Abstract
Human pituitary adenoma is one of the most common intracranial tumors with an incidence as high as 16.7%. Recent evidence has hinted a relationship between growth factors of pituitary or hypothalamic origin and proliferation of human pituitary adenoma cells. This study explores the effects of small interfering RNA–mediated silencing of basic fibroblast growth factor gene on the proliferation, migration, and invasion of human pituitary adenoma cells. Human pituitary adenoma tissues were collected to obtain human pituitary adenoma cells. The basic fibroblast growth factor silencing interference plasmid was constructed, and the human pituitary adenoma cells were transfected and assigned as basic fibroblast growth factor–small interfering RNA, negative control–small interfering RNA, and blank groups. The quantitative real-time polymerase chain reaction and Western blotting were carried out to detect the expression of basic fibroblast growth factor, pituitary tumor transforming gene, vascular endothelial growth factor, cluster of differentiation 147, and matrix metalloproteinase 9. Cell Counting Kit-8 assay was conducted to observe cell proliferation at 0, 24, 48, and 72 h. Flow cytometry was used to determine cell cycle. Transwell and scratch test were applied to detect the invasion and migration of pituitary adenoma cells. Protein kinase C activity was detected. In comparison with the blank group, the basic fibroblast growth factor–small interfering RNA group showed reduced messenger RNA and protein expression of basic fibroblast growth factor, reduced cell viability at 24, 48, and 72 h, increased cells in G0/G1 stage, declined cells in S and G2/M stages, decreased number of cell migration, shortened migrating distance, reduced protein kinase C activity, and decreased expression of pituitary tumor transforming gene, vascular endothelial growth factor, cluster of differentiation 147, and matrix metalloproteinase 9. However, the negative control–small interfering RNA group had no evident differences in basic fibroblast growth factor expression, cell viability, cell cycle, number of cell migration, migrating distance, protein kinase C activity, and expression of pituitary tumor transforming gene, vascular endothelial growth factor, cluster of differentiation 147, and matrix metalloproteinase 9 compared with the blank group. The study provides evidence that small interfering RNA–mediated silencing of basic fibroblast growth factor gene inhibits the proliferation, migration, and invasion of human pituitary adenoma cells.
Keywords
Introduction
Human pituitary adenoma, caused by abnormal growth of the pituitary cells, is one of the most frequent intracranial tumors.1,2 Epidemiologic study has showed that incidence of human pituitary adenomas is as high as 16.7% within general population. 3 It is commonly believed that changes in pituitary cells, hypothalamic dysfunction, and inactivation of adenoma-related X-chromosome are the contributors resulting in human pituitary adenomas. 1 Human pituitary adenomas can bring about neuroendocrine disorders and severe dysfunctions. 4 Management of human pituitary adenomas involves radiotherapy, neurosurgery, and biological therapies. 5 With low morbidity and mortality, trans-sphenoidal surgery of surgical treatment has become the most popular choice over the last four decades. 6 However, the ratio of recurrence of pituitary adenoma patients over a follow-up period of 23.1 months (range: 4–62.5 months) is demonstrated to be 20% after trans-sphenoidal surgery. 7 It has been reported that growth factors of pituitary or hypothalamic origin may also promote proliferation of human pituitary adenoma cells. 8 Therefore, investigation on the growth factor might be a promising area for the treatment of human pituitary adenomas.
Basic fibroblast growth factor (bFGF), a member of the FGF family, is regarded as a potent mitogen. 9 bFGF is highly expressed in a variety of tumor cells and/or tissues and plays a significant role in stimulating the migration, differentiation, and proliferation of cells of neuroectodermal and mesenchymal origin. 10 Besides, bFGF is a protein secreted by the ureteric bud and is necessary for inducing cellular aggregation. 11 In addition, it has been recognized that bFGF promotes neurite extension and stimulates Schwann cell proliferation, which indicates that it promotes nerve regeneration. 12 It is illustrated that FGFs and the FGF signaling pathway not only benefit the process of wound healing but also have positive effects on tumor development and progression. 13 As it would contribute to the proliferation, angiogenesis, and survival of glioblastoma multiforme (GBM), bFGF is expected to improve the prognosis for GBM. 14 Another study indicates that there exists a correlation between bFGF and angiogenin in A375 human melanoma cells. 15 It is also suggested that in the process of epithelial morphogenesis, bFGF signaling contributes to the regulation of basement membrane formation. 16 Furthermore, bFGF has been proved to have correlation with pituitary tumor transforming gene (PTTG), and positive nuclear expression of PTTG is observed in human pituitary adenomas.17,18 With the participation in cells and tumor growth and the possible relationship with PTTG, bFGF is considered to play a key role in human pituitary adenomas. Therefore, this study proposed a hypothesis that bFGF silencing may have effects on the proliferation, migration, and invasion of human pituitary adenoma cells.
