Abstract
The role and mechanism of miR-452-5p in lung adenocarcinoma remain unclear. In this study, we performed a systematic study to investigate the clinical value of miR-452-5p expression in lung adenocarcinoma. The expression of miR-452-5p in 101 lung adenocarcinoma patients was detected by quantitative real-time polymerase chain reaction. The Cancer Genome Atlas and Gene Expression Omnibus databases were joined to verify the expression level of miR-452-5p in lung adenocarcinoma. Via several online prediction databases and bioinformatics software, pathway and network analyses of miR-452-5p target genes were performed to explore its prospective molecular mechanism. The expression of miR-452-5p in lung adenocarcinoma in house was significantly lower than that in adjacent tissues (p < 0.001). Additionally, the expression level of miR-452-5p was negatively correlated with several clinicopathological parameters including the tumor size (p = 0.014), lymph node metastasis (p = 0.032), and tumor–node–metastasis stage (p = 0.036). Data from The Cancer Genome Atlas also confirmed the low expression of miR-452 in lung adenocarcinoma (p < 0.001). Furthermore, reduced expression of miR-452-5p in lung adenocarcinoma (standard mean deviations = −0.393, 95% confidence interval: −0.774 to −0.011, p = 0.044) was validated by a meta-analysis. Five hub genes targeted by miR-452-5p, including SMAD family member 4, SMAD family member 2, cyclin-dependent kinase inhibitor 1B, tyrosine 3-monooxygenase/tryptophan 5-monooxygenase activation protein epsilon, and tyrosine 3-monooxygenase/tryptophan 5-monooxygenase activation protein beta, were significantly enriched in the cell-cycle pathway. In conclusion, low expression of miR-452-5p tends to play an essential role in lung adenocarcinoma. Bioinformatics analysis might be beneficial to reveal the potential mechanism of miR-452-5p in lung adenocarcinoma.
Keywords
Introduction
The incidence and mortality of lung cancer is the highest among all malignant neoplasms in China as well as in the world.1–3 Lung cancer can be divided into two histological categories: non-small-cell lung cancer (NSCLC) and small-cell lung cancer (SCLC). NSCLC accounts for about 80% of lung cancer cases, including lung adenocarcinoma (LUAD), lung squamous cell carcinoma (LUSC), and lung large-cell cancer (LULC).4,5 The onset of LUAD is occult and is more common than other histological subtypes of lung cancer because of its high incidence. Together with research on the biological behavior of lung cancer and targeted drugs, the molecular targeting therapy of LUAD has recently made remarkable progress.6–8 However, resistance to targeted drugs is still high, and the applicable patient population is limited. Therefore, it is urgent to study the molecular mechanism of LUAD and to explore new methods of its diagnosis and treatment.
MicroRNA (miRNA) is a class of endogenous non-coding RNA molecules that regulate protein translation, with a length of approximately 22 nucleotides.9,10 Studies have shown that miRNAs silence genes by inhibiting the synthesis of related proteins or degrading the messenger RNA (mRNA) of its target gene and thus play an important role in the regulation of growth, aging, cell proliferation, differentiation, apoptosis, and even development of cancer.11,12 Many studies have reported the role of miRNA in the development of LUAD and its important value in the diagnosis, treatment, and prognosis of this disorder.13–17
MiR-452-5p, previously also named as miR-452, is an RNA molecule obtained by modification of the 5′ end of pre-miR-452. Previous studies have reported that miR-452-5p expressions in different neoplasms are distinct, which is upregulated in urinary tract epithelial tumors, esophageal cancer, and liver cancer,18–20 but downregulated in breast cancer, prostate cancer, and glioma.21–23 Currently, there have been only a few reports regarding the relationship between miR-452-5p and lung cancer. The clinical significance and possible molecular function (MF) of miR-452-5p in NSCLC was reported by Zhang et al. 24 and He et al.25,26 However, clinical samples in their research were restricted by region and quantity, and the complicated mechanism of miR-452-5p in LUAD remained unclear. The Cancer Genome Atlas (TCGA) and Gene Expression Omnibus (GEO) aim to achieve a comprehensive understanding of cancer at the molecular level using genomic analyses, including large-scale genome sequencing and microarray, for improving the diagnosis, treatment, and prevention of cancer. In this study, we determined the expression levels of miR-452-5p in paraffin-embedded samples from 101 patients with LUAD. In order to reduce the errors caused by sample size and study population, we validated the expression level of miR-452-5p with LUAD using GEO and TCGA databases. Combined with several online databases and bioinformatic software, the function and signal pathway enrichment analyses of miR-452-5p target genes were further performed to provide possibilities for future clarification of the molecular mechanism of miR-452-5p in LUAD.
