Abstract
The small-molecule inhibitors of p53–murine double minute 2 interaction, such as Nutlin-3, are effective against cancers bearing wild-type p53. However, murine double minute 2 inhibitors often are unable to completely eliminate solid tumor cells. To address this issue, we investigated the anticancer effects of Nutlin-3 in combination with Oridonin in osteosarcoma cells. We found that Oridonin at sub-toxic concentrations synergistically enhanced Nutlin-3-mediated cell viability inhibition in wild-type p53 U2OS and SJSA-1, but not in p53-mutant MNNG/HOS and in null-p53 Saos-2 osteosarcoma cell lines. Importantly, in the presence of Oridonin, Nutlin-3 could completely abolish cell viability in the wild-type p53 osteosarcoma cell lines. Western blotting analysis showed that Oridonin treatment rapidly and distinctly increased the levels of all three forms of Bim and also markedly reduced the levels of Bcl-2 and Bcl-xl in osteosarcoma cells. Western blotting analysis further showed that Oridonin considerably enhanced Nutlin-3-triggered activation of caspases-9 and -3 and poly(ADP-ribose) polymerase cleavage. Flow cytometry assay showed that Oridonin significantly enhanced Nutlin-3-mediated apoptosis in wild-type p53 osteosarcoma cells. Overall, our results suggest that the combined treatment of Nutlin-3 plus Oridonin may offer a novel therapeutic strategy for osteosarcoma.
Introduction
Osteosarcoma (OS) is a common primary bone tumor that predominantly occurs in adolescents and young adults. This malignancy is thought to arise from mesenchymal cells and appears in diverse pathological and clinical forms with diverse degrees of aggressiveness. Multi-agent chemotherapy and aggressive surgical resection have significantly increased the overall survival rate. However, patients with recurrent disease or having metastatic lesions still lack effective therapy. New therapies are therefore urgently needed for this subset of OS patients.1,2
Murine double minute 2 (Mdm2) is an oncogene overexpressed in numerous types of cancers, notably in human sarcomas due to gene amplification or aberrant post-translational protein modifications.3,4 By inhibiting tumor suppressor function of wild-type (wt) p53, Mdm2 plays important roles in cancer development and progression. 4 Hence, restoration of wt-p53 function by Mdm2 inhibitors, such as Nutlin-3, has emerged as a novel cancer therapeutic strategy. To date, numerous Mdm2 inhibitors have been synthesized and tested, and several of them have shown encouraging results in human cancer clinical trials. These evidences suggest that Mdm2 inhibitors hold great promise for treating cancer. 5 Nevertheless, previous studies also indicated that Mdm2 inhibitors induced modest cell killing effect in most tumor cells and had limited single-agent effectiveness in human tumors.6,7 These facts have stimulated great efforts to explore combination strategy for improving the efficacy of Mdm2 inhibitors in cancer treatment.7–9
Oridonin is a natural diterpenoid compound purified from the Chinese medicinal herb Rabdosia rubescens, which has been used for the treatment of various human cancers, especially esophageal cancer in our Henan province by Traditional Chinese Medicine physicians for a very long time. 10 Previous studies revealed that Oridonin has a variety of biological effects, such as anti-inflammatory, anti-viral, and anti-bacterial activities. 11 Extensive attention has been paid to the anticancer activity of Oridonin. It was demonstrated that Oridonin as a single agent had a profound anti-proliferative effect on cancer cells.12,13 Moreover, it was reported that Oridonin was able to reverse resistance of cancer cells to multiple chemotherapeutic agents.14,15 In this study, we investigated whether Oridonin could promote Nutlin-3-mediated anticancer activity in OS cells.
Materials and methods
Cell culture
Human OS cell lines SJSA-1, U2OS, MNNG/HOS, and Saos-2 were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). The cells were cultured in RPMI 1640 supplemented with 10% fetal calf serum (FCS; Sigma-Aldrich, Shanghai, China) at 37°C under 5% CO2 in a humidified atmosphere. Exponentially growing cells were treated by the drugs for the indicated time periods.
Chemicals and antibodies
Nutlin-3 was obtained from Shanghai Selleck Chemicals Co (Shanghai, China). Z-VAD-FMK was obtained from R&D Systems China Co. (Shanghai, China) and dissolved in dimethyl sulfoxide (DMSO) at 10 mM concentration and stored in −20°C. Oridonin was purchased from Shanxi Huike Plants Exploitation (Xian, China) and was dissolved in DMSO with a stock concentration of 1 mM and stored at −20°C.