Materials and methods
Ethical statement
This study with all the protocols was approved by the Ethics Committee of our hospital and all patients signed the informed consents.
Cell culture and selection
Fresh human pituitary adenoma tissues were obtained from 63 patients in our hospital and verified by clinical manifestation and histopathological examination. The tumor tissues were first stored in sterile saline. After washing with DF12 (Gibco, Grand Island, NY, USA) for three times, the tumor tissues were stored in a bottle of penicillin (100 U/mL) and then cut into small pieces. The 5 mL of 2.5 g/L trypsin (Sigma-Aldrich, St. Louis MO, USA), 6.7 mkat/L of collagenase (Sigma-Aldrich), and 3.3 mkat/L of DNA (Sigma-Aldrich) were added successively to the tissue samples. The samples were digested in a 37°C water bath for 25 min. After adding the fetal bovine serum (FBS; Gibco), the samples were filtered by 200 meshes and centrifuged at 1000 r/min for 5 min. Next, the supernatant was abandoned. Centrifugation was repeated again after adding the DF12 culture solution and then the supernatant was abandoned. Finally, DF12 (containing 100 mL/L FBS) was added to adjust the cell concentration. Then, the cells were seeded onto the culture dish and incubated under the condition of 37°C, 50 mL/L CO2, and saturated humidity. Solution was changed after 24 h for the first time and the following change occurred 48 h later.
Construction of bFGF interference plasmid
The 799–819 region of bFGF coding gene (GenBank ID: NC_000004) was defined as a target sequence, and the small interfering RNA (siRNA) sequence was designed as 5′-ACTACAATACTTA-CCGGTCAA-3′, the loop construction sequence was CTCGAG, named bFGF-siRNA. The
Cell transfection and grouping
Human pituitary adenoma cells at logarithmic phase were collected. After digesting with pancreatic enzyme, they were seeded onto the six-well plate. Once cell growth achieved 60%–80% confluence, transfection was performed. The 4 µg bFGF-siRNA and NC-siRNA were diluted in 250 µL of Opti-MEMI medium (Gibco). After the samples were mixed completely, the transfection reagent Lipofectamine™ 2000 (Shanghai Genechem Co., Ltd) was diluted by 250 µL Dulbecco’s Modified Eagle’s Medium (DMEM) medium (Gibco). The transfection reagent was mixed respectively with plasmid diluent of bFGF-siRNA and NC-siRNA and placed at room temperature for 20 min. The mixture was then added onto six-well culture plates for transfection. After 4 h, the culture solution was replaced with DMEM (containing 10% FBS) nutrient solution. After 48 h, the cells were observed under an inverted microscope. Finally, the cells were assigned to blank group, NC-siRNA group, and bFGF-siRNA group according to corresponding transfection methods.
Quantitative real-time polymerase chain reaction
According to the kit instructions (QIAGEN, Hilden, Germany), the total RNA of cells in each group was extracted. Ultraviolet spectrophotometer was chosen to detect the optical density (OD) value (260/280) of extracted RNA, and the concentration of RNA was calculated. Samples were stored at −80°C for preparation. The reverse transcription of complementary DNA (cDNA) was operated in accordance with kit instructions (QIAGEN). Based on the gene sequence published by GenBank database, Primer 5.0 software was applied for primer designing (Table 1). The primers were synthesized by Shanghai Sangon Biological Engineering Technology & Services Co., Ltd (Shanghai, China). The quantitative real-time polymerase chain reaction (qRT-PCR) reaction system was of 20 µL: 10 µL of SYBR PremixExTaq, 0.8 µL of forward primer, 0.8 µL of reverse primer, 0.4 µL of ROX Reference Dye II, 2 µL of DNA template, and 6.0 µL of ddH2O. Reaction conditions were as follows: 95°C pre-degeneration for 30 s and 40 cycles of 95°C degeneration for 5 s, 60°C annealing for 30 s, and 72°C extension for 30 s. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal reference and the solubility curve was drawn to evaluate the reliability of PCR results. The cycle threshold (Ct) value (at a peak of an amplification curve) was collected and relative gene expression was calculated based on 2−ΔΔCt.
