Abstract
Signal transducer and activator of transcription 5 and Akt pathways, implicated in signaling transduction downstream of BCR–ABL, play critical roles in the pathogenesis of chronic myeloid leukemia. Therefore, identification of novel compounds that modulate the activity of such pathways could be a new approach in the treatment of chronic myeloid leukemia. Previous studies have demonstrated that indole-3-carbinol inhibits the proliferation and induces apoptosis of various tumor cells. However, its anticancer activity against chronic myeloid leukemia cells and the underlying mechanism remain unclear. Our data revealed that indole-3-carbinol promoted mitochondrial apoptosis of chronic myeloid leukemia–derived K562 cells, as evidenced by the activation of caspases and poly (ADP-ribose) polymerase cleavage. Treatment with indole-3-carbinol was found to be associated with a decrease in the cellular levels of phospho-Akt and phospho–signal transducer and activator of transcription 5. In addition, real-time polymerase chain reaction analysis showed that the downregulation of genes is regulated by Akt and signal transducer and activator of transcription 5. We also found that treatment with indole-3-carbinol resulted in the activation of the p38 mitogen-activated protein kinase and reduced expression of human telomerase and c-Myc. Collectively, these results demonstrate that the oncogenic signal transducer and activator of transcription 5/Akt pathway is a cellular target for indole-3-carbinol in chronic myeloid leukemia cells. Thus, this clinically tested natural compound can be a potential candidate in the treatment of chronic myeloid leukemia following confirmation with clinical studies.
Keywords
Introduction
Chronic myeloid leukemia (CML) is a clonal disease characterized by the incidence of the (9; 22) (q34; q11) translocation, which generates the BCR–ABL fusion gene. BCR–ABL is a constitutively active tyrosine kinase present in more than 90% of the patients, which promotes the survival and proliferation of neoplastic cells through the upregulation of multiple downstream pathways. 1 Numerous signal transduction pathways activated by BCR–ABL have been implicated in the transformation and control of proliferation and differentiation of CML cells.2–4 Janus kinase/signal transducer and activator of transcription (JAK/STAT) and PI3/AKT are the most common signaling pathways constitutively activated by BCR–ABL.4,5 STAT5, a STAT family member, is necessary for the transformation and cell cycle progression of CML cells.5,6 STAT5 and Akt activation occurs in over 90% of CML cells, which can regulate transcriptional activation of the genes involved in cell growth, proliferation, survival, and apoptosis resistance.4,5 Emerging evidence suggests that the activation of STAT5 and Akt signaling pathways contributes to disease progression and poor survival in CML.7,8 Therefore, inhibition of STAT5 and Akt signaling proteins may be an interesting approach against CML cells. p38 mitogen-activated protein kinase (MAPK) is a class of MAPK responsive to multiple stress stimuli; it is involved in cell differentiation, apoptosis, and autophagy. 9 A new finding has raised the possibility of p38 MAPK acting as a critical negative regulator of BCR-ABL-mediated leukemogenesis in CML cells treated with imatinib. 10 Furthermore, the activation of p38 MAPK pathway helps in mediating the antileukemic effect of dasatinib. 11 Thus, finding various means and methods of activating p38 MAPK in BCR–ABL expressing cells may be a promising strategy in the treatment of CML. The clinical use of tyrosine kinase inhibitors (TKIs), such as imatinib, as the standard first line of therapy in CML has improved prognosis, response rate, and overall survival. 12 However, the complete elimination of CML clones in patients receiving TKIs is limited by the primary and secondary resistance mainly due to the upregulation of BCR–ABL 13 as well as extra mutations in the BCR–ABL oncoprotein. 14 Therefore, in light of resistance to TKIs in some patients with CML, augmenting the therapeutic armamentarium against CML is critical. Much evidence has indicated that natural products can exhibit the anticancer effect by perturbing multiple cellular signaling pathways in tumor cells.15–17 Indole-3-carbinol is a natural compound derived by hydrolysis from glycobrassicin found in cruciferous vegetables. 18 A broad and potent antitumor activity of indole-3-carbinol has been demonstrated in multiple human cancers such as those of breast, colon, and prostate. 19 Moreover, experiments have demonstrated the therapeutic benefits of indole-3-carbinol in clinical trials for cervical dysplasia, 20 breast cancer, 21 and vulvar intraepithelial neoplasia. 22 To the best of our knowledge, to date, no study has addressed the effects of indole-3-carbinol and its mode of action on the proliferation and survival of CML cells. In this study, we examined the effects of indole-3-carbinol on oncoproteins regulated by BCR–ABL in CML-derived K562 cells. We revealed that indole-3-carbinol as a single agent exhibits a potent antitumor activity through the regulation of various downstream effectors and signaling pathways activated by BCR–ABL in K562 cells. We delineated that the inhibitory effect of indole-3-carbinol was accompanied by the repression of STAT5 and Akt signaling pathways and upregulation of phospho-p38 MAPK.
