Abstract
MicroRNAs are widely involved in cancer progression by inhibiting the expression levels of oncogenes or tumor suppressor genes, and dysregulation of microRNAs may contribute to tumorigenesis. Here, we found that overexpressed miR-208b can reduce the proliferation of human osteosarcoma cell lines U-2OS and Saos-2 by arresting cell cycle progression. The in vivo xenograft tumors induced by Saos-2 cells overexpressing miR-208b had smaller size and grew more slowly than those induced by the control cells. The mobility of U-2OS or Saos-2 cells was also downregulated by miR-208b. MiR-208b targeted a site in the 3′ untranslated region of receptor tyrosine kinase–like orphan receptor 2. Inhibition of receptor tyrosine kinase–like orphan receptor 2 suppresses osteosarcoma metastasis in vitro. Recovering the expression levels of receptor tyrosine kinase–like orphan receptor 2 in miR-208b-overexpressed U-2OS or Saos-2 cells attenuated the inhibitory effects of miR-208b. In addition, the expression levels of miR-208b are significantly reduced in human osteosarcoma tissue samples compared to normal tissue samples, and miR-208b levels correlated inversely with receptor tyrosine kinase–like orphan receptor 2 levels. On these bases, we identified that miR-208b targets receptor tyrosine kinase–like orphan receptor 2 gene by which miR-208b can regulate the development of osteosarcoma.
Keywords
Introduction
Osteosarcoma (OS) is one of the most frequent primary sarcoma of the skeleton in children, adolescents, and young adults, and it is a kind of destructive malignant tumors. 1 Surgery was the only method to treat OS before 1970s. The cure rate was low, and the mortality rate was about 80&x37;. In 1970s, chemotherapy came out, providing a new treatment option for patients with OS. Benefited from the new therapy, the livability increased from about 20% to 65%–70%. 2 To date, multi-agent chemotherapy is a common therapeutic option for the OS as it has a high tendency to metastasize.3,4 So more effective treatment methods with much fewer side effects still need to be improved in the future.
MicroRNAs (miRNAs) are highly conserved, very short non-coding nucleic acid chains comprising 18 to 24 nucleotides, which are naturally produced by the genome. MiRNAs cannot be translated into proteins or oligopeptides, and they generally suppress gene expression at the post-transcriptional level.
5
In general, the 3′ untranslated region (3′-UTR) of messenger RNAs (mRNAs) can provide target sites for miRNAs.
6
In the cytoplasm, RNA-induced silencing complex (RISC) containing mature miRNAs can bind to the messenger RNAs
7
and block the mRNA translating into protein or speed-up the degradation of the mRNA via binding to the target sites of mRNAs in various biological processes.
8
In recent years, some studies indicate that miRNAs may contribute to the inhibition of cancer. For example, miR-22 has a directed target: adenosine triphosphate (ATP) citrate lyase. The expression level of miR-22 and tumor growth and metastasis show a negative correlation.
9
However, some miRNAs contribute to the development of cancer. For example, miR-21 contributes to the development of human laryngeal carcinoma by binding to the target site of
Receptor tyrosine kinase–like orphan receptor 2 (ROR2) is a member of the receptor tyrosine kinase (RTK) family that regulate cell proliferation, polarity, differentiation, migration, metabolism, and survival.13,14 Lu et al. 15 investigated WNT-5a and ROR2 expression using immunohistochemistry in 42 OS and 12 osteochondroma specimens and reported that expression of both WNT-5a and ROR2 was correlated with Enneking surgical stage and tumor metastasis. ROR2 could enhance cell migration, and the WNT5B/ROR2 signaling pathway is a potential therapeutic target for OS. 16
In this study, we used molecular biology and cell biology methods to determine the role of miR-208b in the regulation of OS. The regulating effect of miR-208b on OS was identified by various experimental methods in vivo or in vitro. We explored the regulating effect of miR-208b on OS progression and confirmed a functional target—
Experimental procedures
Clinical tumor specimens
A total of 15 OS samples were obtained from Jilin Central Hospital. All the samples were obtained with the patients’ informed consent and approved by the Research Ethics Committee of Jilin Central Hospital.
Cell culture and transfection
Human OS cell lines Saos-2 and U-2OS were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cultures of the cell lines were maintained in a humidified, 37°C, 5% CO2 incubator. McCoy’s 5A Media (modified with tricine; Sigma, St. Louis, MO, USA) with 10% fetal bovine serum (FBS; Gibco, Carlsbad, CA, USA) was used for cell culture. To generate the protein overexpression, the coding sequences of ROR2 without 3′-UTR were cloned into pcDNA3.0 (+). Transfections were performed with the Lipofectamine 2000 Reagent (Invitrogen, Waltham, MA, USA) following the manufacturer’s protocols.