Primer sequences for qRT-PCR.

qRT-PCR: quantitative real-time polymerase chain reaction; bFGF: basic fibroblast growth factor; F: forward; R: reverse; PTTG: pituitary tumor transforming gene; VEGF: vascular endothelial growth factor; CD147: cluster of differentiation 147; MMP-9: matrix metalloproteinase 9; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Western blotting
According to the bicinchoninic acid (BCA) kit instructions (Wuhan Boster Biological Technology Co., Ltd, Wuhan, China), protein in each group was extracted and the corresponding concentration was detected. The extracted protein was added into the loading buffer and then boiled for 10 min at 95°C, after which each well was added with 30 µg loading buffer. Protein separation was operated by 10% polyacrylamide gel (Wuhan Boster Biological Technology Co., Ltd) with electrophoretic voltages changing from 80 to 120 V. The wet transmembrane worked at 100 mV for 45–70 min, after which protein was transferred onto the polyvinylidene fluoride (PVDF) membrane. Samples were blocked in 5% bovine serum albumin (BSA) at room temperature for 1 h. The primary antibodies of bFGF (1: 1000; Abcam, Cambridge, MA, USA), PTTG (1: 1000; Abcam), vascular endothelial growth factor (VEGF; 1: 1000; Abcam), cluster of differentiation 147 (CD147; 1: 1000), matrix metalloproteinase 9 (MMP-9; 1: 1000; Abcam), and GAPDH (1: 1000; Santa Cruz Biotechnology, Santa Cruz, CA, USA) were added and incubated at 4°C overnight. Samples were then washed with Tris-buffered saline with 0.1% Tween (TBST) three times (5 min/time) and the secondary antibody was added for 1 h and incubated at room temperature, after which the membrane was washed three times (5 min/time) and chemiluminescence reagent was added. GAPDH was used as the internal reference. The Bio-rad Gel Dol EZ imager (Bio-Rad, Hercules, CA, USA) was used for development. ImageJ software (National Institutes of Health, Bethesda, MD, USA) was conducted for gray value analysis of target band.
Cell Counting Kit-8 assay
The Cell Counting Kit-8 (CCK-8) assay (Dojindo Molecular Laboratories, Kumamoto, Japan) was employed for detecting the proliferation of human pituitary adenoma cells. After digesting with 0.25% pancreatic enzymes, the cells (105/mL) at logarithmic phase were seeded onto the 96-well plate with 100 µL in each well and the marginal wells were filled with phosphate-buffered saline (PBS). The samples were deposited in an incubator for cell adherence. When cell fusion reached 80%, the cells were handled according to the above grouping and six parallel wells were set up in each group. After 24, 48, and 72 h culture, CCK-8 solution (10 µL) was added into each well and the plates were stored at a 4°C in a refrigerator for incubation. After 1–4 h of incubation, Varioskan Flash (Thermo Fisher Scientific, Waltham, MA, USA) was used to measure the OD value at 450 nm. The experiment was repeated three times and the average OD value was obtained. Cell survival rate = OD value of the experimental group/OD value of the blank group.
Flow cytometry
Pituitary adenoma cells were fixed in ice-cold absolute ethyl alcohol at 4°C overnight. After washing with PBS, the cells were centrifuged at 2000 r/min and the supernatant was discarded. Each sample was added with 500 µL of 1× fluorescence-activated cell sorting (FACS) buffer (containing PBS, 0.1% BSA, and 0.01% NaN3) and 2.5 mL of Rnase A (10 mg/mL). After complete mixing, the samples were stored at room temperature for 15 min. The 25 µL of propidium iodide (PI; 1 mg/mL; Beyotime Biotechnology, Shanghai, China) was added into each sample and incubation in dark at room temperature for 15 min. The cell cycle was detected by flow cytometry (FACSC anto II; Becton, Dickinson and Company, Franklin Lakes, NJ, USA).