Materials and methods
I3C, tetrazolium salt (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)), and propidium iodide (PI) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Primary antibodies for Western blotting, including caspase-9 (9502), cleaved caspase-3 (9661), c-Myc (13987), STAT5 (9363), phospho-STAT5 (9351), Akt (9272), phospho-Akt (9271), p38 (9212), phospho-p38 (9211), phospho-Crkl (3181), and β-actin (8H10D10) were obtained from Cell Signaling Technology (Danvers, Massachusetts, USA). Anticleaved poly (ADP-ribose) polymerase (PARP) antibody (ab32561) was from Abcam (Cambridge, UK). Secondary antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, California, USA).
Methods
Cell culture
K562 cells (CML-derived cell line) were cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS), 100 units/mL penicillin, 100 μg/mL streptomycin, and 0.05%
Cell viability assay
MTT colorimetric method was used to assay the effect of various concentrations of I3C on cell viability of CML cells. Briefly, K562 cells were seeded onto a 96-well culture plate (10 × 103 cells/well) following incubation with desired concentrations of I3C (100, 200, and 400 μM) for 24 and 48 h. After removing the medium, the cells were incubated with 10 µL MTT solution (5 mg/mL in phosphate-buffered saline (PBS)) for 4 h at 37°C, and the resulting formazan was solubilized with dimethyl sulfoxide (100 µL). The absorbance of each well was measured at 570 nm in an enzyme-linked immunosorbent assay reader.
Cell apoptosis assay
The effect of I3C on cell death of K562 cells was assayed by the Annexin V/PI apoptosis kit according to the manufacturer’s instructions (BD Biosciences, San Diego, CA, USA). In brief, K562 cells were seeded onto 12-well cell culture plate at a density of 2 × 105 cells/well. After 24 and 48 h treatment with I3C (200 and 400 μM), the cells were collected and washed with cold PBS. Then, the cells were resuspended in 100 μL reaction buffer containing 5 μL Annexin V–fluorescein isothiocyanate (FITC) and 5 μL PI. After incubation for 15 min at room temperature in the dark, 400 μL of the binding buffer was added to each sample. The stained cells were then analyzed through Becton–Dickinson fluorescence-activated cell sorting (FACS; BD Biosciences, San Jose, California, USA). Annexin V–positive and PI-negative cells were considered to be in early apoptotic phase, whereas cells having positive staining both for Annexin V and PI were deemed as undergoing late apoptosis.
RNA extraction and preparation of complementary DNA
TriPure isolation reagent (Roche Applied Science, Penzberg, Germany) was used for RNA isolation from K562 cells at 32 h after treatment with 400 µM I3C according to the manufacturer’s instructions. The RNA concentration and quality was determined by Nanodrop Spectrophotometer (Thermo Fisher Scienic, Waltham, MA, USA). Complementary DNA (cDNA) synthesis was performed with 1 μg total RNA using RevertAid™ First Strand cDNA Synthesis Kit (Fermentas, Ontario, Canada). The quality of generated cDNAs was determined by polymerase chain reaction (PCR) using human glyceraldehyde 3-phosphate dehydrogenase (GAPDH) specific primer.
Quantitative real-time PCR
Amplified cDNAs were subjected to real-time PCR using specific gene primers and SYBR Green PCR Master Mix (Ampliqon, Odense, Denmark) by means of the Rotor-Gene 6000 Real-Time PCR System (Corbett Research, Mortlake, Australia). PCR amplification was performed at 95°C for 15 min followed by 40 cycles including a denaturation step at 95°C for 15 s and a combined annealing/elongation step at 60°C for 60 s. Finally, a melting curve analysis was performed to exclude unspecific products or primer dimer formation and verify the specificity of the products. Each sample was analyzed in triplicate, and fold difference or fold-change in gene expression was calculated using the comparative Ct (2−ΔΔCT) method. The list of primers used and their respective sequences are given in Table 1.
The forward and reverse primer sequences used for real-time PCR.

PCR: polymerase chain reaction; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; hTERT: human telomerase reverse transcriptase; XIAP: X-linked inhibitor of apoptosis protein.