Materials and methods
Clinical tissue samples
For this retrospective study, the paired LUAD and adjacent non-cancerous lung tissue were obtained from 101 LUAD patients (56 males and 45 females; mean age = 61.10 years; range = 23–90 years) in the Department of Pathology, First Affiliated Hospital of the Guangxi Medical University (Nanning, Guangxi, China) between January 2012 and June 2014. All specimens were fixed in 10% formalin and embedded by paraffin (FFPE). The study protocol was approved by the ethics committee of the First Affiliated Hospital of Guangxi Medical University, with informed consent signed by all participants. All the specimens were evaluated and diagnosed by two independent pathologists. In Table 1, clinicopathological features of all LUAD patients were summarized.
Relationship between the expression of miR-452-5p and clinicopathological parameters in LUAD.

LUAD: lung adenocarcinoma; miRNA: microRNA; SD: standard deviation; TNM: tumor–node–metastasis.
Student’s paired t-test was used for the comparison between two groups.
One-way analysis of variance (ANOVA) test was performed.
Statistically significant (p < 0.05).
Quantitative real-time polymerase chain reaction detection of miR-452-5p in LUAD
Total RNA was extracted from FFPE tissue using the miRNeasy Kit (Qiagen, KJVenlo, the Netherlands), and the concentration and purity of RNA were determined by NanoDrop 2000 (Wilmington, DE, USA), in accordance with our previous reports.15–17,27,28 The internal reference was the combination of miR-191 and miR-103, whose stability had been verified previously.15–17,27,28 Sequences of the miRNAs in this study were as follows: AACUGUUUGCAGAGGAAACUGA (miR-452-5p); CAACGGAAUCCCAAAAGCAGCU (miR-191); and AGCAGCAUUGUACAGGGCUAUGA (miR-103). The miR-452-5p expression was computed with the formula 2−Δcq.27,29,30
Verification of miR-452-5p expression level in LUAD using the TCGA and GEO databases
We extracted miR-452-5p (previously named as miR-452) expression and the clinical information including prognosis of LUAD patients from TCGA and GEO. Then, we summarized, calculated, and analyzed these data by Microsoft Office Excel and SPSS software. The primary expression data of miR-452-5p without being standardized were log2-transformed for further study.
Prediction of miR-452-5p target genes
The prediction was performed using prediction software, including TargetScan, PicTar2, miRanda by RNAhybrid, miRDB, RNA22-HAS, TargetMiner, EMBL-EBI, miRWalk, DIANA-microT, mirBridge, miRMap, miRNAMap, and PITA. At the same time, target genes of miR-452-5p, which had been confirmed by experiments, were searched in PubMed and EMBASE databases. Potential miR-452-5p target genes that were positive in all seven software programs or were confirmed by experiment were used for further analyses.
Bioinformatic analysis of miR-452-5p target genes
The potential miR-452-5p target genes were subjected to Gene Ontology (GO) and pathway analyses, using Database for Annotation, Visualization and Integrated Discovery (DAVID) database; EnrichmentMap (a plug-in of Cytoscape); and Molecular Signatures Molecular Tag Database (MSigDB) database derived from Gene Set Enrichment Analysis (GSEA). 31 Target genes were also mapped to the database of Kyoto Encyclopedia of Genes and Genomes (KEGG) to identify the signaling pathways; p < 0.05 was regarded as a significant threshold to identify gene sets with high frequency of annotation in its related pathway. STRING database was utilized for the selection of hub genes which were probably involved in the strategic pathway related to LUAD. Hub genes were identified by the combined score summarized in STRING with Cytoscape.32,33
Statistical analyses
Statistical analyses were carried out with SPSS 22.0 (SPSS Inc., Chicago, IL, USA), StataSE 12.0 (StataCorp LP, College Station, TX, USA), GraphPad Prism 5 (GraphPad Software, Inc., San Diego, California, USA), R version 3.3.0, and Microsoft Office 2007 software packages. Student’s t-test or Mann–Whitney test was chosen to study the significance of difference between two groups in view of the normal distribution curve. One-way analysis of variance (ANOVA) test was used when comparison existed among three groups. Spearman correlation was applied to analyze the relationship between miR-452-5p expression and clinicopathological parameters. Receiver operating characteristic (ROC) curve analyses were used with the area under the curve (AUC) calculated for the differential diagnostic value of miR-452-5p to differentiate LUAD from non-cancer lung tissues. The effect of miR-452-5p expression on the survival of patients was statistically analyzed using Kaplan–Meier curves and the log-rank test. The Cox proportional hazard regression model was also used to examine the risk factors of LUAD. Differences with p < 0.05 were considered statistically significant. The meta-analysis was applied with StataSE 12.0. If observation of standard mean deviation (SMD) <0 and with its 95% confidence interval (CI) not crossing zero, indicated that miR-452-5p had lower expression in cancerous specimens than that in non-cancerous specimens. In contrast, if the overall SMD >0 and with its 95% CI not crossing zero, miR-452-5p was considered as highly expressed in cancerous specimens. Publication bias was detected by Begg’s funnel plot and Egger’s test.