Scratch test
Cells were seeded onto the six-well plate at a density of 5 × 10 cells/well. When the cells merged into a monolayer, a 2-mm scratch wound was made across the diameter of each plate and the samples were cultured for 24 h. After treatment based on above grouping procedures, the cells were cultured for another 24 h. Phase-contrast microscopy was used to observe the scratches, and photographs were taken both immediately and 24 h after wounding. The images were analyzed by Image-Pro Plus 6.0. Cell migrating distance (mm) = scratch distance (0 h) − scratch distance (24 h).
Transwell assay
The packing box containing Transwell chambers (Millipore, Bedford, MA, USA) was rewarmed and put into the 24-well plate. The culture medium was placed in an incubator for pre-warming. A 0.5 mL of culture medium was added to the upper and lower chambers separately and hydrated in the incubator for 2 h, after which liquid in the upper and the lower chambers was abandoned. After digestion of cells, the cell suspension (5 × l04 cell/mL) was prepared. The hydrated chambers were transferred to the 24-well plate once the 0.5 mL of complete medium was added, and then, 0.5 mL of diluted cell suspension was added to the chambers. Liquid in the upper and lower chambers was abandoned after incubation for 24 h. Cells in the upper membrane were wiped gently with cotton, washed with PBS three times, fixed with pre-cooled paraformaldehyde for 30 min, stained with 1% crystalline violet for 10 min, and rinsed by running water. The dried crystalline violet was observed under an inverted microscope, and the average number of migration cells after 24 h was calculated.
Detection of protein kinase C activity
Protein kinase C (PKC) activity was measured according to the instructions of the kit purchased from Gibco. Human pituitary adenoma cells at logarithmic growth phase were selected and the nutrient solution was abandoned. Next, the cells were washed with ice-cold Tris buffer (containing 20 mmol/L Tris (pH 7.4), 0.5 mmol/L ethylenediaminetetraacetic acid (EDTA), and 0.5 mmol/L ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid (EGTA)), finally ended with 1 mL Tris buffer. Cells were centrifuged at 1000 r/min for 5 min and the supernatant was abandoned, and then stored at −70°C. A 0.5 mL of buffer A was added into each tube and then the cells were destroyed by ultrasound. Centrifugation was conducted at 1000 r/min for 5 min to separate the supernatant and precipitate, the supernatant was cell cytoplasm. Buffer A was added to the precipitate according to the instructions; after shaking thoroughly for 45 min and centrifuging for 20 min, the supernatant (cell membrane) was extracted. Isotope 32P was added after protein was measured by Ultraviolet spectrophotometer. Reaction system with the same amount of the protein was built in accordance with the instructions. Then, 1 mL scintillation liquid was added after pointing membrane, washing membrane and drying process were completed. Before calculating the variation ratio, PKC molar concentration per volume was obtained. Variation ratio (%) = (PKC molar concentration of the experiment group/PKC molar concentration of the control group) × 100%.
Statistical analysis
SPSS version 18.0 (SPSS Inc., Chicago, IL, USA) was used to analyze the obtained results. Measured data were displayed as mean ± standard deviation. The differences of data accorded with normal distribution between two groups were analyzed by
Results
Transfection efficiency of human pituitary adenoma cells
Transfection efficiency of pituitary adenoma cells was observed under the fluorescence microscope (Figure 1). According to the result, non-transfected cells were in the blank group and a large number of transfected cells were viewed in both the NC-siRNA group and bFGF-siRNA group. The transfection efficiency was higher than 80%, indicating that the cell transfection was successful.

Comparison of the transfection efficiency in the (b) blank, (c) NC-siRNA, and (d) bFGF-siRNA groups under the fluorescence microscope (200×). (a) Microscopic observation of human pituitary adenoma cells.