Cell Counting Kit-8 cell viability assay
Cell viability was evaluated using Cell Counting Kit-8 (CCK-8; Sigma-Aldrich, Shanghai, China). All assays were performed in triplicate, and all experiments were performed at least three times. After drug treatment, cells were incubated at 37°C with CCK reagent for 60–120 min, and the optical density (OD) value was measured at 450 nm wavelength according to the manufacturer’s instructions. The combination index (CI) was calculated using an equation based on the Chou–Talalay method for drug combination. 16
Apoptosis assay
Apoptosis was performed by flow cytometry assay using Annexin V–fluorescein isothiocyanate (FITC) and propidium iodide (PI; catalog no. 11858777001). OS cells were treated as indicated; the cells were collected and subjected to Annexin V-FITC/PI double staining for flow cytometry. The lower left quadrant (Annexin V-FITC−/PI−) was considered as live cells, the lower right quadrant (Annexin V-FITC+/PI−) was considered as early-stage apoptotic cells, the upper right quadrant (Annexin V-FITC+/PI+) was considered late-stage apoptotic cells, and the upper left quadrant (Annexin V-FITC−/PI+) was considered as necrotic cells.
Western blotting analysis
Western blotting analysis was performed as described previously. 12 Antibodies such as anti–poly(ADP-ribose) polymerase (PARP; 9542), anti-caspase-3 rabbit polyclonal antibody (9662), anti-Bcl-2 rabbit polyclonal antibody (2762), anti-Bad rabbit polyclonal antibody (9292), anti-Bim (2819), and anti-Tubulin (2144) were purchased from Cell Signaling Technology (Shanghai, China); anti-p53 (OPG03) from EMD Millipore (Shanghai, China); anti-Mdm2 (IF2, 33-7100) from Thermo Fisher Scientific (Shanghai, China); and horseradish peroxidase (HRP)-conjugated secondary anti-mouse, anti-goat, and anti-rabbit antibodies were purchased from Santa Cruz Biotechnology Shanghai Co. Ltd. (Shanghai, China).
Quantitative real-time reverse transcription polymerase chain reaction
Total RNA from the cultured cells was obtained by using the RNeasy Mini Kit (Qiagen, Inc., Shanghai, China). Approximately 1–2 µg of total RNA was used for reverse transcription to complementary DNA (cDNA) using the SuperScript II Reverse Transcriptase system (Invitrogen, Shanghai, China). Quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) was performed with iQ SYBR-Green Supermix (Bio-Rad, Pudong Shanghai, China) on a Bio-Rad iCycler Real-Time PCR machine. The following primers were used: Puma (BCL2 binding component 3): 5′-ACCTCAACGCACAGTACGAG-3′(forward) and 5′-CCCATGATGAGATTGTACAGGA-3′ (reverse). All values were normalized to the housekeeping gene glyceraldehyde-3-phosphate dehydrogenase (GAPDH; forward primers: 5′-GTCAGCCGCATCTTCTTT-3′; reverse primers: 5′-CGCCCAATACGACCAAAT-3′). Each qRT-PCR figure is the average of three independent experiments.
Statistical analysis
Statistical analyses were performed by one-way analysis of variance (ANOVA) using SPSS (version 13.0; SPSS Inc., Chicago, IL, USA); p < 0.05 was considered statistically significant and p < 0.01 was considered very statistically significant.