Western blot analysis
K562 cells were treated with I3C, and then, the cells were harvested at the indicated times, washed with cold PBS, and lysed (5 × 106 cells/aliquots) in 0.2 mL of radioimmunoprecipitation assay (RIPA) buffer (10 mM Tris–HCl, pH 7.4; 150 mM NaCl; 5 mM ethylenediaminetetraacetic acid (EDTA), 1% Triton X-100, 0.1% sodium dodecyl sulfate (SDS), and 0.5% sodium deoxycholate) supplemented with protease and phosphatase inhibitor cocktails (Sigma-Aldrich, St. Louis, MO, USA). After centrifugation at 13,000 r/min for 20 min at 4°C, cell-free supernatant was collected and protein concentration of the supernatant was determined by the Bradford protein assay. The equal amounts of each protein samples were subjected to electrophoresis on 10% SDS–polyacrylamide gel electrophoresis (PAGE), followed by transfer to a nitrocellulose membrane (Hybond-ECL; Amersham Corp., Little Chalfont, UK). Thereafter, the membrane was incubated in blocking buffer (1× Tris-buffered saline (TBS), 0.1% Tween 20 with 5% non-fat dry milk) for 1 h and then probed overnight at 4°C with specific primary antibodies. After removing primary antibody solution, the membrane was washed five times in 1× TBS containing 0.1% Tween 20 (TBST), followed by incubation for 1 h at room temperature with horse radish peroxidase (HRP)-conjugated secondary antibody. A chemiluminescence detection system (Amersham ECL Advance Kit; GE Healthcare, Little Chalfont, UK) was used to visualize the immunoreactive proteins.
Statistical analysis
Two-tailed Student’s t test was used to determine statistically significant difference among the groups. p < 0.05 was considered to be statistically significant.
Results
Indole-3-carbinol induces apoptosis through caspase activation and poly (ADP-ribose) polymerase cleavage
To assess the effects of indole-3-carbinol (I3C) on cell survival, K562 cells were subjected to 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. K562 cells were treated with various concentrations (100, 200, and 400 µM) of I3C for 24 and 48 h. Viability of K562 cells treated with I3C significantly decreased in a dose- and time-dependent manner. I3C at 100 and 200 µM did not have a significant cytotoxic effect on these cells after treatment for 24–48 h. Over 50% decrease in the viability was observed in cells treated with 400 µM I3C within 48 h (Figure 1(a)). Thus, I3C exerted a significant cytotoxic effect on K562 cells. To further confirm our data, we performed Annexin V/propidium iodide (PI) double staining followed by flow cytometric analysis after treating the cells with 200 and 400 µM I3C for 24 and 48 h. As shown in Figure 1(b), the percentage of Annexin V/PI double-positive cells increased among I3C-treated K562 cells compared with untreated control cells. The percentage of cells undergoing early-stage apoptosis (Annexin V–fluorescein isothiocyanate (FITC) positive) increased at 400 µM after 24 h. However, with treatment for 48 h, a significant number of cells underwent late-stage apoptosis. To investigate the potential molecular mechanisms responsible for I3C-induced apoptosis of K562 cells, we detected the expression of cleaved products of caspase-3, caspase-9, and poly (ADP-ribose) polymerase (PARP) by western blotting after treating the cells with 200 and 400 µM I3C for 18 and 32 h. As presented in Figure 1(c), I3C treatment led to a significant increase in cleaved products of caspase-3, caspase-9, and PARP compared with the control group in a dose- and time-dependent manner. Collectively, these findings suggest that I3C may promote apoptosis through the mitochondrial pathway of apoptosis of K562 cells.