The messenger RNA and protein expression of bFGF in human pituitary adenoma cells in each group
After transfection, the expression of bFGF in each group was detected by qRT-PCR. Compared with the blank group, no significant difference of the messenger RNA (mRNA) expression of bFGF was noted in the NC-siRNA group (
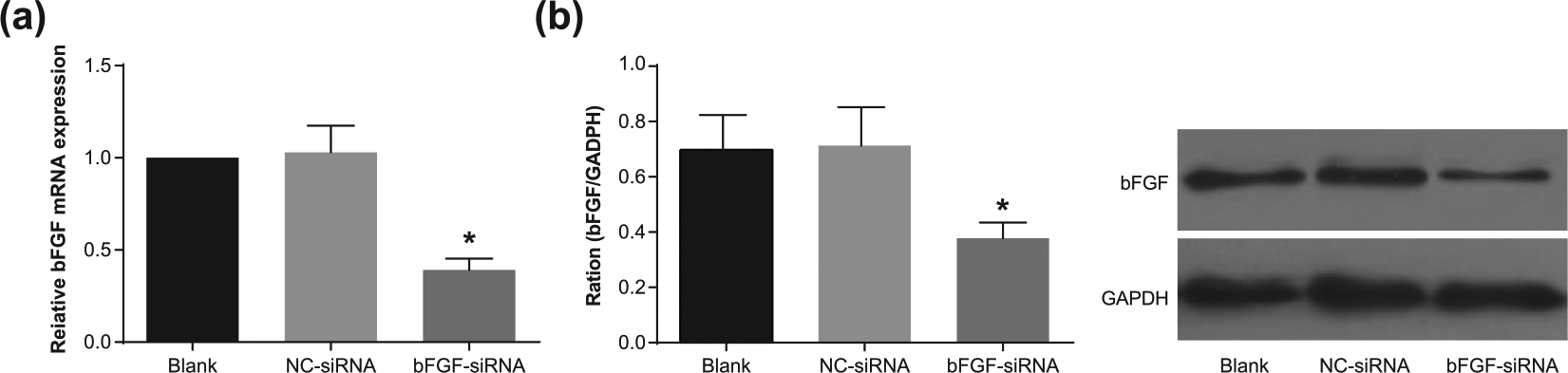
Expression of (a) bFGF mRNA and (b) protein in each group by qRT-PCR and Western blotting.
bFGF silencing inhibited the proliferation of human pituitary adenoma cells
Cell viability reduced at 24, 48, and 72 h in the bFGF-siRNA group compared with the blank group (all

Comparison of cell proliferation in the blank, NC-siRNA, and bFGF-siRNA groups detected by CCK-8.
bFGF silencing blocked cell cycle of human pituitary adenoma cells in G0/G1 stage
There were increased cells at G0/G1 stage and declined cells at S and G2/M stages in the bFGF-siRNA group compared with the blank group (all
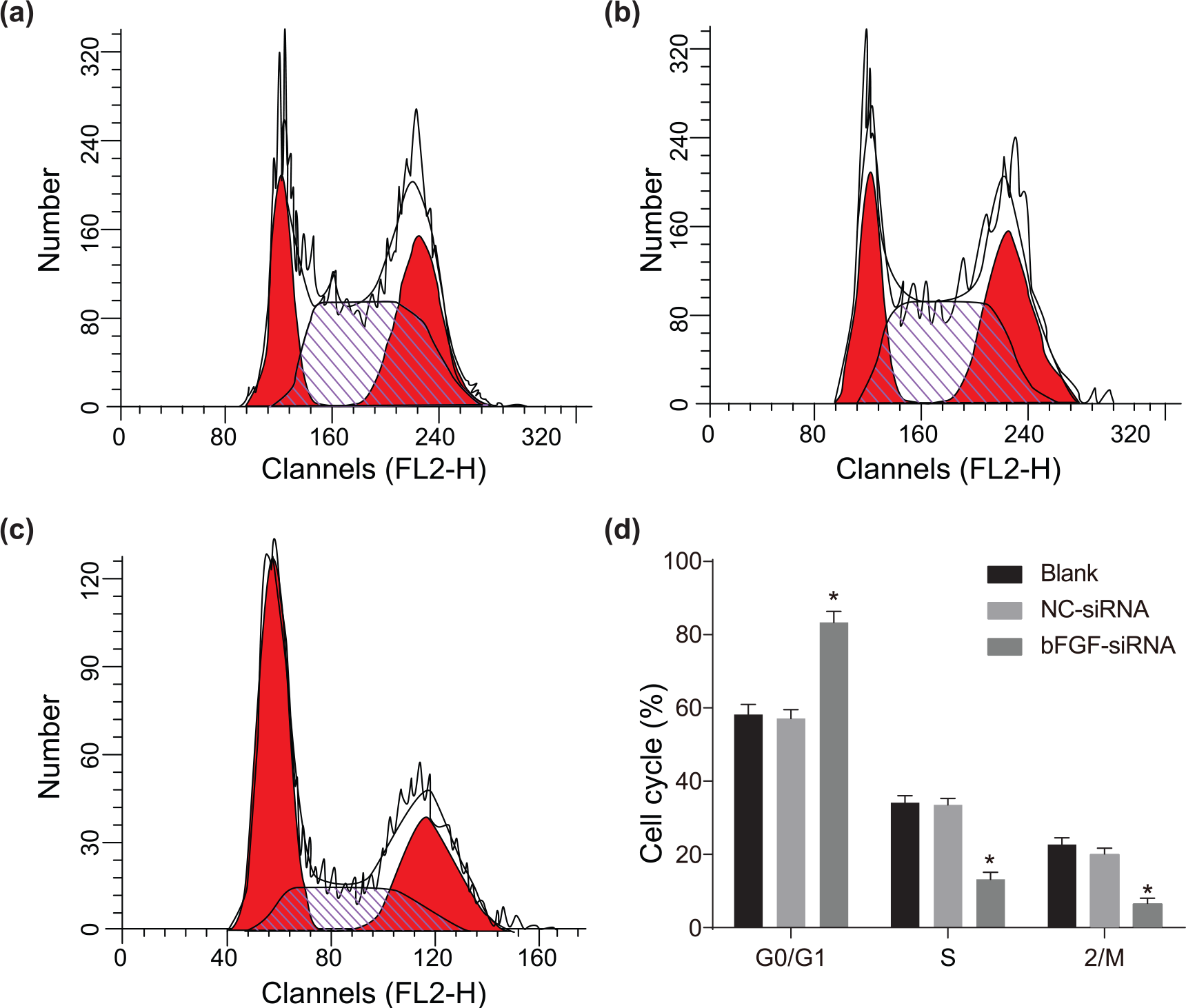
Comparison of cell cycle in the (a) blank, (b) NC-siRNA, (c) bFGF-siRNA groups detected by flow cytometry, and (d) comparison of cell cycle among groups.
bFGF silencing inhibited the invasion of human pituitary adenoma cells
There were a large number of invasive cells in the blank group and the NC-siRNA group, but the number of invasive cells in the bFGF-siRNA group was comparatively smaller (Figure 5(a)). Compared with the blank group, invasive cell number was significantly decreased in the bFGF-siRNA group (

Comparison of invasion of human pituitary adenoma cells in the blank, NC-siRNA, and bFGF-siRNA groups detected by Transwell assay. (a) Invasion map of three groups. (b) Invasive cell numbers of three groups.
bFGF silencing inhibited the migration of human pituitary adenoma cells
Dynamic observation at 0 and 24 h on scratches in each group showed that no significant difference was noted in terms of migrating distance and

Comparison of migration of cells in the blank, NC-siRNA, and bFGF-siRNA groups detected by scratch test. (a) Scratch chart of three groups. (b) Migration distance of three groups.
bFGF silencing inhibited the PKC activity of human pituitary adenoma cells
Significantly decreased PKC activity of membrane, cytoplasm, and total PKC activity was noted in the bFGF-siRNA group compared with the blank group (all
PKC activity of human pituitary adenoma cells in the blank, NC-siRNA, and bFGF-siRNA groups.

PKC: protein kinase C; NC: negative control; siRNA: small interfering RNA; bFGF: basic fibroblast growth factor.