Results
Diagnostic value of miR-452-5p expression in LUAD by quantitative real-time polymerase chain reaction
As shown in Figure 1(a) and Table 1, the expression of miR-452-5p in LUAD was lower than that in adjacent tissues (2.2952 ± 1.4518 vs 5.3319 ± 3.0569, p < 0.001). The AUC was 0.855 (95% CI: 0.805–0.906, p < 0.001), with a sensitivity of 85.1%, specificity of 76.2%, and diagnostic threshold value of 2.8 (Figure 1(b)).

MiR-452-5p expression was reduced in human LUAD tissues and its diagnostic value. (a) The expression levels of miR-452-5p in 101 LUAD tissues and the paired non-cancerous lung tissues detected by RT-qPCR. (b) ROC curve analysis of miR-452-5p expression levels for the discrimination between LUAD tissues and non-cancerous lung tissues.
The relationship between miR-452-5p expression and clinicopathological features of LUAD by quantitative real-time polymerase chain reaction
The expression levels of miR-452-5p in LUAD groups with different tumor size, lymph node metastasis, and tumor–node–metastasis (TNM) stage showed statistical significance (Figure 2). As shown in Table 1, miR-452-5p expression in LUAD patients with tumor diameters ⩽3 cm was higher than that in LUAD patients with tumor diameters >3 cm (p = 0.045). Also, miR-452-5p expression in LUAD patients with lymph node metastasis was lower than that in LUAD patients without lymph node metastasis (p = 0.018). MiR-452-5p expression in LUAD patients with vascular invasion was lower than that in LUAD patients without vascular invasion (p = 0.009). In addition, among the different TNM stages, relative miR-452-5p expression in stage III–IV LUAD patients was significantly lower than that of LUAD patients at stages I and II (p = 0.016). Spearman’s correlation analyses showed that the expression of miR-452-5p was negatively correlated with tumor size (r = −0.243, p = 0.014), lymph node metastasis (r = −0.214, p = 0.032), and TNM stage (r = −0.209, p = 0.036). MiR-452-5p was not significantly correlated with age, gender, smoke, pathological grading and vascular invasion.
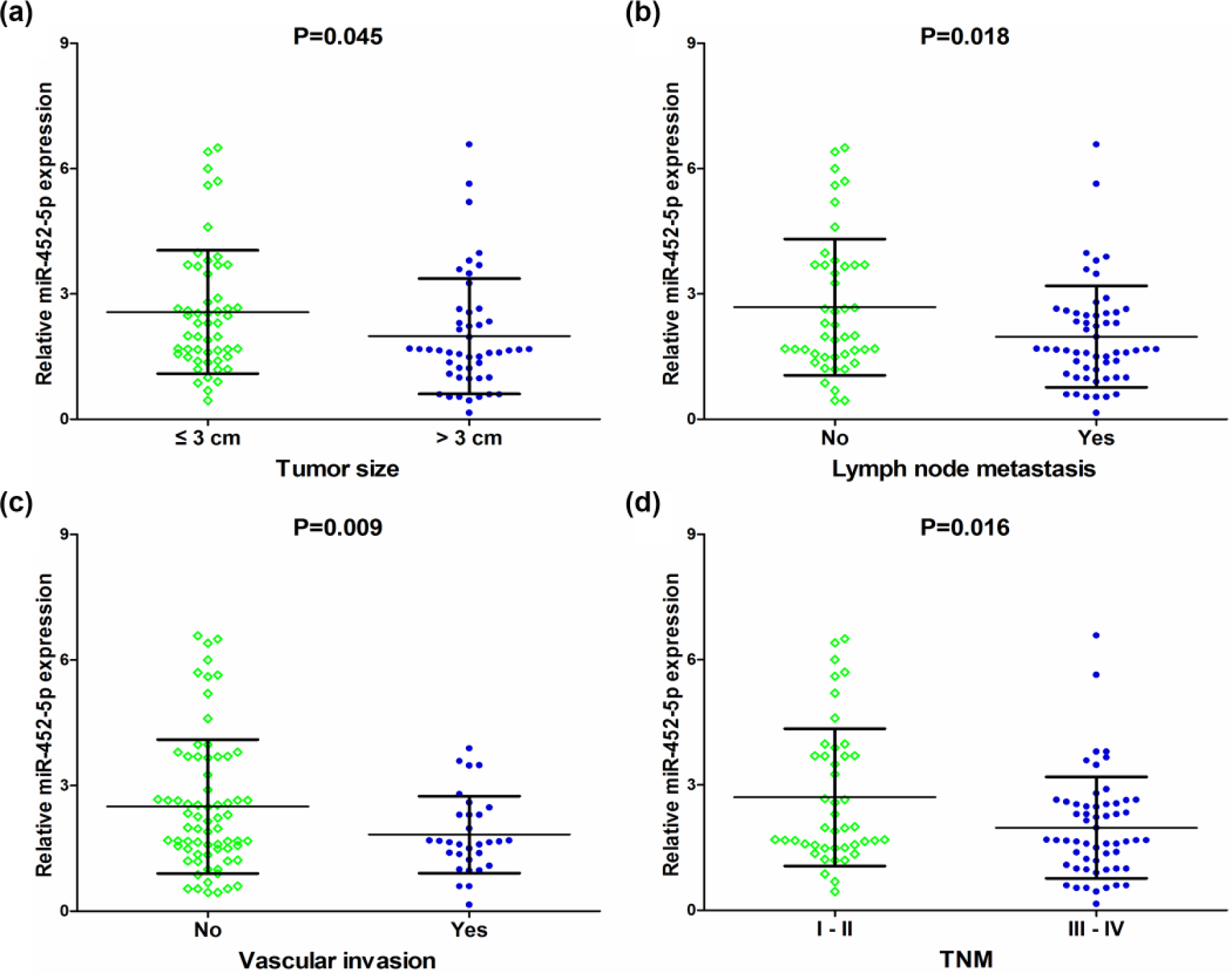