Cell proliferation assay
The cell counting kit-8 (CCK-8; Dojindo Laboratories, Kumamoto, Japan) was used for proliferation assay. A volume of 1 × 103 U-2OS or Saos-2 cells treated with miRNA-208b mimics or miRNA control were cultured on 96-well culture plates for 6, 12, 24, or 48 h. After adding 10 µL of the CCK-8 solution to each well, cells were incubated at 37°C for 2 h. A Multiskan GO (Thermo Fisher Scientific, Vantaa, Finland) was used for recording the optical density values at 450 nm.
Cell migration and invasion assays
Transwell chambers (Millipore, Billerica, MA, USA) were used for invasion assays. After transfection, 1 × 105 cells in 50 µL FBS-free Dulbecco’s Modified Eagle’s Medium (DMEM) were added to the upper chamber containing 50 µL Matrigel. A volume of 450 µL DMEM containing 10% FBS was placed into the lower chamber. The cells in 24 well-plates with transwell chambers were cultured in a humidified atmosphere for 24 h at 37°C with 5% CO2. When the tumor cells located on the bottom surface of the membrane, 100% methanol was used for fixing. After 30 min, the cells were stained by 0.5% crystal violet (Sigma-Aldrich, St Louis, MO, USA) for 20 min and then counted under a phase-contrast microscope (Olympus, Tokyo, Japan). Similar to invasion assays, migration assays were executed with 24-well plates without Matrigel.
Cell cycle analysis
Cells were cultured for 2 days after transfection. The cells were fixed with ice-cold 95% ethanol, and propidium iodide/RNase buffer (BD Biosciences, NJ, USA) was used for cell staining. The cells were analyzed by flow cytometry after incubating for 30 min at 4°C in dark.
Quantitative reverse transcription polymerase chain reaction
Total RNA was extracted from cultured cells or tissue samples using TRIzol reagent (Invitrogen). To get complementary DNA (cDNA), reverse transcription (2 µg of total RNA was used to synthesize cDNA) was performed using the miScript II RT Kit (Qiagen, Valencia, CA, Denmark). Then, quantitative reverse transcription polymerase chain reaction (qRT-PCR) of miRNA was carried out using a SYBR PrimeScript miRNA RT-PCR Kit (TaKaRa, Otsu, Japan). U6 was used for normalization. The expression levels of ROR2 mRNA were quantified using the SYBR Green PCR core reagents (Bio-Rad Laboratories, Hercules, CA, USA) at the following reaction conditions: 95°C for 2 min, 40 cycles of denaturation at 95°C for 25 s, primer annealing at 60°C for 25 s, and extension at 72°C for 20 s. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) served as an internal control, and the relative mRNA expression was calculated using 2−ΔCt method.
Western blot analysis
Cells were scraped from the wells, and total proteins were extracted using ice-cold radioimmunoprecipitation assay (RIPA) lysis buffer (Solarbio, Beijing, China) with protease inhibitor. Total proteins were separated on 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to nitrocellulose membrane (Bio-Rad Laboratories). The membrane was then blocked with 5% non-fat milk and incubated with the primary antibodies (Abcam, Cambridge, UK). The membranes were washed and treated with a horseradish peroxidase–conjugated secondary antibody subsequently (ICL Lab, Portland, Oregon, USA). After washing with Tris-buffered saline (TBS), the bands were visualized by chemiluminescence (Millipore). The strength of GAPDH bands was regarded as the standard.