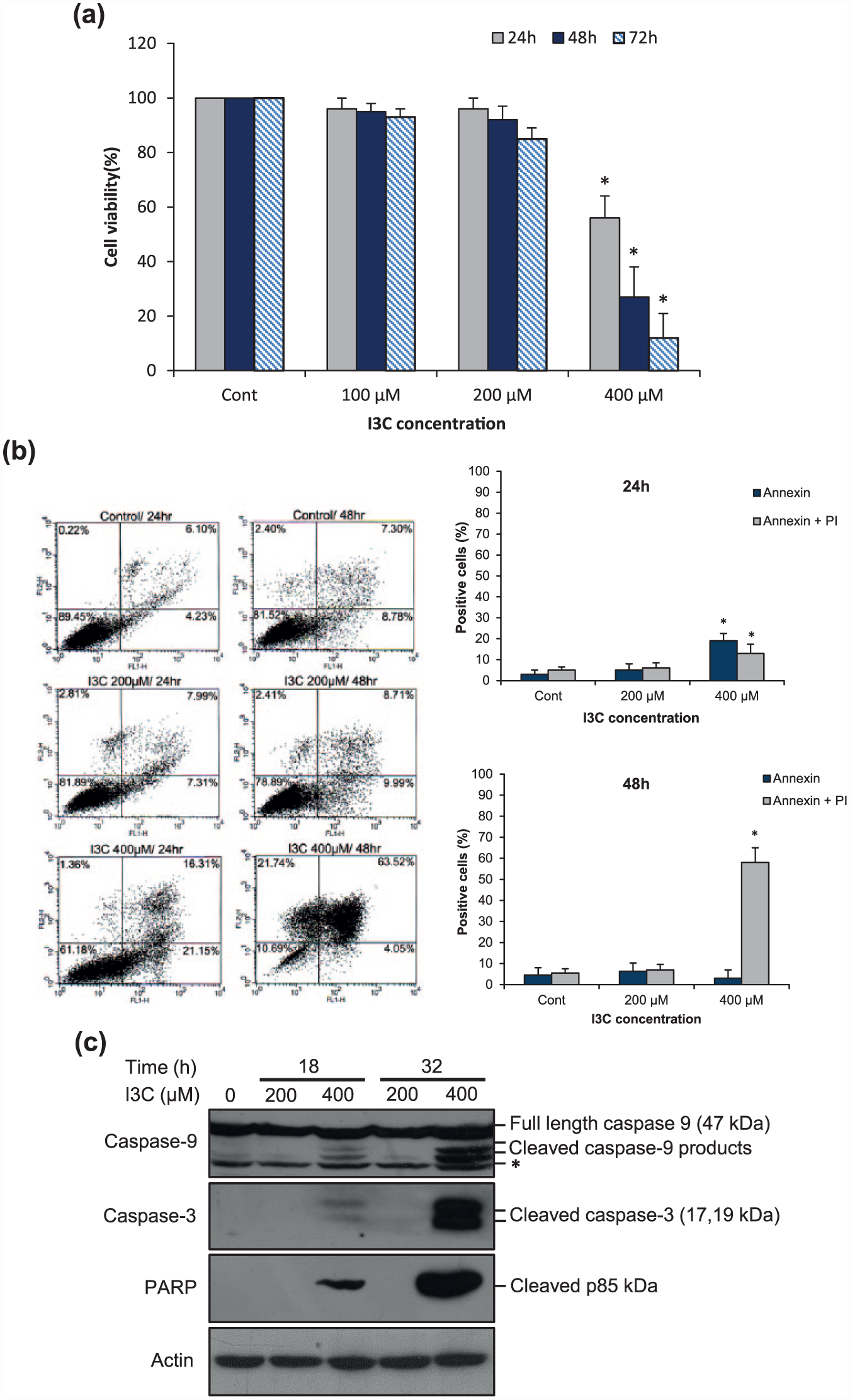
I3C accentuates caspase-mediated apoptosis of K562 cells. (a) I3C decreases cell viability of K562 cells. Cells were incubated with various concentrations of I3C for 24, 48, and 72 h and subjected to MTT colorimetric method. The value of untreated control cells was set to 100%. The data are expressed as mean ± SD of at least three independent experiments (*p < 0.05 compared with untreated control cells). (b) I3C promotes a dose- and time-dependent increment of apoptotic cells in K562 cells. The cells were treated with 200 and 400 µM I3C for 24 and 48 h. Cells were then analyzed for Annexin V–FITC and PI by flow cytometry (n = 3; *p < 0.05 relative to untreated cells). (c) Effects of I3C on caspase activation and PARP cleavage. K562 cells were treated with 200 and 400 µM I3C for 18 and 32 h. The total cell lysates were subjected to western blot analysis to assess the cleavage of caspase-3, caspase-9, and PARP. The asterisk indicates a non-specific band detected by the caspase-9 antibody. β-actin serves as loading control to normalize protein levels. One representative western blot image of three independent experiments is presented.
I3C suppresses STAT5 activation but has no effect on Crkl phosphorylation
The persistent activation of the transcription factor STAT5 is a molecular hallmark of CML. 6 STAT5 is constitutively phosphorylated on tyrosine and transcriptionally activated by BCR–ABL. 23 Therefore, we wished to ascertain whether I3C could inhibit STAT5 activity in K562 cells. As shown in Figure 2(a) and (b), western blot analysis revealed that treatment with 400 µM I3C leads to a substantial reduction in p-STAT5 levels in K562 cells, while the total STAT5 protein level remains unchanged. In addition, the effect of I3C on the expression levels of antiapoptotic genes, Bcl-xL and Mcl-1 (downstream effectors of STAT5), was determined by real-time polymerase chain reaction (PCR). As presented in Figure 2(c), the messenger RNA (mRNA) expression levels of Bcl-xL and Mcl-1 were downregulated in K562 cells treated with 400 µM I3C for 32 h. Collectively, these results indicate that I3C could inhibit STAT5 in CML cells. As STAT5 phosphorylation was reduced in K562 cells treated with I3C, we postulated the possibility of I3C inhibiting BCR–ABL kinase activity. The Crkl protein is a downstream signaling substrate of BCR–ABL, and its tyrosine phosphorylation (p-Crkl) serves as a specific indicator of BCR–ABL kinase activity in CML. 24 Therefore, the phosphorylation status of Crkl was evaluated in I3C-treated K562 cells by western blotting. As presented in Figure 2(d) and (e), I3C treatment had no effect on the phosphorylation of Crkl.