Relationship between miR-452-5p expression and clinicopathological parameters in LUAD. (a) MiR-452-5p expression levels in patients with tumor diameters ⩽3 cm and with tumor diameters >3 cm. (b) MiR-452-5p expression levels in patients with lymph node metastasis and without lymph node metastasis. (c) MiR-452-5p expression levels in patients with vascular invasion and without vascular invasion. (d) MiR-452-5p expression levels in stage-III and -IV patients and in stage-I and -II patients.
The ROC was used to verify the predictive performance of miR-452-5p expression levels in various clinical parameters of LUAD. The AUC of miR-452-5p expression for LUAD patient’s tumor size was 0.640 (95% CI: 0.532–0.749, p = 0.015). Furthermore, in patients with lymph node metastasis and vascular invasion, the AUC was 0.624 (95% CI: 0.514–0.735, p = 0.033) and 0.610 (95% CI: 0.499–0.722, p = 0.078), respectively. Moreover, the AUC was 0.622 (95% CI: 0.511–0.733, p = 0.037), for TNM stage of LUAD (Figure 3).

ROC curve analyses of miR-452-5p expression levels for clinicopathological parameters of LUAD. (a) ROC curve of miR-452-5p expression levels for tumor size. (b) ROC curve of miR-452-5p expression levels for lymphatic metastasis. (c) ROC curve of miR-452-5p expression levels for vascular invasion. (d) ROC curve of miR-452-5p expression levels for TNM stage.
Prognostic value of miR-452-5p in LUAD by quantitative real-time polymerase chain reaction
Clinical follow-up data of 57 patients were used for survival analyses. To evaluate the relationships between miR-452-5p expression levels and patient survival, a median value of 1.98 was selected as the miR-452-5p expression level threshold. The mean survival time of LUAD patients with low miR-452-5p expression (n = 29) was longer than that with high miR-452-5p expression (n = 28), however, without being statistically significant (Figure 4; 19.908 ± 2.969 months vs 17.938 ± 3.789 months, p = 0.322). Cox regression analyses indicated that miR-452-5p was not a statistically significant prognostic indicator for patients with LUAD (univariate: hazard ratio (HR) = 0.729, 95% CI: 0.388–1.371, p = 0.326; multivariate: HR = 0.611, 95% CI: 0.297–1.254, p = 0.179; Table 2).