Expression of PTTG, VEGF, CD147, and MMP-9 in human pituitary adenoma cells in each group
Compared with the blank group, no obvious difference was noted in terms of the protein expression of PTTG, VEGF, CD147, and MMP-9 in the NC-siRNA group (all
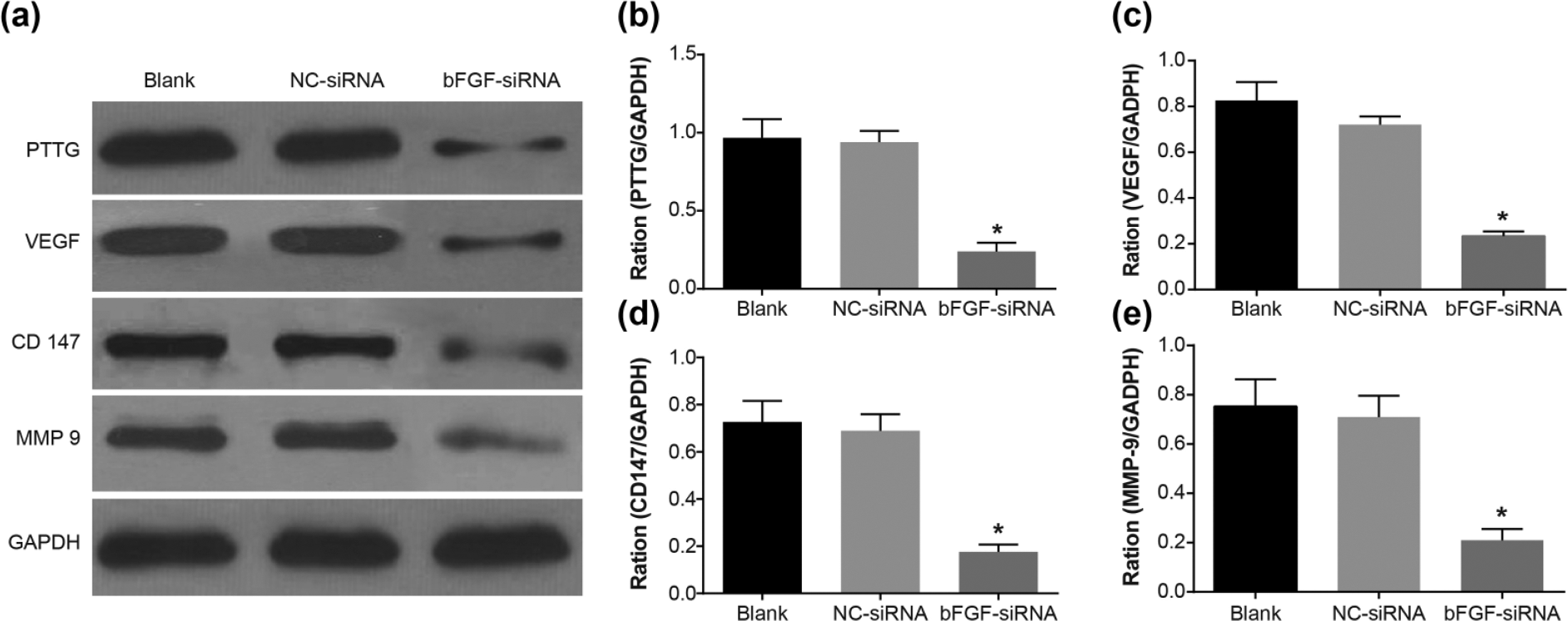
Comparison of the expression of PTTG, VEGF, CD147, and MMP-9 by Western blotting. (a) Band patterns of the expression of PTTG, VEGF, CD147, and MMP-9. (b) The cartogram of the PTTG protein expression. (c) The cartogram of the VEGF protein expression. (d) The cartogram of the CD147 protein expression. (e) The cartogram of the MMP-9 protein expression.
The mRNA expression of PTTG, VEGF, CD147, and MMP-9 detected by qRT-PCR.

mRNA: messenger RNA; PTTG: pituitary tumor transforming gene; VEGF: vascular endothelial growth factor; CD147: cluster of differentiation 147; MMP-9: matrix metalloproteinase 9; qRT-PCR: quantitative real-time polymerase chain reaction; NC: negative control; siRNA: small interfering RNA; bFGF: basic fibroblast growth factor.
Discussion
Human pituitary adenomas, widely recognized as the commonest type of pituitary disorder, may present symptoms related with endocrine dysfunction, including infertility, galactorrhea, and neurologic symptoms including headache or visual problems. 19 Although the prevalence of clinically-relevant human pituitary adenomas is about 1:1000–1300 of the population, importance should be attached to human pituitary adenomas with the increased morbidity and mortality.20,21 bFGF, SP1, and transcription factors have been reported to be closely related with human pituitary adenomas. 1 The target of this study is to investigate the effects of bFGF silencing on the proliferation, migration, and invasion of human pituitary adenoma cells, thereby providing a promising new target for human pituitary adenoma therapy. The results of a series of experiments showed that bFGF silencing could inhibit the proliferation, migration, and invasion of human pituitary adenoma cells.