Results
Oridonin increases the levels of Bim and decreases the levels of Bcl-2 and Bcl-xl in OS cells
We and others have reported that Oridonin could modulate the levels of Bcl-2 family proteins in melanoma and leukemia cells.12,13 We initiated our study by investigating whether Oridonin had similar effects on the levels of Bcl-2 family proteins in OS cells. We performed CCK-8 cell viability assay to determine a sub-toxic concentration range of Oridonin in wt-p53 U2OS and SJSA-1 and p53-mutated MNNG/HOS and p53-null OS Saos-2 cell lines. We found that Oridonin at 0.5–6 µM dose-dependently and partially inhibited cell viability in all four OS cell lines (Figure 1). We next treated the OS cell lines with three sub-toxic concentrations of Oridonin (1, 2, and 4 µM) for 48 h and then examined the levels of several Bcl-2 family proteins and the level of p53 in the treated cells by western blot analysis. The results showed that Oridonin treatment had no effect on p53 level and had little effect on Bad level in the OS cell lines. In contrast, the treatments distinctly and dose-dependently increased the levels of all three forms of the pro-apoptotic protein Bim, including BimEL (extra-long form), BimL (long form), and BimS (short form) and also markedly reduced the levels of Bcl-2 and Bcl-xl in the OS cells (Figure 2(a), Supplementary Figure 1). A time course analysis showed that Oridonin rapidly increased the levels of Bim (3–8 h). In contrast, the inhibitory effect of Oridonin on the levels of Bcl-2 and Bcl-xl occurred relatively slowly, and obvious reduction in Bcl-2 and Bcl-xl levels was detected only at 24 h after treatment (Figure 2(b)). With qRT-PCR assay, we analyzed the impact of Oridonin on the expression of Puma and found that Oridonin did not significantly alter the messenger RNA (mRNA) expression on this pro-apoptotic Bcl-2 family member (Figure 2(c)).

Oridonin inhibits cell viability of OS cells in a dose-dependent manner. (a) U2OS, (b) SJSA-1 (c) Saos-2, and (d) MNNG/HOS cell lines were treated by serial dilutions of Oridonin for 24 or 72 h. Cell viability was determined by CCK-8 assays. Representative inhibitory curves from three independent experiments are shown for each cell line.

Oridonin modulates the levels of multiple Bcl-2 family proteins in OS cells. (a) U2OS and SJSA-1 cell lines were treated by Oridonin (ORI) at 1, 2, and 4 µM for 24 h. The protein levels of p53, Bad, BimEL, BimL, BimS, Bcl-2, and Bcl-xl were examined by western blotting analysis. Tubulin was used as a loading control. (b) U2OS and SJSA-1 cell lines were treated by 2 µM ORI for 3, 8, and 24 h. The protein levels of BimEL, BimL, BimS, Bcl-2, and Bcl-xl were examined by western blotting analysis. Tubulin was used as a loading control. (c) U2OS and SJSA-1 cell lines were treated by Oridonin (ORI) at 1, 2, and 4 µM for 24 h. The mRNA level of Puma was examined by qRT-PCR assay.
Oridonin synergizes with Nutlin-3 to inhibit cell viability in wt-p53 OS cells
To investigate the potential interaction between Nutlin-3 and Oridonin in OS cell lines, we treated the cells with serial dilutions of Nutlin-3 (0.1–100 µM) alone or combined with 1, 2, or 4 µM Oridonin for 72 h and then examined cell viability with CCK-8 assay. We observed that Nutlin-3 alone could potently inhibit cell viability in U2OS and SJSA-1 cell lines, consistent with previous findings that these two wt-p53 cell lines were sensitive to Mdm2 inhibitors.5–7 Nevertheless, about 10% OS cells still survived after treated by Nutlin-3 alone even up to a high concentration of 30–100 µM for 3 days, suggesting a relatively modest cytotoxic effect in these OS cells. The cell viability assays further showed that in combination with Oridonin, Nutlin-3 much more effectively inhibited cell viability in both wt-p53 OS cell lines (Figure 3(a)). CI analyses suggest that the combination have a synergistic effect in both cell lines (Figure 3(b)). Importantly, in the presence of Oridonin, cell viability in both OS cell lines could be completely abolished by Nutlin-3 at much lower concentrations (3–10 µM; Figure 3(a)). These results indicate that the combination have a strong cytotoxic effect in OS cells.

Oridonin synergizes with Nutlin-3 in inhibition of cell viability of OS cells. (a) U2OS, SJSA-1, Saos-2, and MNNG/HOS cell lines were treated by serial dilutions of Nutlin-3 (0.1–100 µM) alone or in combination with 1, 2, or 4 µM Oridonin for 72 h. Cell viability was determined by CCK-8 assay. Representative inhibitory curves from three independent experiments are shown for each cell line. (b) CI was calculated based on IC50 values and listed in the table. The data were averages of three independent experiments.