Effects of I3C on STAT5 signaling pathway and Crkl protein. (a) I3C inhibits phosphorylation/activation of STAT5 in K562 cells. Cells were treated with 200 and 400 µM I3C. After 18 and 32 h, equal amounts of total cell lysates (50 µg) were subjected to immunoblotting with specific antibodies against p-STAT5 and total STAT5. β-actin was used for normalization of protein levels. One representative western blot image of three independent experiments is presented. (b) The relative expression of each protein was quanied by densitometric analysis using ImageJ software and calculated by dividing the intensity of each band by the respective intensity of actin. (c) I3C downregulates expression of STAT5 target genes. K562 cells were treated with 400 µM I3C for 32 h. After RNA isolation and cDNA synthesis, the expression of the Bcl-xL and Mcl-1 genes was determined using SYBR Green real-time PCR, and all values were normalized to GAPDH (n = 3; *p < 0.05 compared with untreated control cells). (d) I3C treatment exhibits no significant effect on Crkl phosphorylation in K562 cells. Equal amounts of total cell lysates were subjected to immunoblotting with specific antibodies against p-Crkl and β-actin. One representative western blot image of three independent experiments is presented. (e) The intensity of the bands was quanied using ImageJ software and the relative expression of p-Crkl was calculated after normalizing to actin.
I3C inhibits Akt and its downstream target genes
The Akt pathway or PI3K-Akt pathway is an important signal transduction pathway that plays fundamental roles in cell survival, growth, proliferation, and apoptotic processes. 4 To explore the inhibitory effects of I3C on Akt pathway, the phosphorylation status of Akt at Ser473 was evaluated in K562 cells treated with 200 µM and 400 µM I3C by western blot analysis. As shown in Figure 3(a) and (b), the phosphorylation of Akt decreased in I3C-treated cells, which indicated the inhibitory effect of I3C on Akt protein. The downregulation of Akt phosphorylation was observed with 400 µM I3C after 32 h. Moreover, the expression of antiapoptotic Akt target genes, including Bcl-2, X-linked inhibitor of apoptosis protein (XIAP), and survivin in K562 cells was analyzed by real-time PCR. In parallel with the inhibition of Akt protein, I3C treatment significantly decreased gene expression of Bcl-2, XIAP, and survivin in K562 cells (Figure 3(c)). These data indicate that I3C can repress Akt signaling in CML cells.

Effects of I3C on AKT signaling pathway. (a) I3C attenuates phosphorylation of Akt. K562 cells were incubated with 200 and 400 µM I3C for 18 and 32 h. The total cell lysates were prepared and western blot analysis was performed using antibodies against total AKT and phospho-Akt (Ser 473). Data from a representative experiment are shown from a total of three independent experiments. (b) The relative expression of each protein was quanied using ImageJ software and calculated by dividing the intensity of each band by the respective intensity of actin (n = 3; *p < 0.05 compared with untreated control cells). (c) I3C downregulates expression level of Akt target genes. After treatment of K562 cells with 400 µM I3C for 32 h, the cells were harvested and subjected to SYBR Green real-time PCR using gene specific primer for Bcl-2, XIAP, and survivin. GAPDH was used as a housekeeping gene for normalization (n = 3; *p < 0.05 compared with untreated control cells).
I3C induces phosphorylation/activation of p38 MAPK in K562 cells
p38 MAPK regulates cell differentiation, growth, and apoptosis. 9 Evidence supports that the activation of the p38 MAPK signaling cascade plays an important role in potentiating the effects of imatinib on BCR-ABL-expressing cells. 10 Therefore, we were interested in ascertaining the effects of I3C on the phosphorylation/activation of p38 MAPK. Thus, K562 cells were treated with 200 and 400 µM I3C for 18 and 32 h, and the kinase activation was determined by western blotting using phospho-specific antibody against p38 (Thr-180, Tyr-182). As demonstrated in Figure 4(a) and (b), K562 cells treated with 400 µM I3C displayed an elevated level of phospho-p38 compared with the untreated cells.

I3C induces phosphorylation/activation of p38 MAPK in K562 cells. (a) After treatment with I3C (200 and 400 µM) for 18 and 32 h, the cells were harvested for protein analysis. A volume of 50 µg of protein from cell lysates was analyzed by SDS-PAGE and immunoblotted with specific antibodies against p38 MAPK and phospho-p38 MAPK. Western blot image is a representative of three independent experiments. (b) The relative expression of each protein was quanied by densitometric analysis using ImageJ software and calculated by dividing the intensity of each band by the respective intensity of actin (n = 3; *p < 0.05 compared with untreated control cells).