Initially, compared with the blank group, the mRNA and protein expression of bFGF were decreased after bFGF silencing. bFGF, also called FGF-2, takes responsibility for cell growth, adhesion, migration, and differentiation and chemotaxis and matrix composition in different types of cells. 22 In addition, it is shown that decreased protein and mRNA expression of PTTG–1 is accompanied by reduced bFGF expression. 23
The most important finding was that bFGF silencing restrained the proliferation, migration, and invasion of human pituitary adenoma cells. The experiment observed that the cell viability at 24, 48, and 72 h in the bFGF-siRNA group was significantly reduced, which indicated that bFGF silencing could effectively suppress the occurrence of pituitary adenoma. A cell culture experiment demonstrated that cell number was higher in the bFGF-loaded nanofiber than in the bFGF-free nanofiber, which indicated that bFGF played a role in regulating cell proliferation. 24 Furthermore, bFGF silencing also influenced the cell cycle. The result showed that human pituitary adenoma cells were blocked in the G0/G1 stage and thus the cells in the S and G2/M stages were reduced. It is noted that the FGF-2/FGF receptor (FGFR) signaling axis can trigger antagonistic cell cycle regulatory pathways thus blocking cell proliferation and causing G2/M arrest. 25 Decreased healing rate, reduced invasive cell number, and shortened migrating distance were also noted in the bFGF-siRNA group. Blair et al. 26 disclosed that bFGF could promote cellular invasion by activating phosphoinositide 3-kinase (PI3K)/Akt and extracellular signal–regulated kinase (ERK) pathway. bFGF has been reported to stimulate cell motility of fibroblasts and invasion of various kinds of tumors, and the motility and invasion of Ewing’s sarcoma family of tumors (ESFT) were affected through the function of PI3K-Rac1 signaling pathway downstream of FGFR. 27 Besides, another finding introduced that through the urokinase-type plasminogen activator (uPA) and Ets-1, bFGF enhanced the invasion ability of ovarian cancer cells. 28 Furthermore, we found that the bFGF-siRNA group also showed decreased PKC activity. PKC has acted as a receptor for the tumor-promoting phorbol esters since it was discovered. 29 It is illustrated that PKC plays a part in regulating the growth and differentiation of various tissues, including anterior pituitary gland cells, and it was inferred that PKC might play a key part in signal transduction in the human pituitary adenomas. 30 One study has supported the previous evidence that reduced PKC enzyme activity was noted in primary human colon tumors. 31
Findings also indicated that bFGF silencing was accompanied by decreased expression of PTTG, VEGF, CD147, and MMP-9. PTTG has been recognized to be tumorigenic and has important correlation with bFGF in human pituitary adenomas.32,33 It is also exposed that PTTG can be upregulated by bFGF. 34 VEGF, a vasculogenic factor, functions well in stimulating endothelial cell mitogenesis and promoting cell migration. 35 Furthermore, FGF signaling is likely to build a favorable environment for vasculature growth. 36 CD147 was reported to be related with many physiological functions, and also the development of cancer. 37 CD147 has been proved to play a critical role in the production of VEGF. 38 MMP-9 is a key component in cell invasion and migration. 39 It is also admitted that induction of MMP-9 is especially important for the invasion of various types of tumor cells. 40 Therefore, the expression of PTTG, VEGF, CD147, and MMP-9 is closely related with cell migration and invasion and downregulation of these parameters further demonstrates restrained migration, invasion, and proliferation abilities of human pituitary adenoma cells mediated by bFGF silencing.
In conclusion, we found that bFGF silencing suppressed the proliferation, migration, and invasion of human pituitary adenoma cells in vitro, which provided a new target for the treatment of human pituitary adenomas. This is a promising area for human pituitary adenoma treatment, and further studies will be conducted in the future based on larger sample and detailed methods.
Footnotes
Acknowledgements
The authors thank the reviewers for providing helpful comments in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study with all the protocols was approved by the Ethics Committee of The First Affiliated Hospital of Xinjiang Medical University, and all patients signed the informed consents.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Natural Science Foundation of Xinjiang Uygur Autonomous Region (No. 2013211A078).