In mutated-p53 MNNG/HOS and null-p53 Saos-2 cell lines, it appeared that Oridonin also enhanced Nutlin-3-mediated cell viability inhibition, but CI analysis indicated that the two agents had no synergistic activity (Figure 3(a) and (b)). Altogether, these results suggest that Oridonin synergizes with Nutlin-3 to inhibit cell viability in OS cells in a p53-dependent manner.
Oridonin enhances Nutlin-3-mediated apoptosis in OS cells
We next investigated whether apoptosis played a role in the synergy. To that end, we treated two wt-p53 OS cell lines with 3 µM Nutlin-3 alone, 2 µM Oridonin alone, or both. After treatment for 24 h, we noted that the combination, but not the single agents, triggered typical apoptosis morphological alterations, such as cell shrinking and floating in OS cells (data not shown). We then stained the treated cells with Annexin-V-FITC and PI and performed flow cytometry analysis to quantify the percentage of apoptotic cells. We found that Oridonin alone induced apoptosis only in <5% and Nutlin-3 alone in 6%. In contrast, the combination induced apoptosis in about 38% of U2OS cells, showing a significant improvement in apoptotic activity (Figure 4(a)). In SJSA-1 cell line, the pro-apoptotic activity of two-drug combination also was much stronger than the single agent (Figure 4(b)). These results suggest that apoptosis played a significant role in the synergy.
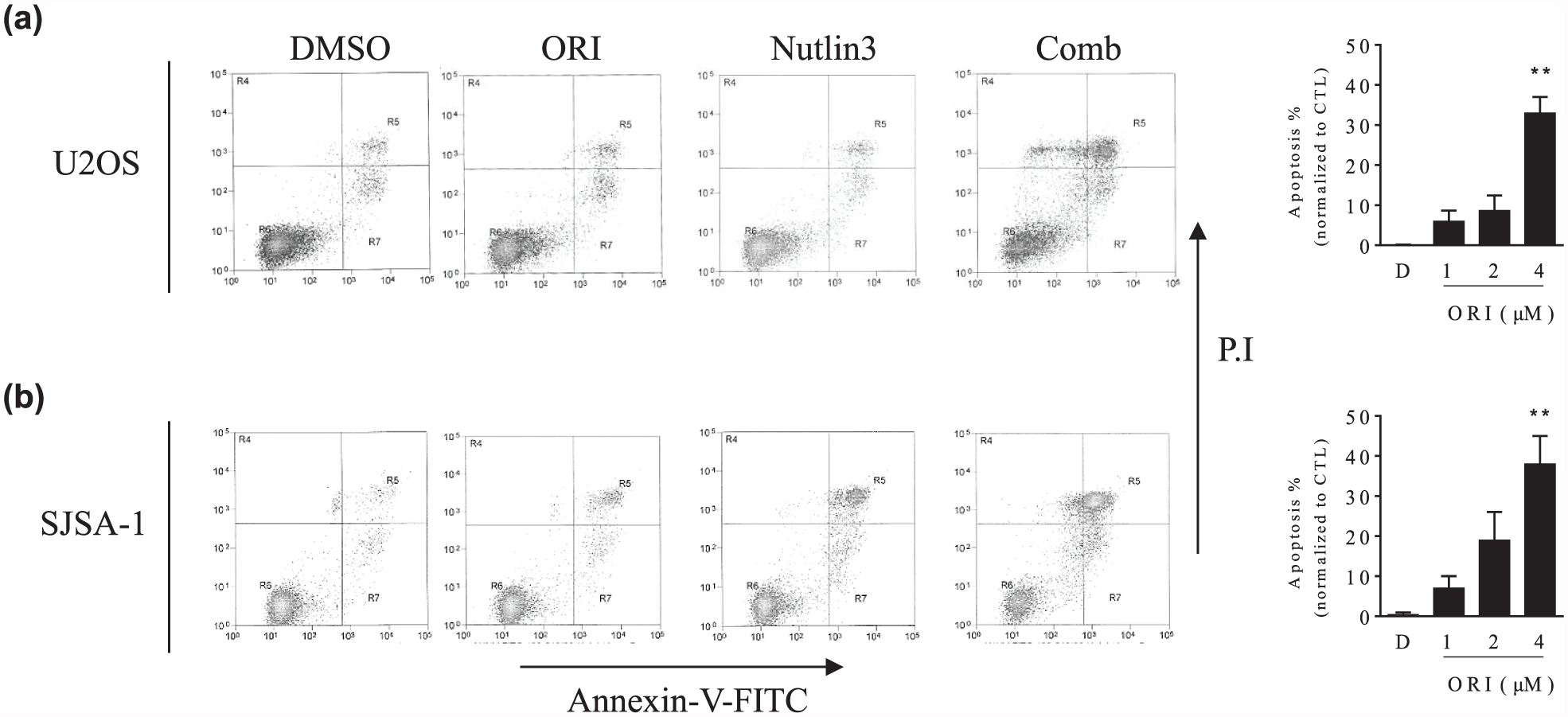
Oridonin enhances Nutlin-3-mediated apoptosis in OS cells. (a) U2OS and (b) SJSA-1 cell lines were treated by 3 µM Nutlin-3 alone, 2 µM Oridonin (ORI) alone, or their combination (Comb) for 24 h. DMSO was used as a control treatment. Treated cells stained with Annexin-v/PI were examined by flow cytometry. (Left panels) Representative plots of apoptosis from three independent experiments are shown for U2OS and SJSA-1 cell lines, respectively. (Right panels) The average percentages of U2OS and SJSA-1 cell lines in apoptosis are shown. The data were presented as mean ± standard deviation of three independent experiments (**p < 0.01).