I3C downregulates expression of c-Myc and human telomerase reverse transcriptase genes
Human telomerase reverse transcriptase (hTERT) is a key component of the human telomerase complex that controls telomerase activity and plays a critical role in tumorigenesis. 25 c-Myc is a transcription factor that can regulate hTERT gene transcription. 26 To investigate the potential effects of I3C on hTERT and c-Myc, K562 cells were treated with 400 µM I3C for 32 h and analyzed for the expression of these genes using quantitative real-time PCR. As shown in Figure 5(a), a significant decrease in mRNA expression levels of hTERT and c-Myc was observed in K562 cells compared with the control cells. Western blot analysis confirmed that the expression level of c-Myc protein was also reduced in I3C-treated cells (Figure 5(b) and (c)). These results reveal that I3C may decrease the telomerase activity by downregulating hTERT transcription through c-Myc suppression in K562 cells.

I3C downregulates expression levels of c-Myc and hTERT. (a) K562 cells were treated with 400 µM I3C for 32 h, following which RNA was extracted and the expression of c-Myc and hTERT genes was measured using quantitative real-time PCR. GAPDH was used as a housekeeping gene for normalization (n = 3; *p < 0.05 compared with untreated cells). (b) K562 cells were incubated with 200 and 400 µM I3C for 18 h and 32 h. Cell lysates were prepared and western blot analysis was performed with indicated antibodies (data from a representative experiment are shown from a total of three independent experiments). (c) The relative expression of c-Myc protein was quanied using ImageJ software and calculated by dividing the intensity of each band by the respective intensity of actin (n = 3; *p < 0.05 compared with untreated control cells).
Discussion
Natural anticancer products may exhibit antitumor properties and induce apoptosis of various human cancer cells. The selected anticancer natural compounds activate cell death and induce apoptosis by perturbing multiple cellular signaling pathways in tumor cells. 27 I3C is a natural product derived from glucosinolates and is found in widely consumed vegetables of the Brassica genus such as broccoli, Brussels sprouts, and cabbage. 18 Previous in vitro studies have shown that I3C and its derivatives promote apoptosis and inhibit the growth of various human cancer cells without affecting normal cells. 19 Emerging evidence suggests that the therapeutic efficacy of I3C against cancer is mediated by the alteration of the activity of various signaling pathways such as nuclear factor kappa B (NF-κB), Akt, MAPK, Wnt, and Notch.19,28 Our data demonstrated that I3C-induced apoptosis proceeds through caspase-3 and caspase-9 activation as well as PARP cleavage in K562 cells (Figure 1(c)). Our results corroborate with the previous studies which reported that I3C and its derivatives induce apoptosis through PARP cleavage and caspase activation in acute myeloid and acute lymphoblastic leukemia cells.29,30 The leukemogenic effect of BCR–ABL is mediated through the activation of several downstream signaling pathways.2–4 Multiple lines of evidence indicate that STAT and Akt signaling pathways are crucial for disease evolution and progression in CML.2,7 Among the seven STAT proteins, the persistent activation of the transcription factor STAT5 is a molecular hallmark of CML that plays a critical role in the pathogenesis of this leukemia.5,6 Cumulative evidence suggests that STAT5 is necessary for transformation and cell cycle progression of BCR-ABL-positive cells, thereby underpinning STAT5 as a molecular target in the treatment of CML.5,6,31 Our data reveal that phospho-STAT5 as a key effector molecule in the oncogenic network of BCR–ABL can be potentially targeted by I3C in K562 cells. A recent report supports our findings that diindolylmethane (DIM), a natural compound derived from I3C, can inhibit STAT signaling pathway in human bladder cancer, leading to cell cycle arrest and apoptosis. 32 Furthermore, Lian et al. 33 reported that phosphorylation and subsequent activation of STAT3 transcription factor could be inhibited by I3C in human pancreatic cancer cells. The antiapoptotic effect of STAT5 is mediated by the upregulation of several target genes, including Mcl-1and Bcl-xL. There is evidence indicating that Mcl-1 and Bcl-xL contribute to the survival of Philadelphia chromosome-positive (Ph+) leukemic stem cells and disease progression in CML.34,35 Moreover, it has been demonstrated that Bcl-xL expression is downregulated during etoposide-induced apoptosis of myeloid leukemia K562 cells. 4 Importantly, our data reveal the repressive effect of I3C on Bcl-xL and Mcl-1 expression. The tyrosine phosphorylation and activation of STAT5 commonly occur by cytokine signaling through JAK/STAT pathway. 36 However, in CML cells, oncogenic BCR–ABL causes the tyrosine phosphorylation and activation of STAT5, which stimulates the expression of genes that promote cell growth and survival.6,23 Thus, we hypothesized that the suppressive effect of I3C on STAT5 may be associated with the inhibition of the BCR–ABL kinase activity. To this end, we assessed the Crkl phosphorylation as a specific indicator of the BCR–ABL kinase activity. Our findings demonstrated that I3C treatment had no effect on the phosphorylation status of Crkl in K562 cells (Figure 2(d) and (e)). These results suggest that I3C does not act as a direct inhibitor of the BCR–ABL kinase activity.