Statistical analysis
The numerical results are presented as mean ± standard deviation. Statistical significance between groups was determined by one-way analysis of variance (ANOVA). Statistically significant differences of
Results
MiR-208b suppresses OS progression both in vitro and in vivo
Previous study showed that miR-208b expression significantly decreased in OS cells. 17 To explore the role of miR-208b in OS development, transfection with miR-208b mimics was executed and progress of tumor was tested both in vitro and in vivo. The efficiency of miR-208b was confirmed by qRT-PCR in the OS cells—U-2OS and Saos-2 (Figure 1(a)). The overexpression of miR-208b negatively regulated the proliferation of U-2OS and Saos-2 cells (Figure 1(b) and (c)). After miR-208b transfection, the colony number reduced from 56 ± 7.3 to 26 ± 4.9 in U-2OS and from 51.5 ± 9.1 to 27.8 ± 3.2 in Saos-2 (Figure 1(b)) cells. Cell viability weakened in both U-2OS and Saos-2 at different time points of treatment (Figure 1(c)). Following miR-208b mimics transfection, the proportion of cells in G1 phase increased significantly, indicating a G1 arrest (Figure 1(d)). To further compare the proliferation, Western blotting was performed subsequently. Our results showed that the expression of proliferation-associated genes cyclin E and cyclin D1 was downregulated in both the miR-208b-treated U-2OS and Saos-2 cells. However, the expression of CDK4 was not significantly altered (Figure 1(e)).
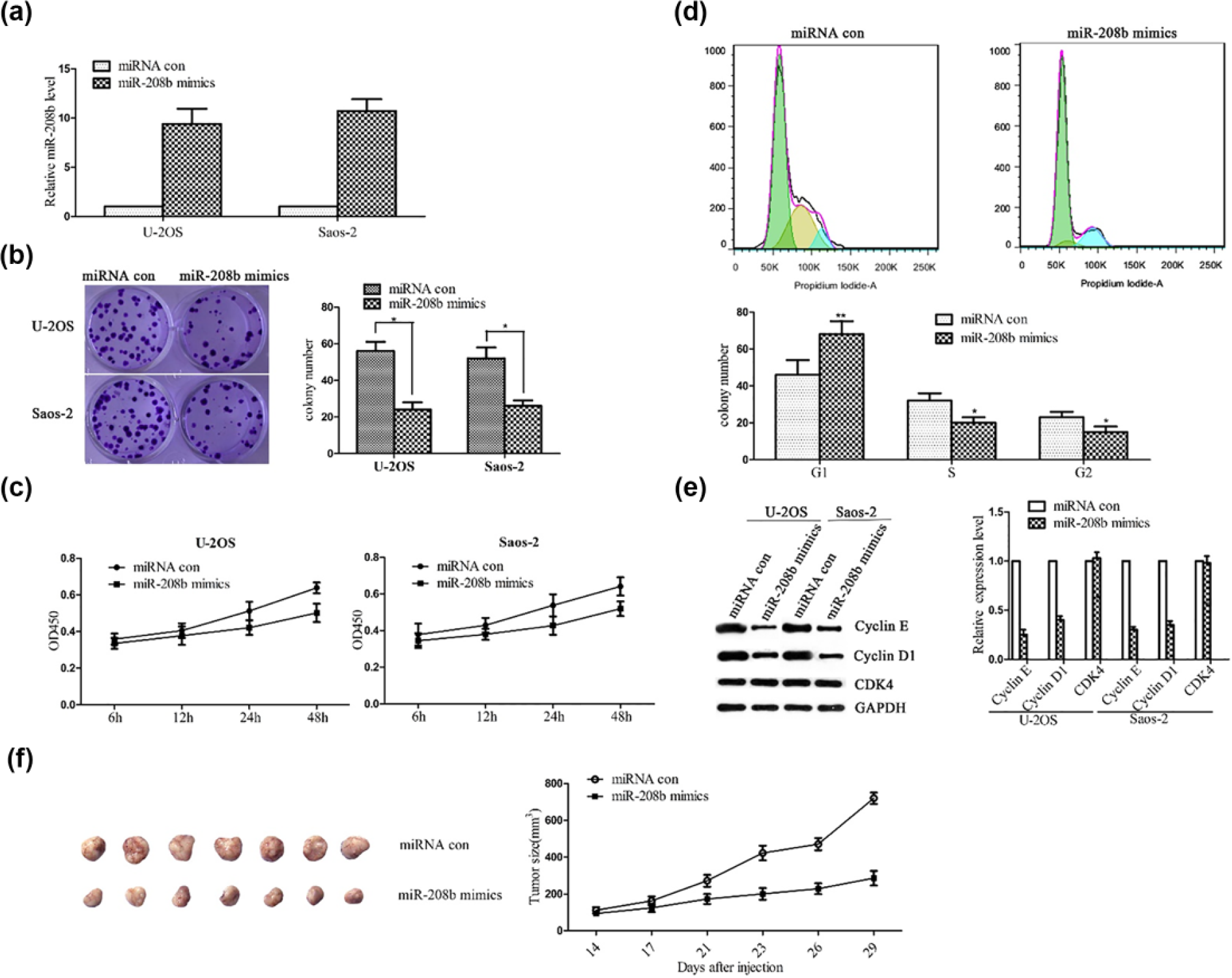
Inhibiting effects of miR-208b on osteosarcoma progression. (a) U-2OS and Saos-2 cells were treated with miR-208b mimics, and expression level of miR-208b in U-2OS and Saos-2 cells was detected by quantitative PCR. (b) The proliferation of U-2OS and Saos-2 cells was detected by colony formation assay. (c) Viabilities of U-2OS and Saos-2 cells were detected by CCK-8 cell proliferation assay at different time points. (d) Cell cycle distribution was analyzed by flow cytometry. (e) Cyclin E, Cyclin D1, and CDK4 protein levels were detected by Western blotting. (f) Hypodermic implantation of Saos-2 cells infected with control vector or miR-208b mimics was performed to build the osteosarcoma mouse model. The size of subcutaneous tumors in these two groups was calculated and compared (*
We also built subcutaneously implanted models of Saos-2 cells. After 14 days of implantation, the sizes of tumors were measured at various time points. Our results showed that tumors induced by the cells overexpressing miR-208b had smaller size and grew more slowly than those induced by the scramble cells (Figure 1(f)).These findings suggest that miR-208b has a negative role in tumor progression both in vivo and in vitro.