Oridonin has no impact on levels of Mdm2 and p53 in OS cells
The apoptotic activity by Mdm2 inhibitors mainly relies on p53 activation and the upregulation of a classic pro-apoptotic p53-target genes Puma (Supplementary Figure 2(a) and (b)).4,5 We next investigated whether Oridonin had impact on p53 activation. Western blotting and qRT-PCR assays showed that Oridonin had no influence on the levels of Mdm2, p53, and Puma expression and also did not affect the ability of Nutlin-3 in inducing Mdm2 and p53 accumulation and Puma upregulation in OS cell lines (Figure 5(a)–(d)). These results suggest that the mechanism underlying synergistic antitumor effect of Nutlin-3 and Oridonin is not through increased p53 activation.

Oridonin has no effects on protein levels of Mdm2, p53, and the mRNA level of Puma in OS cells. (a) U2OS and (b) SJSA-1 cell lines were treated by 3 µM Nutlin-3 (N) alone, 2 µM Oridonin (O) alone, or their combination (C) for 24 h. DMSO (D) was used as a control treatment. Treated cells were lyzed and the protein level of Mdm2 and p53 was analyzed by western blotting analysis. Tubulin was used as a loading control. (c and d) The relative level of Puma mRNA was examined by qRT-PCR assay. The data were presented as mean ± standard deviation of three independent experiments.
Oridonin enhances Nutlin-3-triggered activation of caspases in OS cells
Activation of caspases and cleavage of PARP are biochemical hallmarks of apoptosis and were used as biomarkers of apoptosis induction.17,18 We then treated the cells with either agent or both for 24 h and examined whether caspase-3 was activated and PARP was cleaved in OS cells by western blot analysis. The results showed that as compared to the minimal effects by either single agent, the combination resulted in a marked decrease in initiator pro-caspase-9 and a robust accumulation of activated caspase-3 in both wt-p53 OS cell lines. Moreover, the combination also more efficiently cleaved full-length PARP than either single agent in the OS cell lines (Figure 6(a)).

Oridonin markedly promotes Nutlin-3-triggered activation of apoptosis signaling in OS cells. (a) U2OS and (b) SJSA-1 cell lines were treated by 3 µM Nutlin-3 (N) alone, 2 µM Oridonin (O) alone, or their combination (C) for 24 h. DMSO (D) was used as a control treatment. Treated cells were lyzed and the protein level of full-length PARP (fl-PARP), cleaved PARP (cl-PARP), pro-Caspase-9, and cleaved Caspase-3 (cl-Casp3) was analyzed with western blotting analysis. Tubulin was used as a loading control. (c) U2OS and (d) SJSA-1 cell lines pre-treated by pan-caspase inhibitor Z-VAD-FMK for 1 h were treated by the combination of 3 µM Nutlin-3 and 2 µM Oridonin for 48 h. Cell death was determined by trypan blue exclusion assay. The data were presented by mean ± standard deviation of three independent experiments (*p < 0.05; **p < 0.01).