Akt activation has been idenied as a key signaling molecule in BCR-ABL-mediated leukemogenesis. 4 Molecular studies on CML indicate that on treatment with a growth factor, Akt and STAT5 remained phosphorylated and activated in CML CD34+ cells even in the presence of BCR–ABL inhibitors. 37 There is evidence suggesting that the cooperation of STAT5 and Akt signaling pathways is critical in the disease progression and evolution of drug-resistant CML. 7 Therefore, it appears that the simultaneous inhibition of STAT5 and Akt signaling pathways can lead to a significant suppression of CML cell growth. In parallel to the inhibition of STAT5 phosphorylation, exposing K562 cells to I3C resulted in a significant decrease in the phosphorylation of Akt protein (Figure 3(a)). Similar results were observed in the Akt inhibition by OSU-A9, an indole-3-carbinol derivative, in the AML cell lines, HL-60 and THP-1. 29 Furthermore, Chinni and Sarkar 38 demonstrated that the inhibition of Akt activation is a key event in I3C-induced apoptosis of prostate cancer cells. AKT signaling promotes cell growth and survival through the activation of downstream effector molecules such as XIAP, Bcl-2, and survivin. XIAP was found to be overexpressed in cells from CML patients and associated with an unfavorable prognosis. 39 Similarly, high levels of survivin expression correlated with BCR–ABL expression levels in patients with CML. 40 Our results demonstrated that the mRNA expression levels of XIAP, Bcl-2, and survivin significantly decreased in I3C-treated cells (Figure 3(c)). Therefore, Akt signaling pathway as a significant therapeutic target in CML can be potentially suppressed by I3C in K562 cells.
p38 MAPK is a class of MAPKs responsive to multiple stress stimuli and involved in cell differentiation, apoptosis, and autophagy. 9 p38 can play a dual role as a regulator of cell death in CML through various mechanisms. On one hand, p38 mediates cell survival and inhibits apoptosis in CML cells treated with arsenic trioxide (AT). 41 On the other hand, some reports suggest that p38 exerts a proapoptotic role in CML cells treated with STI-57 and dasatinib.10,11 In agreement with the latter reports, in our study, I3C activated the p38 signaling pathway in K562 cells, as demonstrated by the increase in phospho-p38 levels (Figure 4(a) and (b)). Therefore, our results indicate that p38 can play an ambivalent role in apoptosis, depending on the cell context and death stimulus.
Human telomerase is a specialized enzyme composed of two core subunits: hTERT and human telomerase RNA template (hTR). hTERT, a catalytic subunit of the enzyme telomerase, maintains telomere length and controls telomerase activity. 25 hTERT expression and telomerase activity are increased in the bone marrow cells of CML patients. Bock et al. 42 reported that hTERT expression is higher in patients with CML and refractory anemia (RA) than in those with other hematopoietic stem cell disorders and non-neoplastic hematopoiesis. Therefore, telomerase can be considered as an attractive target for the diagnosis and treatment of CML. BCR–ABL is known to play an important role in the regulation of telomerase activity through the JAK/STAT pathway. 43 Several lines of evidence highlight a critical role of STAT5 in hTERT gene expression and telomerase activity in BCR-ABL-positive cells.44,45 Adler et al. 46 showed that I3C can inhibit telomerase activity and hTERT mRNA expression in prostate cancer cell lines. In this study, we revealed that hTERT expression is significantly repressed in K562 cells following exposure to I3C (Figure 5(a)). Recent studies have idenied that the hTERT gene is a direct target for transcriptional regulation by c-Myc.26,47 c-Myc upregulation is correlated with the activation of STAT5 signaling in BCR-ABL-positive cells, which indicates that c-Myc is a molecular target for STAT5. 48 In agreement with the effect of I3C on the expression of hTERT, the mRNA and protein expression levels of c-Myc was also reduced in I3C-treated cells compared with the control cells (Figure 5(a)–(c)). Thus, the most likely explanation for our results is that I3C-induced suppression of STAT5 may give rise to the downregulation of c-Myc and subsequent inhibition of hTERT expression (Figure 6).