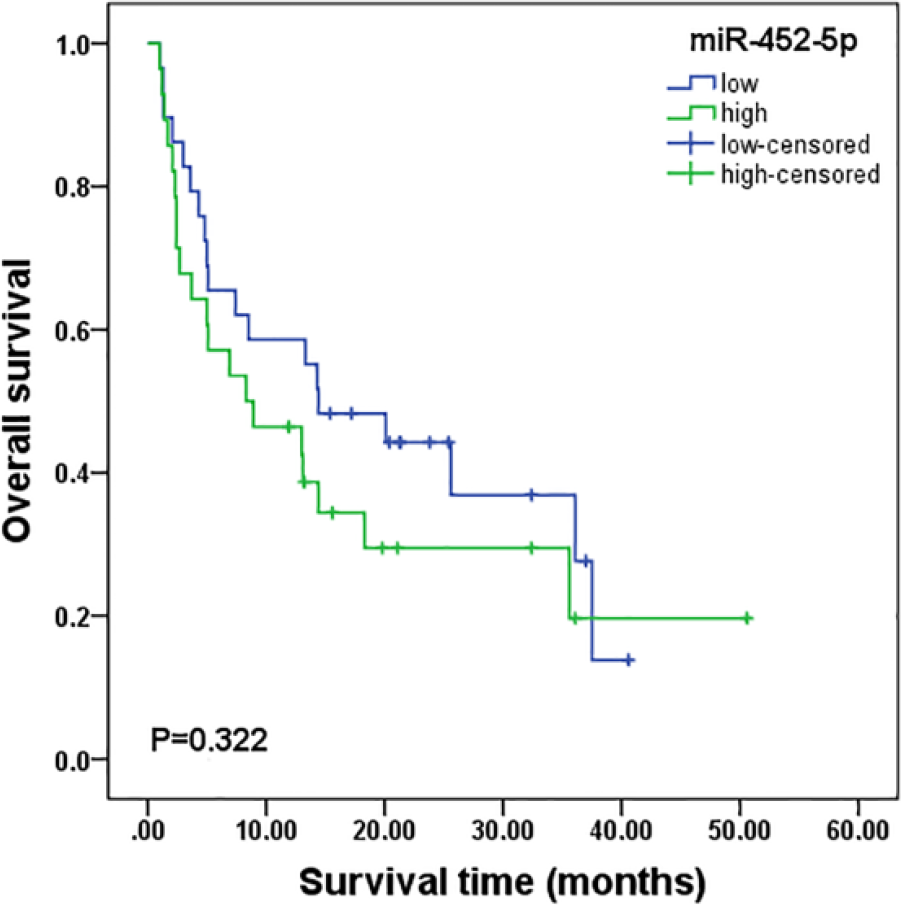
Correlation of miR-452-5p expression with survival. The Kaplan–Meier survival analysis showed that the mean survival time of LUAD patients with relative low miR-452-5p expression was longer than LUAD patients with relatively high miR-452-5p expression, but there was no statistical significance (19.908 ± 2.969 months vs 17.938 ± 3.789 months, p = 0.322).
Multivariate analysis of clinicopathological parameters for overall survival in LUAD.

HR: hazard ratio; 95% CI: 95% confidence interval; LUAD: lung adenocarcinoma; TNM: tumor–node–metastasis.
Statistically significant (p < 0.05).
Diagnostic value of miR-452 in LUAD analyzed with TCGA data
Since TCGA did not provide the data of expression level of miR-452-5p, we calculated the clinical value of miR-452. A total of 504 cases of LUAD (458 cancers and 46 adjacent tissues) patients with differential miR-452 expression were extracted from the TCGA. As shown in Figure 5(a) and Table 3, the expression of miR-452 in LUAD was lower than that in adjacent tissues (8.3539 ± 1.40738 vs 8.7608 ± 2.06101, p < 0.001). The AUC of miR-452 expression for LUAD was 0.678 (Figure 5(b); 95% CI: 0.587–0.769, p < 0.001), with a sensitivity of 63.3%, specificity of 80.4%, and diagnostic threshold value of 8.8267.

Diagnostic significance of miR-452 expression in LUAD analyzed with TCGA data. (a) The expression levels of miR-452 in 458 LUAD tissues and 46 non-cancerous lung tissues. (b) ROC curve analysis of miR-452 expression levels for the discrimination between LUAD tissues and non-cancerous lung tissues.
The relative expression of miR-452-5p in LUAD analyzed with data of TCGA and GEO.

LUAD: lung adenocarcinoma; TCGA: The Cancer Genome Atlas; GEO: Gene Expression Omnibus; SD: standard deviation.
Detection of the miR-452 expression.
Mann–Whitney test was used for the comparison between two groups.
Student’s paired t-test was used for the comparison between two groups.
Statistically significant (p < 0.05).
Prognostic value of miR-452 in LUAD analyzed with TCGA data
From the TCGA database, 431 cases of LUAD were obtained for Kaplan–Meier curve survival analyses, with a mean value of 8.3768 was selected as the miR-452 expression level threshold (Figure 6). Although the results indicated that LUAD patients with low miR-452 expression (n = 210) obtained a longer mean survival time than LUAD patients with high miR-452 expression (n = 221), it was unable to determine whether miR-452 could be a prognostic factor in patients with LUAD (82.320 ± 12.962 months vs 62.658 ± 8.951 months, p = 0.714).