To determine whether caspases were required for the combinational effect, OS cell lines were pre-treated with 50 µM pan-caspase inhibitor (Z-VAD-FMK) for 1 h before the addition of two drugs concomitantly (Figure 6(b)). Cell death assay showed that inhibition of caspase activity by Z-VAD significantly attenuated combination-mediated cell killing effect. Collectively, these results suggest that Oridonin greatly enhances Nutlin-3-triggered cell death in OS cells through a caspase-dependent apoptotic signaling pathway.
Discussion
Small-molecule Mdm2 inhibitors have recently emerged as an important class of anticancer agents. It has been found that small-molecule Mdm2 inhibitors exert potent anticancer activity against OS and many other cancers.1,2 However, a major issue for this class of drugs is the fact that Mdm2 inhibitors as single agents have limited antitumor activity in clinical trials.1,2 This issue has considerably hindered the translation to the clinic.1,2 To address this issue, a number of groups have been exploring ways to promote the anticancer activity of Mdm2 inhibitors through combination with other agents. Studies in neuroblastoma, breast cancer, ovarian cancer, acute myeloid leukemia, and chronic myeloid leukemia cells showed that the anticancer activity of Mdm2 inhibitors could be considerably enhanced through combination with chemotherapeutic and other anticancer agents.9,19–22 We here reported a novel strategy to improve the activity of Mdm2 in OS cells. This strategy consists of Mdm2 inhibitors Nutlin-3 and Oridonin, a bioactive constituent of the Chinese medicinal herb. It was noteworthy to mention that simultaneous treatment with both agents not only synergistically inhibited cell viability but also displayed a strong cytotoxic effect in OS cells. Besides, Oridonin has long been used in clinic as an alternative medicine for cancer treatment.10–13 Therefore, this combination strategy is warranted for further in vivo studies and even clinical studies in patients with OS.
Our data suggest that apoptosis plays a significant role in the synergy between Nutlin-3 and Oridonin in OS cells. This is supported by the results that Oridonin substantially enhances Nutlin-3-triggered caspase activation and PARP cleavage and also by the finding that a caspase inhibitor significantly attenuates the cell death induction by the combination. Because apoptosis is regulated by Bcl-2 family proteins, we analyzed the effect of the two drugs on the expression of several important Bcl-2 family members in OS cells. Consistent with previous reports, our data show that Nutlin-3 induces upregulation of the pro-apoptotic Puma, a p53-target gene.4–6 However, Mdm2 inhibitors had little effect on other Bcl-2 family proteins in OS cells. 9 According to recent reports, Puma alone cannot fully neutralize the highly expressed anti-apoptotic Bcl-2 family members in many tumor cells.23–25 This may be a potential reason for the modest apoptotic effect of Mdm2 inhibitors as a single agent in OS and other tumor cells. In the analysis of the effect on Bcl-2 family proteins by Oridonin, we found that this drug significantly increases the level of pro-apoptotic protein Bim and also considerably inhibited the expression of anti-apoptotic proteins Bcl-2 and Bcl-xl in OS cells. The sum outcome of combination treatment is, therefore, a substantial increase in the pro-/anti-apoptotic Bcl-2 ratio, thus leading to robust apoptosis. Importantly, Bim levels increase very rapidly after Oridonin treatment, suggesting that this pro-apoptotic Bcl-2 family protein may be involved in the initiation of apoptosis by the combination. Additionally, our data show that the synergy between Nutlin-3 and Oridonin only occurs in OS cells harboring wt-p53, but not in p53-deficient OS cells. This provides evidence showing that the upregulation of Puma plays an essential role in synergistic induction of apoptosis by Nutlin-3 and Oridonin.
Previous studies have revealed that Oridonin is a natural multi-target agent. However, the exact mechanisms by which Oridonin exhibits anticancer activity are not well understood.10,11 Recent studies showed that Oridonin is involved in the post-translational modifications of Bcl-2 family members by binding to and inhibiting microRNA clusters. For instance, Weng et al. 13 reported that Oridonin increased Bim expression by inhibiting miR-17 and miR-20a in leukemia cells. Yang et al. 26 showed that Oridonin increased the expression of Bim and Bax by decreasing miR-32 expression in colon cancer cells. Moreover, we recently found that Oridonin significantly induced upregulation of Bim in melanoma cells. 12 Together with our finding in this study, it is suggested that Bim upregulation by Oridonin might be a broad-spectrum activity in cancer cells, to which should be paid special attention in future anticancer research.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