MiR-208b suppresses motor abilities in both U-2OS and Saos-2 cells
To study the effect of miR-208b on motor abilities of tumors, the migration and invasion assays were performed. According to the results, the motor abilities of U-2OS and Saos-2 cells were inhibited by overexpression of miR-208b (Figure 2(a) and (b)). The migration of U-2OS and Saos-2 cells was measured after being cultured for 24 h (Figure 2(a)). The results show that the cell migration was inhibited in both U-2OS and Saos-2 cells treated with miR-208b mimics. In addition, the cell number was counted in the invasion assays, and overexpression of miR-208b suppressed the cell invasion (Figure 2(b)). The expression of motor ability related genes was detected subsequently, and the expression of C-MYC and p-JNK was restrained in both U-2OS and Saos-2 cells treated with miR-208b mimics (Figure 2(c)). Our results show that overexpressed miR-208b can inhibit motor abilities of the OS cell lines.
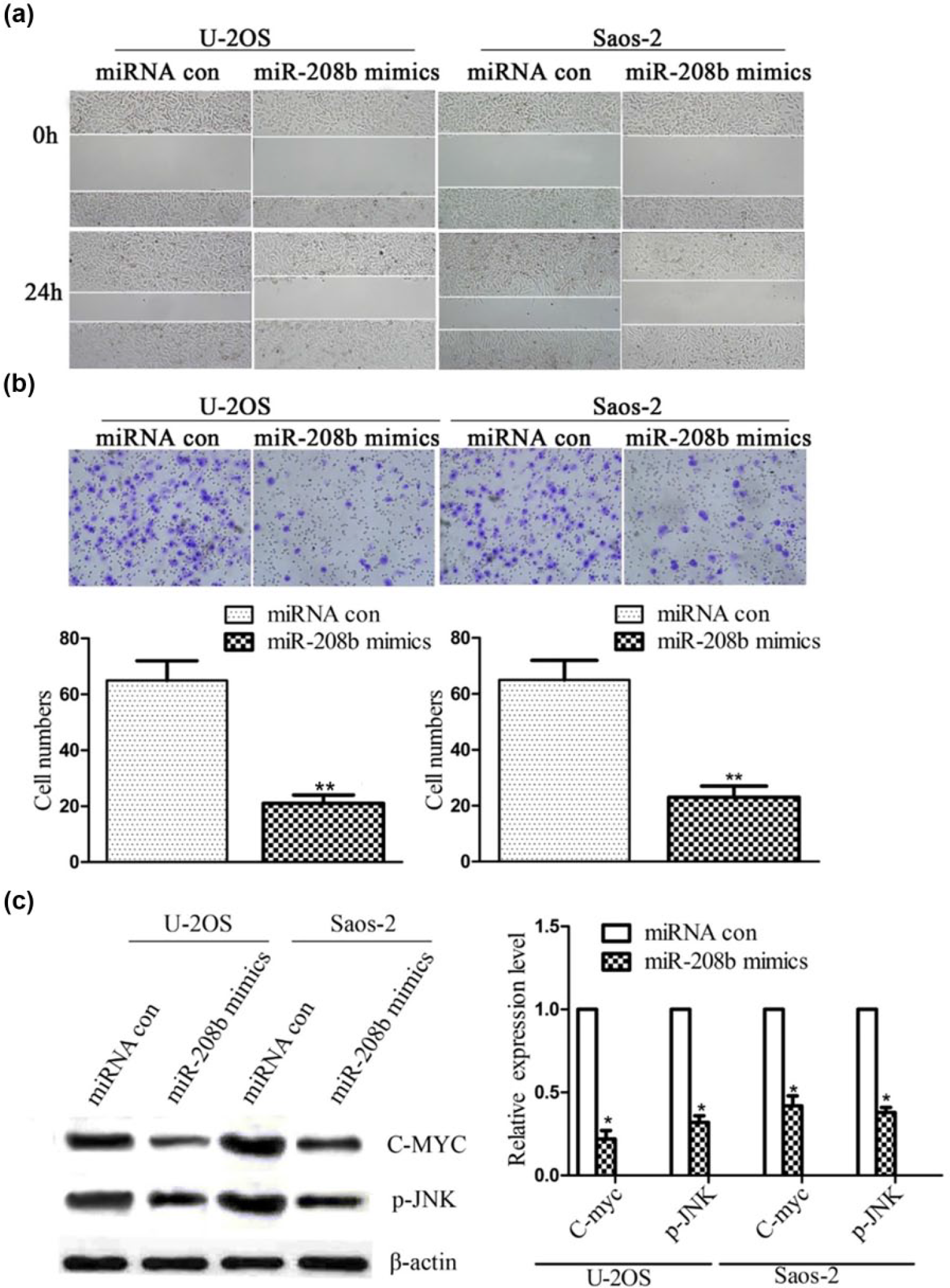
MiR-208b inhibits motor abilities of U-2OS and Saos-2 cells. (a) Migration and (b) invasion of U-2OS and Saos-2 cells were detected. The average number of invasive cells per field was counted. (c) C-MYC and p-JNK protein levels were detected by Western blotting (*
MiR-208b targets ROR2 and inhibits its expression
Bioinformatics tool TargetScan was used for forecasting the putative miR-208b targets, and miR-208b has a predictive target—

MiR-208b targets
Inhibition of ROR2 suppresses OS metastasis in vitro