Correlation of miR-452 expression with the survival of LUAD patients of TCGA. The Kaplan–Meier survival analysis showed the mean survival times of LUAD patients with relative low miR-452 expression were longer than LUAD patients with relative high miR-452 expression, but there was also no statistical significance (82.320 ± 12.962 months vs 62.658 ± 8.951 months, p = 0.714).
Verification of miR-452-5p expression in LUAD via a meta-analysis
A total of 1202 cancer and non-cancer samples from nine eligible datasets, including 496 cases from seven eligible GEO datasets, 504 cases from the TCGA dataset, and 202 cases from the clinical samples in house, were screened in the final meta-analysis (Supplementary Table S1). Additionally, the relationship between miR-452-5p and patients with LUAD in each of 7 GEO datasets was also illustrated (Figure 7 and Table 3). In the meta-analysis, the random-effects model was selected to calculate the pooled SMD and 95% CI because of the noticeable heterogeneity (I2 = 82.6%, p < 0.001). The result showed that the expression of miR-452-5p in LUAD was significantly lower than that in control groups (Figure 8(a), SMD = −0.393, 95% CI: −0.774 to −0.011, p = 0.044). Subgroup analysis indicated that detection methods of miR-452-5p expression may be the source of heterogeneity because of the reduction of heterogeneity in the miRNA array group (I2 = 51.4%, p = 0.084). The studies were symmetrically arranged in the funnel plot (p = 0.458), indicating no publication bias in the research (Figure 8(b)).

Relative expression of miR-452-5p in LUAD with 7 GEO datasets. In the 7 GEO datasets, significant downregulation of miR-452-5p in the patients with LUAD was detected in GSE47525 (2.3333 ± 0.19664 vs 2.8929 ± 0.51361, p = 0.020), GSE48414 (−0.9802 ± 2.76291 vs 0.4707 ± 0.44619, p = 0.005), and GES63805 (5.2197 ± 0.43217 vs 5.4962 ± 0.55624, p = 0.032), respectively. The results of GSE14936, GSE25508, GSE40738, and GSE74190 showed no statistical significance.

Meta-analysis of miR-452-5p expression in LUAD combined with GEO, TCGA, and our clinical specimens. (a) Forrest plot of SMD for the illustration of miR-452-5p expression level in LUAD compared with non-cancerous group. Subgroup analysis suggested that the source of heterogeneity might generate from detection methods including miRNA array, RNAseq, and RT-qPCR. (b) Funnel plot of nine datasets included in the meta-analysis of miR-452-5p expression in LUAD.
Potential target genes of miR-452-5p
A total of 17,094 candidate target genes were screened using a total of 14 prediction programs. The positive candidates verified by at least seven programs or validated by experiments were further analyzed. Thus, a total of 249 target genes of miR-452-5p were selected and subjected to GO and pathway analyses.
GO and KEGG pathway analyses
The predicted 249 target genes of miR-452-5p were inputted into the DAVID database for GO analyses. In addition, the results showed that at the level of biological process (BP), target gene sets of miR-452-5p were closely related to regulation of transcription and commissural neuron axon guidance and transcription (p < 0.01). Based on cellular component (CC), they were mainly involved in the assembly of cellular structures such as nucleoplasm, nucleus, and cytoplasmic vesicle membrane (p < 0.01). Regarding MF, they were mainly enriched in protein binding, chromatin binding, and zinc ion binding (p < 0.01). Simultaneously, the visualization function of Cytoscape was used to graphically display the results of GO enrichment annotation analyses (p < 0.01, Q < 0.1, overlap coefficient > 0.5), allowing more intuitive presentation of the results (Figure 9(a)).

GO and KEGG pathway analyses with the prospective target genes of miR-452-5p. (a and b) A total of 17 annotation items of GO (p < 0.01, Q < 0.1, overlap coefficient > 0.5) and 6 annotation items of KEGG (p < 0.05, Q < 0.05, overlap coefficient > 0.5) pathways were connected using the EnrichmentMap (a plug-in of Cytoscape) due to the overlapped target genes. (c) The GO (p < 0.01) and KEGG (p < 0.05) annotation items of miR-452-5p target gene set. The intensity of the color indicates the p value, and the size of nodes and edges is representative of the number of genes.