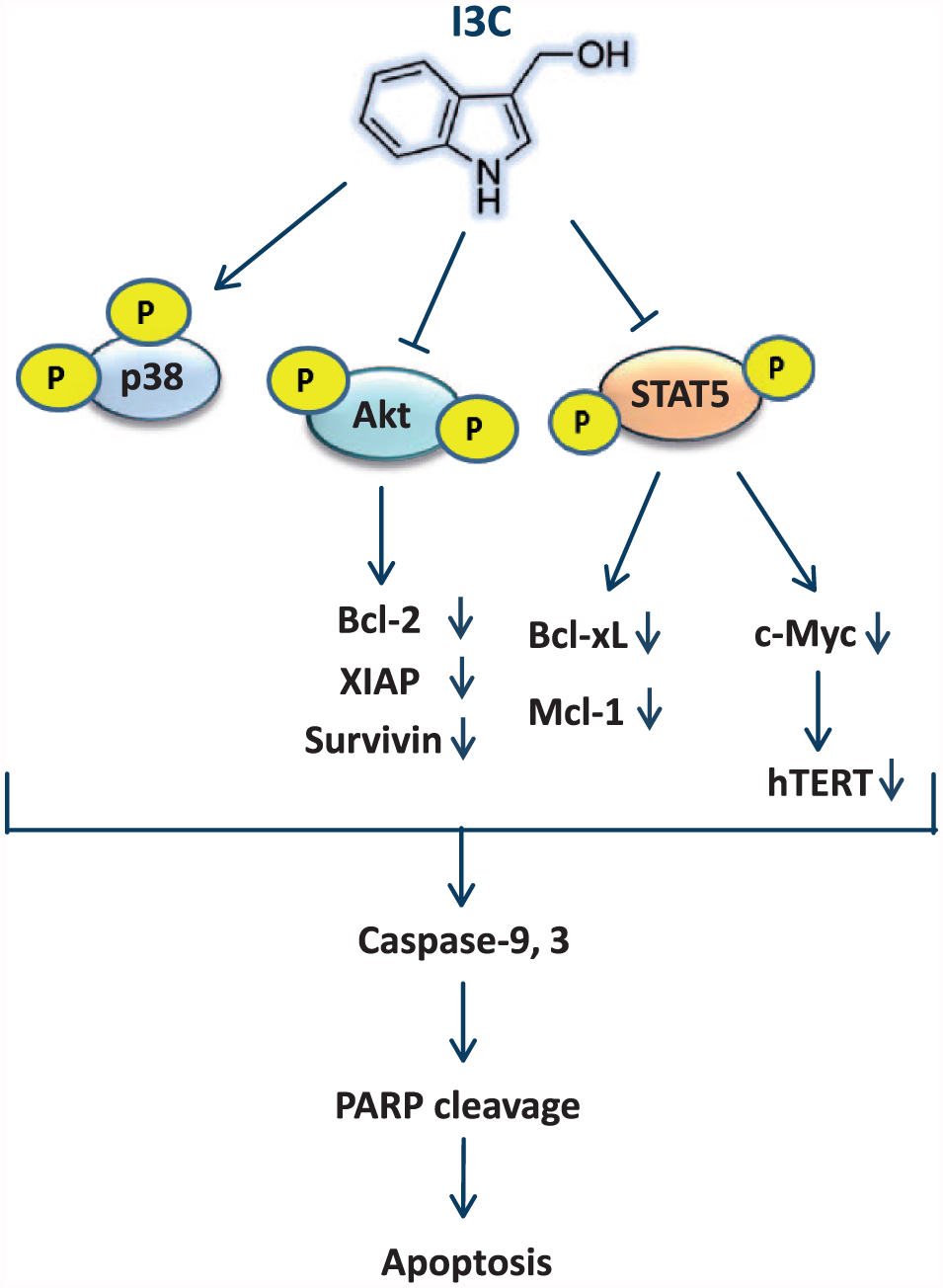
Schematic representation of the plausible molecular mechanism proposed for the induction of apoptosis by I3C in K562 cells. This molecular mechanism is regulated via the activation of p38 and repression of Akt and STAT5 signaling pathways. Subsequently, in response to the repression of Akt and STAT5, the expression of antiapoptotic (Mcl-1, Bcl-xL, Survivin, Bcl-2, XIAP) and proliferative (c-Myc, hTERT) gene products were decreased, resulting in the activation of a cascade of caspases and PARP cleavage.
In conclusion, to the best of our knowledge, these data provide mechanistic evidence for the first time that the natural compound I3C potently induces apoptosis of CML cells through the activation of the caspase cascade and perturbation of critical signaling pathways that are implicated in the pathogenesis of CML. Considering the safety and tolerability of I3C proved in clinical trials, 49 along with its targeted ability to induce apoptosis of CML cells, I3C could be a potential therapeutic candidate for the treatment of CML. However, further in vivo studies are required to determine whether I3C has potential in the treatment of CML.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Grant No. 25317 from Iran University of Medical Sciences.
