in cells transfected with anti-miR-503 detected by (c and e) qPCR and (d and f) western blot analysis (N = 3; *p < 0.05 vs control).
WEE1 siRNA abrogates the effect of anti-miR-503 on the sensitivity of laryngeal carcinoma cells to radiation
To determine whether WEE1 is involved in the regulation of radiosensitivity in laryngeal carcinoma cells, the expression of WEE1 was detected after radiation. As shown in Figure 4(a) and (b), the mRNA and protein levels of WEE1 were higher in Hep-2 and TU212 cells than in BEAS-2B cells after 4 h post-radiation with 2 Gy (p < 0.05), implying that WEE1 may be engaged in regulation of the radiosensitivity of laryngeal carcinoma cells. To further confirm this role for WEE1, WEE1 siRNA and anti-miR-503 were co-transfected into Hep-2 and TU212 cells, respectively. Western blot and qPCR results showed that miR-503 inhibition led to a significant upregulation of WEE1 level in both of the cell lines (Figure 4(c) and (d)). In addition, the improvement in survival fraction caused by anti-miR-503 was markedly inhibited by WEE1 knockdown in Hep-2 (Figure 4(f)) and TU212 cells (Figure 4(g)). Moreover, cell proliferation (Figure 4(h) and (i)), cell cycle (Figure 4(e)), and apoptosis (Figure 4(j)) were all reversed in cells co-transfected with WEE1 siRNA and anti-miR-503 after radiation for 4 h with 2 Gy. These results indicated that the inhibition of WEE1 by siRNA abrogated anti-miR-503-induced radioresistance.

WEE1 is required to enhance the radiosensitivity of miR-503 in laryngeal carcinoma cells. Hep-2 and TU212 cells were transfected with control anti-mRNAs (anti-miR Ctrl), anti-miR-503, and nonspecific siRNA (anti-miR-503 + siNC) or anti-miR-503 and WEE1 siRNA (anti-miR-503 + siWEE1), respectively, for 48 h. The (a) mRNA and (b) protein levels of WEE1 in Hep-2 and TU212 cells were higher in BEAS-2B cells after 4 h post-irradiation with 0, 2, 4, 6, or 8 Gy, as analyzed by qPCR and western blot analysis (#p < 0.05 vs BEAS-2B cells). (c) Western blot and (d) qPCR analysis of WEE1 protein expression in different groups in Hep-2 and TU212 cells after 4 h post-irradiation with 2 Gy. (f and g) The inhibitory effect of anti-miR-503 on radiosensitivity was rescued when WEE1 siRNA expression vector was co-transfected. WEE1 siRNA reversed the effect of anti-miR-503 on (h and i) cell proliferation, (e) cell-cycle distribution, and (j) cell apoptosis after irradiation for 4 h with 2 Gy (#p < 0.05 vs anti-miR Ctrl; *p < 0.05 vs anti-miR-503 + siNC).
Discussion
We demonstrate herein a potential role of miR-503 in the radiosensitivity of laryngeal carcinoma cells. We first provide evidence that miR-503 was decreased in two human laryngeal carcinoma cell lines Hep-2 and TU212 and miR-503 was also downregulated after radiation. Further analysis revealed that upregulation of miR-503 inhibited proliferation, but promoted cell-cycle progression after radiation and sensitized cells to radiation, whereas anti-miR-503 had the opposite effect. Additionally, we identified that WEE1 was a direct target gene of miR-503, and WEE1 knockdown abrogated the inhibitory effect of anti-miR-503 on radiosensitivity. These findings suggest a potential role of miR-503 in regulating the radiosensitivity of laryngeal carcinoma through WEE1 inhibition.
More recent evidence suggests a role for miRNA in the radiosensitivity of laryngeal carcinoma. Xu et al. 29 showed that miR-24 was able to enhance radiosensitivity in laryngeal squamous cell carcinoma by targeting X-linked inhibitor of apoptosis protein (XIAP). MiR-296-5p was found to be a predictive marker for radiotherapy resistance in early-stage laryngeal carcinoma. 30 A series of studies have shown the aberrant expression of miR-503 and its effect on various cancer.31–33 In this study, miR-503 expression was significantly downregulated in laryngeal carcinoma cells and this was closely associated with the radiation resistance of Hep-2 and TU212 cells. Furthermore, miR-503 inhibition induced an arrest at the G2/M checkpoint and restrained the apoptosis of Hep-2 and TU212 cells after radiation, whereas miR-503 overexpression abrogated G2/M arrest and promoted apoptosis of cells after radiation. Our data provide the first insights into the function of miR-503 in regulating the sensitivity to radiotherapy.
Identifying the specific target mRNA is crucial for studying the molecular mechanisms of miR-503. To better understand the mechanisms of miR-503 in response to radiation, the specific target mRNA of miR-503 was identified and characterized by Dual-Luciferase Reporter Assay, qPCR, and western blot analysis. Our study revealed that WEE1 was a functional target of miR-503, and miR-503 negatively regulated the expression of WEE1 in Hep-2 and TU212 cells. DNA damage induced by radiation exposure is responsible for the radiosensitivity of cancer cells. 34 Cancer cells mainly depend on the G2 checkpoints to gain time for DNA repair after DNA-damaging treatments such as irradiation. 35 Therefore, preventing cancer cells from repairing the damaged DNA during G2 arrest is one strategy to sensitize laryngeal carcinoma cells to radiation. Previous studies have reported that WEE1, a protein kinase and a G2/M checkpoint regulator, 36 plays an important role in the radiosensitivity of laryngeal cells and that WEE1 inhibition influences the radiation-induced G2/M checkpoint,37,38 thus preventing mitotic entry and sensitizing cells to apoptosis.15,16 In this study, we discovered increased expression of WEE1 in laryngeal cells, which was inversely correlated with miR-503 level. Moreover, knockdown of WEE1 reversed the increased efficacy of radiation induced by anti-miR-503.
In conclusion, our study is the first to demonstrate that miR-503 contributes to the radiosensitivity of laryngeal carcinoma cells possibly through WEE1 inhibition. These findings identify a potential role of miR-503 in enhancing the radiosensitivity in laryngeal carcinoma cells and suggest that miR-503 could be a potential therapeutic target for laryngeal carcinoma.
Footnotes
Compliance with ethical standards
This article does not contain any studies with human participants or animals performed by any of the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