The role of ROR2 on progression of OS cell lines was studied by small interfering RNA (siRNA). When transfected with

Silencing
MiR-208b mediates OS metastasis by targeting ROR2
MiR-208b binds to the predicted

The clinical OS has a higher expression level of ROR2 and a lower expression level of miR-208b
Immunohistochemical staining was performed in clinical OS tissues (Figure 6(a)). The number of ROR2-positive cells in OS tissues was more than that in normal tissues. To explore the role of miR-208b-ROR2 regulation in tumor, quantitative PCR (qPCR) was performed in both the OS tissues and benign tissues (including giant cell tumor I also known as GCT I and GCT II–III and synovial osteochondroma also known as SOC; Figure 6(b) and (c)). The results showed that in OS, the level of miR-208b was downregulated, but the level of
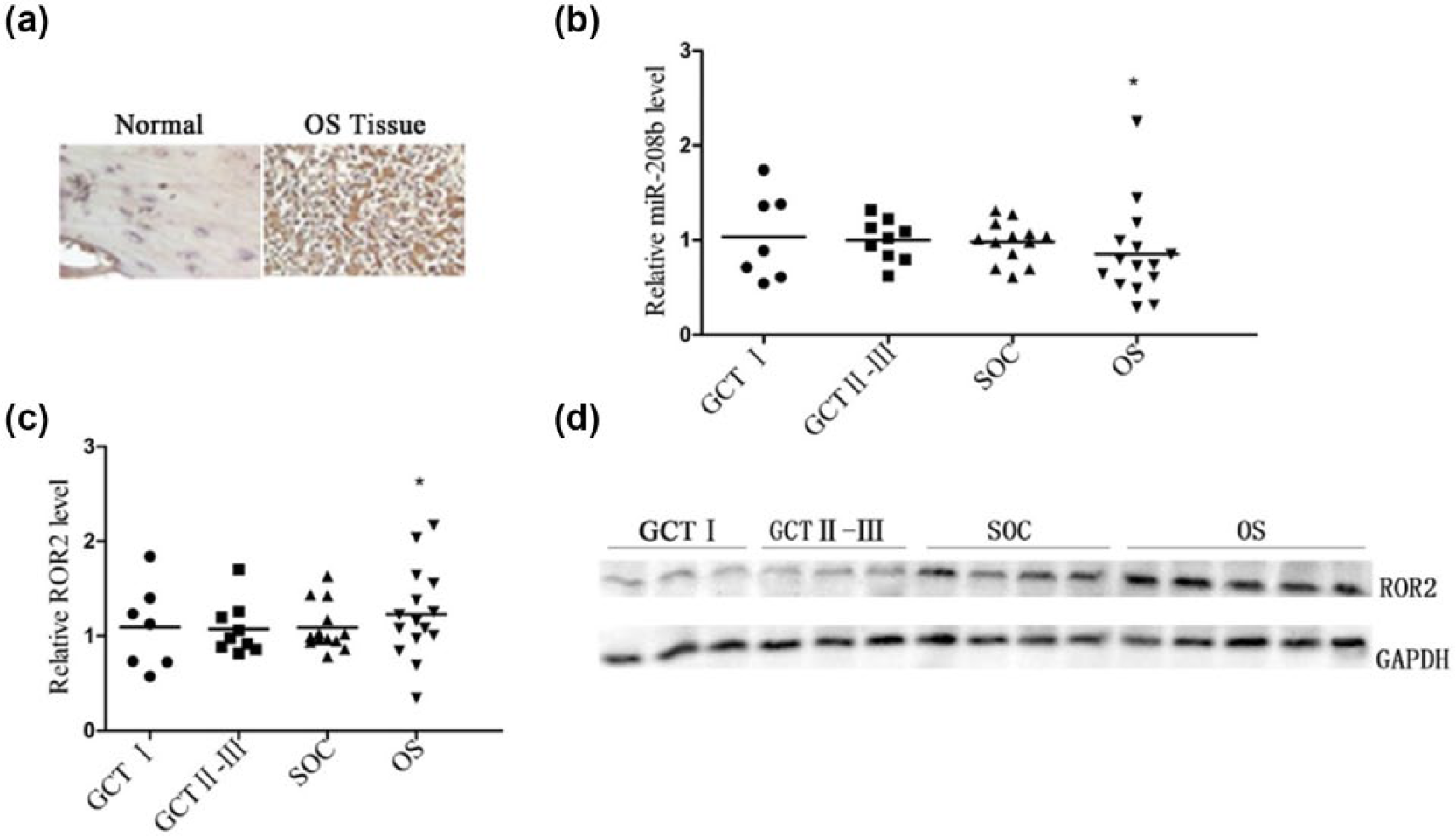
Low miR-208b level activates
Discussion
OS is a sarcoma of the bone which is a common primary malignant bone tumor in children, adolescents, and young adults.
1
Until recent years, due to the use of multi-agent chemotherapy and other treatment means, the overall survival of localized OS is above 70%.3,4 However, prognosis is unfavorable due to OS recurrence and metastasis. So treatment methods still need to be improved in the future. In our study, we used molecular biology and cell biology methods to determine the role of miR-208b in the regulation of OS. The regulating effect of miR-208b on OS was identified by various experimental methods in vivo or in vitro. Exogenous