As shown in Figure 9(b), combined with DAVID database and Cytoscape, pathway enrichment analyses of the gene sets were performed to identify biological pathway information for the above target gene set in the KEGG pathway database. The results showed that the predicted miR-452-5p target gene sets were significantly enriched in signaling pathways regulating pluripotency of stem cells, oocyte meiosis, and cell cycle (Figure 9(c) and Supplementary Table S2; p < 0.05, Q < 0.5, overlap coefficient > 0.5).
In addition, the gene expression profile graph from the Global Cancer Map was also processed in MSigDB. So that we could identify the expression of significantly altered genes among the miR-452-5p target genes, as well as their relative tendencies of expression in the defined functional groups (Figure 10).

Compendia expression profiles of miR-452-5p targets adapted from Global Cancer Map. The heatmap shows different expression levels of miR-452-5p target genes between experimental group and control group in various neoplasms. Gene family classification of the target genes and the sorted gene list calculated by the enrichment score were beside the heatmap.
Network analysis for selection of hub genes
The network was consisted of 249 nodes and 118 edges. Among 249 miR-452-5p target genes, 95 target genes were connected based on experiment, co-expression, neighborhood, or some other evidences summarized by the STRING. The disconnected target genes (n = 154) were removed from the network (Figure 11). Finally, we screened out 10 hub genes (nodes connected with more than six edges) from the 95 target genes. These hub genes were SMAD family member 4 (SMAD4); myocyte enhancer factor 2C (MEF2C); tyrosine 3-monooxygenase/tryptophan 5-monooxygenase activation protein epsilon (YWHAE); SMAD family member 2 (SMAD2); SWI/SNF-related, matrix-associated, actin-dependent regulator of chromatin, subfamily a, member 4 (SMARCA4); tyrosine 3-monooxygenase/tryptophan 5-monooxygenase activation protein beta (YWHAB); platelet activating factor acetylhydrolase 1b regulatory subunit 1 (PAFAH1B1); protein kinase cyclic adenosine monophosphate (cAMP)-dependent type-II regulatory subunit beta (PRKAR2B); cyclin-dependent kinase inhibitor 1B (CDKN1B); and replication protein A1 (RPA1). Five hub genes including SMAD4, SMAD2, CDKN1B, YWHAE, and YWHAB were significantly enriched in cell-cycle pathway.

Network analysis of miR-452-5p target genes by STRING. The 10 hub genes highlighted by red are (nodes connected with more than 6 edges) screened out from the network. Network nodes represent proteins, and network edges represent protein–protein associations. Interactions are built on the evidences such as experimentally determined, gene neighborhood and co-expression. The hub genes were selected among the nodes interacted by more edges.
Discussion
This study showed that miR-452-5p expression in LUAD was significantly downregulated than that in non-cancerous lung tissues, and the diagnostic value of miR-452-5p expression for LUAD was also confirmed with clinical detection in house, data digging with TCGA and GEO as well as a comprehensive meta-analysis. Correlation analysis showed that the expression of miR-452-5p was closely associated with tumor size, lymph node metastasis, and TNM stage. Survival analysis revealed that LUAD patients with low miR-452-5p expression could obtain a longer mean survival time. These findings indicate that miR-452-5p could play an essential part in the oncogenesis and progression of LUAD. Moreover, five hub target genes of miR-452-5p involved in the cell-cycle pathway were predicted and screened for the guidance of future study to elucidate the mechanism of LUAD.
The miRNAs mainly perform their biological functions by complete or incomplete complementary binding with the 3′-untranslated region (3′-UTR) of mRNAs.34–37 MiR-452-5p is located in Xq28 of human being, which can target multiple genes and thus play a potentially important role in the BP of tumors through a variety of mechanisms. Hu et al. 21 reported that miR-452-5p expression was downregulated in breast cancer, which may lead to adriamycin resistance by targeting the insulin analogue, insulin like growth factor 1 receptor (IGF-1R), and the docetaxel resistance of breast cancer cells by targeting the anaphase promoting complex 4 (APC4). Liu et al. reported that the expression of multifunctional stem cell regulatory factors such as SRY-box 2 (SOX2) was downregulated after upregulating the expression of miR-452-5p in glioma cell lines. And they confirmed the downregulation of miR-452-5p in glioma cells and tissues was correlated with promoter methylation. 23 Zheng et al. 20 reported that the high expression of miR-452-5p was observed in hepatocellular carcinomas, suggesting that miR-452-5p can promote hepatocellular carcinomas by targeting CDKN1B. Zhang et al. 24 reported that miR-452-5p inhibited the proliferation, invasion, and migration of NSCLC cells via epithelial–mesenchymal transition (EMT) process. Meanwhile, He et al. 25 suggested that miR-452-5p enhanced the invasion capability of NSCLC cells by targeting BMI1 proto-oncogene, polycomb ring finger (BMI1). However, the possible function and mechanism of miR-452-5p expression in LUAD remain unclear.