miR-208b can reduce the proliferation of U-2OS or Saos-2 cells in vitro, and the tumor cells overexpressing miR-208b showed a slow growth rate in vivo. Furthermore, we also proved that overexpression of miR-208b obviously decreased proliferation by arresting cell cycle in G1 phase. In addition, the mobility of U-2OS or Saos-2 cells was downregulated by the overexpression of miR-208b. ROR2 was downregulated after being transfected with miR-208b mimics in both U-2OS and Saos-2 cells. After
Both knockdown of
Our results showed that the overexpression of miR-208b can effectively inhibit the expression levels of endogenous
MiRNAs are required for various biological processes. Overexpression of miR-208b can improve myocardial functions via GATA4, and miR-208b plays a protective role in post-infarction myocardial fibrosis. 28 What is interesting is that miRNAs also take part in skin development and neural development. Wang et al. 29 identified that miR-205 can mediate the repression of phospho-Akt and restrict the proliferation of skin stem cells by targeting and modulating the phosphoinositide 3-kinase (PI3K) pathway. MiR-219 and miR-338 are required for oligodendrocyte differentiation and play their role via SOX6 and HES5.30,31 Although there are many studies about miRNAs, more research is needed to apply to clinical application.
Taken together, our studies support that
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Project in the Science and Technology Development Plan of Jilin Province (20120941).