Up to now, only He et al. 26 reported that the downregulated expression of miR-452-5p was related to the clinicopathological features in patients with NSCLC. Although the theme of our research was similar with He et al., 26 our study has its own novelty and advantages. First, this study analyzed a larger sample size utilizing meta-analysis, and the expression level of miR-452-5p in LUAD was confirmed using a total of 1202 samples from TCGA, GEO, and our clinical samples. The heterogeneity generated from the detection methods of miR-452-5p was also analyzed by subgroup analysis. Second, we combined 14 online prediction tools to screen target gene sets of miR-452-5p. Using network, GO, and pathway analyses of miR-452-5p target genes, we performed a systematic determination of possible molecular mechanisms of miR-452-5p in the biological progresses of LUAD.
As a result of bioinformatic analysis of 249 miR-452-5p target genes, we found that miR-452-5p-related target genes were mainly functioned by binding with protein, chromatin, or other biological molecules, which are involved in the BP of transcriptional regulation. In addition, the miR-452-5p target gene set was significantly enriched in the cell-cycle pathway which is closely related to the prognosis of LUAD. 38 And five hub genes including SMAD4, SMAD2, CDKN1B, YWHAE, and YWHAB were significantly enriched in the cell cycle. These results identified possible functions and mechanisms of miR-452-5p and provided a direction for future experimental studies in LUAD. For example, previous studies reported that SMAD2 and SMAD4 could directly affect the signal transduction of transforming growth factor beta 1 (TGF-β1), which plays an important role in the development and metastasis of lung cancer;39,40 MiR-452-5p can promote hepatocellular carcinomas by targeting CDKN1B; 20 and YWHAE and YWHAB were reported to associate with the aggressive behavior and poor prognosis of endometrial stromal sarcomas and the migration and invasion of human hepatocellular carcinoma cells, respectively.41,42 But the similar roles and mechanisms of miR-452-5p and these five hub genes in LUAD are still unknown. Thus, it was presumed that miR-452-5p might play a critical role in LUAD by targeting these genes involved in cell cycle. Further in-depth experimental studies of the relationships between these genes and miR-452-5p should therefore help to elucidate the mechanism of carcinogenesis of LUAD.
Several limitations still exist in this study, First, heterogeneity may inevitably generate in the meta-analysis of 1202 samples, not only result from the detection method but also due to the difference in region, gender, age, stage, and the sample type of LUAD patients. Second, miRNAs regulate target genes to function during tumor development through different biological molecular pathways, to form a complex regulatory network of interactions involving biological molecules. Generalized exploration of the mechanisms of molecular biology is time-consuming. We therefore choose to predict the function of target genes using bioinformatic methods, including conducted GO and pathway analyses, as well as construct an interaction network model of biological molecules. However, in order to identify the mechanisms of miR-452-5p involved in LUAD, further verification is still needed to identify new targets for possible treatments of LUAD patients, including in vitro and in vivo experiments as well as clinical trials.
In summary, differences in miR-452-5p expression levels between LUAD and its adjacent non-cancerous tissues were detected by quantitative real-time polymerase chain reaction (RT-qPCR), followed by validation of the low expression of miR-452-5p in LUAD patients with the TCGA and GEO data via meta-analysis. An intuitive MF network model of miR-452-5p target gene sets was also constructed using bioinformatics to comprehensively reveal the functions and possible mechanisms of miR-452-5p in the LUAD carcinogenesis. With the rapid development of molecular bioinformatic databases and the release of a large number of public databases, a combination of bioinformatic and experimental research becomes important in cancer research. Such an approach may contribute to provide a new direction for the development of early diagnosis and targeted treatment of LUAD.
Footnotes
Acknowledgements
The funders had no role in the study design, the data collection and analysis, the decision to publish, or the preparation of the manuscript. X.-n.G. and J.L. contributed equally as co-first authors, and T.-q.G. and G.C. contributed equally as co-corresponding authors of this paper.
Compliance with ethical standards
The study protocol was approved by the Ethics Committee of the First Affiliated Hospital of Guangxi Medical University, with informed consent signed by all participants.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by a Fund of the Guangxi Provincial Health Bureau Scientific Research Project (Z2013201, Z2014055), a Fund of the National Natural Science Foundation of China (NSFC81360327, NSFC81560469), the Natural Science Foundation of Guangxi, China (2015GXNSFCA139009), the Scientific Research Project of the Guangxi Education Agency (KY2015 LX062), and the Scientific Research Project of the Basic Ability Promoting for Middle Age and Youth Teachers of Guangxi Universities (KY2016YB077).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
